Abstract
The aim of this study was to compare the recovery cycle of somatosensory evoked potentials (SEPs) in children with migraine without aura before and after treatment with topiramate. Eleven migraine children were studied before and after a 3-month treatment with topiramate at the average dose of 1.3 mg/kg/day. We calculated the SEP latency and amplitude modifications after paired electrical stimuli at 5, 20 and 40 ms interstimulus intervals, comparing them with a single stimulus condition assumed as baseline. In nine patients, who had a significant reduction in headache frequency after treatment, the recovery cycles of the P24 (P = 0.03) and N30 (P < 0.005) potentials were longer after than before topiramate treatment. In two migraineurs who did not show any improvement, the recovery cycles of the cortical SEP components were even shorter after treatment. Our results suggest that topiramate efficacy in paediatric migraine prophylaxis is probably related to restored cortical excitability.
Introduction
In migraine, the most reproducible neurophysiological finding during the interictal phase is reduced habituation of cortical evoked potentials to repeated stimuli when compared with healthy subjects. Habituation is a common physiological feature of cortical responses to visual, auditory, olfactory and somatosensory stimuli and is characterized by a gradual decrease in the amplitude of cortical responses to repetitive stimuli. The lack of habituation in migraineurs is considered as a marker of abnormal cortical excitability, although there is no agreement on whether it may be referred to hypo- or hyperexcitability of the cerebral cortex (1–8). The response pattern of the somatosensory cortex in migraine has also been studied by analysing the somatosensory evoked potential (SEP) recovery cycle. In healthy subjects, the most characteristic finding during repetitive electrical nerve stimulation is a reduction of SEP component amplitude as a function of decreasing time between stimulus presentation. When SEPs are obtained by two paired electrical stimuli at different interstimulus intervals (ISIs), the SEP amplitude evoked by the later stimulus is smaller than that recorded from a single stimulus. The longer the ISI, the higher the amplitude of the SEPs recorded after the second stimulus, until complete amplitude recovery is observed. The SEP amplitude decrease in a paired-stimuli protocol is caused by complex inhibitory mechanisms within the cortex that reduce the excitatory postsynaptic potentials as well as by synaptic refractoriness of networks and relays generating individual peaks (9). The SEP recovery cycle can, thus, be considered as a marker of primary somatosensory cortex excitability. Indeed, a shortened cortical SEP recovery cycle has been previously demonstrated in diseases that are characterized by central nervous system hyperexcitability, such as cortical myoclonus, subacute spongiform panencephalitis (10,11), or by disinhibition of the somatosensory system, such as dystonia (12). In a previous study, we found that children with migraine have a shorter SEP recovery cycle when compared with healthy, age-matched controls, indicating disinhibition of the somatosensory system (13).
Topiramate (TPM) is an antiepileptic drug that has been proven to be effective in the preventive treatment of migraine in adults. Two randomized controlled trials have confirmed these results in children and adolescents (14–16). TPM has a wide variety of actions, including: a negative modulatory effect on the AMPA/kainate subtype of glutamate receptors, a positive modulatory effect on γ-aminobutyric acid (GABA) A receptors, use- and time-dependent blockade of voltage-activated Na+ channels, a negative modulatory effect on a neuronal L-type high-voltage-activated Ca2+ channel and an inhibitory effect on carbonic anhydrase isozymes (17). However, the precise mechanism accounting for TPM's efficacy in migraine prophylaxis is still uncertain, although it seems to be linked to neuronal excitability reduction (18–20). Although Aurora et al. (21) have suggested that TPM may reduce the excitability of the visual cortex, after having studied two patients with probable chronic migraine, a possible effect of TPM on somatosensory cortex excitability has not been investigated thus far.
The aim of this study was to compare the SEP recovery cycle in a group of children with migraine without aura before and after treatment with TPM.
Methods
Patients
Inclusion criteria
We selected 11 consecutive children (three boys, eight girls, mean age 11.8 ± 2.2 years, range 8–14) who referred to the Headache Centre of Paediatric Hospital ‘Bambino Gesù’ in Rome and fulfilled the criteria of the International Classification of Headache Disorders: 2nd edition (2004) (22) for migraine without aura. They suffered from at least four monthly migraine episodes and were studied with the same SEP protocol before and after a 3-month treatment with TPM at the average dose of 1.3 mg/kg/day.
Exclusion criteria
Subjects were excluded if they experienced cluster headache or chronic (transformed) migraine, had had > 15 headache days during the prospective baseline phase, overused analgesics or acute migraine treatments (> 12 days/month of analgesics or > 8 days/month of ergot or triptans), due to the increased potential for medication overuse headache. Other reasons for exclusion included previous failure of two or more adequately dosed migraine preventive medications, previous failure of TPM therapy for migraine prevention, use of TPM or any other migraine preventive medication within 14 days of the prospective baseline phase, and a history of nephrolithiasis. The patients enrolled in this study were allowed to take acute abortive medications for symptomatic relief of migraine and non-migraine headaches.
The children, with the help of their parents, had kept a headache diary for 2 months before entering the study and continued to do so during the 3-month treatment with TPM. The target daily dose of TPM was reached by means of weekly increases of the drug dose by approximately one-quarter of the definitive dose for the first 4 weeks.
None of the patients was on preventive medication and the mean number of headache episodes/month at baseline was 6.73 ± 1.95. SEPs were recorded no sooner than 72 h from the last episode and no migraine attacks occurred in the 48 h after the recording session.
The study was performed in agreement with the Declaration of Helsinki and was approved by the local Ethics Committee. Informed consent to be included in the study was obtained from the children and their parents.
SEP recording technique
For SEP recording, subjects lay on a bed in a quiet and semi-darkened room. Right and left median nerves were stimulated at the wrist by surface electrodes; stimulation intensity was fixed at the motor threshold (stimulus duration 0.2 ms, stimulus rate 2 Hz). SEPs were recorded from four electrodes (impedance < 5 kΩ) placed: (i) at Erb's point, ipsilateral to the stimulation (Erbi), referred to an electrode at the contralateral Erb's point (Erbc); (ii) over the sixth cervical vertebra (C6), referred to an anterior electrode above the thyroid cartilage (AC); (iii) over the parietal area contralateral to the stimulation (P3/P4); (iv) over the frontal midline (Fz). The scalp electrodes were referred to an electrode at the earlobe ipsilateral to the stimulated side (Au1/Au2). The ground was at the stimulated arm. The analysis time was 100 ms, with a sampling rate of 5000 Hz. The amplifier bandpass was 10–2000 Hz (12 dB roll-off).
Experimental procedure
The following procedure was carried out before and after 3 months' treatment with TPM—averages of 800–1000 runs were recorded in four experimental conditions: (i) baseline (single stimulus), which was taken as control; (ii) paired stimuli at 5 ms ISI; (iii) paired stimuli at 20 ms ISI; and (iv) paired stimuli at 40 ms ISI. In the double-stimulus traces, the responses following the second stimulation were obtained by subtracting the control SEP waveforms from the waveforms following each double stimulus.
Analysis of clinical efficacy
We compared the headache frequency at baseline, relative to the 2 months preceding the trial, with the headache frequency at 3 months, relative to the third month of treatment with TPM, using Student's t-test.
SEP analysis
SEPs were identified according to their latency and polarity. The peak latencies and peak-to-peak amplitudes of the SEP components were measured. The investigator who measured the SEP components (R.M.) was blind for condition (pre vs. post treatment with TPM).
The peripheral N9 potential, generated by the brachial plexus volley, was recorded by the Erbi electrode, whereas the spinal N13 response, generated within the cervical grey matter (23), was identified in the C6-AC trace. Both scalp electrodes (P3/P4 and Fz) recorded the P14 component, originated from the medial lemniscus and measured in the frontal trace where it shows the highest amplitude (24). The parietal N20 and P24 responses, generated by neurons in the primary somatosensory area, were recognizable in the parietal trace, whereas the N30 potential, whose origin is still debated (in the pre-central or post-central cortex), was recorded by the frontal lead. SEP latencies and amplitudes following a single stimulus, recorded after left- and right-side stimulation, were compared by using a paired Student's t-test and the Wilcoxon test, respectively.
In the single stimulus condition, paired Student's t-test and Wilcoxon test were performed to compare SEP latencies and amplitudes before and after treatment.
Two-way
In order to reveal possible differences between the SEP amplitude recovery cycle
of children with migraine before and after treatment with TPM, the SEP
amplitudes in the double stimulus conditions were normalized as the percentage
of the corresponding SEP amplitude obtained after a single stimulus, which was
assumed as 100%. The SEP amplitudes to double stimuli at different ISIs were
then compared by means of two-way
Results
Clinical efficacy and tolerability
After the 3-month treatment period, headache frequency decreased significantly from 6.73 ± 1.95 to 3.64 ± 4.34 monthly episodes (P = 0.02). Of the 11 patients, nine (81%; seven female, two male, mean age 11.67 ± 2.39 years) were considered responders (> 50% decrease in headache frequency), with a mean frequency reduction of 71.4%, whereas two patients (one male, one female, mean age 12.5 years) did not respond to the treatment. In one of the non-responders, headache frequency increased (from seven to 15 monthly crises) and in the other the headache frequency decreased from 11 to eight monthly crises (27% frequency reduction). TPM was generally well tolerated: one patient experienced weight loss (−1 kg) and another patient complained of fatigue.
Neurophysiological results
SEP amplitudes and latencies
In the single-stimulus condition, the comparison of SEP amplitudes and latencies recorded from the two sides showed no differences (P > 0.05). Furthermore, there were no significant differences in SEP amplitudes and latencies between before and after treatment in the single-stimulus condition (P > 0.05).
SEP recovery cycle
There were no significant modifications of the SEP latencies at different ISIs before and after treatment (P > 0.05).
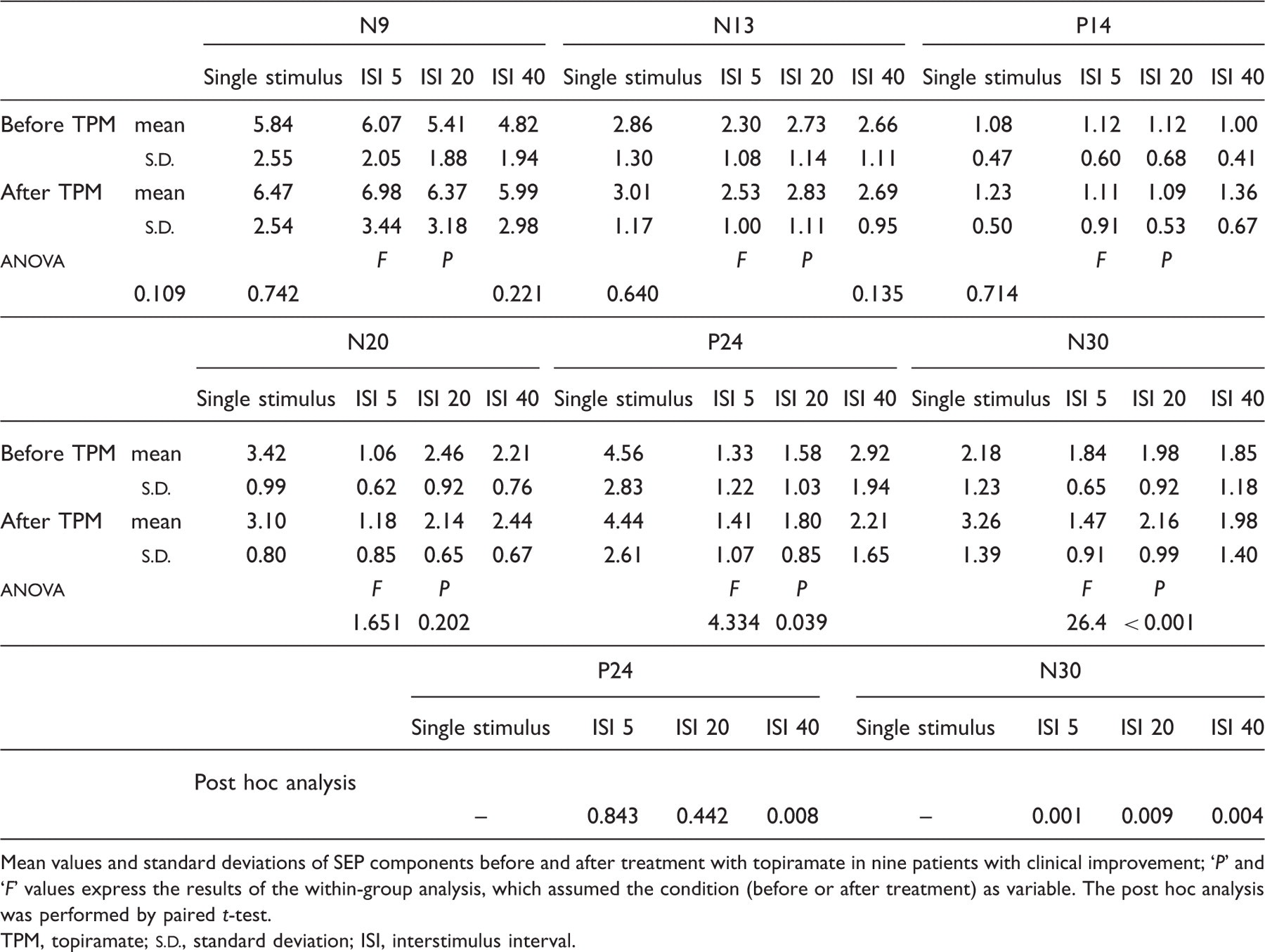
In nine patients, who had a significant reduction in headache frequency
(> 50%) after treatment with TPM, the recovery
cycles of the parietal P24
(F = 4.3,
P = 0.03) and N30
(F = 26.4,
P < 0.005)
potentials were significantly longer after treatment when compared with
baseline (Fig. 1). Somatosensory evoked potential (SEP) recovery cycle in a patient
before and after treatment with topiramate. SEPs were recorded
after a single stimulus (baseline), and after two paired stimuli
at 5, 20 and 40 ms ISIs. In the double-stimulus
traces, the responses following the second stimulation were
obtained by subtracting the baseline SEP waveforms from the
waveforms that follow each double stimulus. Notice that after
treatment the P24 and N30 responses show a lower recovery at 20
and 40 ms ISIs when compared to before
treatment.
Post hoc analysis showed that at 40 ms ISI the P24 amplitude was
higher after treatment when compared with before treatment
(P = 0.004) and that at
all ISIs the N30 amplitude was significantly higher after treatment when
compared with before treatment
(P < 0.01) (Table 1,
Fig. 2). In contrast, in two migraineurs who did not
show any improvement the recovery cycles of the cortical SEP components were
even shorter after the pharmacological treatment (Table 2, Fig. 3). Pre-treatment (A) and post-treatment (B) mean somatosensory
evoked potential (SEP) amplitudes at different ISIs in patients
with clinical improvement. The SEP amplitudes in the
double-stimulus conditions are expressed as percentage of the
corresponding baseline amplitudes, considered as 100%. Vertical
bars indicate standard deviations. Notice the faster recovery
cycle of the P24 and N30 responses before than after
treatment. Pre-treatment (A) and post-treatment (B) mean cortical
somatosensory evoked potential (SEP) amplitudes at different
ISIs in two patients without clinical improvement. The same
presentation as in Fig. 2. Notice that the recovery cycles of
the cortical SEP components were not lengthened by treatment, as
compared to before treatment. Mean somatosensory evoked potential (SEP) amplitudes in responder
patients Mean values and standard deviations of SEP components before
and after treatment with topiramate in nine patients with
clinical improvement;
‘P’ and
‘F’ values
express the results of the within-group analysis, which
assumed the condition (before or after treatment) as
variable. The post hoc analysis was performed by paired
t-test. TPM, topiramate; Somatosensory evoked potential (SEP) amplitudes in patients
without clinical improvement Absolute SEP amplitudes before and after treatment with
topiramate in two patients without clinical improvement. TPM, topiramate; ISI, interstimulus interval; NR pt 1,
non-responder patient 1; NR pt 2, non-responder patient
2.





Discussion
The main finding of our study is represented by the lengthening of the recovery cycle of the P24 and N30 SEP components in children affected by migraine without aura who had responded to a 3-month treatment with TPM. Interestingly, in the two patients whose headache frequency did not decrease during treatment, the SEP recovery cycle did not differ significantly between before and after treatment.
Effect of topiramate on cerebral cortex excitability
The effect of TPM on human cortex excitability has been previously studied using transcranial magnetic stimulation (TMS) in healthy subjects and in patients with probable chronic migraine (PCM) and chronic neuropathic pain (21,25–27). The effect of single oral doses of TPM on intracortical inhibition (ICI) and intracortical facilitation was investigated in 20 healthy subjects in a double-blind, placebo-controlled, crossover study (25). A significant dose-dependent increase of ICI was noticed after 200 mg TPM compared with placebo. The authors conclude that a single dose of TPM selectively increases ICI by GABAA-ergic mechanisms and/or glutamatergic antagonism. Inghilleri et al. (26) studied 23 patients with neuropathic pain before and after a 1-week course of treatment with carbamazepine, gabapentin, TPM and placebo. Antiepileptic treatment, but not placebo, abolished the normal repetitive TMS (rTMS)-induced facilitation of motor evoked potentials (MEPs), but left the progressive lengthening of the cortical silent period during the rTMS train unchanged. These results suggest that carbamazepine, gabapentin and TPM modulate intracortical excitability by acting selectively on excitatory interneurons. In patients with chronic neuropathic pain, the same authors (27) evaluated the effects of TPM at different doses (25, 50, 75 and 100 mg) on cortical excitability variables tested with rTMS. In patients receiving TPM at higher doses (75 and 100 mg), rTMS failed to elicit the MEP facilitation, suggesting that TPM modulates the excitatory intracortical interneurons and impairs rTMS-induced synaptic potentiation. Finally, Aurora et al. (21) tested occipital cortex excitability by using the magnetic suppression of perceptual accuracy (MSPA) profiles in two patients with PCM before and after treatment with TPM. In both patients, MSPA profiles appeared to normalize at a 100-mg dosage of TPM. The authors postulate that the efficacy of TPM in reducing headache frequency is due to its GABAergic activity, which reduces cortical excitability.
The effect of TPM has also been studied on the rat cortex, where it caused inhibition of transient and persistent Na+ current fractions (28), whereas in another study it was shown that several drugs that are widely prescribed for migraine prevention (TPM, valproate, propranolol and amitriptyline) are able to elevate the cortical spreading depression (CSD) threshold (29). CSD is characterized by a slowly spreading wave of neuronal and glial depolarization and is thought to be implicated in migraine pathogenesis (30). The authors concluded that TPM may suppress cortical excitability and thus elevate the CSD threshold via actions on multiple sites, including ion channels and neurotransmitter receptors (29).
Migrainous somatosensory cortex and topiramate
Although diverse neurophysiological techniques have been used to study cortical excitability in migraine (motor, visual, auditory and somatosensory evoked potentials) (31), the effect of TPM has been investigated only on the motor system excitability in healthy subjects and in patients with neuropathic pain and on visual cortex excitability in two patients with PCM (21,25–27,32,33). This is the first study to assess the effect of TPM on the somatosensory system of patients with episodic migraine. The somatosensory system processes information on several modalities of somatic sensation, including pain, which is the main symptom in migraine. Neurophysiological techniques exploring the somatosensory system are, thus, more appropriate in the study of migraineurs than those assessing motor, visual or auditory pathways. The study of the SEP recovery cycle, which has been shown to be shortened in pathological conditions characterized by central nervous system disinhibition, including migraine (10–13), is probably the most suitable method to investigate the effects of drugs on the migrainous somatosensory system.
One of the possible mechanisms that could explain a shortened SEP recovery cycle in migraine is an increase in intracellular sodium concentrations (34), which causes a depolarized resting neuronal membrane and, thus, neuronal hyperexcitability. Abnormal neuronal excitability is implicated in the pathogenesis of all forms of familial hemiplegic migraine, due to mutations of the gene encoding for the neuronal voltage-dependent P/Q-type calcium channels (FHM1), for the α2-subunit of the Na+/K+ ATPase ionic pump (FHM2), and for the α1-subunit of neuronal sodium channels (FHM3) (35).
The effect of TPM on the SEP recovery cycle could be explained by its action on voltage-dependant sodium channels. TPM's ability to inhibit voltage-gated sodium currents has been demonstrated in rat cerebellar and pyramidal cells (28). The blockage of voltage-dependant sodium channels might reduce the intracellular Na+ concentration, thus decreasing the neuronal rest potential. This could explain the effectiveness of TPM in lengthening the SEP recovery cycle.
It is not clear why TPM is only able to ‘normalize’ the recovery cycle of the P24 and N30 components, while it does not seem to have any effect on the recovery cycle of the N13 or N20 components. We have no satisfactory explanation for this phenomenon: the first hypothesis is that the N13 generator (cervical dorsal horn cells) is not directly implicated in migraine pathogenesis.
Alternatively, TPM is able to reduce the frequency of migraine episodes, but does not render patients completely free of migraine attacks. In other words, TPM is not able to revert the migrainous cortex disinhibition at all levels, which probably explains why it can reduce the frequency of migraine attacks without abolishing them.
Another mechanism that can explain the reversal of cortical disinhibition after TPM treatment is the effect of this drug on the GABAA-ergic inhibitory circuits. Indeed, TPM increases intracortical inhibition, as demonstrated by a TMS study, by acting on the cortex GABAA-ergic tone (25).
The shortening of the SEP recovery cycle in migraine patients is not necessarily due to somatosensory system hyperexcitability, but could merely represent a neurophysiological marker of abnormal somatosensory gating. This hypothesis is consistent with the previous evidence of a lack of auditory sensory gating in migraine patients. Abnormal gating mechanisms in both the somatosensory and auditory systems may be due to a hypofunction of monoaminergic subcortico-cortical pathways (36). From this point of view, TPM would act on this subcortico-cortical system to restore the physiological gating, thus leading also to SEP recovery cycle lengthening.
A shortened SEP recovery cycle in our migraine patients could be, at least in part, due to an habituation deficit. However, there is no agreement in previous studies investigating SEP habituation in migraine. Ozkul and Uckardes (37), by using a 3-Hz stimulus rate, showed significant habituation of both the N13 and N20 potentials in healthy subjects, whereas migraineurs showed even a potentiation. In contrast, Lang et al. (38) analysed 18–20 trial blocks recorded by a 1-Hz stimulus rate and did not find any habituation of the early somatosensory evoked components in either healthy subjects or migraine patients. Further studies are needed to clarify this issue.
Limitations of the study
The present study has some limitations that must considered in the interpretation of the results. (i) The main limitation is the low number of non-responders (only two), due to the efficacy of TPM in migraine prophylaxis (81% of responder rate). This prevented any statistical comparison between responders and non-responders. Thus, we decided to use within-group analysis to investigate the effect of TPM on the SEP recovery cycle only in patients who had improved after pharmacological treatment. Unfortunately, we could merely show preliminary observational data concerning the SEP recovery cycle modifications in non-responders. A further study, which takes the high efficacy of TPM in migraine prophylaxis into account and enrols a larger sample of patients, is needed to draw definitive conclusions on the possible effect of TPM on non-responders' brain excitability. (ii) The present study was performed only in patients with migraine without aura, due to the much lower frequency of migraine with aura in the paediatric population (39). Therefore, our results need to be confirmed in patients with migraine with aura. (iii) The effect of TPM on the SEP recovery cycle was explored in a paediatric population. Childhood migraine may be a good model for the study of the pathophysiological basis of the disease, because many factors may occur during the course of life, such as concomitant diseases, effects of drugs and environmental influences, changing the phenotypic expression of migraine (40). However, whether the present results may also apply to adult migraineurs has to be investigated by further studies.
Conclusions
Our results have shown that a 3-month treatment with TPM lengthened the recovery cycle of the P24 and N30 SEP components in all children who had improved clinically. Since the SEP recovery cycle may be assumed as a marker of somatosensory system excitability or of the effectiveness of somatosensory gating, it is conceivable that the efficacy of TPM in the preventive treatment of paediatric migraine is related to its ability to reduce cortical disinhibition or to modify the sensory gating at somatosensory level. However, if TPM's effect in migraine consisted merely of increasing the inhibitory mechanisms of the central nervous system, the SEP recovery cycle after treatment in non-responders would be difficult to explain. Indeed, in our two non-responder patients the SEP recovery cycle seemed even shortened after TPM. The hypothesis can be made that in migraine the main dysfunction of cortical input processing concerns the capability of the cortex to tune its excitability according to the external requests. This may explain TPM's effect in non-responders and the paradoxical effect of rTMS on cortex excitability (41,42).
