Abstract
Background: Data on the association between TNFα and TNFβ gene polymorphisms and migraine are conflicting.
Methods: We performed a systematic review and meta-analysis of studies published until January 2011. We used data from published papers and as provided after contact with the authors. We calculated study specific odds ratios (OR) and 95% confidence intervals (CI) assuming additive, dominant, and recessive genetic models as well as pooled effect estimates.
Results: Among the ten studies identified, the best evidence is available for the TNFα −308G>A and TNFβ 252A > G polymorphisms indicating no overall association with migraine. Subgroup analyses suggested that the A allele of the TNFα −308G > A variant more than doubles the risk for migraine among populations with a heterogeneous ethnic background, which was driven by associations for migraine without aura (additive model: pooled OR = 2.87, 95% CI 1.86–4.43). Further, the risk for migraine with aura was increased among Asian populations (additive model: pooled OR = 1.71, 95% CI 1.07–2.71). Both observed effects were stronger among females than males.
Conclusions: Our results indicate no overall association between TNFα and TNFβ gene variants and migraine. However, associations differed among specific populations. Our findings need to be treated with caution and further targeted research is warranted to evaluate population-specific effects including population stratification.
Introduction
Migraine is a common neurological disorder affecting 10–20% of the population and women 3–4 times more often than men (1). Clinically migraine presents with recurrent headache attacks, associated symptoms of vegetative disturbance, and hypersensitivity of various functional systems of the nervous system (1,2). Further, about one-third of migraineurs experience transient neurological symptoms mostly involving the visual system prior to or during a migraine attack, which are known as migraine aura (2).
Migraine pathophysiology is incompletely understood. Current concepts view migraine as an inherited brain disorder intermittently leading to neuronal dysfunctions of various parts of the nervous system (1,3). A ‘neurogenic inflammation’ appears to be a key mechanism in activating the trigeminal system and causing the headache (4).
Tumour necrosis factor α (TNFα) is an important inflammatory cytokine and modulator of immune responses, which also appears to play a role in migraine. For example, studies have reported changes in serum (5–7) and urine concentrations (8) of TNFα as well as altered serum concentrations of the soluble TNFα receptor (9) among migraineurs. A link between TNFα and migraine is further plausible, since TNFα can stimulate transcription of calcitonin gene-related peptide (CGRP), which plays a pivotal role in migraine pathophysiology (10).
The genes coding for TNFα and the closely related TNFβ (lymphotoxinα, LTα) are located in the class III gene cluster of the major histocompatibility complex on chromosome 6 (11). Variants in these genes have been shown to modulate cytokine levels of TNFα and TNFβ (12,13); hence, various polymorphisms in the TNFα and TNFβ genes have been investigated among migraineurs. The most frequently studied variants are the −308G > A polymorphism in the promoter region of the TNFα gene (rs1800629) and the 252A > G polymorphism in intron 1 of the TNFβ gene (rs909253). However, results are conflicting with some studies suggesting associations with migraine for the TNFα −308G > A (14–18) and TNFβ 252A > G polymorphisms (19,20), while others do not (14,17,19–23).
We sought to summarize the current evidence on the association between polymorphisms in the TNFα/TNFβ gene cluster and migraine including migraine with aura (MA) and migraine without aura (MO) by systematically reviewing the literature and performing a meta-analysis.
Methods
Selection of studies
We followed the guidelines for systematic reviews of genetic association studies (24). Two investigators (MS, PMR) independently searched MEDLINE, EMBASE, and Science Citation Index from inception to January 2011 combining text words and MESH terms, where appropriate, for tumour necrosis factor and lymphotoxin (‘tumour necrosis factor’ or ‘tumour necrosis factor-alpha’ or ‘tumour necrosis factor-beta’ or lymphotoxin or ‘lymphotoxin-alpha’ or ‘lymphotoxin-beta’ or TNF or LTA) with terms for genetic variations (‘gene’ or ‘polymorphism’ or ‘genetic variation’) and terms for headache and migraine (‘headache’ or ‘headache disorders’ or ‘migraine’ or ‘migraine disorders’). The search terms were combined with the ‘explode’ feature where applicable. We considered all publications without language restrictions. In addition, we manually searched the reference list of all primary articles and review articles.
A priori, we defined the following criteria for inclusion:
Studies must have a cross-sectional, case–control, or cohort design. Authors must investigate patients with migraine, diagnosed according to the criteria of the International Headache Society (IHS) (25,26) or according to modified IHS criteria and healthy control subjects. Authors must investigate genetic variants located in the TNFα and/or TNFβ gene. Information on genotype frequencies for the polymorphisms investigated among migraineurs and non-migraineneurs must be presented in the publication or be obtainable from the authors upon request. In publications with overlapping cases and/or controls the largest study with usable genetic data was included.
In a first step, two investigators (MS, PMR) by consensus identified all studies not meeting any of the pre-specified criteria by screening the title and abstracts. These studies were excluded. In a second step, the same investigators evaluated the full-paper publications of the remaining studies. Studies were excluded if they did not meet all criteria.
Data extraction and contact with authors
Two investigators (MS, PMR) independently extracted data from the published studies and entered them into a customized database. Disagreements were resolved by consensus. The extracted data included authors’ names and title of study, year of publication, country of origin, setting (clinic vs. population), study design, genotyping method, migraine status (migraine, MA, MO), age range and gender of study individuals, study size, allele and genotype frequencies, and additional potentially relevant information. We sought to collect genetic information for all migraineurs and migraineurs with and without aura separately as well as for the whole study population and females and males separately. If not presented in the paper, allele and genotype frequencies were calculated where possible. For studies, which did not allow extraction of all relevant information including genotype and allele frequencies, we contacted the authors to obtain the missing information. We wrote emails to the corresponding authors explaining our project and asking them to provide the additional data. Authors not responding within 2 weeks were sent up to four reminder emails.
Statistical analysis
We first used logistic regression to calculate odds ratios (ORs) and 95% confidence intervals (CIs) for the association between genetic polymorphisms and migraine assuming additive, dominant, and recessive genetic models for each study. The additive model assumes that the risk for migraine among carriers of the heterozygous genotype is half way between carriers of the homozygous genotypes. While the dominant model assumes that carriers of the heterozygous and homozygous variant genotypes have the same risk of developing migraine compared with carriers of the homozygous wild-type genotype, a recessive model assumes that carrying the homozygous variant genotype is necessary to alter the risk for migraine compared to carriers of the heterozygous and homozygous wild-type genotype. We also determined Hardy−Weinberg equilibrium (HWE) for each study. We investigated (overall) migraine, MA, and MO. We further performed analyses stratified by gender and country of origin [countries with European populations vs. countries with Asian populations vs. countries with populations of heterogeneous ethnic background (Turkey and Iran)], where applicable.
We weighted the log of the ORs by the inverse of their variance to obtain pooled estimates. We ran random-effects models, which include assumptions about the variability between studies. We performed the DerSimonian and Laird Q test for heterogeneity and also calculated the I2 statistic for each analysis (27). We used Galbraith plots to visually examine the impact of individual studies on the overall homogeneity test statistic. We further used meta-regression to investigate whether gender, country of study origin, or clinical phenotype (MA, MO) significantly impact the pooled results. We evaluated potential small study effects such as publication bias statistically with the methods described by Begg and Mazumdar (28) and Egger et al. (29).
All analyses were performed using SAS version 9.1 (SAS Institute, Cary, NC, USA) and STATA 10.1 (Stata, College Station, Texas, USA).
Since we only utilized data from previously published studies, we did not obtain approval of an ethics committee or written informed consent.
Results
Figure 1 summarizes the process of identifying eligible studies. After title and abstract evaluation we were left with 11 records (14–23,30). We identified one additional publication by manually searching the reference lists of the records (31). Of the 12 records, we excluded two: one abstract (30), because detailed results were later published in full (19) and one paper (31), because the study population overlapped with that of a record already included for our analysis (20). We were finally left with ten studies (14–23).
Process of identifying studies.
Contact with authors
In eight papers, genotype and allele frequencies were only reported for all participants and/or for overall migraine, but not stratified by gender and/or aura (16–23). After contacting the authors, we could obtain complete data for five studies (18–22) but not for two other studies (16,23). We also included genotype and allele frequencies from our previous study (17).
Study characteristics
Characteristics of the included studies according to the polymorphisms investigated




IL-1, interleukin 1; MA, migraine with aura; MO, migraine without aura; NA, not available; TNF, tumour necrosis factor.
Eight studies investigated mixed female and male populations (14–16,18–20,22,23), while two study populations consisted only of females (17,21). Migraineurs in six studies consisted of migraineurs with aura and without aura (14,16,17,20,21,23), while four studies only investigated MO (15,18,19,22). One study had a cohort design (17), the remainder were case–control studies (14–16,18–23). One study was performed among children/adolescents (22), the other studies among adults (14–21,23). Five studies were performed in European populations (16,17,19,20,22), two in Turkish populations (18,23), two in Asian populations (14,21), and one in a Iranian population (15).
Allele and genotype frequencies as well as Hardy-Weinberg Equilibrium according to the investigated polymorphisms
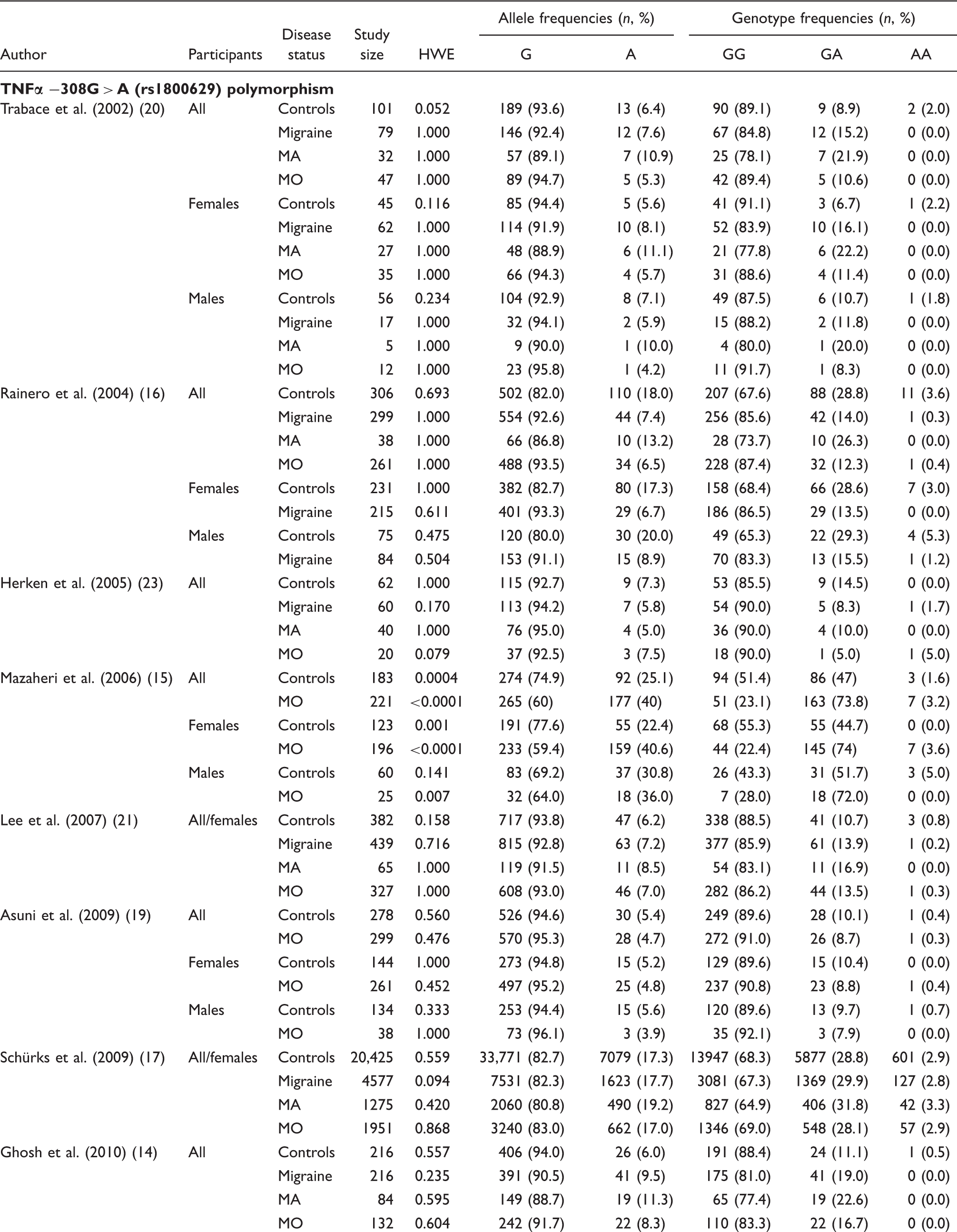


HWE, p-value from exact test for the Hardy–Weinberg equilibrium; MA, migraine with aura; MO, migraine without aura; TNF, tumour necrosis factor.
Odds ratios (95% confidence intervals) for the association between the investigated polymorphisms and migraine

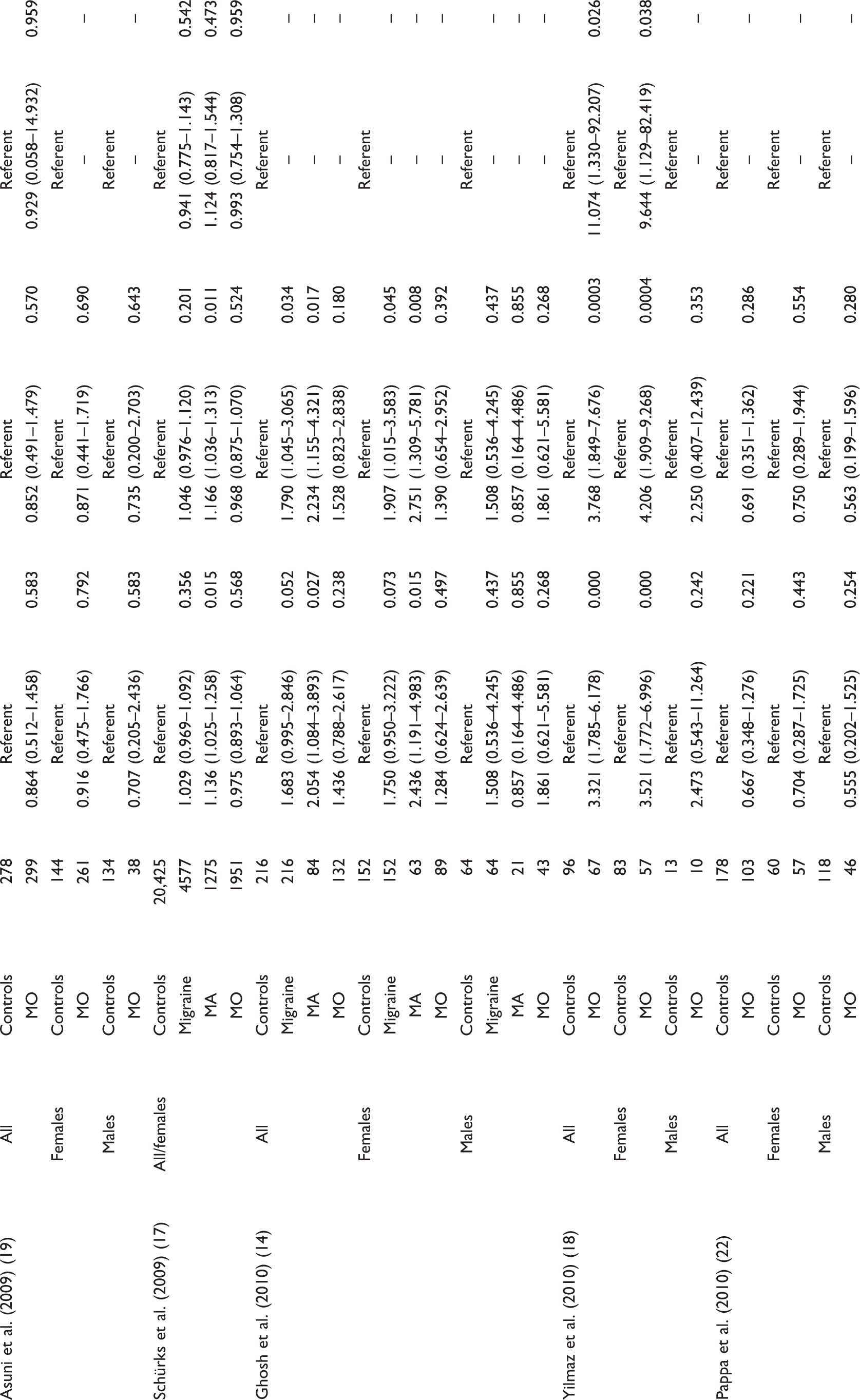

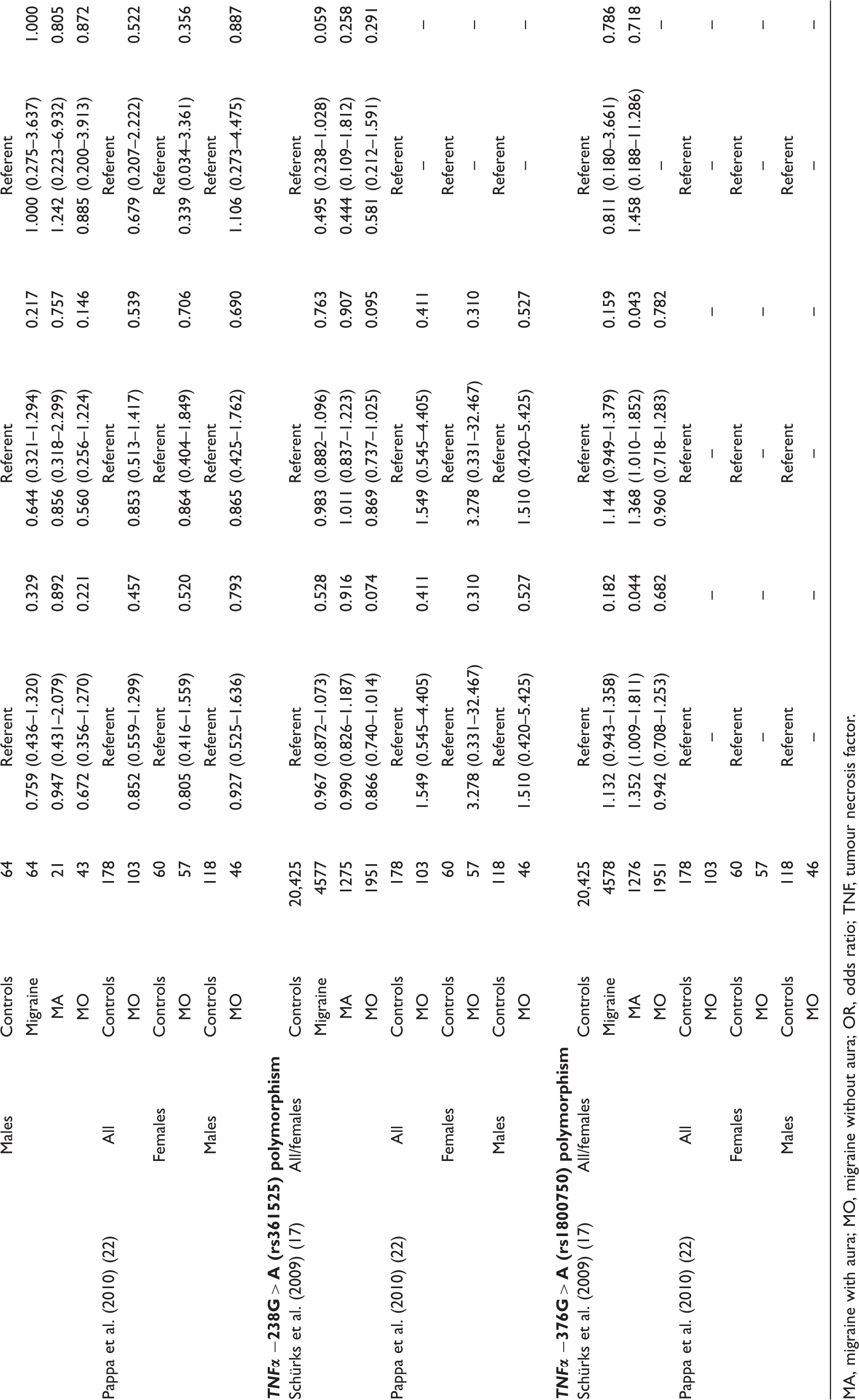
MA, migraine with aura; MO, migraine without aura; OR, odds ratio; TNF, tumour necrosis factor.
Association between the TNFα −308G > A (rs1800629) polymorphisms and migraine from random effects model, heterogeneity, and small study effects
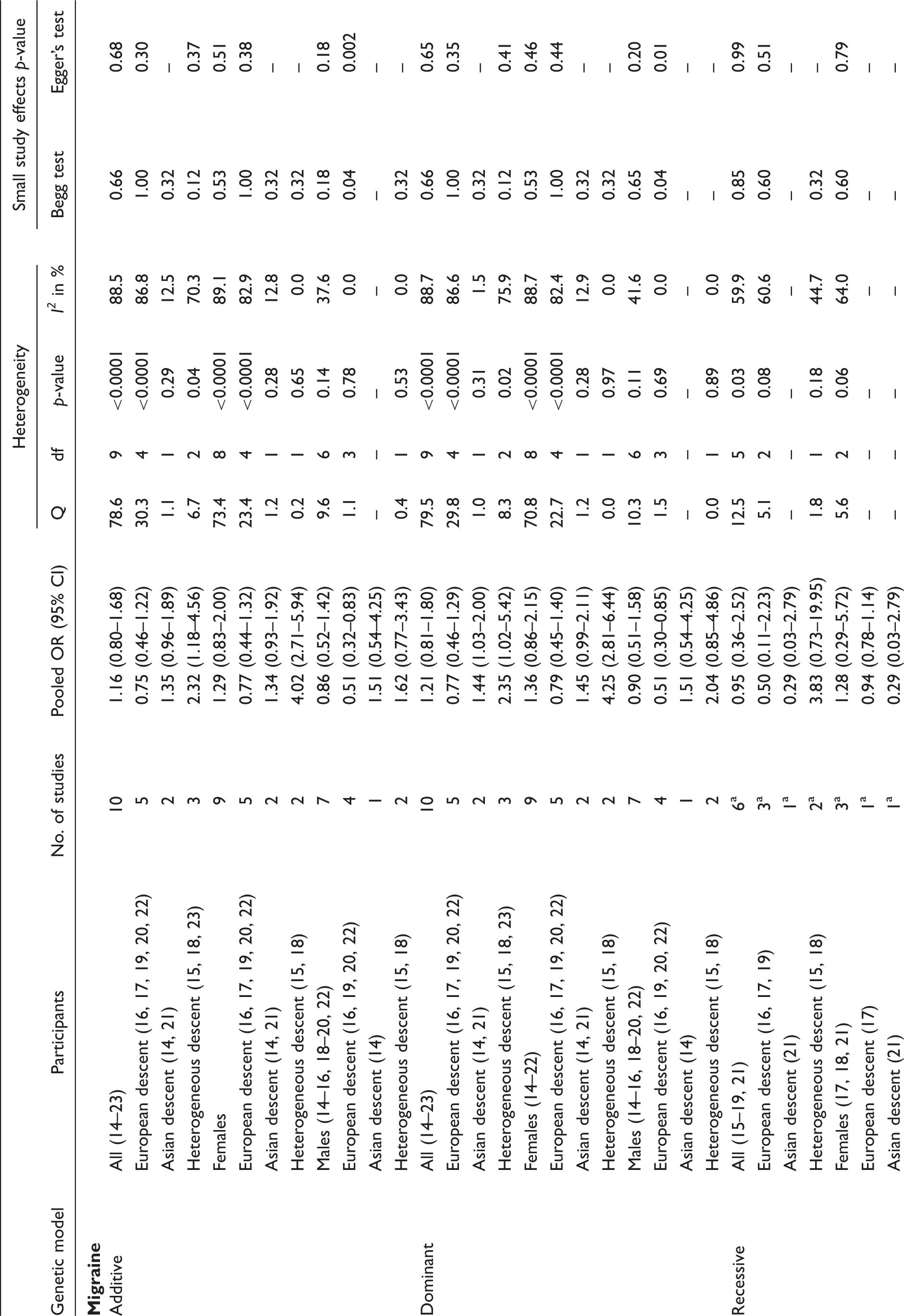
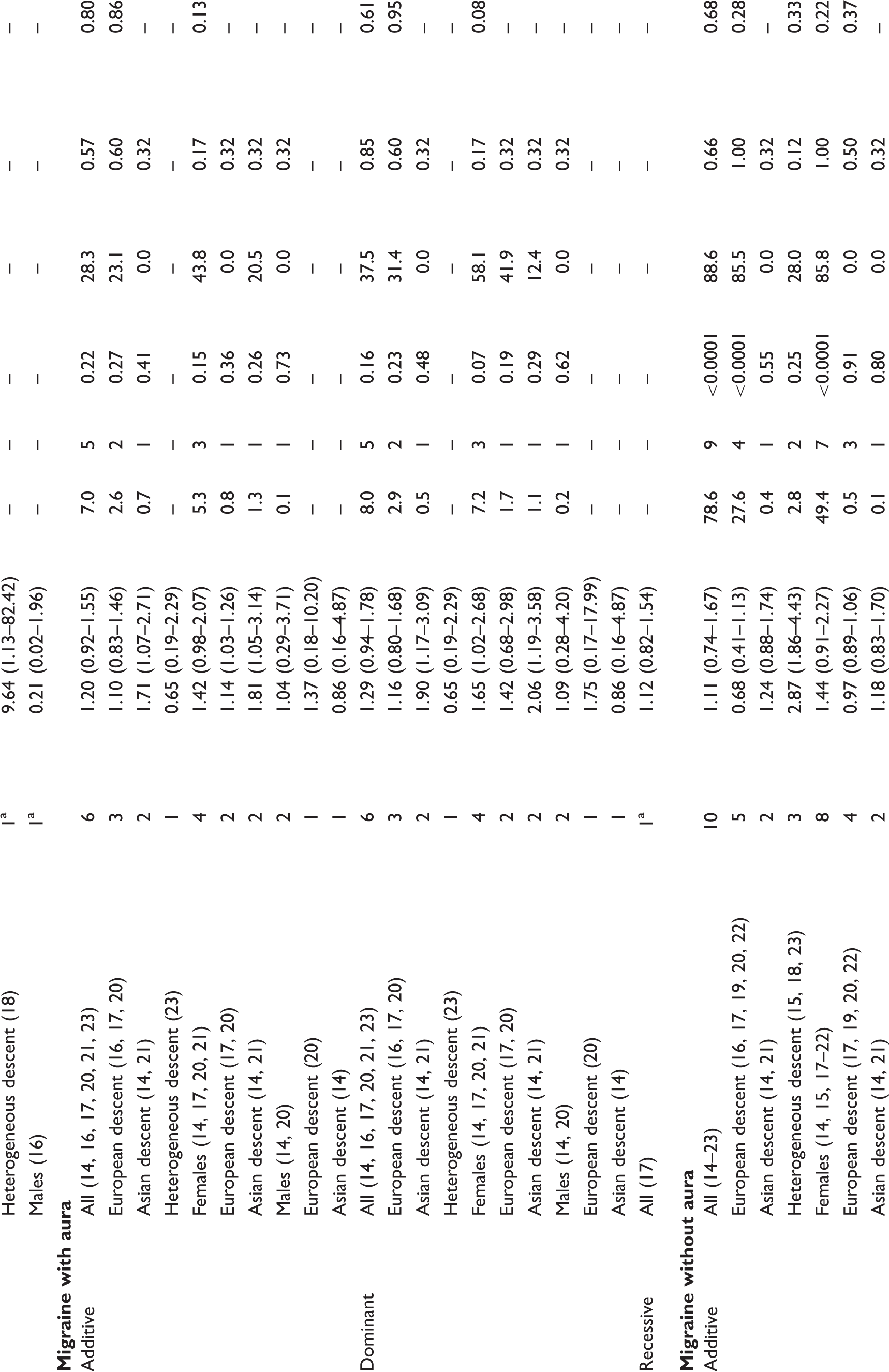

OR, odds ratio.
Effect estimates for some studies could not be calculated due to small numbers.
Association between the TNFβ 252A > G (rs909253) polymorphisms and migraine from random effects model, heterogeneity, and small study effects
Effect estimates for some studies could not be calculated due to small numbers.
Association between the TNFα−308G > A (rs1800629) polymorphism and migraine
Genotype distributions in control and migraine populations were in HWE in all but one study (Table 2). Five studies suggested an increased risk of migraine assuming additive and dominant models (14,15,17,18,20) (Table 3). In contrast, one study suggested a reduced risk (16). The effects were stronger among females than males. The remaining four studies did not suggest an association between any of the genotypes and migraine (19,21–23). Effect estimates for some of the associations assuming a recessive model could not be calculated, due to missing observations for the AA genotype in some studies.
Our overall pooled analyses do not suggest an association between the TNFα −308G > A polymorphism and migraine (Figure 2, Table 4). The pooled OR (95% CI) from an additive model for migraine was 1.16 (0.80–1.68). Effect estimates were similar when assuming a dominant model and lower in recessive models. There was high heterogeneity among all participants (additive model: I2 = 88.5%). Since meta-regression indicated that country of study origin had a significant modifying effect on the overall pooled OR (European populations: reference; Asian populations: p = 0.18; populations of heterogeneous ethnic background: p = 0.02) we performed pooled analyses among studies in these populations separately. The effect estimates were lower among Europeans than among Asians, a pattern most pronounced among males. Analyses among populations of heterogeneous descent suggested a significantly increased risk for migraine assuming an additive model (pooled OR = 2.32, 95% CI 1.18–4.56), which was driven by a four-fold increased risk among females (pooled OR = 4.02, 95% CI 2.71–5.94).
Odds ratios for the association between the TNFα −308G > A polymorphism and migraine assuming an additive model from individual studies and from the pooled analysis. CI, confidence interval; ES, estimate (indicates odds ratio).
Analyses stratified by clinical phenotype also do not suggest an association between the TNFα −308G > A polymorphism and MA (all participants, additive model: pooled OR = 1.20, 95% CI 0.92–1.55) or MO (all participants, additive model: pooled OR = 1.11, 95% CI 0.74–1.67) (Table 4). This agrees with results from meta-regression indicating that clinical phenotype is not a significant source of heterogeneity (all p > 0.23). While heterogeneity among studies investigating MO was high (all participants, additive model: I2 = 88.6%), it was low among those investigating MA (all participants, additive model: I2 = 28.3%). Pooled results of the two studies among Asian populations suggested an increased risk assuming additive and dominant models, specifically for MA and were most pronounced among females assuming a dominant model (pooled OR = 2.06, 95% CI 1.19–3.58).
For migraine analyses among males suggested potential small study effects (additive model: Begg’s test, p = 0.04; Egger’s test, p = 0.002). When considering gender-stratified results meta-regression did not indicate that gender significantly impacts pooled results for migraine, MA, or MO in additive or dominant models (all p-values ≥0.34). Meta-regression among effect estimates from recessive models could not be performed.
Association between the TNFβ 252A > G (rs909253) polymorphism and migraine
Genotype distributions in all control and migraine populations were in HWE (Table 2).
One study suggested a decreased risk of migraine assuming additive and dominant models for MO (20) (Table 3). In contrast, effect estimates suggested an increased risk for MA among females. Effect estimates for some of the associations could not be calculated due to missing observations for some of the genotypes. Two studies suggested an increased risk for migraine (19,21). Three studies did not suggest an altered risk for migraine for any of the genotypes (14,17,22).
Our overall pooled analyses do not suggest an association between the TNFβ 252A > G polymorphism and migraine (Figure 3, Table 5). The pooled OR (95% CI) from an additive model for migraine was 1.02 (0.87–1.21). The effect estimates were similar for females and males and results did not change in dominant or recessive models. There was moderate heterogeneity among all participants (additive model: I2 = 66.0%), which only appeared among males (additive model: I2 = 60.8%), but not females (additive model: I2 = 0.0%).
Odds ratios for the association between the TNFβ 252A > G polymorphism and migraine assuming an additive model from individual studies and from the pooled analysis. CI, confidence interval; ES, estimate (indicates odds ratio).
Study results stratified by clinical phenotype also do not suggest an association between the TNFβ 252A > G polymorphism and MA (all participants, additive model: pooled OR = 1.03, 95% CI 0.95–1.12) or MO (all participants, additive model: pooled OR = 1.00, 95% CI 0.83–1.22) (Table 5). The results were similar in dominant and recessive models as well as for females and males. This agrees with results from meta-regression on clinical phenotype (all p-values > 0.39). Studies among participants with MA did not suggest heterogeneity (all participants, additive model: I2 = 0.0%). In contrast, for MO there was moderate heterogeneity (all participants, additive model: I2 = 69.3%), which only appeared for males (additive model: I2 = 56.8%), but not females (additive model: I2 = 0.0%).
For MA there was indication for a small study effect among females assuming a recessive model (Begg’s test, p = 0.04; Egger’s test, p = 0.005). Meta-regression did not indicate that country of study origin significantly impacts the pooled results for migraine, MA, or MO (all p-values >0.10). In addition, when considering gender-stratified study results, meta-regression did not indicate that gender significantly impacts the pooled results for migraine or MO (all p-values ≥0.68). Meta-regression among effect estimates from gender-stratified analyses could not be performed.
Association between the TNFα −238G > A (rs361525) polymorphism and migraine
The genotype distribution among controls deviated from HWE in one study (p = 0.006) (17) (Table 2). In the second study, the Hardy–Weinberg principle was given among migraineurs and controls (22). The minor allele frequency in both studies was low (≤5.1%).
Results from both studies do not suggest a statistically significant association between the TNFα −238G > A polymorphism and migraine (Table 3).
Pooled results did not suggest an association for migraine (all participants, additive model: pooled OR = 0.97, 95% CI 0.88–1.08) or MO (all participants, additive model: pooled OR = 0.91, 95% CI 0.66–1.26). There was no indication for heterogeneity among the studies (additive model, migraine: I2 = 0.0%; MO: I2 = 14.0%). Only one study investigated associations in MA (17), suggesting no association (Table 3).
Association between the TNFα −376G > A (rs1800750) polymorphism and migraine
In one study HWE and effect estimates could not be calculated (22) (Tables 2 and 3). In the second study the genotype distribution among controls was not in HWE (p = 0.002) and the minor allele frequency was <2% (17). Results suggested an increased risk for all three models. Pooled analyses could not be performed.
Sensitivity analyses
Galbraith plots
For some of our analyses, Galbraith plots identified individual studies as important sources of heterogeneity. Hence, we performed sensitivity analyses by excluding studies that fell outside the margin set by the z score±2 standard deviations.
With regard to the TNFα −308G > A polymorphism Galbraith plots identified studies introducing heterogeneity for migraine, MO, and among females only for MA. After excluding these studies, associations remained statistically insignificant. For example, among all participants and assuming an additive model the pooled OR (95% CI) was 1.04 (0.94–1.15) for migraine and 0.98 (0.90–1.06) for MO after excluding three studies (15,16,18) and 1.15 (1.04–1.27) for females with MA after excluding one study (14).
With regard to the TNFβ 252A > G polymorphism Galbraith plots identified studies introducing heterogeneity for migraine and MO. Excluding these studies did not change the associations. For example, among all participants and assuming an additive model the pooled OR (95% CI) for migraine was 1.02 (0.96–1.08) and for MO 1.02 (0.94–1.10) after excluding two studies (19,20).
Excluding other studies
We further investigated if study design and age of the study population impacts the pooled results by excluding the cohort study (17) and the study among children/adolescents (22), respectively. For the TNFα −308G > A and the TNFβ 252A > G polymorphisms this did not change the associations with migraine, MA, or MO for any of the models assumed (data not shown). Further, excluding the study where genotype distribution of the TNFα −308G > A polymorphism deviated from HWE (15) did not change the results (e.g. additive model, all participants: OR = 1.02, 95% CI 0.72–1.44).
Discussion
The overall results from this meta-analysis do not indicate an association between any of the investigated polymorphisms and migraine, which did not change in sensitivity analyses. However, among studies investigating the TNFα −308G > A polymorphism the association differs by country of origin and clinical phenotype has a modifying effect. Compared to studies among European and Asian populations those among populations of heterogeneous ethnic background (Turkey and Iran) suggested an increased risk for migraine and MO. The effect was stronger among females than males translating into a four-fold increased risk. In addition, results for MA suggested a two-fold increased risk among Asian populations which may be stronger in females than males. The results from these subgroup analyses, however, need to be treated with caution due to potential population stratification and small study numbers.
An inflammatory process is important in the pathophysiology of migraine and has been named ‘neurogenic inflammation’ (4). TNFα is an inflammatory cytokine with a plausible link to migraine. First, an increased release of TNFα has been reported in MO (5,6) and TNFα plasma levels are higher during than between migraine attacks (7). Further, lower serum concentrations of the soluble receptor for TNFα have been reported in migraineurs compared to controls, while the serum concentrations of TNFα did not differ (9). In contrast, among women with menstrual migraine urine secretion of TNFα appears to be markedly decreased (8). Second, TNFα was shown to stimulate CGRP gene transcription and CGRP plays a key role in the pathophysiology of migraine (10). Third, TNFα is crucial in the development of inflammatory hyperalgesia (32), a phenomenon related to allodynia experienced by some migraine patients.
Levels of TNFα and the closely related TNFβ are under genetic control. The TNFα −308G > A polymorphism modulates transcriptional activity of the TNF gene (12) and TNFα and TNFβ polymorphisms have been shown to determine secretion of TNFα and TNFβ (13). Hence, studies have investigated the role of gene polymorphisms in the TNFα and the TNFβ gene in migraine (Table 1). However, despite biological plausibility, study results were conflicting and our meta-analysis does not support an overall association between any of the investigated gene variants and migraine including its subtypes.
There are two possible explanations for the observed overall lack of associations. Either, there may be truly no association and in some studies positive results may have occurred due to chance or certain study characteristics or the pattern of association may be more complex involving additional factors. In this context the following considerations need to be discussed.
First, pooling study results on the TNFα −308G > A and TNFβ 252A > G polymorphisms indicated remaining moderate to high heterogeneity for migraine and MO which may be due to differences between subgroups. This is plausible given that migraine susceptibility differs for example by gender (33) and ethnicity (34). For the TNFα −308G > A variant analyses stratified by country of study origin eliminate part of that heterogeneity. We observed that Asian populations are at increased risk for MA, the effects being stronger among females than males. We also found that populations with a heterogeneous ethnic background have an increased risk for migraine and MO compared to European and Asian populations. We cannot exclude that this is a spurious finding due to population stratification. Population stratification denotes a systematic difference in allele frequencies between subpopulations in a population for example due to different ancestry, nonrandom mating, and physical separation. If cases and controls stem from different subpopulations and a marker under investigation has different allele frequencies in these subpopulations, an association (or lack of association) may occur reflecting the ancestral difference rather than the disease susceptibility.
Second, these differential associations were based on small numbers of studies. While we made the effort to gather the maximum amount of information including data stratified by gender and clinical phenotype, additional information was unavailable from two studies for the TNFα −308G > A polymorphism (16,23). This information would have been valuable in sketching more clearly associations with migraine among populations of different descent.
Third, although all studies used strict or modified IHS classification criteria (25,26), there is the potential for migraine as well as MA and MO to be misclassified given its biologically heterogeneity and wide clinical spectrum (35).
Fourth, the investigated gene variants may be associated with certain migraine traits or measures of migraine severity like attack frequency rather than IHS-defined migraine status.
Fifth, we have included all available genetic association studies and not prematurely excluded any study based on HWE. This approach increases the total sample size and thus the power to detect a potential association and also allows for greater flexibility at the analysis level by performing sensitivity analyses. This is in keeping with recommendations for performing meta-analyses (36). Our sensitivity analyses indicate that excluding studies where HWE among controls is deviated does not change the results.
Sixth, the available studies varied in sample size. In particular one cohort study (17) was much larger than the other case–control studies. While a very large study may dominate the overall pooled results in a meta-analysis, there was no such indication from our analyses. Results from sensitivity analyses excluding the cohort study were very similar to the overall results for theTNFα −308G > A and TNFβ 252A > G polymorphisms.
Seventh, almost 200 known single nucleotide polymorphisms (SNPs) have been reported in the TNFα and TNFβ genes (37,38), which may affect gene transcription and TNF level. Some SNPs may have a low minor allele frequency as seen in some of the included studies (Table 2), obviating reliable analyses; however, such variants are unlikely to strongly influence a common and complex disorder like migraine. While there are single reports suggesting an association between other SNPs with migraine, no follow-up studies are available yet (Table 1b). Many of the gene variants, however, including gene interactions, have not been investigated yet.
Eighth, TNFα’s and TNFβ’s roles in migraine are likely not just determined by their levels, but also by their functional effect at the receptor level, which is likewise under genetic control and unaccounted for in all studies.
Finally, many neurotransmitters like CGRP, serotonin, dopamine, orexin, and glutamate are important for migraine pathophysiology (39). While a functional interaction between TNFα and CGRP has been shown (10), many more interactions between inflammatory markers and neurotransmitters are likely involved in migraine.
Additional targeted research is warranted to further clarify a potential role of TNFα and TNFβ gene variants in migraine. An important next step is performing genome-wide association studies in large populations. Two studies have recently been published, implicating the glutamate pathway (40,41), a pathway implicated in neuropathic pain (41), and another yet unknown mechanism (41). While other genome-wide association studies are under way, these are unlikely to answer questions raised by our meta-analysis, namely a possible genetic effect among non-European populations, since all are performed in populations of European descent. Hence, additional large studies such as genome-wide association studies need to be performed in other populations that also account for potential population stratification, in order to discern potential genetic differences in migraine susceptibility.
Footnotes
Acknowledgements
We would like to gratefully acknowledge the following colleagues and researchers, who supported this meta-analysis by kindly providing us with additional data from their published studies (in alphabetical order):
Greece: Maria Hatzistilianou (MD), School of Medicine, Aristotle University, Thessaloniki.
Italy (Cagliari, Sardinia): Carlo Asuni (MD), Unit of Clinical Pharmacology, Teaching Hospital of Cagliari; Donatella Congiu (MSc) and Maria Del Zompo (MD), Department of Neurosciences ‘B.B. Brodie’; University of Cagliari.
Italy (Rome): Patrizia Lulli (PhD) and Paolo Martelletti (MD), Department of Clinical and Molecular Medicine; Simonetta Trabace (PhD), Department of Molecular Medicine; Sapienza University of Rome.
South Korea: Jong-Won Kim (MD, PhD), Department of Laboratory Medicine, Samsung Medical Center, Sungkyankwan University School of Medicine, Seoul.
Turkey: Sema Erol Çakmak (MD), Aynur Özge (MD, PhD), Osman Özgür Yalin (MD), İbrahim Arda Yilmaz (MD), Department of Neurology; M. Emin Erdal (PhD), Department of Medical Biology and Genetics; Mersin University Medical Faculty. Tuba Gökdogan Edgünlü (PhD), Department of Medical Genetics, Mugla University School of Medical Healthy Science.
Funding
P Rist is funded by a training grant from the National Institute of Aging (AG00158).
Conflicts of interest
In the last 2 years: MS has received an investigator-initiated research grant from the Migraine Research Foundation and honoraria from LEK Consulting for telephone surveys and from the American Academy of Neurology for educational material. RYLZ Zee has received research support from the National Heart, Lung, and Blood Institute, the Doris Duke Charitable Foundation, the Leducq Foundation, the Donald W. Reynolds Foundation, and Roche. DIC has received research support from Celera. TK has received investigator-initiated research funding from the French National Research Agency, the US National Institutes of Health, Merck, the Migraine Research Foundation, and the Parkinson's Research Foundation. Further, he is a consultant to World Health Information Science Consultants, LLC; he has received honoraria from the American Academy of Neurology and Merck for educational lectures and from MAP Pharmaceutical for contributing to an scientific advisory panel. None of the disclosures for any of the authors constitutes a conflict of interest with regard to this meta-analysis.
