Abstract
Thunderclap headache (TCH) is a neurological emergency that warrants immediate and comprehensive diagnostic determination. When no pathology can be identified the condition is classified as primary TCH, which is considered benign and self-limiting. TCH has also been reported as the initial symptom of reversible cerebral vasoconstriction syndrome (RCVS), which subsumes a variety of conditions, inconsistently coined Call-Flemming syndrome, benign angiopathy of the central nervous system, drug-induced arteritis, or migrainous vasospasm. Serious complications such as borderline ischaemic stroke have been reported. Although no standardized treatment regime exists, one commonly described but unproven therapy is parenteral or oral application of the calcium channel blocker nimodipine. Here, we report on a case of RCVS, where a progressive course prompted intra-arterial application of nimodipine, which resolved vasoconstriction immediately. We discuss the use of intra-arterial nimodipine application as a potential emergency treatment for a complicated or treatment-refractory course of RCVS.
Keywords
Introduction
Reversible cerebral vasoconstriction syndrome (RCVS) has been proposed as the unifying term for conditions characterized by acute and severe headache, facultative neurological symptoms and the radiological finding of a self-limited vasoconstriction of the cerebral arteries (1).
Historically, descriptive terms include Call–Flemming syndrome (2), benign angiopathy of the central nervous system (3, 4), postpartum angiopathy (5), migrainous vasospasm or migraine angiitis (6), drug-induced cerebral arteritis or angiopathy (7) and thunderclap headache with reversible vasospasm (8), but all reports are probably describing a spectrum of related disorders.
The clinical and radiological features of RCVS have recently been reported in a comprehensive study that prospectively included 67 patients (9). The clinical course is almost invariably marked by recurring thunderclap headaches (TCH), a term that describes severe headache with hyperacute onset (10). Frequently associated symptoms include nausea, vomiting and agitation. Early complications may involve circumscribed cortical subarachnoid or intracerebral haemorrhage, and changes consistent with a reversible posterior leukoencephalopathy. Some patients experience transient focal neurological deficits or seizures, but few sustain parenchymal damage due to ischaemic stroke (9, 11).
Imperative to the immediate diagnostic work-up for TCH is the exclusion of brain haemorrhage by brain computed tomography (CT) as well as the exclusion of a ‘CT-negative’ subarachnoid haemorrhage and inflammatory conditions by cerebrospinal fluid (CSF) analysis (10). In cases of negative findings, neurovascular examination by CT angiography or magnetic resonance angiography (MRA) should be performed to exclude cerebral venous sinus thrombosis, arterial dissection, aneurysms or vasospasm. Since subtle changes of distal branches can be missed by MRA, the gold standard to obtain qualitative and quantitative parameters of vascular status remains conventional angiography. Furthermore, the identification of characteristic patterns of vasoconstriction are important for the diagnosis, as in RCVS vessels typically show a ‘string and beads’ appearance. When vasoconstriction is excluded, the diagnostic criteria of primary TCH are fulfilled. Interestingly, Chen et al. propose primary TCH and RCVS to be spectra of the same disorder (11).
The underlying cause of RCVS is still poorly understood and causative treatment is not available. The onset can be triggered by exposure to vasoactive substances and is likely to be influenced by hormonal as well as sympathetic factors, as indicated by the striking female gender bias and frequent onset during exertive activities. Consequently, vasoactive medications have to be stopped when applicable. Treatment is empirical and includes simple observation, corticosteroids and the calcium-channel blocker nimodipine. Although not yet supported by controlled clinical studies, case reports and smaller retrospective analysis seem to suggest that intravenous (i.v.) infusion of nimodipine, followed by oral administration, is best (11–14). The course can be monitored clinically and using transcranial Doppler (TCD) (15), and complete resolution of vasoconstriction within 3 months confirms the diagnosis of RCVS. One caveat is that TCD may not be sufficient to monitor vasoconstriction in the cerebral vascular capillary network. In a few cases the condition may progress even under such clinical treatment, resulting in multiple cerebral infarctions with clinical deterioration or death (9).
We present a case of RCVS with a complicated and malignant course, leading to progressive ischaemic events. The rapidly progressive course prompted intra-arterial application of nimodipine subsequent to diagnostic angiography, which led to immediate resolution of vasoconstriction and rapid clinical improvement.
Case report
A 50-year-old woman had a history of migraine headaches but had been headache-free for the last 20 years. Cardiovascular risk factors included untreated borderline hypertension, hyperlipidaemia and a past history of smoking. She was treated with a statin and had been taking a common herbal remedy for menopausal symptoms isolated from Cimicifuga racemosa (possibly containing phyto-oestrogens) for 2 years. In her past medical history a rheumatoid disorder had been considered due to incidental elevations of erythrocyte sedimentation rate (ESR) and leucocyte count, but she did not suffer from any relevant symptoms. Prior to admission she had made an emergency call after the acute onset of a severe occipital headache (verbal analogue scale 9/10), accompanied by nausea and vomiting. She was seen by an emergency physician who found an elevated blood pressure (220/110 mmHg) and prescribed nifidepine and non-steroidal anti-inflammatory drugs, which brought temporary relief. A recurrent episode led to hospitalization, where a CT scan showed no pathology. The episode was considered a hypertensive crisis and she was discharged with pain medication at the patient's request. Two days later she reported to the hospital because of another recurrent attack and was admitted to the neurological ward for pain management and diagnostic work-up. The CT scan was normal and a lumbar puncture showed slightly elevated protein (48 mg/dl) as the only abnormality (reference range 15–45 mg/dl). Magnetic resonance imaging (MRI) disclosed no parenchymal pathology. Vascular vasoconstriction was already seen in the MRA, but was interpreted as insignificant. Gabapentin was started on the assumption of a neuralgiform headache, and she was discharged home after her symptoms resolved over a period of 1 week. Five days later she developed a progressive fluctuating confusional state and was admitted to our stroke unit. On examination she displayed pronounced disorientation and mnestic deficits (mini-mental state test 14/30, Structured Interview for the Diagnosis of Dementia of the Alzheimer Type, Multi-Infarct Dementia, and Dementias of Other Aetiology 21/55), but no other focal neurological deficits. Cranial MRI including diffusion-weighted images revealed acute ischaemic lesions bilaterally in the occipital lobe and left frontal lobe (Fig. 1). Additionally, generalized vasoconstriction was indicated on MRA examination. Accordingly, TCD showed elevated velocities in the middle cerebral (MCA; VMCA R, 210 cm/s; L, 200 cm/s), anterior cerebral (ACA; VACA R+L, 180 cm/s) and basilar arteries (BA; VBA, 160 cm/s). A second lumbar puncture showed a lymphomonocytic pleocytosis of 54 cells/cm3 (reference range < 45 cells/cm3), but unchanged protein or glucose in the CSF. Due to the history of elevated ESR, i.v. treatment of 1 g/day methylprednisolone was started on the assumption of a cerebral angiitis. The confusion partially resolved and no other deficits were detected on repeated neurological testing. Nevertheless, a control CT scan 2 days into the treatment disclosed a new parasagittal ischaemic lesion in the territory of the left MCA. Thus, conventional digital subtraction panangiography was initiated, which showed severe generalized vasospasms with subtotal stenosis of the A1 segment of the left ACA and MCA (Fig. 2). To prevent further ischaemic infarction due to the extensive vessel narrowing, it was decided to commence further interventional treatment. Anaesthesiological stand-by was provided. Intra-arterial infusion of nimodipine into the left internal carotid artery (ICA) was started at a rate of 1 mg/h via a saline-flushed 5-F catheter under systemic heparinization. After 30 min of application, significant improvement was seen in vessel diameter (Fig. 3). Therefore, two additional catheters were introduced in the left vertebral artery (VA) and the right ICA and treatment was continued for a total of 90 min until radiological signs of vasospasm had disappeared. The infusion rate did not exceed 2 mg/h and cardiovascular parameters remained stable during the intervention. Blood pressure was supported by treatment with noradrenalin (0.35 mg/h). Intra-arterial infusion of nimodipine was then stopped and replaced by i.v. application at a rate of 2 mg/h. A fall in blood pressure led to this being adjusted to 1 mg/h. The left-sided sheath in the femoral vein was left in place for a control angiography the following day, which showed a stable status with no indication of vasospasm. In agreement, TCD showed lower peak velocities (VMCA R: 140 cm/s, L: 130 cm/s; VACA R: 130 cm/s, L: 60 cm/s; VBA: 50–120 cm/s). High-dose treatment with 1 g/day of methylprednisolone was discontinued at this point. Over the following days TCD was used for monitoring and showed incremental normalization of blood flow velocities. After 4 days, i.v. treatment was changed to p.o. medication at 240 mg/d nimodipine, which was accompanied by transient paresis of the right arm. A repeated MRI 6 days later showed no further progression of the strokes seen on the initial MRI, except for the new left parasagittal lesion detected on the CT scan, which had prompted the angiography. After discharge the patient was rehabilitated for 6 weeks. On reassessment she did not complain of headaches, but there was still an incomplete hemi-anopsia, and she reported problems in multitasking compared with before.
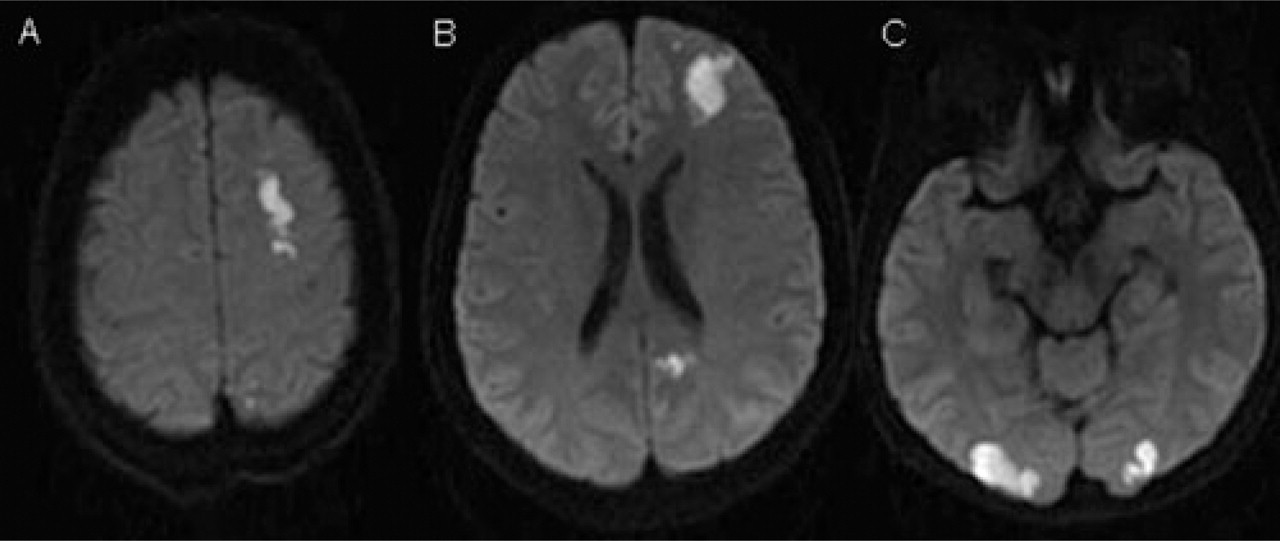
Diffusion-weighted images show acute ischaemic lesions bilaterally in the occipital lobe and in the left frontal lobe.

Digital subtraction angiography with selective catheterization of the left carotid artery in anterior-posterior (A) and oblique (B) projections reveals high-grade stenosis of the A1 segment of the left anterior cerebral artery (ACA; normal arrows) and the M1 segment of the left medial cerebral artery (dotted arrows), as well as alternating stenosis with dilation of the higher-order branches of the ACA (arrowheads).

Digital subtraction angiography series of the left carotid artery performed 30 min after intra-arterial infusion of 1 mg/h of nimodipine over a saline-flushed 5-F catheter under systemic heparinization shows significant improvement in vessel diameter. Normal arrows: A1 segment of the left anterior cerebral artery (ACA); dotted arrows: M1 segment of the left medial cerebral artery; arrowheads: higher-order branches of the left ACA.
Discussion
Generally considered a rare and ill-defined condition, RCVS attracts increasing attention, and recent studies imply that RCVS is more common and distinct than previously appreciated (1, 9). Following the first systematic review by Call et al. (2), reports of patients presenting with the clinicoradiological constellation of recurring TCH and diffuse reversible vasospasm continued to be published under various nosologies by physicians of various backgrounds (for a comprehensive review see Calabrese et al. (1)). Due to this Babylonian confusion no clear awareness has been raised of this disorder, even among neurologists. Furthermore, routine diagnostic tests, such as CT and CSF, are frequently normal, and due to the usually benign course of the condition many patients probably remain undiagnosed. However, even when angiographic techniques are applied and show vasoconstriction, RCVS can easily be mistaken for a primary angiitis of the central nervous system (PACNS), due to similar angiographic features (16). As a consequence, corticosteroids or cytotoxic drugs may be administered, with no proven effect in RCVS. In the case of a relevant medical history of migraines, which is frequently seen, the condition might be mistaken for a migraine attack and migrainous infarction.
The complications seen in a proportion of patients are potentially pernicious. Pharmacological treatment is inconsistent and not backed up by study data, as no large clinical trials have been conducted for the treatment of RCVS. Obviously, causative factors need to be eliminated. In our case, the herbal remedy for menopausal symptoms was discontinued because it may contain active phyto-oestrogens. Observation can be sufficient in some cases, but good results are reported for treatment with the calcium-antagonist nimodipine, although superiority of this approach needs to be demonstrated in larger studies (4, 12, 13, 17, 18). Current observational reports suggest that nimodipine is effective for headache relief, and it could be more effective when delivered in the i.v. than in the oral form (13). Both forms of application are well tolerated, the only caveat being a fall in blood pressure, which needs to be avoided because of the potential decrease in cerebral perfusion pressure. Whereas there seems to be no risk for stroke or the development of posterior leukoencephalopathy in primary TCH (17), in RCVS brain infarction is likely to result from hypoperfusion distal to severe vasoconstriction. Interestingly, in a systematic study using TCD for monitoring mean flow velocity of the MCA and the Lindegaard Index, no immediate effect on vasospasm could be shown after initiation of treatment (19). In rare cases, even progression of strokes was seen in patients receiving i.v. nimodipine (9).
One of the main pitfalls in the described clinical scenario is the differentiation between RCVS and PACNS (1, 16). Oral or i.v. nimodipine treatment was not initiated in our patient due to the preliminary diagnosis of PACNS. Although corticosteroids can be a treatment option in RCVS (4), the clinical course worsened in our case, but would have been expected to be beneficial in PACNS. Clinical factors that support the diagnosis of RCVS are the acute rather than subacute to chronic onset of the headache that qualifies as TCH. CSF examination is usually normal or near-normal in RCVS, where it is abnormal in > 90% of PACNS. In our patient CSF examination showed only a slightly elevated cell count in the second examination (54 cells/cm3, reference range < 45 cells/cm3), as occasionally seen after repeat lumbar punctures or as a pleocytosis after cerebral infarction. Thus, these findings did not support the diagnosis of PACNS in our case. Whereas sensitivity of angiography in RCVS is very high, it lacks specificity, and even during angiography RCVS can easily be mistaken for PACNS. The most specific finding in RCVS is the resolution of vascular abnormalities within days or weeks. In this patient, immediate resolution of vasospasm was seen in the vascular territory where nimodipine was applied. The combination of the above features and the resolution after nimodipine infusion here confirmed the diagnosis of RCVS in our patient.
This is the first report on intra-arterial application of nimodipine following diagnostic angiography in a patient with RCVS. The reversal of vasospasm, monitored by conventional angiography, confirms the instant effectiveness of this application route. Although it cannot be ruled out that either the spontaneous clinical course might have been benign or the effect of i.v. treatment could have been similarly effective, this approach may have prevented further progression of stroke. In the repeated MRI 10 days after the intervention no progression of the ischaemic lesions was noted.
In earlier studies, treatment with intra-arterial nimodipine has shown a direct positive response in patients with vasospasm due to subarachnoid haemorrhage, although the effect was somewhat restricted to a subgroup and was only temporary in some patients (20–22). Aside from the adverse events well established for conventional diagnostic angiography, i.e. vessel dissection, rupture or bleeding (< 1%), intracranial hypertension has been reported in one patient in these studies. Additional risks of nimodipine infusion may include oedema or haemorrhagic infarction from transient relief of vasoconstriction.
We propose that, depending on the degree of vasoconstriction in RCVS and after individual assessment of contraindications, one possible therapeutic approach might be the parenteral or oral application of nimodipine. In progressive or treatment-refractory cases, intra-arterial application of nimodipine should be considered, although only as emergency therapy. Further clinical studies are warranted to determine the best possible therapeutic approach.
