Abstract
Objective
To investigate the effect of the H1-antihistamine clemastine on the migraine-inducing abilities of pituitary adenylate cyclase activating peptide-38.
Methods
We conducted a double-blind, randomized, placebo controlled two-way cross-over study. Twenty migraine without aura patients were randomly allocated to receive bolus clemastine 2 mg (1 mg/ml) or bolus saline 2 ml intravenously over 2 min on two study days. Following each bolus injection, 10 pmol/kg/min of pituitary adenylate cyclase activating peptide-38 was administered intravenously over 20 min. We recorded migraine/headache characteristics every 10 min until 90 min after the start of infusion, and collected blood to investigate mast cell degranulation and the inflammation markers tryptase and tumor necrosis factor-alpha before and after infusion of pituitary adenylate cyclase activating peptide-38.
Results
After clemastine pretreatment, five out of 20 participants developed a migraine-like attack in response to a pituitary adenylate cyclase activating peptide-38 infusion compared to nine out of 20 after placebo pretreatment (p = 0.288). Following clemastine pretreatment, 15 out of 20 participants reported headache in response to a pituitary adenylate cyclase activating peptide-38 infusion, whereas 19 out of 20 participants did so following placebo pretreatment (p = 0.221). We found no difference in area under the curve 12 h for headache intensity between the two experimental days (p = 0.481). We found no difference in area under the curve 180 min for tryptase (p = 0.525) or tumor necrosis factor-alpha (p = 0.487) between clemastine and placebo pretreatment days.
Conclusion
H1-antihistamine, clemastine, failed to prevent migraine or headache after pituitary adenylate cyclase activating peptide-38 infusion, thus making a role for histamine release or mast cell degranulation in pituitary adenylate cyclase activating peptide-38-induced migraine less likely.
Introduction
Pituitary adenylate cyclase-activating peptide-38 (PACAP38) induces long lasting dilation of extracranial arteries (1,2) and migraine-like attacks (3,4). The molecular mechanisms responsible for PACAP38 induced migraine and the extent to which prolonged dilation of extracranial arteries contributes to provoked migraine remain unknown. Mast cell degranulation and subsequent activation of peripheral trigeminal sensory fibers may play a role in migraine pathophysiology (4). In rats, mast cell degranulation was shown to cause prolonged vasodilation and activation of meningeal nociceptors as well as downstream activation of the spinal trigeminal nucleus (5). PACAP38 induces histamine release from human skin mast cells (6) and mast cell mediated wheal after cutaneous injection (7).
While often referred to as H1-receptor antagonists, the H1-antihistamines are inverse agonists that downregulate constitutive activity of the H1-receptor (8). Histamine induces migraine-like attacks in migraine without aura (MO) patients and pretreatment with an H1- antihistamine prevents the provoked attacks (9).
Here we investigated the effect of the H1-antihistamine clemastine on the migraine-inducing abilities of PACAP38 in a double-blind, randomized, placebo-controlled two-way crossover study. We hypothesized that pretreatment with clemastine would prevent the development of a migraine-like attack after intravenous infusion of PACAP38. In addition, we hypothesized that clemastine pretreatment would inhibit the increase in biomarkers of mast cell degranulation; tryptase (10) and tumor necrosis factor alpha (TNF-alpha) (11).
Materials and methods
We randomly allocated 20 patients with MO (13 women, seven men) to receive either bolus clemastine 2 mg (1 mg/ml) or bolus saline 2 ml intravenously over 2 min on two study days separated by at least one week. Dose of PACAP38 was chosen based on previous migraine provocation studies (3,12). Following each bolus injection, 10 pmol/kg/min of PACAP38 (Calbiochem) was administered intravenously over 20 min. The solutions of bolus injections looked identical. Randomization was done by medical staff not involved in the study. The randomization code remained in the hospital during the study and was not available to the investigators until the study was completed.
Clemastine and PACAP38 were purchased from Merck Millipore (Hellerup, Denmark) and analyzed by Calbiochem. PACAP38 was prepared for infusion and randomized in a balanced fashion by the central pharmacy. All stock solutions were stored at −20℃ before usage.
Inclusion criteria for the study were: MO-patients fulfilling the International Headache Society (IHS) criteria of headache classification (13), ages 18–60, and weight 50–100 kg. Fertile women were included only if using either oral contraceptives or intrauterine devices or if sexually inactive. Exclusion criteria were: Tension-type headache exceeding five days a month; any other primary headache; headache or intake of analgesics less than 48 h and migraine less than 72 h before trial days; daily intake of any medication, including migraine prophylactic drugs (oral contraceptives were allowed); intake of any medication other than oral contraceptives less than four times the half-life of the medication before study days; pregnancy or breastfeeding; history or clinical findings suggestive of hypertension (systolic blood pressure > 150 mmHg and/or diastolic blood pressure > 100 mmHg), hypotension (systolic blood pressure < 90 mmHg and/or diastolic blood pressure < 50 mmHg); mental illness or substance abuse; or cardiovascular disease, including cerebrovascular disease or any other type of illness that the investigating medical doctor deemed relevant for participation in the trial. A full medical history was taken, a medical examination was performed, and electrocardiography (ECG) was recorded to detect any evidence of cardiovascular disease.
Standard protocol approvals, registrations, and patient consents
All participants were enrolled via the websites sundhed.dk and forsøgsperson.dk during July 2015 through April 2017 (Figure 1). All participants gave their written informed consent before inclusion. The ethics committee of Copenhagen approved the study (Journal no.: H-4-2014-103) and it was conducted according to the Helsinki II declaration. The study was registered at ClinicalTrials.gov (NCT02364453).
Flow chart of recruitment process.
Experimental protocol
The study was performed at the Danish Headache Center. Participants arrived non-fasting at the clinic between 9:00 and 10:00 am on each study day. Due to logistical difficulties, one participant arrived at 1:00 pm on one study day. Before study start, participants were 72 h migraine free and 48 h headache free, and free from intake of any analgesics. Fertile women took urine pregnancy tests before experiments. Participants lay in the supine position in quiet surroundings resting for 15 min before having baseline values measured twice (T-10 and T0). A single intravenous cannula inserted into the antecubital vein was used both for blood sampling and administration of saline/clemastine and PACAP38. Following each blood sampling, the cannula was irrigated using saline.
Each PACAP38-infusion began at T0 and lasted 20 min. Vital signs and a headache questionnaire were recorded every 10 min from T-10 until 90 min after the infusion started. ECG monitoring was used during the entire experiment. Intravenous saline 500 ml was given to 16 of 20 participants on both study days to prevent postural dizziness during mobilization after the 70 min observational period. Afterwards, the participants were offered lunch and awaited the final blood sample in the trial room. The participants filled out the first two columns of their self-administered questionnaire (see paragraph on headache questionnaire below) before leaving the hospital, and thus were able to ask questions about how to fill it out. The following day, patients returned for an additional blood sample taken 24 h after infusion start. At this time, headache questionnaires were returned.
Blood sampling
Blood was obtained four times, using a syringe from a venous catheter, at 0, 60, 180 min and 24 h after the start of infusion. Before each blood sampling of 20 mL, 3 mL of blood was discarded from the catheter because it had been flushed with isotonic saline after the last sampling. The collected blood was immediately transferred into one 10 mL vacutainer containing clot activator and gel for serum separation and one 10 mL vacutainer containing ethylenediaminetetraacetic acid (EDTA) for plasma separation. On the day of infusion, vacutainers for serum rested at room temperature and vacutainers for plasma at 5℃ from 30 min till 210 min after infusion. Vacutainers with blood collected 24 h after infusion start rested for 30 min. All vacutainers with blood were centrifuged at 4℃ for 10 min at 1600 g. Subsequently, serum and plasma were transferred into polypropylene tubes and stored at −20℃ until further analyses.
Determination of tryptase and TNF-alpha
Serum tryptase was quantified by use of a fluorescence immunoassay for the Phadia 250 analyser (Phadia, Thermo Fisher Diagnostics, Allerød, Denmark). Plasma TNF-alpha was quantified by use of the high sensitivity human TNF-alpha Quantikine immunoassay kit (R&D Systems, Bio-Techne, Abingdon, United Kingdom). The procedures of the kit inlay were followed.
Headache intensity and questionnaire
Headache intensity was recorded at baseline (T-10, T0) and every 10 min up to 90 min on a verbal rating scale from 0 to 10. Zero means no headache, 1 represents a very mild headache (including a sensation of pressing or throbbing or otherwise altered sensation in the head not associated with pain), 5 is headache of moderate intensity and 10 is the worst headache imaginable (14). In addition, headache localization, characteristics and associated symptoms were recorded.
After the 90 min of in-hospital monitoring by the investigator, participants were carefully instructed to continue recording at home using a self-administered questionnaire. Self-recording began at 2 h and continued to 12 h post-infusion or until bedtime. Recordings at 2 h and 3 h were done while still in the hospital. Remaining recordings were done at home. The questionnaire recorded headache characteristics and associated symptoms according to the International Headache Society (IHS) criteria (13). It also included questions concerning adverse events, premonitory symptoms (unusual fatigue, yawning, neck stiffness, mood swings, difficulty concentrating, hunger, or thirst) and if the reported headache mimicked the participants’ spontaneous migraine attacks.
The participants could take rescue medication or their usual anti-migraine treatment at any time. Ethically, having given them a headache-inducing substance, we could not instruct them to abstain from taking analgesics even if the pain did not mimic their usual migraine.
Migraine-like attack criteria
As experimentally provoked migraine attacks by pharmacological substances are not spontaneous, they cannot fulfill strict IHS criteria for migraine without aura (13). Thus, we used the following definition for a migraine-like attack:
Migraine-like attack fulfilling either 1 or 2:
Headache fulfilling criteria C and D for migraine without aura according to the IHS criteria (13). Headache described as mimicking the usual migraine attack and treated with acute migraine medication (rescue medication).
Vital signs
Heart rate and blood pressure were measured at baseline timepoints (T-10, T0) and every 10 min during the observation period of 90 min after PACAP38 administration using an auto-inflatable cuff (Microlife®). ECG monitoring was performed throughout the entire trial using Kivex Cardiofax V.
Statistical analyses
Headache intensity scores are presented as median (range). Heart rate and mean arterial blood pressure data are presented as median values. We calculated median peak headache intensity and median time to onset of headache as well as mean time to onset of immediate and delayed migraine-like attacks.
Based on previous studies reporting migraine induction in 60–70% of participants after PACAP38 (3,4,15), calculation of sample size was based on detection of a difference between treatments in migraine-incidence of 65% on the placebo day and 20% on the clemastine day at a 5% significance level with 80% power. Sample size was calculated to 14 participants, and 20 participants were included to ensure power.
The primary end-points were incidence of migraine-like attacks and the difference in area under the curve (AUC) for headache intensity scores (0–12 h) between clemastine and placebo. Secondary end-points were difference in incidence of adverse events, changes over time for mean blood pressure and in heart rate from 0–90 min after start of infusion. Differences in AUC for headache intensity scores were tested by the Wilcoxon signed rank test and the incidence of migraine-like attacks was analyzed as categorical paired data with McNemar’s test.
Analyses were not corrected for multiple testing and were all performed with GraphPad Prism 5 for Windows. p < 0.05 was considered to be the level of significance.
Results
Twenty MO patients with a median migraine attack frequency of 2.5 days per month (range 1–5) completed the study. One participant was prescribed daily medication after the first experimental day and therefore excluded and replaced. Mean age was 29 years (range 21–54) and mean weight was 73.5 kg (range 55–94). One participant previously participated in a PACAP38 provocation study with lower doses (4, 6 and 8 pmol/kg/min) of PACAP38 (12) than used in the present study. This patient did not report migraine-like attacks on either experimental day.
Incidence of migraine-like attacks
Clinical characteristics and associated symptoms of usual and provoked migraine-like attacks and/or headaches in 20 MO patients. On both of the two study days, participants received pretreatment of intravenous bolus clemastine 2 mg (1 mg/ml) or intravenous bolus saline 2 ml over 2 min followed by 10 pmol/kg/min of PACAP38. Red text represents migraine-like attacks.

Localization/intensity/quality (throb: throbbing; pres: pressing)/aggravation (by cough during in-hospital phase and by movement during out-hospital phase).
Nausea/photophobia/phonophobia.
Migraine-like attacks are defined according to criteria, described in methods.
Pain freedom or pain relief (≥50% decrease in intensity) within 2 h.
+: present; −: not present; N/A: not applicable.
This patient fulfilled criteria for migraine-like attacks at three separate time points on this study day.
Incidence of migraine-like attacks, headache, intake of simple analgesics and triptans, and effect of simple analgetics and triptans. On two study days the participants, 20 MO patients, received pretreatment of intravenous bolus clemastine 2 mg (1 mg/ml) or intravenous bolus saline 2 ml over 2 min followed by 10 pmol/kg/min of PACAP38 on both study days.
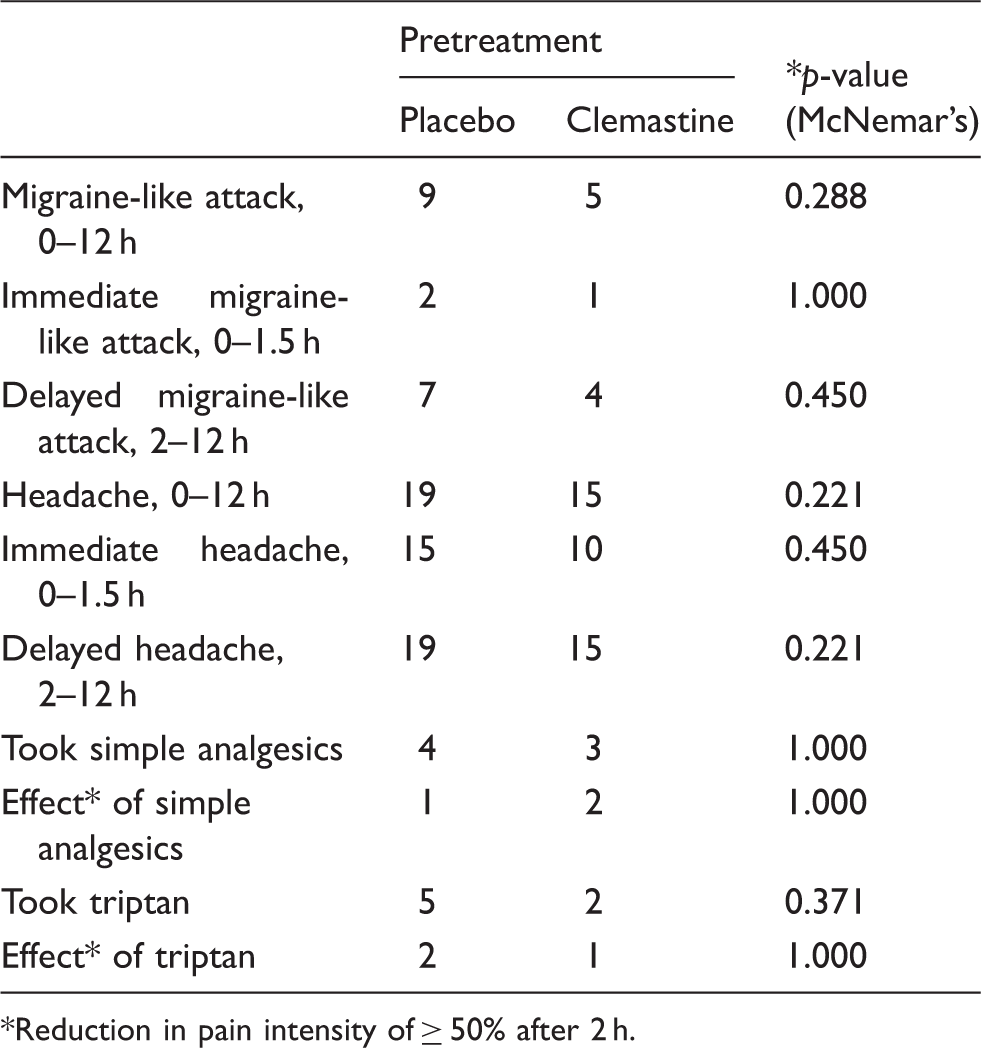
Reduction in pain intensity of ≥ 50% after 2 h.
Incidence and intensity of headache
Following clemastine pretreatment, 15 out of 20 (mean 75%, 95% CI 54–96%) participants reported headache following PACAP38 infusion, whereas 19 out of 20 (mean 95%, 95% CI 85–100%) participants did so following placebo pretreatment (McNemar test, p = 0.221). We found no difference in AUC for headache intensity after clemastine vs. placebo pretreatment over the 12 h observation period (p = 0.481) (Figure 2). Exploratory analyses showed no difference in AUC between placebo and clemastine days from 0–120 min (p = 0.755) or from 2–13 h (p = 0.234).
Median (thick lines) and individual (thin lines) headache intensity on clemastine and placebo pretreatment day on a verbal rating scale (VRS) of 0–10. On both of the two study days the participants, 20 MO patients, received pretreatment of intravenous bolus clemastine 2 mg (1 mg/ml) or intravenous bolus saline 2 ml over 2 min followed by 10 pmol/kg/min of PACAP38.
Adverse events
Adverse events incidence (McNemar’s test). Reported symptoms after infusion of intravenous bolus clemastine 2 mg (1 mg/ml) or intravenous bolus saline 2 ml intravenously over 2 min followed by 10 pmol/kg/min of PACAP38.

Rescue medication
Rescue medication was taken during 11 out of 14 migraine-like attacks (see Table 1). After clemastine pretreatment, five participants took analgesics and median time to intake was 6 h (range 3–10 h). After placebo pretreatment, eight participants took analgesics and median time to intake was 8 h (range 2–12 h). In the case of long-lasting and unpleasant flushing or puffing of the face, patients were given a single dose of 100 mg prednisolone to counteract these effects (Table 1).
Heart rate and mean arterial pressure
We found no difference in AUC for heart rate between placebo and clemastine pretreatment (p = 0.261) (see Figure 3). Similarly, there was no difference in AUC for mean arterial pressure (p = 0.729) (see Figure 4).
Median heart rate (HR) on placebo pretreatment (green line) and clemastine pretreatment (pink line) day. Median of the mean arterial pressure (MAP) on placebo pretreatment (green line) and clemastine pretreatment (pink line) day.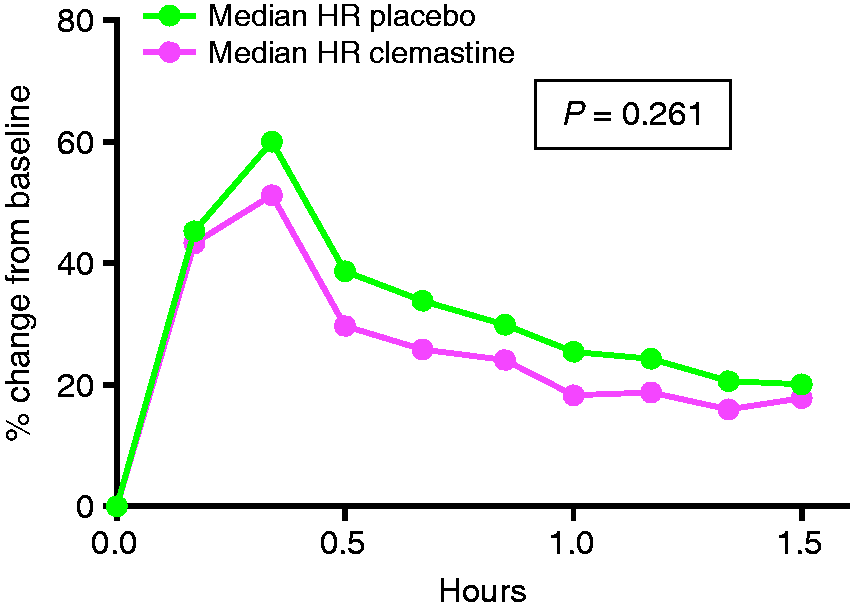

Tryptase and TNF-alpha
We found no difference in AUC0–180 min for tryptase (p = 0.525) or TNF-alpha (p = 0.487) between clemastine and placebo pretreatment day. In the six participants who returned for 24 h blood samples, we found no difference in AUC0–24 h between the clemastine and placebo pretreatment day for tryptase (p = 0.438) or TNF-alpha (p = 0.094). Furthermore, exploratory analysis of baseline versus 60 min of tryptase (p = 0.561) and TNF-alpha (p = 0.791) on placebo pretreatment day showed no difference.
Discussion
In the present study, we found no difference between pretreatment with the H1- antihistamine clemastine and placebo in prevention of PACAP38 induced migraine-like attacks or headache. These data question the importance of histamine release in PACAP38-induced migraine.
The histamine H1-receptor is widely distributed on different cell types including on mast cells, and on endothelial and smooth muscle cells of human cerebral, meningeal and temporal arteries (16,17). In migraine patients, elevated histamine levels (18) and the ability of histamine to induce migraine attacks (9) support a role of histamine in migraine (19). PACAP38 half-life is only 5–10 min (20), but we found the median time to onset of PACAP38-induced migraine-like attacks was 5 h after infusion, in line with previous studies (3,4). Therefore, the provoked migraine-like attacks are likely to involve other mechanisms rather than a direct effect of PACAP38 (21) or PACAP38-induced release of histamine from human skin mast cells (6). In a provocation study in 20 MO patients, both histamine-induced headache and migraine attacks were blocked by mepyramine (an H1-antihistamine) pretreatment (9). Thus, our finding of no blocking effect by the H1-antihistamine clemastine on PACAP38-induced migraine speaks against a role of histamine as mediator of this process.
Leaving aside histamine, the question still remains whether mast cells play a role in PACAP38-induced migraine. Mast cells are found in the dura mater (22) and, once activated, secrete a wide range of mediators including the neurosensitizing protease tryptase and the vasodilatory cytokine TNF-alpha (23). Our serum tryptase and TNF-alpha levels did not differ between placebo and clemastine pretreatment days, suggesting no effect of the H1-antihistamine on release of these mediators. Furthermore, we found no increase in tryptase or TNF-alpha levels from baseline to 60 min on placebo pretreatment day, suggesting no PACAP38-induced release of these mediators. TNF-alpha has been reported to be elevated during spontaneous migraine attacks (24), but recent provocation studies on MO patients showed no increase in serum tryptase and TNF-alpha after PACAP38 infusion (3,25). Collectively, our findings provide no evidence of PACAP38-induced mast cell degranulation.
Importantly, one unexpected factor might have influenced our findings. We found that after clemastine pretreatment 25% participants developed a migraine-like attack compared to 45% after placebo pre-treatment. A migraine induction rate of only 45% is lower than previous PACAP38 provocation studies in MO patients (3,4,15), showing 58–73% migraine induction. This raises an interesting question about placebo response in provocation studies. Anticipation of pretreatment that might block the induced migraine could lower migraine induction rates in the placebo arm (26). Therefore, we speculate that anticipation of pretreatment blocking the induced migraine functions as a placebo response that lowered migraine induction rates in the present study. The unexpectedly low migraine-induction rate of 45% on PACAP38 after placebo pretreatment is a limitation of our study and may have affected our ability to detect a true effect of clemastine pre-treatment. Our power calculations indicated 14 participants would be necessary, but we included 20. However, a post hoc sample size calculation assuming 45% migraine induction on placebo pretreatment day versus 20% on clemastine pretreatment day with the same significance level and power showed we would have needed 39 participants to detect such a difference. Thus, our conclusions about the role of histamine and mast cells in PACAP38-induced migraine must be drawn cautiously. Our strength is our robust double-blind cross-over study design.
Recently, targeting PACAP38 in migraine treatment has gained interest (27,28). PACAP38 has affinity for at least three known receptors: The VPAC1, VPAC2, and PAC1 receptors (29), and shares affinity for the VPAC1-2 receptors with vasoactive intestinal peptide (VIP), a parasympathetic neuropeptide structurally similar to PACAP38 that does not induce migraine in MO patients (30). Consequently, the migraine-inducing abilities of PACAP38 are hypothesized to arise from the PAC1 receptor, for which PACAP38 has 300–1000 times higher affinity than VIP (31). In rats, PAC1 receptor antagonism did not diminish PACAP38 induced mast cell degranulation (32). In addition, the selective PAC1 receptor agonist, maxadilan, did not degranulate mast cells (33). Since our study questioned the role of mast cell degranulation in PACAP38-induced migraine, the present data indirectly support the importance of the PAC1 receptor.
Another interesting aspect that should be considered is the possible interaction between CGRP and PACAP38. Animal studies of rat and rhesus monkey tissue show the presence of PACAP38 and CGRP and their receptors in the trigeminal ganglion and trigeminal nucleus caudalis (34–37). Furthermore, PACAP38 induces CGRP release from the trigeminal nucleus caudalis, but not the trigeminal ganglion in rats (37). In MO patients, infusion of PACAP38 did not alter CGRP levels, regardless of attack status after PACAP38 infusion (25). A newly completed experimental study on the effect of blocking CGRP with AMG334 in PACAP38-induced migraine will provide valuable insight into these mechanisms (38).
Conclusion
We found that the H1-antihistamine, clemastine, did not prevent migraine or headache after PACAP38 infusion, thus making a role for histamine release or mast cell degranulation in PACAP38 induced migraine less likely. Indirectly, this may provide support for the PAC1 receptor as a crucial player in PACAP38-induced migraine. Results from ongoing and planned phase II trials investigating the safety and efficacy of a PAC1 receptor antagonist in migraine treatment (27,28) are eagerly awaited to shed light on the therapeutic potential of this emerging treatment target in migraine.
Clinical implications
The H1-antihistamine, clemastine, does not prevent PACAP38-induced migraine or headache. Histamine and mast cells seem to be unlikely mediators of PACAP38-induced migraine.
Footnotes
Acknowledgements
We thank lab technicians Lene Elkjær and Winnie Grønning for expert assistance, Phadia, Thermo Fisher Diagnostics for assistance with biochemical analyses. Messoud Ashina had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Messoud Ashina is a consultant, speaker or scientific advisor for Allergan, Amgen, Alder, ATI, Eli Lilly, Novartis, and Teva, primary investigator for Amgen 20120178 (Phase 2), 20120295 (Phase 2), 20130255 (OLE), 20120297 (Phase 3), Alder ALD403-CLIN-001 (Phase 3), Amgen PAC1 20150308 (Phase 2 a), and GM-11 gamma-Core-R trials. Luise Haulund Vollesen, Song Guo and Malene Rohr Andersen declare they have no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from Lundbeck Foundation (R155-2014-171) and Research Foundation of Rigshospitalet. The funders did not partake in designing and conducting the study; in the collection, management, analysis, and interpretation of the data; in the preparation, review, or approval of the manuscript; or in deciding to submit the manuscript for publication.
