Abstract
This study aimed to identify the cortical mechanisms underlying the processes of interictal dishabituation to experimental pain in subjects suffering from migraine with aura (MWA). In 21 subjects with MWA and 22 healthy controls, cortical responses to two successive trials of noxious contact-heat stimuli were analyzed using EEG-tomography software.
When compared with controls, MWA patients showed significantly increased pain-evoked potential amplitudes accompanied by reduced activity in the orbitofrontal cortex (OFC) and increased activity in the pain matrix regions, including the primary somatosensory cortex (SI) (
Reduced inhibitory functioning of the prefrontal cortex is a possible cause for disinhibition of the pain-related sensory cortices in migraine. The finding of OFC hypofunction over the disease course is in keeping with current concepts of migraine as a progressive brain disorder.
Introduction
Prolonged or repeated noxious stimulation normally leads to dynamic changes in the neural system associated with either reduced or increased activation in different brain areas (1). Such neural responses are suggested to be subserved by a cortical prefrontal-sensory gating system that modulates sensory processing by either inhibition or facilitation (2,3). Indeed, a functional magnetic resonance imaging (fMRI) study in healthy subjects revealed that repetitive noxious thermal stimulation results in attenuation of activity in the pain-related sensory cortices, concomitantly with increased activity in the cingulo-frontal area, bilaterally (4). Moreover, a magnetoencephalography study has also supported the thesis of significance of the frontal lobe in inhibiting somatosensory input, by demonstrating increased activation of the prefrontal regions in response to electrical stimulation in normal subjects (5).
Misplaced or reduced prefrontal activation could lead to abnormal processing of sensory input. For example, patients with focal prefrontal infarcts show significantly reduced activation over the primary somatosensory cortices during innocuous electrical stimulation (2). These findings indicate the presence of disinhibition of the primary receptive cortex as a possible consequence of the frontal lesion (2). Likewise, major depression patients with possible prefrontal dysfunction show similar reduced habituation of non-painful somatosensory input (6,7).
Dysfunctional sensory processing, reflected by deficient habituation of neural responses, has been widely demonstrated in migraineurs during the attack-free period for different sensory stimuli, including noxious laser stimuli (8,9). These changes have been attributed to either lower-than-normal excitability of the primary sensory cortices (8) or, alternatively, elevated excitability of these areas with a possible disinhibitory element (10).
Keeping in mind the inhibitory role of the prefrontal cortex, as shown in the examples mentioned above, one wonders whether dysfunction in this area might contribute to the lack-of-habituation phenomenon in migraine. We thus hypothesized that decreased sensory inhibition—that is, disinhibition by the dysfunctional prefrontal cortex—is one of the possible causes of increased excitation of the primary sensory cortices in migraine.
We tested our hypothesis by examining cortical evoked responses to repeated noxious contact heat stimuli delivered to the extra-cephalic area (hand) in MWA patients during the pain-free periods, and analyzing the interrelations between cortical generators involved in pain processing, using standardized low-resolution brain electromagnetic tomography (sLORETA) mapping.
Methods
Subjects
Twenty-one MWA patients meeting the International Headache Society criteria (18 women, 3 men, mean age 36 ± 11 years; disease duration 18 ± 12 years; attack frequency 3.4 ± 1.86 attacks/month; longevity range 48–2520 attacks during the disease course) and 22 headache-free individuals (19 women, 3 men, mean age 32 ± 7 years) were enrolled in the study after giving informed consent (11). None of the patients received any preventive medication for six months or analgesic medication 24 hours before the testing. The tests on migraineurs were performed at least two days since the last attack and two days before the next attack. Occurrence of the latter was checked by a telephone call three days after the recording. All subjects were right-handed and free of other neurological, ophthalmological or systemic disease.
The study was conducted according to the Declaration of Helsinki on biomedical research involving human subjects, and the protocol was approved by the Rambam Health Care Campus (Haifa, Israel) Ethics Review Committee. All subjects were instructed about the study, and written informed consent was obtained.
Electrophysiologic stimulation and recording set-up
Analysis
The latencies and peak-to-peak amplitudes for EP components N2 and P2 were identified at the vertex (Cz) electrode.
Source localization of brain electrical activity was done by sLORETA (13). Based on the inverse solution and ‘smoothness’ assumption (neighbouring neuronal generators show highly correlated activity), this method determines standardized three-dimensional intracerebral current density (unit: amperes per square meter, Å/m2) at 6239 voxels (5-mm spatial resolution) of the cortical grey matter and hippocampus. The methodology involved the estimation of the cortical potentials (i.e., cortical generators), by means of a three-shell spherical head model registered to the Talairach human brain atlas (14) digitized by the average of MRI scans from 305 of normal subjects recorded at the Montreal Neurological Institute, and freely downloadable from the sLORETA software website (www.unizh.ch/keyinst/NewLORETA/sLORETA/sLORETA.htm). Individual EEG electrodes were positioned by the Talairach coordinate system according to the spatial association between spherical and realistic head geometry (15). sLORETA software has been already validated in several pain studies (16,17).
Activity in the regions of significant activity (see statistical analysis) were measured at each 2 ms in a time window of 250–700 ms post-stimulus, yielding high-resolution temporal curves. Subsequently, individual inter-train differences (expressed as delta AUC activity) were calculated by subtraction of the areas under the curves (AUCs) between the two stimulation trains (i.e., AUCTrain2 −AUCTrain1
); where positive AUC values indicate increased cortical activity from the first to second train, and vice versa (Figure 1).
Calculation of delta AUC activity. An example of time courses of activity depicting the first (black line) and second (grey line) trains of stimuli in a sLORETA-identified cortical generator, expressed as electrical current density (unit: amperes per square meter, Å/m2) at the maximal voxel of the localized generator. A gap between the curves at the time window of 100 ms (350–450 ms post-stimulus) is quantitatively assessed by the delta AUC (area under curve, Å/m2*ms). Delta AUC value is negative when the activity is lower in the second train than in the first train, and vice versa.
Statistical analysis
Results were expressed as mean ± standard errors. Inter-train and inter-group differences were analyzed using a repeated measures one-way analysis of variance (ANOVA) and Tukey-Kramer post-hoc tests, accordingly (JMP software, SAS Institute, Cary, NC, USA).
For sLORETA, statistical differences were computed as images of voxel-by-voxel t-values. The images were examined to locate regions showing statistically significant effects, using a non-parametric approach thresholded at the 5% probability level determined by 5000 randomizations (13). sLORETA mapping of cortical electrical activity. Neurophysiological maps of the grand average of estimated electrical cortical activity in the first and second stimulation trains and the inter-train comparison (second train minus first train) at the 250–350 ms post-stimulus, in migraine with aura (MWA) and control groups. The activity is expressed as squared magnitude of the computed intracerebral current density (unit: amperes per square meter, Å/m2), with yellow/red for increased activity and blue for decreased activity. Subtracting brain activation during the first train from activation during the second train shows that MWA patients decrease activity in the orbitofrontal cortex (OFC), whereas healthy controls increase activation in this cortical region.
Pearson’s correlation analysis was applied to determine inter-individual interrelations of cortical activity and clinical correlates of migraine (i.e., attack frequency, days since the last attack, disease duration and longevity). Results were considered significant at
Results
Psychophysical and electrophysiologic findings
Mean ratings of pain intensity did not differ between MWA (43.8 ± 15) and control (40.8 ± 15) groups in the first train of stimuli (
For the EP data, N2P2 latencies were not different between the groups in both stimulation trains. There also were no significant inter-group differences in N2P2 amplitudes in the first train ( Inter-train changes for evoked potentials in MWA and control groups Grand average of contact heat-evoked potentials recorded at Cz (referenced to nose-tip) for migraine with aura (MWA) patients and healthy volunteers stimulated with two successive trains of 30 noxious heat pulses each. The potentials comprised a biphasic N2P2 component.
Source localization analysis
sLORETA analysis revealed two time-windows of 250–350 ms and 350–450 ms post-stimulus at which the differences of cortical activity reached statistical significance (
sLORETA statistical maps of inter-train and inter-group comparisons. (A) Statistical maps of inter-train comparisons between the second and first stimulation trains in migraine with aura (MWA) and control groups. Colors in the maps denote significant changes, with yellow/red for increased activity and blue for decreased activity; the critical threshold for statistical significance of 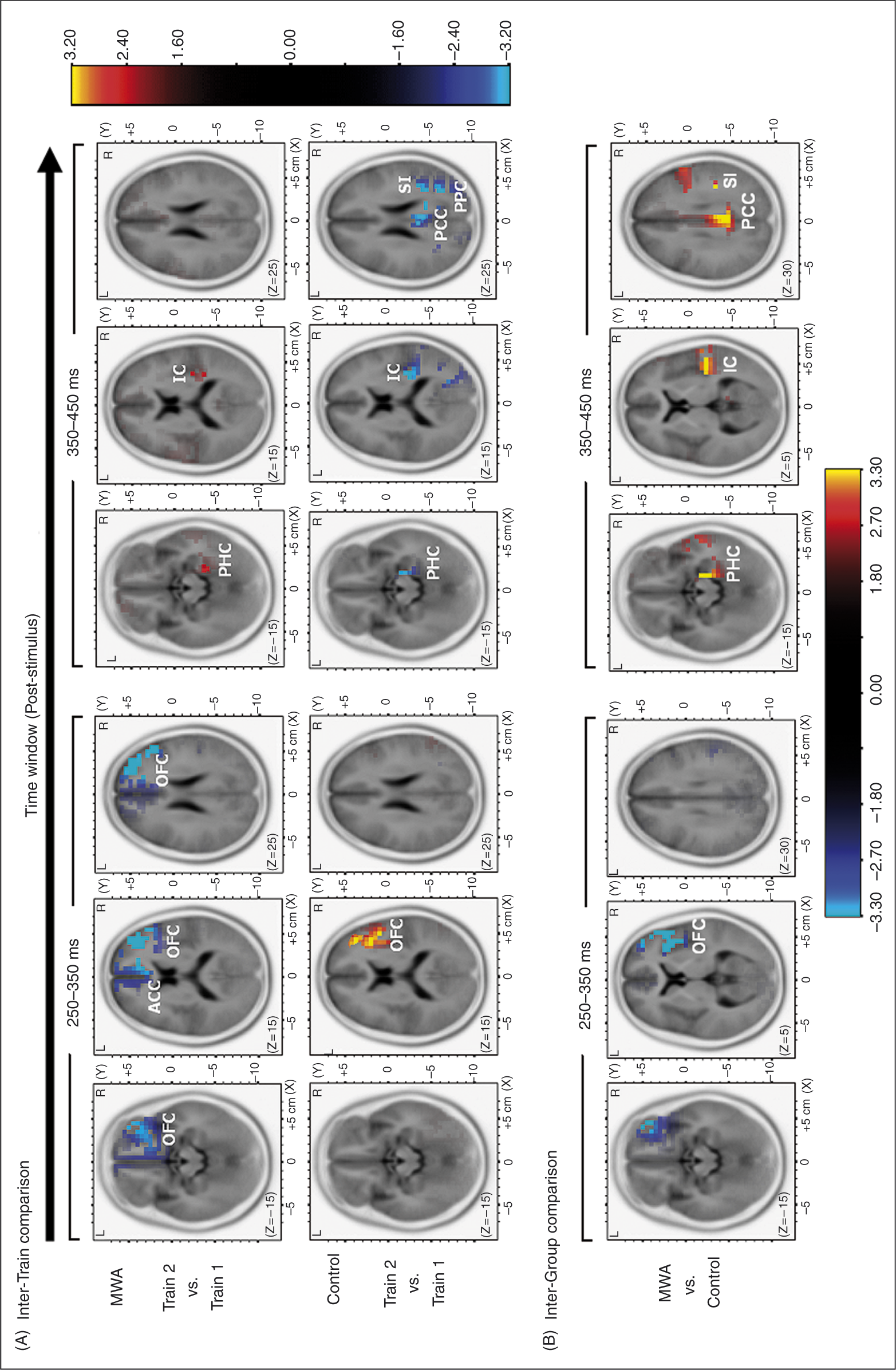
Compared to controls, MWA patients displayed attenuated inter-train activity in the contralateral OFC at 250–350 ms, and augmented activity of the contralateral SI, IC and PHC and bilateral PCC at 350–450 ms (Figure 4B).
Correlations between inter-train (delta AUC) activity and clinical parameters of migraine
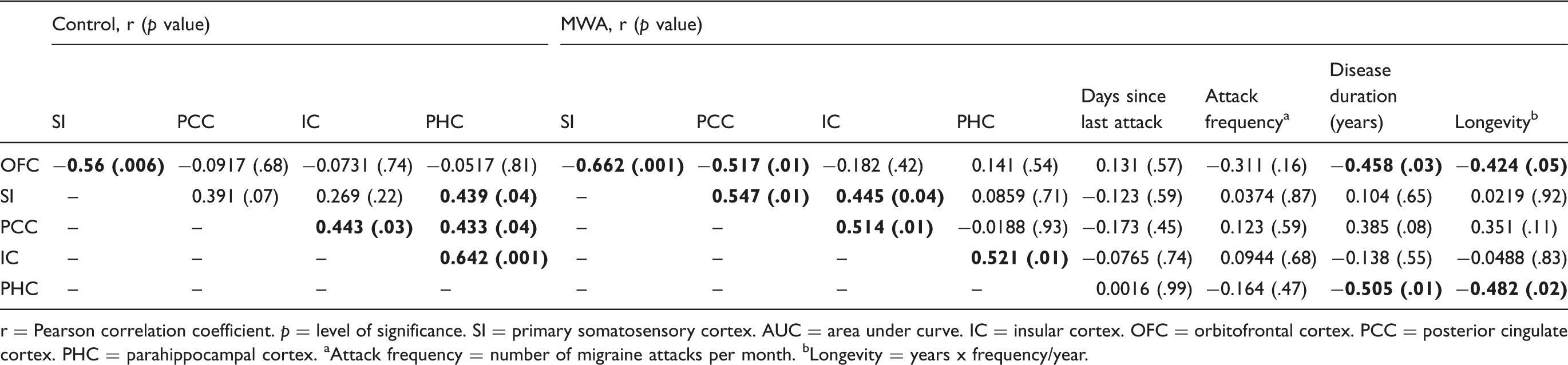
r = Pearson correlation coefficient.
In controls, an inverse correlation between the OFC at 250–350 ms and the SI at 350–450 ms (r = −0.56;
Discussion
In the present study, we identified differences in interictal cortical activity involved in sensory processing induced by repeated noxious input between MWA patients and healthy controls. This was done by using two successive trains of repeated contact heat stimuli, recording EPs, and mapping cortical generators by sLORETA analysis.
Our results suggest that in normal subjects there exists a physiological habituation to noxious heat-evoked stimuli, which is reflected by significant attenuation in N2P2 amplitudes. In MWA patients, however, habituation is replaced by potentiation of these cortical responses, expressed by markedly enhanced subjective pain ratings and increased N2P2 potentials. These results are concordant with previous EP studies implicating deficient habituation, or even potentiation, of nociceptive laser-evoked responses in migraineurs, as compared to healthy subjects (9,18).
Furthermore, the sLORETA software applied in our study made possible the localization of neural structures subserving the noxious heat-evoked responses in MWA and control groups.
Healthy controls, proceeding from the first to second trains of stimuli, displayed increased OFC activation and attenuated activation in the pain-sensory areas. This finding is consistent with a prior fMRI study demonstrating that normal habituation to noxious input is associated with enhanced activation in the ventral frontal regions, including the OFC (4,5). This association has been attributed to OFC-mediated attentional, anticipatory or emotional modulation of the pain cortex (16,19–22). In keeping, the OFC has been proposed to act as a filtering system for aversive sensory inputs via active inhibition of neural circuits in the sensory areas (19).
Anatomical tracing studies in nonhuman primates provided strong evidence for direct neural pathways from the lateral part of the OFC to the SI and posterior insula, whereas the medial OFC is extensively connected with the PCC, as well as with the PHC and hippocampus (23). In view of these findings we confirm that the OFC constitutes one of the central components for the inhibitory modulation of pain sensation by exerting inhibitory effects on remote pain processing structures, in particular the sensory cortices.
With regard to cortical generators in MWA patients, we found a strong focus of reduced inter-train activity in the contralateral OFC followed by enhanced activation in several pain processing regions, including the contralateral SI, IC, PHC and bilateral PCC. As already well described, nociceptive processing takes place within two parallel subsystems. The lateral pain system, projecting through lateral thalamic nuclei to the posterior, somatosensory regions of the cortex, is mainly involved in processing the sensory-discriminative aspects of pain. The medial pain system, in turn, targeting through medial thalamic nuclei the anterior, cingulo-frontal and insular regions of the cortex, is mainly instrumental in processing the affective-motivational aspects of pain (24–26). Both target regions seem to be dysfunctional in MWA patients; increased activation in the SI might point to augmented excitability of the sensory-discriminative cortex, whereas increased activation in the PCC, IC and PHC may contribute to the affective responses to painful stimuli. While the latter regions showed an inter-train increase of activity, the OFC distinguished itself as a single region with inter-train decrease of activity in MWA patients. A similar pattern of cortical activity during noxious stimulation has been previously described in patients with chronic pain disorders, such as fibromyalgia and somatoform pain disorder (27,28). Yet no previous study has reported such a pattern of activation during painful stimulation in MWA patients. Some data exist for cortical responses in MWA patients during innocuous sensory stimulation; a recent fMRI visual stimulation study has demonstrated enhanced interictal activation in the visual cortex and lessened activation in the OFC in MWA patients when compared to controls (29). Thus, our study provides new evidence for differential cortical response to
Analysis of the temporal courses of cortical activity revealed that the inter-train activation changes in the OFC precede the changes in the sensory cortices. Our findings support the hypothesis of an inter-regional connectivity, that is, temporal correlation between spatially remote neurophysiologic events. In this context, the negative temporal correlation between the OFC and SI activity, found in our study, is likely to reflect flow of neural information, that is, inhibitory connectivity between the anterior (frontal) and posterior (sensory) areas (2,19,23).
In both groups, a reciprocal positive inter-regional association was also found in the pain-sensory network, including the SI, IC, PCC and PHC; this observation fits well with the functional roles and anatomic connections of these brain structures (23,26,30). Comparing the inter-train responses between MWA patients and controls, we found similar patterns of inter-regional connectivity. This finding attests that, despite the inter-group differences in the
The mechanism underlying the OFC dysfunction in MWA patients is currently unknown. We speculate that it involves the serotonergic system. Evidence from animal and human studies suggests a role of serotonin (5-HT) in the OFC-mediated descending inhibition of pain (31,32). In migraine, a possible alteration of brain 5-HT neurotransmission is assumed to be linked to hypo-excitability of the visual and auditory cortices (8,33). Our view, however, is that the changes in serotonergic activity in migraine can be referred to the changes of neuronal activity in the OFC. As a result, the ability of the OFC to inhibit—that is, ‘filter out’—the sensory information may be disrupted. Persistent orbitofrontal metabolic abnormality has been previously reported by means of (18) F-fluoro-deoxyglucose positron emission tomography (FDG-PET) in analgesic-abusing migraine patients even after withdrawal of the medication (34). The authors assumed that the OFC hypoactivity is related to vulnerability to substance abuse and drug dependency, suggesting a role of both the serotonergic and dopaminergic systems in migraineurs developing medication overuse headaches (34). It could be thus hypothesized that the OFC dysfunction linked to pain modulation in migraine patients may also involve both serotonergic and dopaminergic modulatory pathways; the involvement of these and other neurotransmitters remains to be further explored.
It has been widely agreed that interictal habituation deficit in migraine accounts for an abnormal state of cortical excitability of the sensory cortex. However, the possible mechanisms for this change in excitability are still debated. One theory postulates a ‘ceiling’ effect phenomenon, in which the level of basal excitability—the pre-activation level—of primary sensory cortices in migraineurs is low (8,33). Hence, in response to repetitive stimulation, there is a progressive increase in cortical excitability up to the ‘ceiling’, resulting in potentiation of neural activity. This is opposed to the habituation effect seen in healthy subjects resulting from a higher, closer to the ceiling, baseline excitability level. Another hypothesis is that the lack of habituation results from enhanced excitability or disinhibition of sensory cortices in migraine (35,36). In our study, localization of generators by the sLORETA methodology revealed anterior prefrontal regions with abnormally reduced activity within the OFC region. Dysfunction of this region may result in an increase of excitability, that is, disinhibition, of the sensory cortices.
Although the sLORETA algorithm used in our study has received comprehensive cross-modal validation (13), it is important to highlight several limitations of this method. First, we superimposed the generators of cortical activity on a standardized (average) brain probabilistic template and derived the EEG electrode coordinates from computed cross-registrations between the spherical and realistic head geometry. This method, however, may represent only a close approximation, but not an exact measurement of the sources of cortical activity; thus, using a non-realistic head model, we only suggest the possible brain generators involved in pain modulation in migraineurs. Clearly, further research comparing individual anatomical MRI scans of these patients with the sLORETA algorithm is expected to validate the results of our study. Second, because sLORETA analysis is limited by relatively low spatial resolution and a tendency to underestimate deep generators in favor of cortical ones, our analysis is pertinent mostly for the cortical regions of the pain network (13). Based on previous work, it is reasonable to hypothesize that subcortical structures, including dorsal raphe nuclei and periaqueductal grey matter, are also involved in the inhibitory-excitatory circuits in migraine (37–39), but this cannot be demonstrated here.
Looking for clinical neurological correlates of migraine, we found a negative correlation between OFC and PHC function and disease longevity and duration, indicating greater hypofunction of the OFC and lesser activation of the PHC along the disease course. Our findings are in agreement with recent voxel-based morphometric studies demonstrating a notable correlation between illness duration and the extent of structural abnormalities in several brain regions, including these specific areas (40,41). Thus, our neurophysiological data suggest that during repetitive occurrence of migraine attacks along the years, in MWA patients, these brain regions seem to experience significant functional and metabolic disturbances leading to morphological changes that accumulate over the disease course. This line of thinking concurs with the current understanding that MWA is a progressive disease, leaving functional, in addition to morphological, residues on the brain.
Disclosures
The authors report no disclosures.
