Abstract
Background: Data on the association between sex hormone receptor polymorphisms and migraine are conflicting.
Methods: We performed a systematic review and meta-analysis on this topic searching for studies published until August 2009. For each study, we calculated odds ratios (ORs) and 95% confidence intervals (CIs) assuming additive, dominant, and recessive genetic models. We then calculated pooled ORs and 95% CIs.
Results and Conclusion: Among the seven genes targeted, four variants were investigated in multiple studies. Effect estimates from an additive model suggest that the ESR-1 594 G > A (pooled OR 1.37; 95% CI 1.02–1.83) and ESR-1 325 C > G (pooled OR 1.16; 95% CI 1.03–1.32) variants are associated with any migraine. This pattern does not differ between migraine with and without aura. In contrast, the ESR-1 Pvu II C > T and PGR PROGINS insert polymorphism do not appear to be associated with migraine. Results were driven by studies among Caucasians and may differ in other ethnic groups.
Introduction
Migraine is a common, chronic disorder characterised by recurrent headache attacks and combinations of gastrointestinal and autonomic nervous system symptoms (1), affecting 10–20% of the population. Up to one-third of migraine patients experience an aura prior to or during the migraine headache.
Population-based, clinical, and physiological studies support an important role for sex hormones in the pathogenesis of migraine. For example, migraine prevalence is 3–4-fold higher among women than men, a subgroup of women suffer from menstrual migraine or menstruation-related migraine, migraine prevalence often changes during pregnancy or after menopause, and both oestrogen withdrawal and changes in oestrogen levels can trigger migraine attacks (2–4). These findings have prompted studies investigating the association of variants in genes coding for proteins in sex hormone receptor pathways and metabolism with migraine.
Gene variants located in the oestrogen receptor 1 gene (ESR-1) (5–11), oestrogen receptor 2 gene (ESR-2) (11), progesterone receptor gene (PGR) (7,8,12), androgen receptor gene (AR) (12), follicle stimulating hormone receptor gene (FSHR) (11), nuclear receptor interacting protein 1 (NRIP1) (11), and cytochrome P450, family 19, subfamily A, polypeptide 1 gene (CYP19A1) (11) have been targeted. However, many results were either contradictory, which may be due to differences in ethnicity, sample sizes, and the proportion of migraine with aura (MA) and migraine without aura (MO) among the study populations, or have not been replicated in independent populations.
We sought to summarise the current evidence on the association between variants in genes coding for proteins in sex hormone receptor pathways and metabolism and migraine including MA and MO by systematically reviewing the literature and performing a meta-analysis.
Methods
Selection of studies
We followed the guidelines for systematic reviews of genetic association studies (13). Two investigators (MS and PMR) independently searched MEDLINE, EMBASE, and Science Citation Index from inception to August 2009 combining text words and MESH terms, were appropriate, for sex hormones (‘hormones’ or ‘sex hormones’ or ‘estrogen’ or ‘progesterone’) with terms for genetic variations (‘gene’ or ‘polymorphism’ or ‘genetic variation’) and terms for headache and migraine (‘headache’ or ‘headache disorders’ or ‘migraine’ or ‘migraine disorders’). The search terms were combined with the ‘explode’ feature where applicable. We did not use any language restrictions. In addition, we manually searched the reference lists of all primary articles and review articles.
A priori, we defined the following criteria for inclusion:
Studies must have a cross-sectional, case-control or cohort design. Authors must investigate patients with migraine and healthy control subjects. Authors must provide information on genotype frequencies of the investigated polymorphisms or sufficient data to calculate these. In studies with overlapping cases and/or controls, the largest study with extractable data was included. Studies must be published as full articles.
In a first step, two investigators (MS and TK) by consensus identified all studies not meeting any of the prespecified criteria by screening the title and abstracts. These studies were excluded. In a second step, the same investigators evaluated the remaining studies in their entirety. Studies were excluded if they did not meet all criteria.
Data extraction
Two investigators (MS and PMR) independently extracted data from the published studies and entered them in a customised database. Disagreements were resolved by consensus. The extracted data included authors and title of study, year of publication, country of origin, ethnicity of population investigated, setting (clinic vs population), study design, genotyping method, migraine status (any migraine, MA, MO), age and gender of study individuals, study size, allele and genotype frequencies, and information on additional genetic variants as well as gene–gene and gene–environment interactions, if investigated. If not given, genotype frequencies were calculated where possible. We did not contact the authors to collect further information.
Statistical analysis
We first used logistic regression to calculate odds ratios (ORs) and 95% confidence intervals (CIs) for the association between sex hormone receptor polymorphisms and migraine assuming additive, dominant, and recessive genetic models. We calculated these for polymorphisms which were investigated in at least two independent study populations. The additive model assumes that the risk for migraine among carriers of the heterozygous genotype is half way between carriers of the homozygous genotypes. While the dominant model assumes that carriers of the heterozygous and homozygous variant genotypes have the same risk of developing migraine compared with carriers of the homozygous wild-type genotype, a recessive model assumes that carrying the homozygous variant genotype is necessary to alter the risk for migraine compared with carriers of the heterozygous and homozygous wild-type genotype. We also determined Hardy–Weinberg Equilibrium (HWE) for the control group in each study. We investigated any migraine, MA, and MO.
We then pooled results from all studies and subsequently stratified analyses by ethnicity and gender where applicable.
We weighted the log of the ORs by the inverse of their variance to obtain pooled relative risk estimates. We ran random-effects models which include assumptions on potential variability across studies. We performed the DerSimonian and Laird Q test for heterogeneity and also calculated the I 2 statistic for each analysis (14). This statistic describes the percentage of total variation across studies that is due to heterogeneity rather than chance (25%, low; 50%, medium; 75%, high heterogeneity). We used Galbraith plots to examine visually the impact of individual studies on the overall homogeneity test statistic. We evaluated potential publication bias visually by examining for possible skewness in funnel plots (15) and statistically with the methods described by Begg and Mazumdar (15) and Egger (16). The latter method uses a weighted regression approach to investigate the association between outcome effects (log odds ratio) and its standard error in each study.
We considered a P-value < 0.05 as statistically significant.
All analyses were performed using SAS v.9.1 (SAS Institute Inc, Cary, NC, USA) and STATA v.10.1 (Stata, College Station, TX, USA).
Since we only utilised previously published data, we did not obtain approval of an ethics committee or written informed consent.
Results
Figure 1 summarises the process of identifying eligible studies. After title and abstract evaluation, we had identified nine studies (5–12,17). We excluded one more study (17) after evaluating the remaining articles in its entirety and were left with eight studies for this analysis.
Process of study selection.
Study characteristics
Seven genes involved in sex hormone receptor pathways and metabolism have been investigated in the identified studies: ESR-1 (5–11), ESR-2 (11), PGR (7,8,12), AR (12), FSHR (11), NRIP1 (11), and CYP19A1 (11). One study further investigated the methylenetetrahydrofolate reductase gene (MTHFR) (9). This gene will not be considered for the present analysis.
Characteristics of the included studies according to the polymorphisms investigated – polymorphisms that have been investigated in at least two papers with extractable data


MA: migraine with aura; MO: migraine without aura; NS: not specified; ESR-1: oestrogen receptor 1 gene; MTHFR: methylenetetrahydrofolate reductase gene; PGR: progesterone receptor gene.
Characteristics of the included studies according to the polymorphisms investigated – polymorphisms that have been investigated in single studies

AR: androgen receptor gene; ESR-1: oestrogen receptor 1 gene; FSHR: follicle stimulating hormone receptor gene; ESR-2: oestrogen receptor 2 gene; CYP19A1: cytochrome P450, family 19, subfamily A, polypeptide 1 gene; NRIP1: nuclear receptor interacting protein 1; MA: migraine with aura; NS: not specified.
For the meta-analysis we have only considered polymorphisms that have been investigated in at least two independent study populations. The data given in 1 (9) of the 2 (7, 9) studies investigating the ESR-1 30 T > C polymorphism did not allow calculating genotype frequencies. Hence, we could not determine pooled relative risk estimates and we did not include this polymorphism in our meta-analysis.
Almost all studies were performed in Caucasian populations. One study was in an Indian population (8). Further, most (5,6,8,10–12), but not all (7,9), studies presented results stratified by gender in addition to results for the overall study population.
Allele and genotype frequencies of the included studies according to the investigated polymorphisms
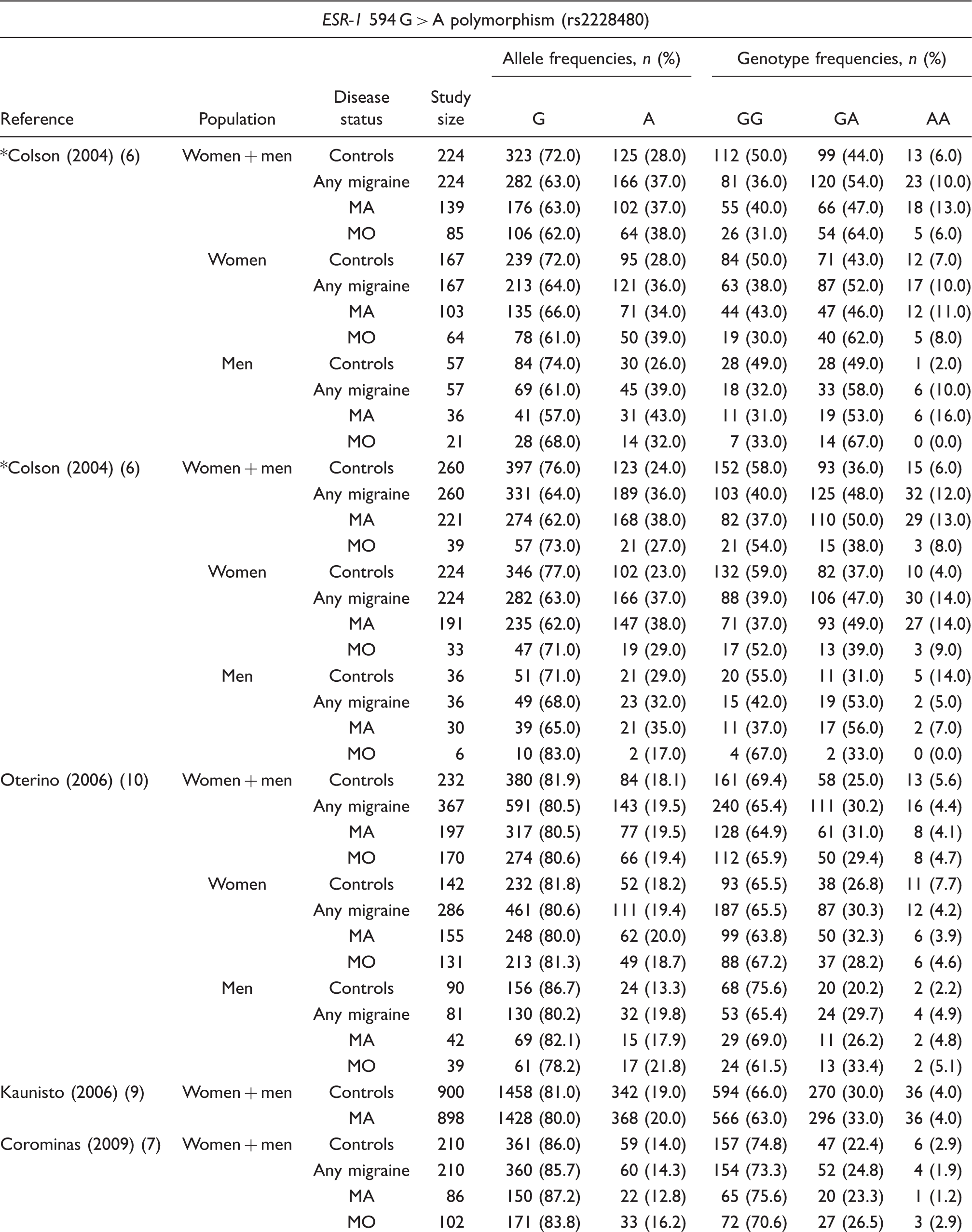


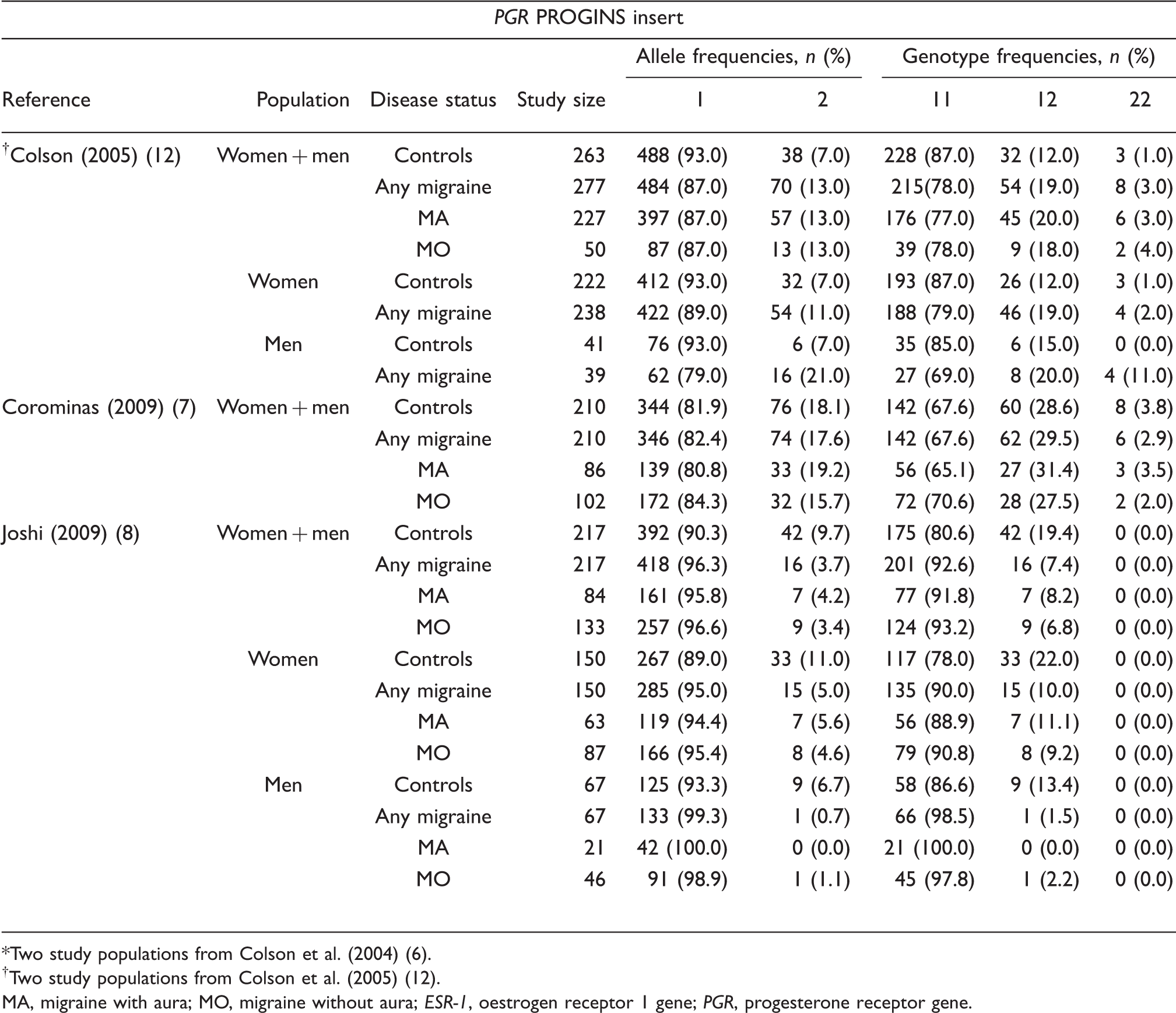
Two study populations from Colson et al. (2004) (6).
Two study populations from Colson et al. (2005) (12).
MA, migraine with aura; MO, migraine without aura; ESR-1, oestrogen receptor 1 gene; PGR, progesterone receptor gene.
Hardy–Weinberg Equilibrium and odds ratios (95% CI) for additive, dominant, and recessive genetic models according to the investigated polymorphisms




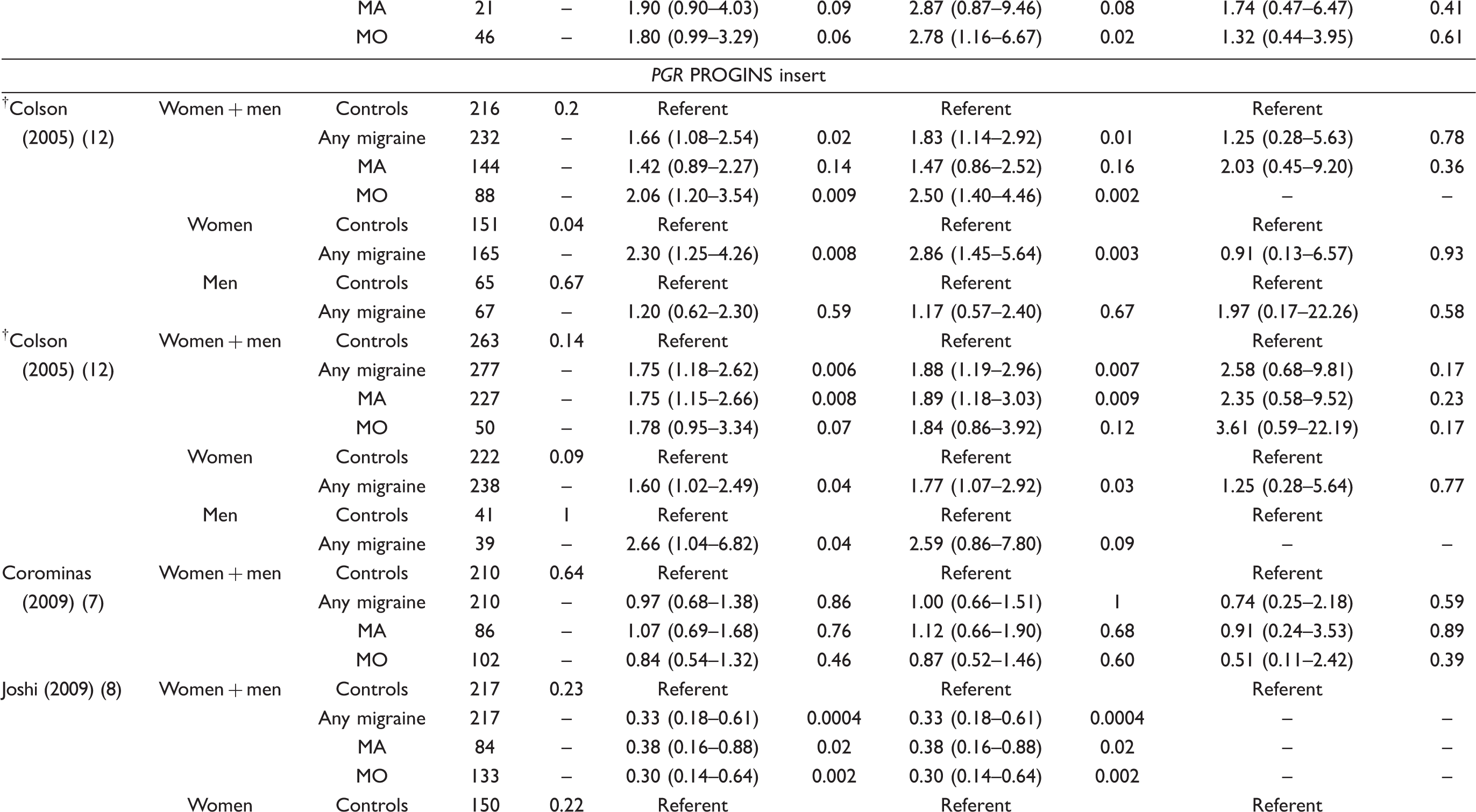
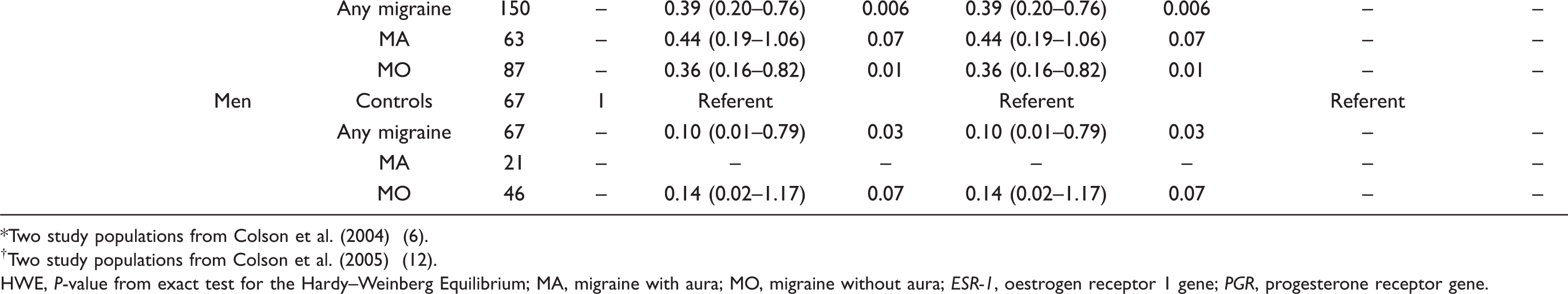
Two study populations from Colson et al. (2004) (6).
Two study populations from Colson et al. (2005) (12).
HWE, P-value from exact test for the Hardy–Weinberg Equilibrium; MA, migraine with aura; MO, migraine without aura; ESR-1, oestrogen receptor 1 gene; PGR, progesterone receptor gene.
Association between sex hormone receptor polymorphisms and migraine, heterogeneity, and publication bias



Association between the ESR-1 594 G > A polymorphism and migraine
Among the five study populations from four studies investigating the association between the ESR-1 594 G > A polymorphism and migraine, there was a statistically significant positive association in two study populations (6) suggesting an increased risk for migraine among carriers of the A allele, which did not appear in the other studies (Table 3) (7,9,10).
The pooled effect estimates among all studies suggest that the A allele is associated with an increased risk for any migraine (additive mode: pooled OR 1.37; 95% CI 1.02–1.83; Table 4). The association appeared most pronounced for carriers of the GA/AA genotype (dominant mode: pooled OR 1.50; 95% CI 1.10–2.06). However, there was medium heterogeneity across all studies (dominant mode: I 2 = 64.5%). Further, the increased risk for the GA/AA genotype appeared to be slightly higher among men (dominant mode: pooled OR 1.80; 95% CI 1.16–2.80) than among women (dominant mode: pooled OR 1.56; 95% CI 0.98–2.48), where it did not reach statistical significance. In addition, heterogeneity was medium among studies investigating women (dominant mode: I 2= 73.5%) and absent among studies investigating men. The results for MA and MO were very similar. Neither Begg’s test nor Egger’s test indicated publication bias for the dominant model.
Association between the ESR-1 325 C > G polymorphism and migraine
Among the six studies investigating the ESR-1 325 C > G polymorphism, two suggested an increased risk for migraine among carriers of the GG genotype (recessive mode), which appeared to be strongest among women (10,11), while the others did not find an altered risk (Table 3) (5,7–9).
The pooled effect estimates suggest that the G allele is associated with a slightly increased risk for having any migraine (additive mode: pooled OR 1.16; 95% CI 1.03–1.32; Table 4). The association was most pronounced for carriers of the GG genotype (recessive mode: pooled OR 1.40; 95% CI 0.93–2.11); however, this result did not reach statistical significance. Further, the effect estimates among studies in Caucasian populations were very similar to the overall result, which included a study in the Indian population. The association between the GG genotype and any migraine was stronger among women than men. Heterogeneity among the studies was low (recessive mode: I 2= 38.9%). The overall association was the same for MA (recessive mode: pooled OR 1.60; 95% CI 1.19–2.17) and MO (recessive mode: pooled OR 1.44; 95% CI 0.97–2.13). In addition, the pattern of a stronger association among women than men also occurred for MA and MO. Neither Begg’s test nor Egger’s test indicated publication bias when assuming a recessive model.
Association between the ESR-1 Pvu II C > T polymorphism and migraine
Among the two studies investigating the ESR-1 Pvu II C > T polymorphism, one found an increased risk for migraine among carriers of the T allele (8), while the other did not (Table 3) (5).
The pooled effect estimates of the two studies neither suggest an association for any of the genotypes with any migraine, MA or MO nor a difference between women and men when looking at any migraine. One study provided gender specific effect estimates for MA and MO, which suggested a higher risk among women than men (8). Heterogeneity between the two studies was high. This is most likely due to the low number of studies and remaining uncertainties which may include genotypic ethnic differences. Formal investigation using Begg’s test did not indicate publication bias.
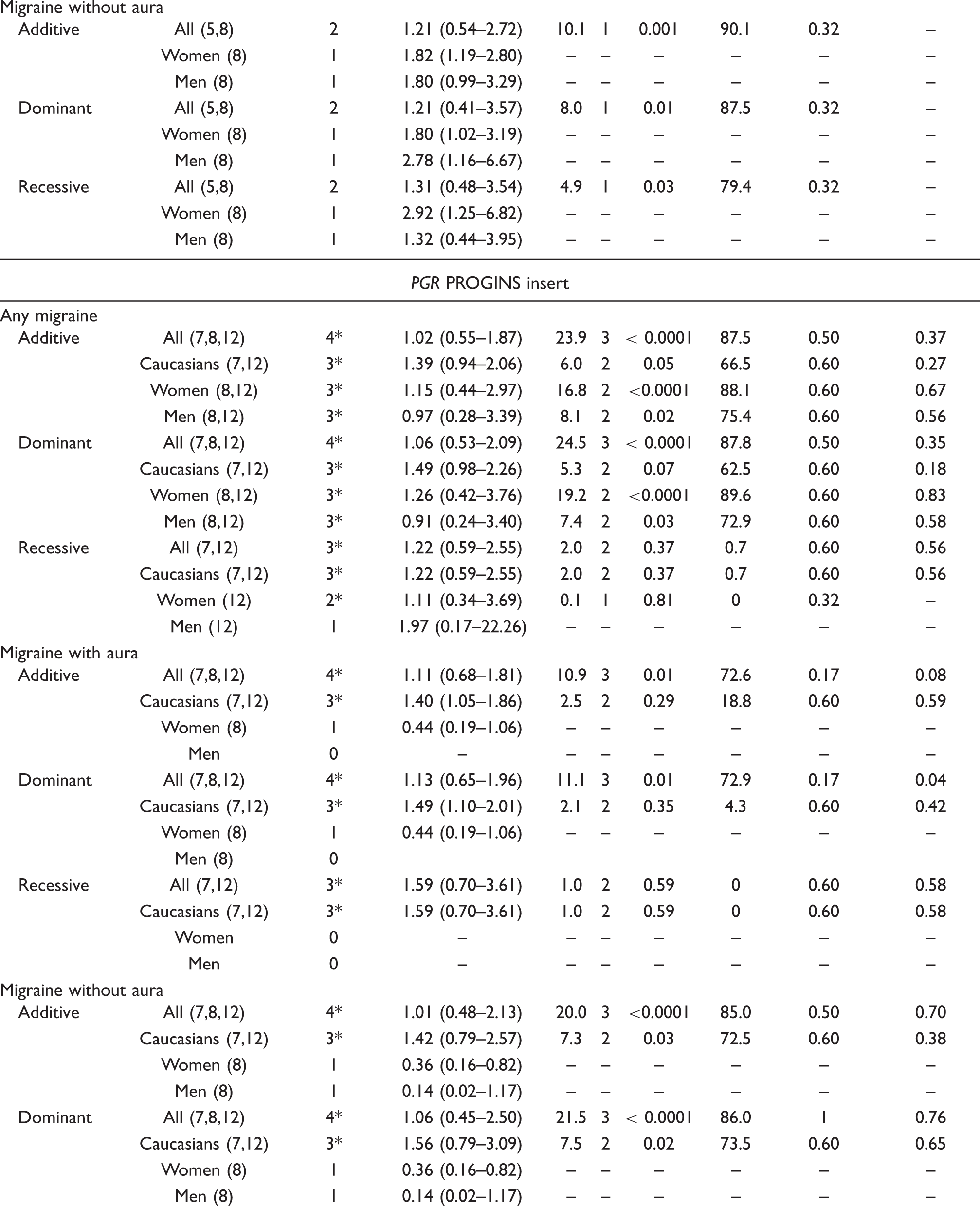
Association between the PGR PROGINS insert polymorphism and migraine
Among the four study populations investigating the PGR PROGINS insert polymorphism, two found an increased risk among carriers of the ‘2’ allele (Alu insert) (12), one found a protective association (8), and one did not find an association (Table 3) (7).
The pooled effect estimates among all studies do not suggest an association between any of the genotypes and any migraine (additive mode: pooled OR 1.02; 95% CI 0.55–1.87; Table 4). This finding did not differ between MA (additive mode: pooled OR 1.11; 95% CI 0.68–1.81) and MO (additive mode: pooled OR 1.01; 95% CI 0.48–2.13). However, further analyses suggested that there may be a moderately increased association for having any migraine among Caucasians, which appeared strongest in a dominant model (pooled OR 1.49; 95% CI 0.98–2.26). While the direction and association of the effect estimates among Caucasians were similar for both migraine subgroups, they only reached statistical significance in MA (dominant mode: pooled OR 1.49; 95% CI 1.10–2.01), but not MO (dominant mode: pooled OR 1.56; 95% CI 0.79–3.09). Heterogeneity across all studies was medium to high for any migraine, MA, and MO; it was low among the studies investigating MA among Caucasians (dominant mode: I 2= 4.3%). This may support the significant results for Caucasians among MA.
Sensitivity analyses
For some of our analyses, Galbraith plots identified individual studies as important sources of heterogeneity. We performed sensitivity analyses by excluding studies that fell outside the margin set by the z score ± 2 SD.
For the association between the ESR-1 594 G > A polymorphism and migraine Galbraith plots did not identify individual studies as significant sources of heterogeneity for any migraine and MO (dominant model). One study (6) was excluded when looking at MA, which lowered the effect estimates; however, the association remained statistically significant (dominant mode: pooled OR 1.18; 95% CI 1.01–1.38).
For the association between ESR-1 325 C > G polymorphism and any migraine, MA, and MO, Galbraith plots did not identify individual studies as important sources of heterogeneity in any of the models.
For the association between the ESR-1 Pvu II C > T polymorphism and migraine, we did not perform a formal sensitivity analysis, because: (i) only two studies were pooled; and (ii) the heterogeneity index was high, suggesting that pooled results need to interpreted with caution.
Effect estimates from the sensitivity analysis did not change the association between the PGR PROGINS insert polymorphism and migraine. They were all slightly higher for any migraine, MA, and MO assuming additive or dominant models. For example, after excluding two studies (8,12), the pooled OR for the association with any migraine assuming a dominant model was 1.34 (95% CI 0.74–2.41).
Discussion
The results of this meta-analysis suggest an association between the ESR-1 594 G > A and 325 C > G polymorphisms and migraine. The risk for MA and MO appears to increase by 40–60% for each of the variants and follows a dominant model in case of the ESR-1 594 G > A and a recessive model in case of the ESR-1 325 C > G polymorphism. In contrast, pooled results for the ESR-1 Pvu II C > T and the PGR PROGINS insert polymorphisms did not suggest an association with migraine. This pattern of association may differ by ethnicity. However, while most studies were conducted in Caucasian populations, only one was done in an Indian population (8), which does not allow an evaluation among non-Caucasian populations. Further, given a lack of replication studies, we cannot conclusively assess an association of additional polymorphisms in ESR-1 (7,9), AR (12), FSHR (11), ESR-2 (11), CYP19A1 (11), and NRIP1 (11) with migraine or migraine subgroups.
Evidence from population-based, clinical, and physiological studies suggests a pivotal role for sex hormones in the pathogenesis of migraine (2–4). In addition, association studies have investigated multiple variants in genes coding for sex hormone receptors or proteins involved in their pathways and metabolism. Among those, multiple studies looked at the ESR-1 594 G > A (6,7,9,10), ESR-1 325 C > G (5,7–11), ESR-1 Pvu II C > T (5,8), ESR-1 30 T > C (7,9), and PGR PROGINS insert (7,8,12) polymorphisms. Apart from the two studies that did not find an association between the ESR-1 30 T > C polymorphism and migraine, results from studies in the other polymorphisms were contradictory.
ESR-1 is located on chromosome 6q25.1 and has eight exons (18). The receptor is expressed, for example, in the hypothalamus, limbic system, hippocampus, and the brainstem of the human brain (19), regions which are implicated in many pain syndromes including migraine. The ESR-1 594 G > A (exon 8) and 325 C > G (exon 4) polymorphisms are synonymous, hence, their functional implication is unknown (20). While our results support that the variant alleles are associated with an increased risk for migraine, they are likely not causative, since they do not alter the amino acid sequence of the receptor. They may be in linkage disequilibrium with another causative variant or set of variants (haplotype) within ESR-1. The Pvu II C > T polymorphism is intronic, thus located in a non-coding region. It does not alter the protein sequence, but may affect splicing and thus modify protein production (21). While our overall pooled results do not support a role for this variant in migraine, the individual results from the two available studies may suggest a difference between Caucasians (5) and Indians (8) (also reflected by the large heterogeneity for the pooled effect estimates). We may speculate that post-transcriptional modification such as splicing differs between ethnic groups. PGR is located on chromosome 11q22 (22). Progesterone receptors are located in various human brain regions (23) and their expression is regulated by oestrogen and progesterone levels (24). The PROGINS polymorphism is a 306-bp long Alu insertion in intron 7 and may negatively affect progesterone receptor expression (25). Our pooled analysis suggests that this Alu insert increases the risk for migraine only among Caucasians.
Study limitations
Some limitations need to be considered:
Migraine is biologically heterogeneous. Although, in all studies, patients were classified according to the criteria established by the International Headache Society (26,27), the clinical spectrum among patients is wide, which may be a source of misclassification. While sample sizes for migraineurs and controls in the studies are about 200 or more (Table 1), power to detect overall and more so gender- or aura-specific associations in subgroups may not be adequate. In addition, not all studies looking at one polymorphism investigated any migraine and also presented stratified analyses according to aura subtype and gender. Further, the total number of studies identified was eight, which is limited. These studies looked at many different gene variants and not all studies investigated the same ones. For example, there were only two studies investigating the ESR-1 Pvu II C > T polymorphism with conflicting results (5,8). The non-significant results from the pooled analysis may be due to insufficient pooled sample size. Power also depends on the minor allele frequencies of the polymorphisms investigated. For example, the minor allele frequency for the PGR PROGINS insert polymorphism is less than 10% in some of the studies leaving few or no observations among the homozygous ‘22’ carriers. Although pooling available study results increases precision and power, there may still be remaining uncertainties. Initial publications of genetic association studies often report positive associations, while successive ones do not find an association. We have performed this meta-analysis at an early stage; however, we still consider it valuable. While the systematic review part allows an overview of the available studies including individual results, the meta-analytic part also enables evaluation of magnitude and direction of combined results from pooled effect estimates including sources of heterogeneity. Ethnicity may be a source of heterogeneity in the association between polymorphisms in genes coding for proteins in sex hormone receptor pathways and metabolism and migraine. The available data suggest this for the ESR-1 Pvu II C > T and PGR PROGINS insert polymorphisms. However, only one study was performed in a non-Caucasian population. Residual heterogeneity among Caucasians for the ESR-1 594 G > A, ESR-1 Pvu II C > T, and PGR PROGINS insert polymorphisms were medium to high, indicating that the effect estimates carry further unidentified sources of uncertainties. In addition, the results from the single study among Indians (8), suggesting an increased risk for migraine among carriers of the ESR-1 Pvu II T allele and a reduced risk among carriers of the PGR PROGINS Alu insertion await replication. In one study (10), genotype distribution of ESR-1 594 G > A and 325 C > G and in another (8) of ESR-1 Pvu II C > T was in Hardy–Weinberg Disequilibrium. We only used extractable data from the papers. One (9) of two (7,9) studies investigating the ESR-1 30 T > C polymorphism did not allow us to extract genotype frequencies; hence, we could not calculate pooled effect estimates. However, both studies did not find an association with migraine, which would likely not change in a pooled analysis. Since we did not have primary data among the studies investigating multiple polymorphisms, we were not able to perform haplotype analyses or investigate potential gene–gene interactions. Such interactions were suggested by individual studies (8,11).
Conclusions and outlook
Additional research is warranted to delineate further the association between gene variants coding for proteins in sex hormone receptor pathways and metabolism and migraine, among Caucasian and more so among non-Caucasian populations. We suggest the following criteria to be applied in future studies:
Studies need to be adequately powered. Power in genetic association studies is determined by both sample size and allele frequencies. Sample sizes of at least several hundred migraineurs and non-migraineurs are needed to detect at least moderate associations. If the minor allele frequency of a polymorphism investigated is low, the sample size must be further increased to have adequate power. Results should not just be presented overall, but also stratified by gender and migraine aura status. This must also be considered with regard to power. Investigators should use standardized migraine classification including aura status. Analyses should focus on main gene effects first, since power to detect gene–gene interactions is often limited.
