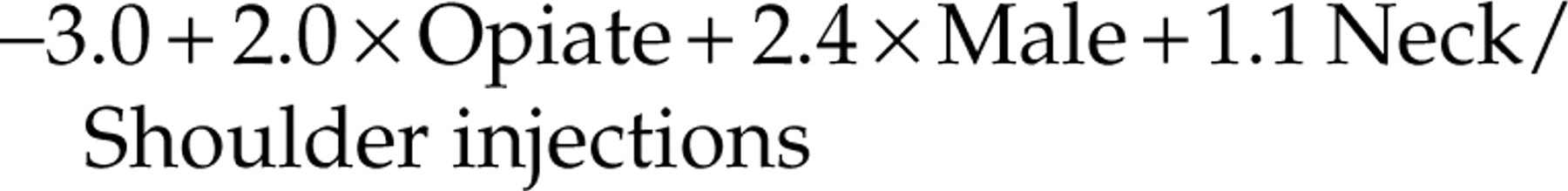Abstract
We set out to identify predictors for the prophylactic effect of placebo injections in subjects with migraine by post hoc analysis of 81 subjects with episodic migraine receiving single-blind placebo injections in a prospective trial of botulinum toxin. Possible predictors of placebo prophylaxis were compared among placebo responders (PRs) and placebo non-responders (PNRs). There were 34 PRs (42%) and 47 PNRs (58%). Male gender [odds ratio (OR) 5.83, 95% confidence interval (CI) 1.12, 30.14, P = 0.022], a history of opioid use (OR 4.44, 95% CI 1.47, 13.41, P = 0.005) and injections in the neck/shoulders (OR 2.44, 95% CI 0.93, 3.19, P = 0.033) were associated with placebo response. Of subjects with two or more of these signs, 88% were PRs compared with 31% of subjects with one or less. Male gender, opioid use and injections in the neck/shoulders are associated with placebo prophylaxis. These findings may have important implications for the design of future clinical trials and for the clinical management of migraineurs.
Introduction
The placebo effect is an outcome that should be maximized during patient care and minimized during placebo-controlled trials. Placebo response rates range from 6 to 47% in the acute treatment of migraine and 20–49% in migraine prophylaxis (1–8). Functional neuroimaging studies have suggested a prominent role of the endogenous opioid system in the neurobiology of placebo analgesia. If predictors of placebo analgesia can be identified, clinicians can use these to their advantage in enhancing treatment outcomes and control for them when designing clinical trials. Reduction of the placebo response rate may maximize the potential to determine the true biological effect of experimental therapies. Identifying predictors of placebo response may also advance our understanding of the biology of this phenomenon.
Methods
This study was investigator-initiated and approved by the Mayo Clinic Institutional Review Board. It was a post hoc analysis of data extracted from a study of 81 subjects who were enrolled in a randomized, placebo-controlled, prospective analysis of botulinum toxin type A for the prophylactic treatment of episodic migraine. All subjects received single-blind injections of 0.9% preservative-free saline (placebo) prior to randomization to botulinum toxin or a second placebo injection. As part of this enriched study design, subjects determined to be placebo responders (PRs) to the first injection were excluded from randomization. During the single-blind phase, all subjects received a single injection into the procerus and each corrugator. Two injections were given into each frontalis and temporalis muscle. All subjects therefore received at least 11 injections. Subjects with tenderness to palpation over the cervical paraspinal muscles received an additional four injections. Subjects with tenderness to palpation over the trapezius muscles also received an additional six injections. Therefore, subjects with tenderness in both of these muscle groups may have received an additional 10 injections. Results in this study were derived from the response to the first placebo injections only.
Subjects were enrolled from our headache subspecialty clinic. They were male or female, 18–65 years of age, with episodic migraine with or without aura (according to International Headache Society criteria) of ≥1 years' duration and with onset prior to 50 years of age (9). Migraine-related disability was greater than or equal to Migraine Disability Assessment Scale (MIDAS) grade III. Subjects were excluded if they used acute headache medication ≥12 days per month or had used more than two prophylactic medications or changed prophylactic medications within 90 days of the screening visit. Changes in prophylactic and acute medications during study participation were not allowed. Subjects were told that they were enrolling in a placebo-controlled trial of botulinum toxin, but were blinded as to the nature of individual injection sessions. Thus, subjects did not know if or when they might receive placebo injections and were not aware of the randomization ratio.
Data were collected regarding subject age, gender, body mass index, number of years with headache, number of prior treatments, percentage of headaches associated with nausea and vomiting, photophobia and phonophobia, percentage that were throbbing, percentage unilateral, presence of aura, acute and prophylactic medication use and comorbid psychiatric disease. Baseline MIDAS, Headache Impact Questionnaire (HImQ) and Migraine Specific Quality of Life Questionnaire (MSQLQ) were collected. All subjects completed a headache diary for the 4 weeks before and the 4 weeks after placebo injection allowing for calculation of headache frequency, severity, duration and antimigraine medications used.
Subjects were considered PRs if they had at least a 30% reduction in diary-recorded headache frequency or severity during the 4 weeks following placebo injections compared with the 4 weeks preceding those injections. We chose a 30% reduction because of the short observation period (1 month) after the single-blind injection compared with the longer observation period in prophylactic studies (>3 months), where a 50% reduction has become a standard end-point.
Statistical analysis used group means with standard deviations. Student's unpaired t-test was used to compare non-categorical group data. Categorical parameters were compared by calculating odds ratios (ORs) with 95% confidence intervals (CIs). A multivariable model was created by using multiple logistic regression and forward selection modelling strategy. Terms were retained in the model if P < 0.050. The result of the multivariable model was cross-validated by using the jackknife (leave-one-out) method.
Results
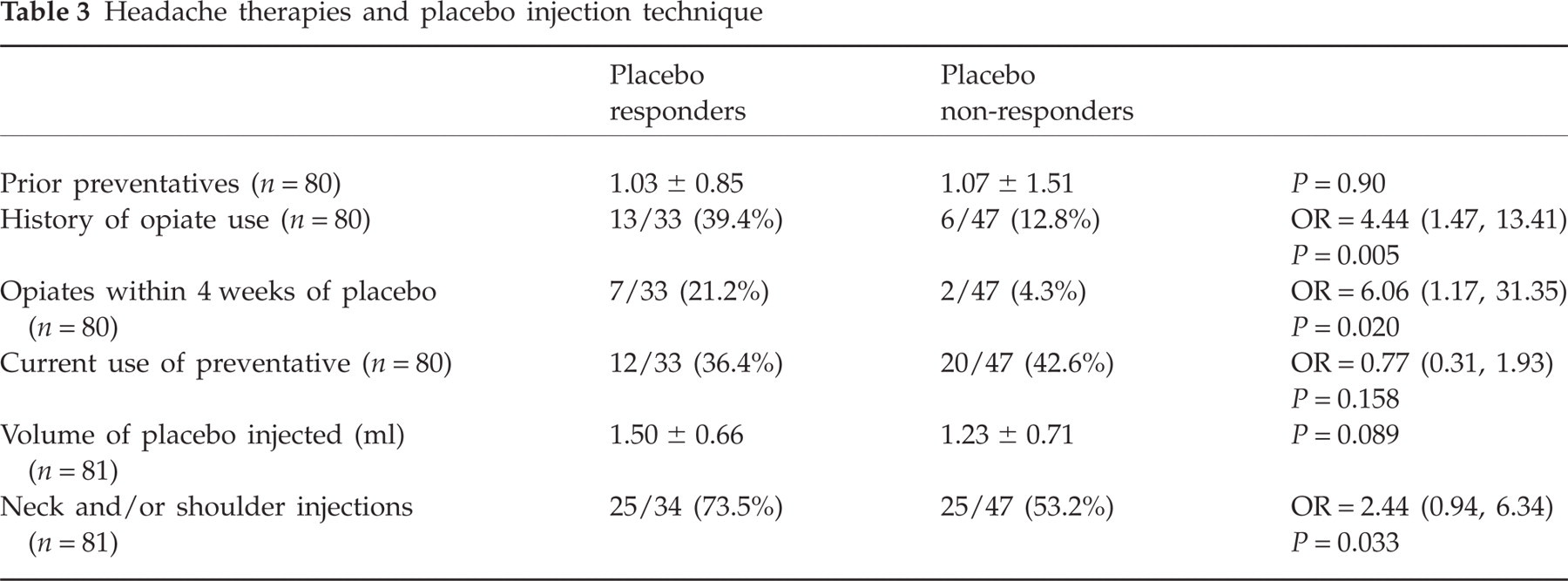
Nine subjects (11.1%) were male and 72 (88.9%) female. The mean age was 44 years. Thirty-four (42.0%) were PRs and 47 (58.0%) were placebo non-responders (PNRs). Whereas 42% had at least a 30% reduction in headache frequency or severity (PRs), only 21% had at least a 30% worsening in headache frequency or severity. Male gender, opioid use and inclusion of neck and/or shoulder injections were associated with an increase in the probability of placebo response. Of 34 PRs, seven (20.6%) were male and 27 (79.4%) female. Among 47 PNRs, two (4.3%) were male and 45 (95.7%) female. The OR of placebo response comparing males with females was 5.83 (95% CI 1.12, 30.14, P = 0.022). A medication use history was available from 33 PRs and 47 PNRs (n = 80). Thirteen of 33 (39.4%) PRs reported a history of opiate use compared with 6/47 (12.8%) PNRs (OR 4.44, 95% CI 1.47, 13.41, P = 0.005). Seven of 33 (21.2%) PRs recorded opiate use within 4 weeks prior to placebo injection compared with 2/47 (4.3%) PNRs (OR 6.06, 95% CI 1.17, 31.35, P = 0.020). Neck and/or shoulder injections were performed on 25/34 (73.5%) PRs and 25/47 (53.2%) PNRs (OR 2.44, 95% CI 0.93, 3.19, P = 0.033). Mean volume was 1.50 ml among PRs and 1.23 ml among PNRs (P = 0.089).
Multiple logistic regression analysis with forward selection modelling with retention of terms with P < 0.05 was performed. A history of opiate use, male gender and inclusion of neck and/or shoulder injections were retained. The best predictor of placebo prophylaxis in this study is determined by the following equation including terms for opiates (Yes = 1, No = 0), gender (Male = 1, Female = 0) and neck/shoulder injections (Yes = 1, No = 0):
The highest likelihood ratio was obtained for a score of 0.00 points. Subjects who had a positive score (more than one of the above signs) had an 88% chance of placebo prophylaxis. Subjects with a score of ≤0.00 (one or less of the above signs) had only a 31% chance of placebo prophylaxis.
There were no other significant differences among parameters measured between PRs and PNRs. This included subject demographics other than gender, such as age and body mass index (Table 1), headache characteristics (Table 2), number of prior prophylactic medications and current use of preventive medications (Table 3), HImQ scores at screening visit, MIDAS at screening visit, MSQLQ scores at screening visit and presence of depression or anxiety (Table 4).
Subject demographics

Headache characteristics

Headache frequency, Days with headache; Recorded severity: 1, mild; 2, moderate; 3, severe; Estimated severity: scale from 0 (no pain) to 10 (worst pain); Migraine aura, at least some headaches with presence of aura.
Headache therapies and placebo injection technique
Headache impact and psychiatric co-morbidity

HImQ, Headache Impact Questionnaire; MIDAS, Migraine Disability Assessment; MSQLQ, Migraine Specific Quality of Life Questionnaire.
Discussion
‘Placebo analgesia’ refers to reduction or abolition of pain following a therapeutic intervention that is not believed to have an independent exogenous effect on pain. Studies examining placebo analgesia consistently show a large placebo response rate in both acute and prophylactic migraine trials (1–8).
Understanding mechanisms mediating placebo analgesia and identifying factors which may predict a placebo response are important in maximizing favourable patient care outcomes. The efficacy of any treatment for pain is likely to be due to a combination of the true biological effect of that intervention and the non-specific effects attributed to that treatment (10). In contrast, minimizing placebo response is important in clinical trials of new therapeutics for migraine. Drug trials of acute and prophylactic migraine treatments require comparison with placebo, a recommendation reinforced by the International Headache Society Clinical Trials Subcommittee (1). However, when placebo response rates are high, the probability of a type II error increases (11). Minimizing the placebo response rate is therefore important in order to identify new therapies with true disease-specific biological activity. In addition, without proper knowledge of the factors that predict placebo response, investigators cannot control for such factors between study groups, thereby creating the potential for unintentionally biasing outcomes.
Several different factors have been considered in this study and by previous investigators as possible predictors of the placebo response. Identified positive predictors have included gender, patient population (North Americans vs. Europeans), younger age, positive patient expectation of outcome, less severe disease, lack of patient knowledge regarding the possibility of receiving placebo (‘hidden placebo’), physician confidence in the efficacy of the therapy used, frequency of administration of the placebo, unbalanced active:placebo randomization ratios, comorbid anxiety and positive response to prior therapies (2, 4, 12–19). Participation in a study may, of itself, result in subject improvement due to education, the caregiver environment and social support (20). Regression to the mean can also account for some degree of improvement in pain studies (21). Painful conditions often follow a fluctuating pattern of severity and patients volunteering to enroll in a study may be more likely to be feeling unwell. Therefore, natural fluctuation back to a less severe state may be responsible for some improvement in both the placebo and treatment groups. However, in this study 21% of subjects had at least a 30% worsening in headache frequency or severity compared with 42% who had at least this much improvement, suggesting that there was a true placebo response.
The pathophysiology of the placebo response, which has only recently begun to be elucidated, is probably dependent upon several modulating systems, including the endogenous opioid, dopaminergic and limbic systems (22, 23). Much recent work in this field has focused on the endogenous opioid system. Animal and human studies have shown that naloxone, an opioid receptor antagonist, can partially or completely inhibit the placebo response (24–27). A positron emission tomography (PET) study comparing brain activation patterns after opioid injection with patterns after placebo injection has shown that the same regions of the brain were activated after both (28). Activation occurred in the rostral anterior cingulate cortex, the periaqueductal grey and pons. These brain structures have high opioid receptor densities and are involved in the descending inhibitory pain system (29). High PRs had greater activation of the rostral anterior cingulate cortex after opioid injection than low PRs. This may reflect genetic variances in µ-opioid receptor expression (30). This suggests that the responsiveness of the opioid system might be a predictor of not only the response to exogenous opioid administration but also placebo response in pain studies. In another study, opioid receptor activity was directly measured by using a combination of molecular imaging with a µ-opioid receptor radiotracer in combination with PET (31). Placebo-induced analgesia was shown to increase µ-opioid receptor transmission in the left dorsolateral prefrontal cortex, rostral anterior cingulate cortex, anterior insula and nucleus accumbens. Such changes correlated with a decrease in perceived pain or the expectation of analgesia. These data provide direct evidence of a link between opioid receptor activity and placebo-induced analgesia. Although there are no data to suggest that these findings would not apply to people with migraine, similar experiments have not yet been replicated in this specific patient group.
Opioid use was associated with placebo response in this study. Our assumption is that subjects using opioids were those who received some benefit from their use. Subjects with greater response to exogenous opioids may be more likely to benefit from placebo injections secondary to endogenous opioid release. This hypothesis is supported by prior studies showing higher placebo response rates in subjects pretreated with opioids (32, 33). The finding in this study that prior or current opioid use may be associated with a significant reduction in the frequency or severity of painful attacks as a result of placebo injections supports the overwhelming experimental, clinical and functional imaging data implicating the endogenous opioid system as an important mediator of placebo analgesia. In addition, if this observation is validated, it may be necessary to exclude or control for subjects who are strong responders to opioids in clinical trials in order to minimize the placebo response.
Male gender was also associated with placebo response. Sex differences in the antinociceptive effects of opioids have been observed in both primates and rodents, with males being generally more sensitive than females (34, 35). While the basis for this difference in opioid responsiveness is not entirely clear, there is ample evidence indicating that pharmacokinetic and pharmacodynamic factors cannot completely account for these differences, as µ-opioids are more potent in males despite comparable brain and plasma levels of morphine after systemic administration in males and females (34). Therefore, the results of this study, demonstrating that male sex may predict a prophylactic response to placebo, is in keeping with an emerging body of evidence which suggests that opioid analgesia is more robust in males. This, of course, should have implications for future clinical trials in migraine and other types of pain, as suggested by others (36).
Finally, a higher number of injections due to inclusion of the neck and/or shoulders was associated with an increased frequency of placebo response. It is unclear if the association with placebo response is due to the increased number of injections or to the addition of injections specifically into the neck and/or shoulders. Although not tested, a greater number of injections may correlate with greater subject expectation of improvement. It has previously been noted that the more invasive a procedure, the greater the placebo response (37). This suggestion is supported by a recently published single-blind randomized controlled trial of arm pain which showed that a validated sham acupuncture device has a greater placebo effect on subjective outcomes than placebo pills (38). We hypothesize that a greater number of injections and, specifically, injections in the cervical paraspinal and trapezius muscles results in more subject discomfort. Higher pain levels would be expected to result in greater release of endogenous opioids, which may then lead to higher placebo response—‘More pain, more gain’.
There are several limitations of this study. The results from this analysis need to be tested in larger populations. In addition, we made the assumption that opioid use correlated with opioid responsiveness. However, we did not directly measure the pain response to opioid administration in these users. Also, some subjects had used opioid in the past but did not use them in the 4 weeks preceding placebo injections, suggesting that perhaps they were not receiving adequate analgesia. The frequency and extent of opioid use was available only for those using opioids during the 4 weeks prior to placebo injection. This group used a mean of five doses during those 4 weeks. Although not reaching significance, of those who used opioid within 5 days prior to placebo, 4/5 were PRs. However, it can be hypothesized that regardless of the analgesic response to exogenous opioid, prior opioid exposure alone could influence the placebo response. This study is also limited by recall bias. Subjects were asked to recall their prior medications from a comprehensive list provided at the time of enrolment into the study, resulting in the possibility that some had used opioid in the past but did not remember.
Conclusions
Male gender, opioid use and an increased number of injections were associated with a prophylactic effect of placebo injections of saline. The integral involvement of endogenous opioid pathways in the placebo response suggests that the analgesic efficacy of exogenous opioids, or possibly exposure to exogenous opioids, may predict the likelihood of placebo response in migraine, and possibly other pain studies. In addition, this analysis suggests that male gender is associated with higher placebo response in migraine prophylactic studies, which is in keeping with robust experimental and clinical evidence for a significant difference in opioid responsiveness in males compared with females. A greater number of injections may increase subject expectation of improvement and thus increase the placebo response rate. If confirmed, these potential predictors of placebo analgesia may have important implications for clinical practice and the design of future placebo-controlled studies in migraine, as well as other pain conditions. Further study is required to confirm these results in a larger sample and address other possible predictors of the placebo response that may not have been considered in this study.