Abstract
The pathophysiology of cluster headache (CH) is only partly understood. Nitric oxide (NO), a potent vasodilator, has been suggested to be involved, and increased plasma levels of nitrite, a stable product on NO degradation, have been identified in the active period and in remission. The aim of our study was to investigate the role of NO in CH by measuring its oxidation products, nitrite and nitrate, in the cerebrospinal fluid (CSF), a biological compartment closer to the supposed focus of the disorder. We collected CSF from 14 episodic CH patients. Lumbar puncture (LP) was performed at two occasions: in active period between headache attacks, and in remission, not earlier than three weeks after the last CH attack. Eleven healthy volunteers served as controls. To estimate NO production, we determined the levels of NO-oxidation end products (NOx), that is, the sum of nitrite and nitrate, by using capillary electrophoresis. CH patients in the active period had significantly increased NOx levels (mean 9.3, 95% confidence interval [CI] 8.5–10.1) compared with those in remission (mean 7.6, 95% CI 6.9–8.2; p < 001) and control subjects (mean 6.2, 95% CI 4.9–7.5; p < 0.001). CH patients also had statistically significant enhanced NOx levels in remission compared with those of control subjects (p = 0.034). CSF was also analysed with regard to inflammatory parameters and protein content. CSF showed signs of pleocytosis or oligoclonal bands or albumin increase in 43% of CH patients although these results were not conclusive. We suggest that CH patients have a generally raised NO tonus, both in the active period and in remission. We interpret these results as indications of a basal hyperfunction of the L-arginine-NO pathway, possibly as an expression of inflammatory activity, and sensitization of pain pathways. This is the first study analysing NOx in CSF in CH, and the results support NO involvement in the pathogenesis of CH.
Introduction
Cluster headache (CH) is characterized by recurrent, unilateral attacks of excruciating pain in the orbital/supraorbital/temporal region. The pathophysiology of CH is in many aspects unknown. However, both the trigeminovascular and the hypothalamic system are presumed to be involved (1,2). A possible inflammatory component has also been discussed. In an affymetric study, upregulation of several pro-inflammatory proteins and cytokines was found (3). Furthermore, interleukin-2 (IL-2) upregulation during the active CH period has been observed (4). On the other hand, several pro-inflammatory adhesion molecules are down-regulated during the active CH period and up-regulated during remission, a reversed pattern observed among patients with superficial temporal arteritis (5). Furthermore, neither clinical symptoms nor systematic changes in routine laboratory tests suggesting a systematic inflammatory disorder were observed among episodic CH patients (6).
Periods with CH are associated with an increased sensitivity to various stimuli which act as attack-promoting, for instance nitroglycerine, a donor of NO that has powerful vasodilator effect (7). Local dilatation of intra- and extracranial vessels may accompany symptoms during CH attacks (8–11). NO is synthesized from L-arginine by a family of enzymes, the nitric oxide synthases (NOS). There are at least three different isoforms of NOS (endothelial, neuronal and inducible NOS). NO does not interact with a specific receptor; instead it diffuses freely across membranes (12). NO has many different physiological effects. For example, the L-arginine-NO pathway regulates vascular tone, inhibits platelet activity and leukocyte adhesion to the endothelium and alters cellular respiration (13–14). Significantly enhanced levels of nitrite in the plasma of CH patients have been observed, both in the active period and in remission (12), although conflicting results have also been reported (15). Decrement of NO was shown in response to hyperbaric oxygen treatment of CH patients compared with patients treated with placebo (16).
Activation of the trigeminovascular system is considered an important step in the development of CH attacks (17). Calcitonin gene–related peptide (CGRP) is a marker of this activation, and increased levels have been found in blood from the external jugular vein during CH attacks provoked by NO donors (17,18). These findings indicate that there could be a link between NO, the trigeminovascular system and CH pathophysiology.
We decided to elucidate the role of NO in episodic CH pathophysiology by analysing NO within the central nervous system, closer to the supposed affected area in the hypothalamus (2), by examining NOx in the CSF of episodic CH patients, both in the active period and in remission. Furthermore, we wanted to compare NO levels and CSF parameters in both phases of the disease.
Materials and methods
Sample collection
Demographic data of cluster headache patients and controls

CH: cluster headache.
The method of capillary electrophoresis
Because NO has a very short half-life (three to five seconds), after which it is converted by oxygen and water into the more stable nitrite and nitrate (20), we measured the levels of these NO-oxidation products. We have combined the levels of nitrite and nitrate and called the sum nitritic oxide products (NOx). The method of capillary electrophoresis separates ions on the basis of their charge ratio and allows individual detection of nitrite and nitrate. Nitrite and nitrate were measured as previously described (21). In short, all vials and filters were carefully washed in deionized, distilled water (Elgast prima 1-3, Buckinghamshire, UK). CSF samples were diluted 1:50 with deionized and distilled water, and ultracentrifuged at 5000 x g through Ultrafree-MC filters (Millipore, Bedford, MA, USA), with a nominal weight cut-off of 5 kDa, and were analysed on a Hewlett-Packard (HP) 3D capillary electrophoresis system (HP, Waldbronn, Germany). The effective length of the capillary was 104 cm. The electrolytes consisted of 25 mM sodium sulphate containing 5% NICE-Pak OFM Anion-BT (osmotic flow modifier) in deionized and distilled water. Samples were injected by electromigration for 20 s at −6 kV and analysed at a negative potential of 300 Vcm−1. Data were acquired at a response time of 0.1 s at 214 nm on an HP 3D Chem Station data system. Each sample was analysed twice and intra-assay precision was assessed from standard curves of nitrite and nitrate in between 0.1 and 100 µM.
Statistical methods
Statistical analysis of NOx levels was performed by using the Student’s t-test. A p value <0.05 was considered statistically significant.
Results
CH patients in the active period had significantly increased NOx evels (mean 9.3, 95% CI 8.5–10.1) compared with in remission (mean 7.6, 95% CI 6.9–8.2; p < 0.001) and control subjects (mean 6.2, 95% CI 4.9–7.5; p < 0.001) (Figure 1). A significant difference was also present between CH patients’ NOx levels in remission and control subjects (p = 0.034), where CH patients in remission had higher NOx levels than the control subjects. Individual NOx changes between the active period and remission are shown in Figure 2 and Table 2. Thirteen of 14 CH patients showed higher NOx levels in the active period compared with remission. Only one CH patient showed higher NOx levels in remission compared with the active period.
Mean levels of nitritic oxide products (NOx) (µM; CI ± 95%) in cerebrospinal fluid from episodic cluster headache patients during both phases of the disease. CI = confidence interval. Individual levels of nitritic oxide products (NOx) during the active period and in remission among episodic cluster headache (CH) patients. Circles in the figure indicate patients who had a CH attack which started as the lumbar puncture (LP) was performed. Unfilled squares indicate patients who had their last attack 24 hours before the LP. Levels of nitritic oxide products estimated by the capillary electrophoresis technique (µM), in episodic cluster headache patients and healthy, headache-free control subjects NOx: nitritic oxide products, CH: cluster headache.
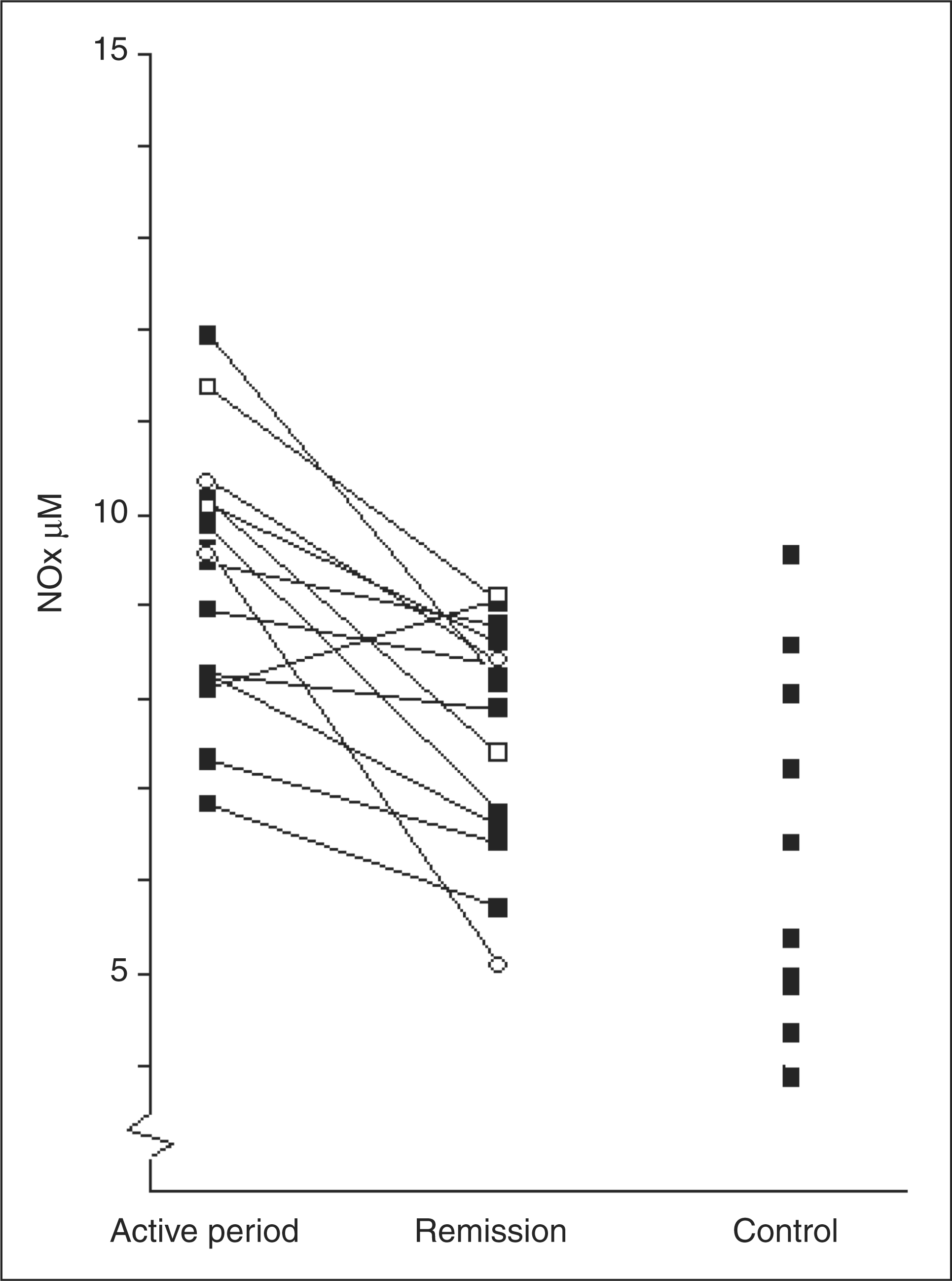

Two of the patients had a CH attack which started as the LP was performed. Their NOx levels were 9.6 µM and 10.3 µM. Two other CH patients had their last attacks 24 hours before the LP, and their NOx levels were 11.3 µM and 10.1 µM. All four patients showed deceased NOx levels in remission. Five patients had a nocturnal CH attack the night before the LP. The mean NOx levels were similar in this group compared with others (9.0 µM vs. 9.7 µM, NS).
Clinically routine laboratory analyses of CSF showed normal levels with regard to protein content and inflammatory parameters in eight of 14 patients (i.e. 57%). A minor albumin increase in CSF was seen in five patients, for two of them both in the active phase and in remission. Two patients showed a modest albumin increase only in the active phase and one patient only in remission. A slight pleocytosis was seen in one patient in both phases of the disease. One patient showed solitary oligoclonal bands in CSF and plasma in remission. Patients with abnormalities in CSF parameters had slightly higher mean NOx levels than other patients (10.0 vs. 8.8 µM), although this difference was not statistically significant. None of the patients with changes in routine laboratory analyses presented any clinical symptoms of concomitant disease.
Smoking habits, other primary headache disorders and results from routine laboratory analyses of cerebrospinal fluid in episodic cluster headache patients

CSF: cerebrospinal fluid, S: smoker, NS: nonsmoker, TTH: tension-type headache, A: active period, R: remission.
Finally, it was explored whether heterogeneity was present between the NO oxidation products, nitrite and nitrate, during the active period and in remission. Nitrite and nitrate levels were compared to the total NOx levels (Figure 3) and found to be of similar distribution.
Mean levels of nitrite, nitrate, and nitritic oxide products (NOx) during the active period and in remission among episodic cluster headache patients and controls.
Discussion
CH is a severe form of headache with profound impact on quality of life. Exploration of its pathophysiology may open the way for new treatments. The role of NO in CH has been increasingly discussed during recent years. D’Amico et al. demonstrated enhanced nitrite levels in plasma among CH patients, both in the active period and in remission (12). The authors suggested that CH patients may have a basal hyperfunction of the L-arginine-NO pathway in both phases of CH. The opposite conclusion was reached by Costa et al., (15) who did not find any difference in plasma nitrate levels in episodic CH and control patients after administration of the NO donor glyceryltrinitrate (GTN). Our findings of significantly enhanced NOx levels in the CSF of CH patients, during both the active period and remission, are in agreement with the findings by D’Amico et al. and add further support to the role of NO in the pathophysiology of CH. The difference between our results and those of Costa et al. may be explained by differences in methodology. Furthermore, we analysed CSF rather than plasma and studied only spontaneous attacks.
Our observation that NOx levels were of the same magnitude regardless of whether the CH attack occurred close to the time of LP speaks against a direct attack-promoting effect of NO.
In a genome scan study using affymetric technique, up-regulation of several pro-inflammatory proteins and cytokines has been proposed (3). Increment in the cytokine IL-2 has also been found during active CH (4). The level of NO production has been shown to correlate with disease activity in several inflammatory disorders, such as cystitis (22), multiple sclerosis (23) and inflammatory bowel disease (24). In our study, the NOx levels were higher during the active period compared to remission and in all CH patients compared with controls. This may suggest that the severity of the condition correlates with inflammatory activity. We suggest that NO participates in CH pathophysiology not only by regulating the vascular tone, but also by participating in an inflammatory vascular response. Furthermore, the severity of the condition correlates with NO levels and thus with the degree of inflammatory activity. According to the model of perivascular neurogenic inflammation, activation of the trigeminovascular system is a crucial step in the development of CH attacks (12). We suggest that the inflammatory activity, and the related level of NO in the brain, must pass a certain threshold level before the trigeminovascular system is activated, which would explain why CH patients develop attacks during the active period and not in remission. One study (25) examined the effect of treatment with 5-isosorbidemononitrite (5-ISMN) 30 mg three times daily for four weeks with the intention of inducing nitrate tolerance in five chronic CH patients. The authors concluded that no effect was seen on the number or intensity of CH attacks, although tolerance was developed for nitrates’ effect on vascular tone. Their observations indicate that the vasodilatation is to be regarded as an epiphenomenon. We believe that the involvement of nitrates may be different with respect to vascular tone and with inflammatory mediated responses. The absence of effect on CH attack frequency by exogenous 5-ISMN could possibly also be a result of NO levels and inflammatory activity already being above the sensitizing threshold, and thus could be seen as a ceiling effect.
Among our patients 50% (or 60% if the four episodic CH patients who discontinued their participation due to development of postpunctional headache are included) suffered from postpunctional headache during the active period, whereas 21% reacted with postpunctional headache in remission. In the literature, the frequency of postpunctional headache varies substantially, from 8.8% to 33% (26,27). The suggested mechanism is leakage through the puncture site, resulting in lowering of CSF pressure (26). Because it is unlikely that CH patients have a higher risk of leakage, this would suggest that they are more pain sensitive and more easily develop postpunctional headache, at least during the active period. However, there are limited data on the pathophysiology of postpunctional headache in general and in CH patients in particular. Our material is small and our observation needs confirmation in larger series.
A slight pleocytosis was found in CSF in one patient in both phases of the disease, and one other patient showed solitary oligoclonal bands in CSF and plasma in remission. We consider these as occasional findings, without likely clinical relevance, because none of the patients had clinical indications of concomitant disease.
A minor albumin increase in CSF was observed among 36% of the CH patients: 28% during the active period and 21% in remission. CSF is continuously formed in the choroid plexus of the intracerebral ventricles. Both active secretion by the choroid epithelium and filtration of plasma across the endothelial wall of the choroid capillary constitute the ventricular CSF (28). The CSF flow rate is the main modulator of blood-derived protein concentrations in CSF (29). The observed increase of albumin in our study may reflect a reduction of CSF flow, or could be caused by increased permeability of the blood-brain barrier (BBB) induced by NO (30). Further studies on the role of the BBB in CH patients are needed to elucidate these suggested explanations.
To our knowledge, this is the first study on NO oxidation products analysed in the CSF of episodic CH patients. The following methodological weaknesses should be considered when interpreting the results of our study:
The small number of patients and control subjects. The control group was as a mean nine years younger and the proportion of male subjects was lower, 38% versus 71%, reflecting the difficulty of achieving a perfect match for patients and controls in a study which includes an invasive diagnostic procedure. There is limited evidence on the effect of age and gender differences on CSF NO levels, and available data from measurements in serum may be confounded by variation in cardiovascular risk factors (31–32), which did not exist among our patients. Each patient also served as its own control when comparing NOx levels during the active period and remission. All patients except one presented decreased NOx levels in remission. The results may also be influenced by changes in smoking and dietary habits not reported by our patients. Nocturnal variations in NOx levels may also have influenced our results. We tried to avoid this problem by performing all LPs between 8 and 10 a.m. However, the fragmented sleeping pattern during the active CH period may have influenced the outcome.
Conclusion
In this first study estimating NO in CSF in episodic CH patients by analysing the end-oxidation products nitrite and nitrate we have found increased NOx levels in CH patients in general and a difference between CH patients in the active period and in remission. However, we have not observed a direct attack-promoting response of NO. Our findings are in agreement with the hypothesis that CH patients might be sensitized for CH attacks by a mechanism related to high NO levels. When the NO level is raised above a certain threshold level, this related mechanism activates the trigeminovascular system. The possibility that NO levels are related to inflammatory activity, varying with CH disease intensity, requires confirmation in future studies.
Footnotes
Acknowledgements
We are grateful to the patients and control subjects who agree to participate in the study. We also wish to thank Mrs Nasrin Bavand for helping us with the analyses using capillary electrophoresis. The study was supported by unconditional grants from GlaxoSmithKline, AstraZeneca and the Tornspiran Foundation. Financial support was provided through the regional agreement on medical training and clinical research (ALF) between the Stockholm County Council and the Karolinska Institute, project number 560664.
