Abstract
There are conflicting data concerning the association between migraine and C677T polymorphism of the MTHFR gene. The C677T polymorphism reduces enzymatic capability by 50% and causes hyperhomocysteinaemia. We performed a meta-analysis of all published studies investigating the association between the MTHFR gene and migraine. Pooled odds ratios (OR) were estimated using random (RE) and fixed effects (FE) models. Among the overall 2961 migraineurs there was no significant difference compared with controls. Only in migraine with aura was the TT genotype associated with a higher risk of disease compared with the CC genotype [FE OR 1.30, 95% confidence interval (CI) 1.06, 1.58; RE OR 1.66, 95% CI 1.06, 2.59]. In the same subgroup a significant difference was observed in the comparison between TT and CT + CC genotypes (FE OR 1.32, 95% CI 1.10, 1.59; RE OR 1.63, 95% CI 1.10, 2.43). This study provides evidence for an association of the MTHFR gene only in migraine with aura.
Introduction
Migraine is a paroxysmal neurological disorder that affects approximately 6% of males and 18% of females in the general population (1, 2). Migraine is a genetically complex and clinically heterogeneous disorder whose features include, in addition to head pain, alterations in sensory sensitivity, autonomic dysfunction, dysregulation of mood and focal neurological symptoms. Formal diagnostic criteria set by the International Headache Society (IHS) classify migraine in two main subtypes, migraine without aura (MoA) and migraine with aura (MA) (3).
Migraine has an important genetic component, although the number and type of genes involved are yet to be elucidated (4). Migraine shows genetic heterogeneity, along with a multifactorial mode of inheritance, whereby genetic factors interact with environmental factors (5, 6). The pathophysiology of migraine is not yet fully understood (7).
The MTHFR gene is located on chromosome 1p36.3, consists in 11 exons and encodes methylenetetrahydrofolate reductase (MTHFR) key enzyme in homocysteine metabolism (8). MTHFR is involved in folate metabolism: it catalyses the formation of 5-methylenetetrahydrofolate from 5,10-methylenetetrafirsthydrofolate, which is the predominant circulatory form of folate and carbon donor for re-methylation of homocysteine to methionine (9). A relatively common functional polymorphism in this gene, C677T, changes an alanine to a valine residue in the catalytic domain, determining a modification in the quaternary structure of the enzyme. This substitution reduces the enzymatic capability by 50%, leading to a mild hyperhomocysteinaemia (10).
The C677T polymorphism of the MTHFR gene has been associated with different diseases such as stroke, coronary artery disease, bipolar disorder, schizophrenia and depression (11–15). Several studies have investigated the association between this polymorphism and migraine, with conflicting results (16–23). Some case–control studies have shown that subjects carrying the TT genotype for this polymorphism have an increased risk for MA (16–21), whereas other studies have found no significant association with migraine or its subgroups (22, 23).
To define further and quantify the disease risk associated with carriage of the 677T allele, we performed a meta-analysis of all existing studies that evaluated allelic and genotypic frequencies of this polymorphism in migraine. Meta-analysis, a statistical tool for combining results across studies, is becoming popular as a method for resolving discrepancies in genetic association studies of complex diseases (24).
Methods
Selection of studies
Eligible studies were identified by searching on Medline for all publications between January 2000 and December 2006, using the search terms ‘MTHFR’, ‘methylenetetrahydrofolate reductase’, ‘677C’ and ‘C677T’ in combination with ‘migraine’, ‘migraine with aura’, ‘migraine without aura’. All case–control studies with extractable data were included. Included studies were published as full-length articles or letters in peer-reviewed journals. The diagnosis of MA or MoA was made according to IHS criteria (3). Only one study, conducted on a population of children or adolescents <18 years old, was excluded (25). We specified two phenotyped groups, MA and MoA, in order to maximize the power of meta-analysis and to distinguish between two, probably different, diseases (26).
Data extraction and quality assessment
From each study the following data were extracted: first author, journal, year of publication, ethnicity of study population, genotyping method, allelic and genotypic frequencies of polymorphism, diagnosis of subgroups and number of cases and controls. Whereas lacking, the allelic and genotypic frequencies of C677T polymorphism for cases and controls and for migraine subgroups were also calculated. In some studies odds ratios (OR) and 95% confidence intervals (CI), or allelic and genotypic frequencies were not provided directly, but could be calculated from the data provided using standard formulae. Tension-type headache patients were excluded.
Data were extracted by a single investigator, not involved in any of the included studies; successively, data were examined by two neurologists expert in headaches.
Statistical analysis
Review Manager 4.2, a statistical software package for managing and analysing all aspects of a Cochrane Collaboration systematic review, was used. The effect of association was indicated as OR with the corresponding 95% CI. Both fixed effect (FE) and random effect (RE) were used to calculate pooled OR. Random effect evaluates the heterogeneity between studies and incorporates between-study variability, representing a more conservative statistical approach. Heterogeneity between studies was tested using the Q-statistic; if P < 0.10, then the heterogeneity was considered statistically significant (27). Heterogeneity was also quantified using the index I2. This index is independent of the number of studies included in the meta-analysis and takes values between 0 and 100% (I2 < 25% indicates the absence of heterogeneity; I2 = 25–50% moderate heterogeneity; I2 > 50% large heterogeneity) (28). We also used funnel plot in order to assess small-study bias and publication bias (29). With the dual aim of maximizing power and accuracy of phenotype characterization, we initially specified an overall group of migraineurs and performed a comparison between allelic and genotypic frequencies in migraineurs and in controls. The best-phenotyped group was then analysed comparing migraine subgroups, MoA and MA, with controls. In line with previous meta-analyses, we initially compared TT genotype with CC genotype and then analysed the recessive (TT vs. CT + CC) and dominant (TT + CT vs. CC) models for allele T.
A previous report has shown that the effect of C677T polymorphism leads to hyperhomocysteinaemia and varies with levels of folate intake (30). In this meta-analysis, because homocysteine concentration or haematic folate levels have been shown by only one study (17), it was not possible to use multivariate analysis to investigate migraine risk associated with TT genotype and the above-mentioned haematic concentrations.
Results
Study inclusion and characteristics
A total of nine published articles reported on the relationship between the C677T polymorphism and migraine. Data from only eight articles met the inclusion criteria. Some studies that investigated C677T MTHFR polymorphism also investigated A1298C polymorphism (17), MTHFD1 (cyclohydrolase formyltetrahydrofolate synthetase) R653Q polymorphism (20). These additional associations were not taken into consideration in the present analysis, and only the association between C677T polymorphism and migraine was analysed. Six studies were conducted on Whites (17, 19–23), one involved Australians of White origin (18) and one study analysed a Japanese population (16). Details are provided in Table 1.
The MTHFR C677T polymorphism was genotyped using polymerase chain reaction followed by HinfI digestion with gel electrophoresis, as previously described (10). Only six studies divided genotypic and allelic frequencies according to sex. We involved in the study 2961 migraineurs, 2170 with MA and 791 with MoA. Two studies considered MA exclusively. Therefore, in order to minimize confounding factors, comparison between total migraineurs and controls was performed excluding Kara's and Kaunisto's studies, which analysed MA exclusively. All eight studies were used when MA was compared with controls.
Meta-analysis
The studies included 2961 migraineurs and 3844 controls genotyped for C677T polymorphism. In both populations, the allele C was the most frequent for the C677T polymorphism. In all studies the distribution of the genotypes in control groups was in Hardy–Weinberg equilibrium (P < 0.05). No evidence was found of publication bias graphically from the funnel plots. The heterogeneity for comparisons between migraineurs and controls was high (I2 > 50%). In subgroup analyses, the heterogeneity in comparisons of genotypic and allelic frequencies in MoA and controls was moderate, and it was high (I2 in a range 54.6–74.9) between MA and controls.
No significant difference was found comparing genotypic and allelic frequencies between migraineurs and controls (Table 2). Exclusively in the comparison between subjects with TT genotype vs. TC + CC, pooled OR with fixed effect was 1.30 (95% CI 1.04, 1.62; P = 0.02, I2 = 56.9%). This significance disappears with random effect (OR 1.40; 95% CI 0.97, 2.02).
Odds ratio and heterogeneity results for C677T MTHFR polymorphism in migraineurs, without and with aura

The corresponding percentages (%) are shown in parentheses.
MA, Migraine with aura; MoA, migraine without aura.
Meta-analysis of subgroups
For additional analyses and in order to determine genetic heterogeneity, MoA and MA were also compared vs. controls. Six articles were used in the comparisons between MoA and controls. We analysed 791 migraineurs without aura and 2319 controls. No comparison yielded results significantly different between cases and controls (Table 2). When MA vs. controls was examined, all studies were included. We analysed 2170 migraineurs with aura and 3844 controls, respectively. The results were significantly different: TT vs. CC OR pooled FE = 1.30 (95% CI 1.06, 1.58, see Fig. 1), with RE 1.66 (95% CI 1.06, 2.59) and TT vs. CT + CC OR pooled FE = 1.32 (95% CI 1.10, 1.59, see Fig. 2), with RE 1.63 (95% CI 1.10, 2.43). There were no statistically significant differences in any other comparison (Table 2). The allelic T model indicated a linear trend of increasing prevalence in MA, but produced no significant association, FE OR was 1.08 (95% CI 0.99, 1.18), with RE OR of 1.21 (95% CI 1.00, 1.48). Graphical analysis of the funnel plot in the comparison of TT vs. CC and of TT vs. CT + CC between MA and controls provided no evidence of selection or publication bias and did not suggest important over-representation of smaller positive studies (see Fig. 3).

Forest plot and pooled odds ratio (OR): risk of MTHFR genotypes T/T vs. C/C on migraine with aura using the fixed-effects model. Each study is shown by an OR estimate with the corresponding 95% confidence intervals.
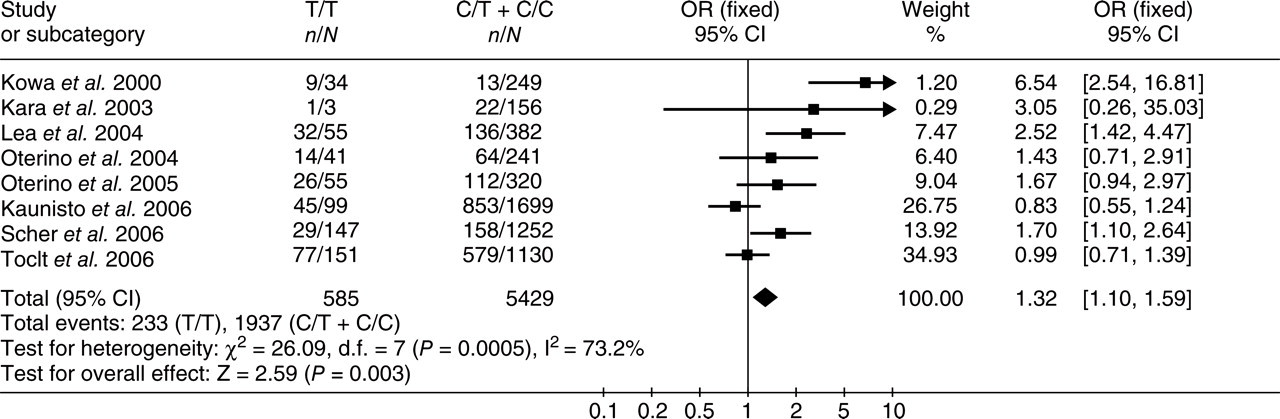
Forest plot and pooled odds ratio (OR): risk of MTHFR genotypes T/T vs. C/T + C/C on migraine with aura using the fixed-effects model. Each study is shown by an OR estimate with the corresponding 95% confidence intervals.
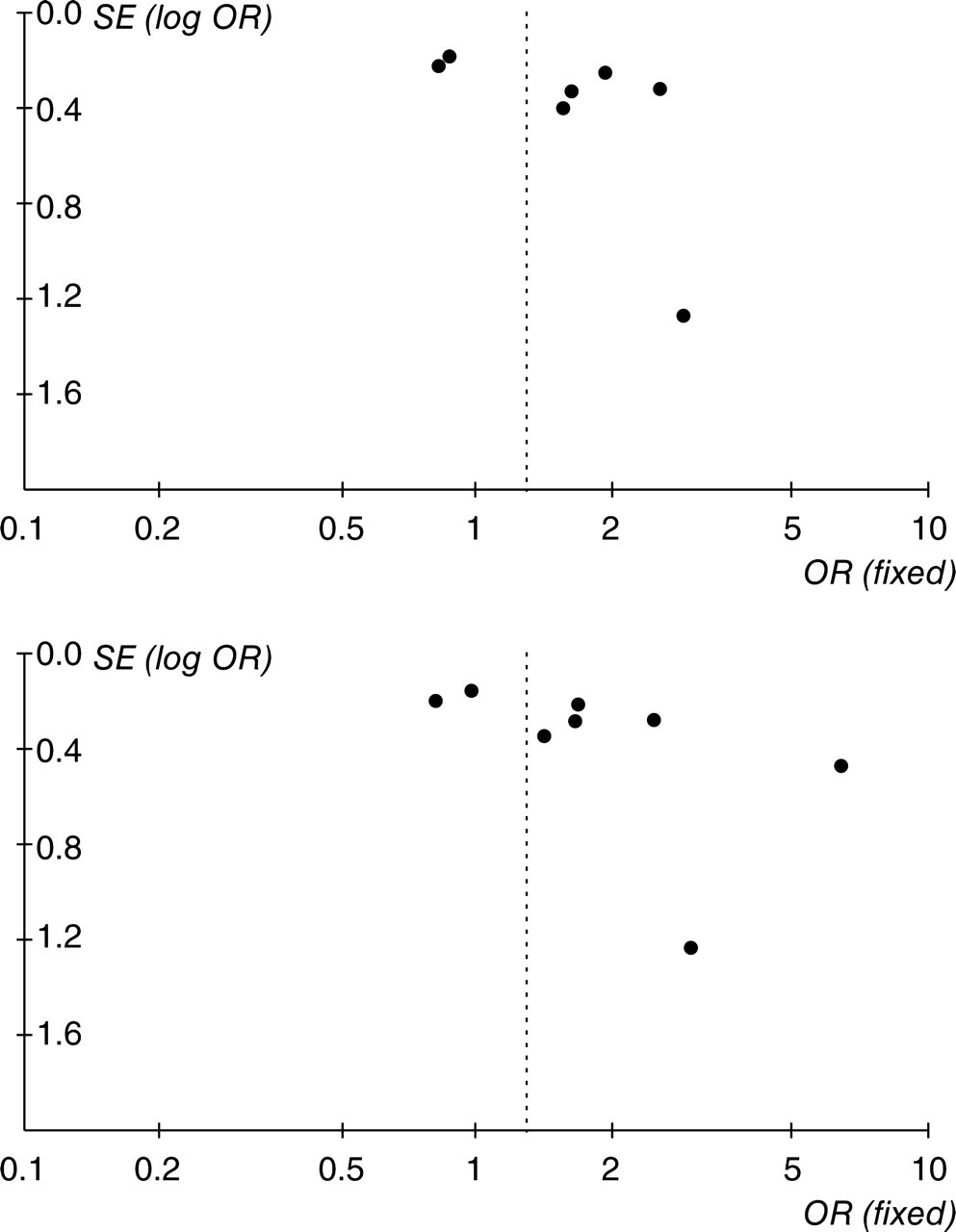
Funnel plots of comparison of T/T vs. C/C (upper) and T/T vs. T/C + C/C (lower) genotypes to determine publication bias.
Discussion
The results of this meta-analysis suggest that there is no association between migraine and the MHFHR C677T polymorphism. However, when migraine subgroups were examined, a significant association was found between MA and the studied polymorphism. Migraineurs with aura carrying the TT genotype, using both fixed and random effects, showed a modest (FE OR 1.3–1.66), but significantly increased disease risk in comparison with CC carriers. This modest effect is the most likely explanation for the lack of association shown in some studies. Our data therefore confirm previous studies suggesting that this polymorphism represents a genetic risk factor for MA.
As for all meta-analyses, the results of this study should be considered cautiously. First, the analysis was performed on a relatively small number of retrospective case–control studies and the possibility of undetected bias cannot be excluded. In addition, only one of the examined studies was performed in a non-White population, so the ethnicity effect was not adequately investigated, whereas racial differences in distribution of this mutation have been described in other studies (31, 32).
The neurobiological basis explaining the association between the MTHFR gene and MA is at present unclear. MA presents significant comorbidity with cardiovascular and cerebrovascular diseases (33, 34) and the 677T allelic variation in the MTHFR gene is involved in the pathogenesis of these diseases (11, 12). A recent large meta-analysis on almost 15 000 patients with stroke has described a moderately increased disease risk, with a pooled OR of 1.17 for T allele and a pooled OR of 1.37 for subjects carrying TT genotype of the MTHFR gene (11). Based on the comorbidity between stroke and migraine and on the possible role of homocysteine in changing cerebral blood flow, few studies have also investigated the relationship between homocysteine and headache. Evers and colleagues have reported elevated serum levels of homocysteine in MA, but not in MoA (35). Store and Goadsby, in the cat model of trigeminal vascular nociceptive activation, have described how DL-homocisteic acid, the oxidized molecule that mimics the effects of homocysteine, increases spontaneous trigeminal cell firing. This suggests hyperhomocysteinaemia sensitizes the dura mater and/or cerebral arteries and leads to migraine attacks (36), playing therefore an active role in migraine pathogenesis.
However, the C667T polymorphism of the MTHFR gene was also found to be significantly associated with several common psychiatric disorders, including unipolar depression, anxiety disorders, bipolar disorder and schizophrenia (13–15). These studies point towards folate or its derivatives as important factors in predicting risk of psychiatric disorders, since the MTHFR C677T influences the function of the folate metabolic pathway. Various somatic and psychiatric conditions have been reported as conditions comorbid with migraine. Some studies have indicated that MA is more strongly associated than MoA with anxiety disorder and depression, but the evidence is not conclusive (37, 38). A recent population-based study found that depression and depression with comorbid anxiety disorder were more likely in women having MA than in those with MoA (39). However, additional studies are needed to clarify this issue.
In conclusion, our study provides additional evidence that the MTHFR gene is a genetic risk factor only for MA. This hypothesis could indicate a different genetic background between MA and MoA. Further investigation conducted in different populations is needed in order to provide a broad insight and to evaluate the precise role of the MTHFR gene in MA.
Footnotes
Acknowledgements
The study was supported by grants from the Ministero dell'Università e della Ricerca Scientifica and the Regione Piemonte (Italy).
