Abstract
Introduction: Reversible cerebral vasoconstriction syndrome (RCVS) comprises a heterogeneous group of acute neurological diseases which are characterized by thunderclap headache and evidence of reversible multifocal constriction of cerebral arteries. A number of precipitating factors have been described in the literature, including recent childbirth and use of vasoactive substances.
Case description: Here we present the case of a female patient with RCVS which occurred in the setting of hormonal ovarian stimulation for intrauterine insemination.
Discussion: This case possibly contributes to the understanding of the pathophysiological mechanisms underlying reversible cerebral vasoconstriction.
Keywords
Introduction
Reversible cerebral vasoconstriction syndrome (RCVS) refers to a group of acute neurological syndromes which are characterized by acute-onset, thunderclap headache and radiological evidence of multifocal reversible vasoconstriction of cerebral arteries (1). It comprises a variety of entities, previously referred to as ‘benign angiopathy of the central nervous system’, ‘Call-Fleming syndrome’ (2) or ‘postpartum angiopathy’ (3). Although RCVS has a benign clinical course in the majority of cases, severe complications have been described, including subarachnoid hemorrhage (SAH), ischemic cerebral infarction, intracerebral hemorrhage (4) and even death (5).
The pathophysiology of RCVS is still not fully understood. Previous case reports as well as a large prospective series of 67 patients (1) reported pregnancy or recent childbirth as well as use of vasoactive substances as common precipitating factors. Here, we present a previously healthy young woman in whom RCVS developed following intrauterine insemination associated with hormone treatment for ovarian stimulation.
Case description
The patient is a previously healthy 36-year-old woman originating from Turkey. Past medical history was unremarkable; in particular, there was no history of an episodic headache disorder. There were no known vascular risk factors, and she was on no regular medication.
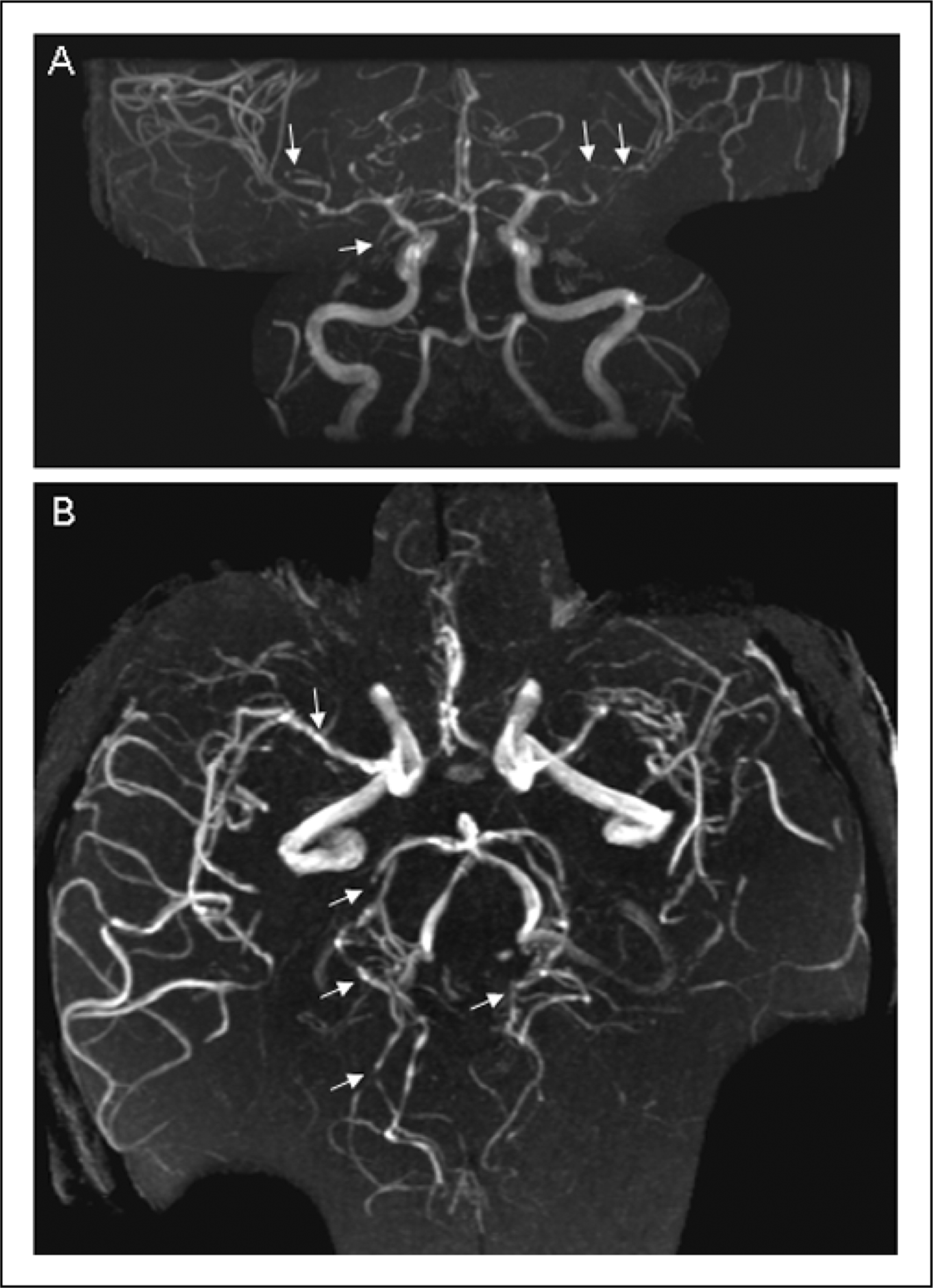
Because of unexplained infertility, she was being treated by a gynecological endocrinologist. She had previously undergone four cycles of ovarian stimulation and intrauterine insemination (IUI)—however, without getting pregnant. During the month before the current illness, a fifth cycle was performed: for ovarian stimulation, she received daily subcutaneous follicle-stimulating hormone (FSH) according to a low-dose protocol for two weeks, followed by human chorionic gonadotropin (HCG) injection for ovulation induction and IUI; no other drugs or substances were taken during this period. Eight days after artificial insemination, the patient developed peracute, explosive headache accompanied by impaired vision (day 0). Initial evaluation at another hospital, including analysis of cerebrospinal fluid (CSF) (day 0) and cranial magnetic resonance imaging (cMRI) (day 1), was unremarkable, and the patient was discharged home in the morning of day 5 after spontaneous remission of the headache syndrome. During the next 1.5 days, progressive psychomotor slowing, difficulties with concentration and blurred vision developed (days 5–6), and the patient presented to the emergency room of our hospital (early afternoon of day 6). She complained of medium-to-severe headache with an occipital maximum (without explosive onset). Physical examination was normal. Gynecological evaluation as well as laboratory testing (normal HCG levels) excluded pregnancy. Neurological examination revealed slight instability of gait and right homonymous lower quadrantanopia. Consistent with this, cMRI revealed an acute ischemic stroke within the left posterior cerebral artery (PCA) territory including the calcarine gyrus as well as multiple small acute ischemic foci also in the right PCA territory (day 6) (Figure 1). On transcranial Doppler sonography, flow velocities in the middle cerebral artery (MCA) were slightly increased bilaterally (170 cm/sec systolically). Time-of-flight (TOF) magnetic resonance angiography (MRA) showed irregularities of the vessel diameters of the major cerebral vessels with multisegmental stenoses bilaterally (‘string-and-beads’) (Figure 2). Laboratory (including serum chemistry, liver function tests, hematology and vasculitis parameters) and CSF analysis were unremarkable. There were no signs of eclampsia such as proteinuria or arterial hypertension. The history of an episode of acutely occurring severe headache (i.e. thunderclap headache) as well as the clinical and imaging findings were overall highly suggestive of RCVS and the patient was transferred for treatment on our stroke unit, where she was monitored with a conservative regimen. Of note, no specific medications (including anti-platelet agents, heparin and nimodipine) were applied.
Diffusion-weighted MRI depicts an acute ischemic stroke in the left posterior cerebral artery (PCA) territory (arrow) and multiple small acute ischemic foci in the right PCA territory (arrowheads). The maximum intensity projection of the time-of-flight magnetic resonance angiography (A: anterior-posterior view, B: cranio-caudal view) reveals irregularities (arrows) of the vessel diameters of the middle, anterior and posterior cerebral arteries bilaterally.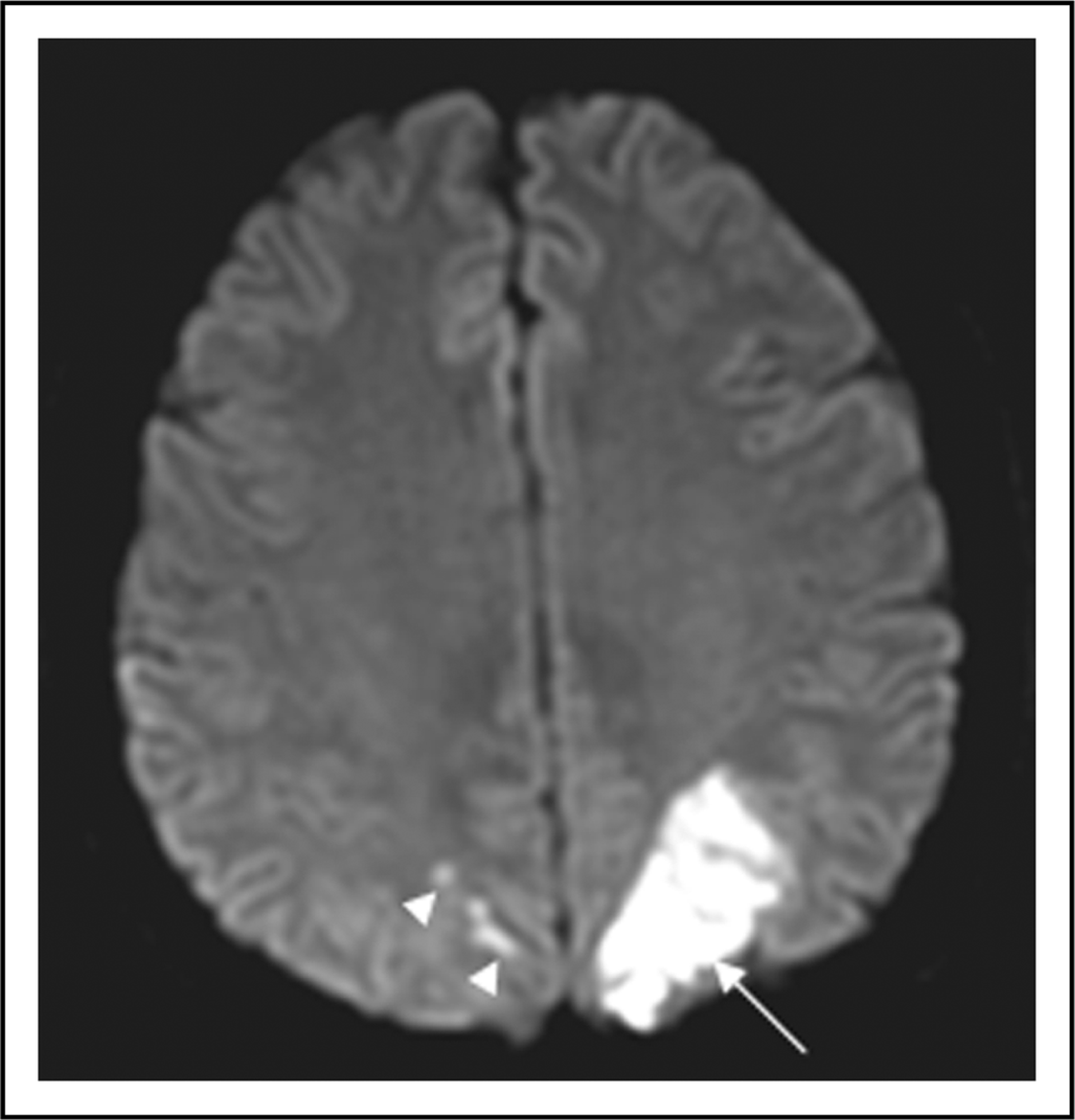
Two days after admission (day 8), another episode of thunderclap headache occurred, without evidence of a hypertensive crisis on continuous non-invasive blood pressure monitoring. Clinical examination did not reveal any new focal neurological deficits. Cranial computed tomography (CT), which was performed immediately after the onset of new thunderclap headache (day 8), revealed localized right fronto-parietal SAH (Figure 3). Due to these new clinical and radiological aspects, conventional digital subtraction angiography (DSA) was performed, which excluded aneurysms, but revealed a ‘string-and-beads’ or ‘sausage-string’ appearance of cerebral vessels, with multiple high-grade stenoses, preferentially affecting the left-sided arteries (Figure 4). Due to these severe angiographic findings, intra-arterial (i.a.) application of nimodipine was performed, which has been reported in this setting by two recent case reports from our center (3,6). Nimodipine was first applied to the right internal carotid artery (ICA) over 30 minutes at a rate of 2 mg/hour, followed by application to the left ICA over another 30 minutes, which resulted in almost complete resolution of the vasoconstriction in both hemispheres (Figure 5). Cardiovascular parameters remained stable throughout the entire intervention.
Native computed tomography scan shows an acute right frontal subarachnoid hemorrhage (black arrows) and the subacute left posterior cerebral artery stroke (white arrowheads). Conventional digital subtraction angiography with selective catheterization of the left internal cerebral artery reveals multiple high-grade stenoses of the left middle and anterior cerebral arteries (arrows). Conventional digital subtraction angiography performed 60 minutes after intra-arterial application of nimodipine at a rate of 2 mg/hour (30 minutes per side) demonstrates an almost complete resolution of the stenoses (arrows).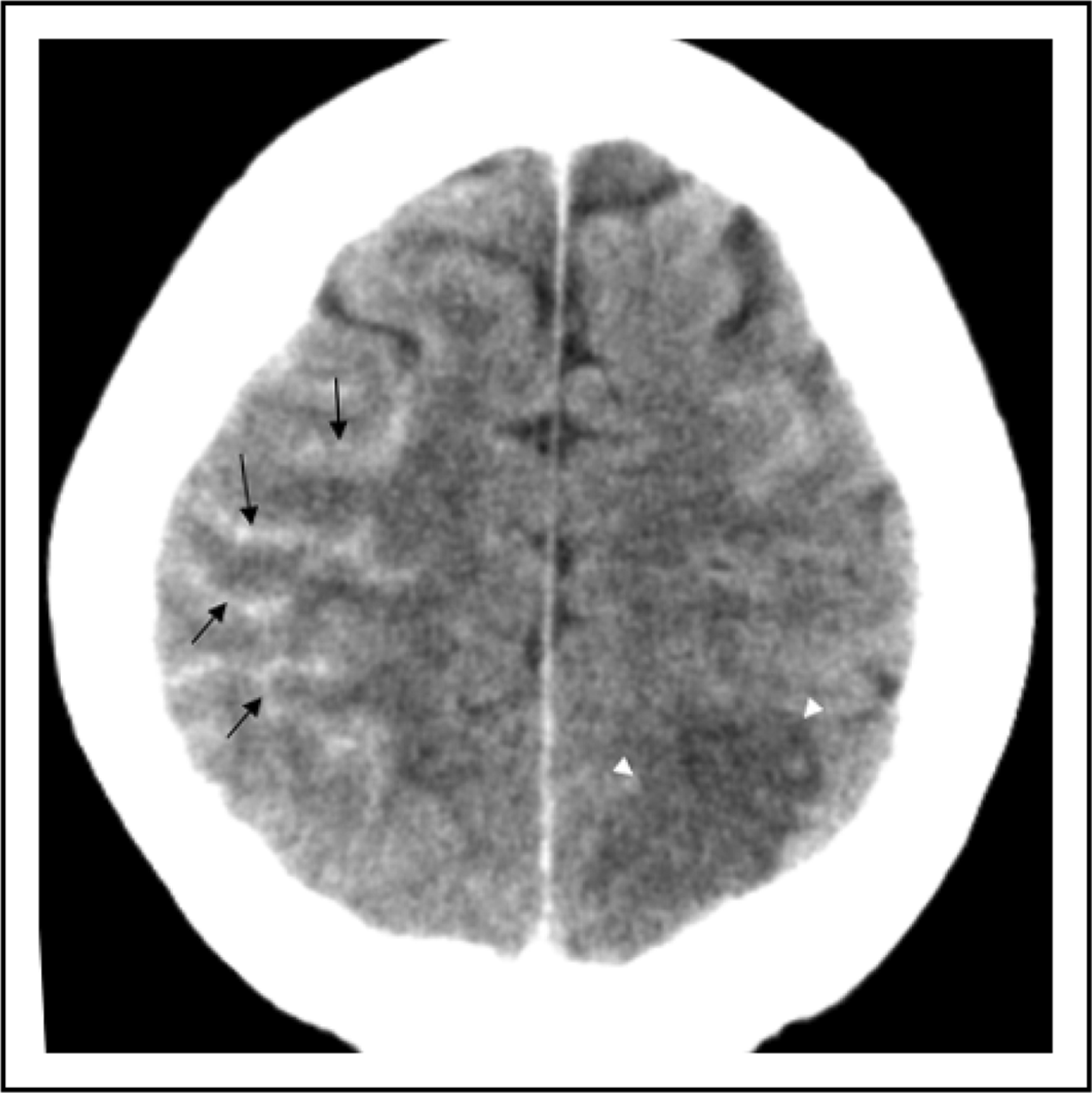


During the next days, the patient continued to complain of continuous medium-to-severe headache, however, without peracute exacerbation of thunderclap episodes. Treatment with nimodipine was continued intravenously (2 mg/h) and, later on, orally, with monitoring of flow velocities in the intracranial vessels by transcranial Doppler sonography (TCD) on an almost daily basis, between d8 and d35. Under this regimen, there was significant clinical improvement, which was reflected by gradual normalization of both MRA findings (data not shown) and TCD: from d8 to d10, maximum systolic MCA flow velocities ranged between 130 and 150 cm/sec; thereafter, the maximum systolic values were below 100 cm/sec. No new focal neurological deficits were observed during the hospital stay, and the patient was discharged home.
Follow-up MRA, which was performed 7 weeks, 11 weeks and 5 months after the initial hospitalization, was without evidence of abnormalities of the intracranial arterial vessels. After 10 months of follow-up, no new neurological symptoms developed.
Discussion
Here, we report a case of a young female patient with a typical clinical and radiological presentation of RCVS: most importantly, there were recurrent episodes of thunderclap headache, and both MRA as well as conventional angiography revealed the classical ‘string-and-beads’ appearance, which was reversible after nimodipine application. Important differential diagnoses, for example cervical artery dissection or cerebral venous thrombosis, which could have been considered from a clinical perspective, were excluded by the initial MRI evaluation. The young age of the patient, the peracute onset of the repeatedly occurring headache attacks, the normal CSF findings, and the reversibility of angiographic findings under nimodipine i.a. argued against a diagnosis of primary angiitis of the central nervous system (PACNS) (7).
Our case highlights many of the clinical features which have been emphasized by previous descriptions of RCVS: visual symptoms, as reported by our patient already on initial presentation, are a very common complaint. Both ischemic infarction, and, in particular, localized SAH (8,9) have been reported as typical complications or RCVS. Furthermore, diagnostic evaluation (including cMRI and MRA) at the time of the initial thunderclap headache was not conclusive; this has been explained by the fact that the vasoconstriction is assumed to progress in a centripetal fashion, that is, involving first the very distal branches, which are difficult to assess by non-invasive imaging techniques (1). Based on these speculations, Ducros and colleagues, in the largest series of RCVS so far, proposed a typical temporal development of complications, with hemorrhagic (i.e. peripheral) complications such as SAH occurring early in the disease course and stroke (i.e. a more ‘central’ manifestation) being a late complication (1). Interestingly, however, this sequence of events is not reflected by our case, where stroke preceded the occurrence of SAH.
One of the most striking features of our case is the close temporal relationship to ovarian stimulation and artificial insemination, which have not been described as precipitating factors in RCVS previously—although RCVS associated with use of oral contraceptive drugs has been recently reported (10). The mechanisms by which these circumstances may potentially cause RCVS are not known. From a pathophysiological perspective, hormone treatment most likely is the key factor. It is well known that ovarian stimulation due to FSH causes a profound rise of estrogen to supraphysiological levels (11). Furthermore, it has been shown in animal and human studies that this rise in estrogen is associated with cerebral vasodilatation, leading to an increase in cerebral blood flow (12,13); this effect is mediated by several factors, including increased expression of endothelial NO synthetase (NOS) (14). Furthermore, it is known that during ovarian stimulation the ovaries also secrete several other factors with vasodilatatory action. Because our patient did not become pregnant, the initially high levels of estrogen (and other vasodilatatory substances) can be expected to have decreased after IUI, particularly at the end of the second half of the menstrual cycle. This would be roughly the time when she developed the first symptoms and presented to the emergency room. Therefore, one may speculate that, in a susceptible individual, the shift from exceptionally high to low estrogen levels (i.e. the loss of the vasodilatatory estrogen effect) may have caused pronounced vasoconstriction as a possible rebound effect. In this context it deserves particular attention that inhibition of neuronal as well as endothelial NOS through endogenous inhibitors has been proposed to be involved in the pathophysiology of arterial vasospasm after SAH (15). Irrespective of this issue, the key pathophysiological role of estrogen withdrawal in our patient resembles RCVS after recent childbirth (i.e. postpartum angiopathy)—which constitutes one of the most common precipitating factors in RCVS (1)—as the puerperium is also characterized by a drastic fall in estrogen levels. Summing up, there is evidence that hormone treatment was an important pathophysiological factor in our patient; however, the precise mechanism remains elusive.
So far, there is no established medical treatment for RCVS. In addition to the elimination of precipitating factors, several authors have previously reported successful use of the calcium channel blocker nimodipine (16–18); in the series by Ducros and colleagues, the vast majority of patients received oral or intravenous nimodipine, with beneficial effects in most of them (1).
Beyond this, several interventional treatment modalities have been described. Intra-arterial application of nimodipine has been successfully used in vasospasm due to SAH (19,20). Based on these experiences, two previous studies from our center reported i.a. use of nimodipine in single cases of RCVS (3,6), with immediate resolution of angiographic findings and positive effects on the further clinical course. Similar observations have been made in a single case of i.a. application of the phosphodiesterase inhibitor milrinone (21). Our case seems to confirm these previous reports. Consistent with Klein et al. (3), initial unilateral nimodipine application on the right side had an equally prompt and strong dilating effect on the contralateral vasculature in addition to the local effect; this is a clear advantage against intracranial balloon angioplasty, which has also been described in the setting of RCVS (22,23). However, despite these observations of a potentially positive effect, it has to be kept in mind that the efficacy of i.a. nimodipine has not been systematically and prospectively evaluated in the context of RCVS. The fact that we observed a sustained normalization of TCD velocities after d10 is no proof of the efficacy of i.a. nimodipine, but may also reflect the continued treatment with IV/oral nimodipine or even the spontaneous course of the disease (24). Finally, we are aware that the positive reports on the use of i.a. nimodipine may be subject to some level of publication bias and that the decision to administer i.a. nimodipine has to be carefully balanced against potential risks and side effects, such as rebound vasoconstriction and reperfusion injury, as reported by Singhal et al. (5). Summing up, i.a. nimodipine may be useful for immediate improvement of vasospasm in RCVS, while no firm conclusions on its overall efficacy are feasible in the absence of systematic data.
In conclusion, we here present a case of RCVS associated with hormone therapy, possibly contributing to the understanding of the pathophysiological mechanisms underlying reversible cerebral vasoconstriction. Further systematic and prospective studies will have to be performed to evaluate the optimal treatment strategy in RCVS.
