Abstract
The aims of the present study were to investigate whether induction of nitrate tolerance is a useful treatment in cluster headache and to correlate any changes in attack frequency of cluster headache and nitrate‐induced headache to the vascular adaptation during continuous nitrate administration. The results were compared to results obtained from studies of nitrate tolerance in healthy subjects. Materials and methods: 5‐isosorbide‐mononitrate (5‐ISMN) 30 mg was administered orally three times daily for 4 weeks in nine sufferers of chronic cluster headache in a double‐blind, randomized placebo‐controlled cross‐over design. Blood velocity in the middle cerebral artery was measured with transcranial Doppler and the diameters of the temporal and radial arteries were measured with high frequency ultrasound. The haemodynamic data were compared to changes in the frequency of cluster headache attacks and interval headaches over time. Results: Tolerance was complete within 24 h in the middle cerebral arteries and after 7 days in the symptomatic temporal artery, while tolerance of the radial artery was not observed within this period. The time profiles of tolerance were almost identical to the time profiles observed in healthy subjects. A close temporal association between the disappearance of nitrate‐induced headache and tolerance of the temporal artery was observed but tolerance had no effect on cluster headache attack frequency. Conclusions: Induction of tolerance to nitrates cannot be used to treat cluster headache. If pain is related to arterial dilatation the results point to extracerebral rather than cerebral arteries as the site of nociception. However, other peripheral and central pain‐modulating effects of nitric oxide, the time courses of which are unknown, should also be taken into consideration.
Histamine and glyceryl trinitrate (GTN) (nitroglycerin) possess a well‐known ability to provoke attacks of cluster headache, which are clinically identical to spontaneous attacks (1, 2). Histamine may act via activation of endothelial nitric oxide synthase (NOS) and endogenous formation of nitric oxide (NO) (3, 4). The organic nitrates, GTN and 5‐isosorbide mononitrate (5‐ISMN), also exert their effect through conversion to NO (5). Both spontaneous and drug‐induced attacks may thus be caused by NO (6).
Due to nitrate tolerance, continuous administration of nitrates in cardiac patients leads to an attenuation of both the cardiac vasodilatory effect and of the induced headache, which is a prominent adverse effect of nitrate therapy (7, 8). The purpose of the present investigation was to study whether nitrate tolerance may represent a new method of treating cluster headache and to describe the appearance of nitrate tolerance in sufferers of chronic cluster headache in order to elucidate the basic mechanisms of NO‐induced cluster headache. We compared the vascular responses of the middle cerebral, temporal and radial arteries of cluster headache patients to those of healthy subjects. The time profile of tolerance to nitrate‐induced headache in cluster headache patients was also compared to that of healthy subjects, and third, any improvement in cluster headache attacks and nitrate‐induced headache was compared to the adaptation of intra‐ and extracranial arteries.
Methods
Study population and design
Nine subjects, seven males and two females, suffering from chronic cluster headache for at least the last 2 years, participated in the study. Characteristics of the participants are presented in Table 1. The subjects were recruited by advertisement in the journal of the Danish Horton Society (patient organization), from the practice of neurologists and from our out‐patient clinic. The diagnosis was based on the criteria of the International Headache Society (IHS) (9) and compared with a diary, recorded by each patient for 1 month prior to inclusion. Excluded were subjects with a history of migraine, abusers of alcohol, morphine or tranquillizers, and subjects on daily medication other than contraceptives. Cluster headache prophylactic medication was discontinued for a wash‐out period of at least 1 month before inclusion. As abortive therapies cannot be withheld when pain is excruciating (10), concomitant acute therapy (sumatriptan, oxygen) was permitted, but after such treatment the haemodynamic measurements were postponed for at least 3 hours. Weak analgesics (paracetamol, acetylsalicylic acid), tea, coffee, alcohol or tobacco were not allowed 8 hours prior to the haemodynamic measurements.
Characteristics of participants

Withdrawals (within 24 h from start of the 5‐ISMN period), excluded from statistical analysis. CH, cluster headache.
The study followed a double blind, randomized, placebo‐controlled crossover design. A 5‐ISMN dose of 30 mg was chosen because a previously performed dose–response study in healthy subjects showed that this dose induced significant changes in the radial and temporal arterial diameters and an acceptable intensity of headache but no major drops in blood pressure or other severe adverse effects (11). 5‐ISMN 30 mg or placebo was administered orally three times a day during two separate periods of 4 weeks, each period being separated from the other by at least 1 month of wash‐out. We deliberately used t.i.d. administration in order to achieve maximal development of tolerance, whereas in cardiological use, 5‐ISMN is administered once daily in order to avoid cardiovascular tolerance. The study followed the Helsinki II declaration and was approved by the National Board of Health and by the Ethics Committee of the County of Copenhagen. All patients gave their informed consent and were free to withdraw at any time and to receive treatment if needed.
Vascular measurements and headache recordings
The time‐averaged mean of the maximal blood velocities (V mean) in both middle cerebral arteries (MCA) was measured with transcranial Doppler (Transscan, EME). V mean is an indirect measurement of change in the diameter of MCA provided that regional cerebral blood flow is unchanged, which is so after GTN (12, 13). The fix‐point for measurements of V mean was a point along the MCA, which was free of the bifurcation between the MCA and the anterior cerebral artery, but as close to it as possible (14). This fix‐point was then used throughout the study in each individual, and every measurement was done after carefully optimizing the signal from this point. The diameters of the left radial artery and both temporal arteries were measured with a high resolution ultrasound scanning (Dermascan C, Cortex Technology). A mean of four measurements performed within 30 s was used to augment precision. An individual fix‐point of each subject along the right frontal branch of the superficial temporal artery and along the radial artery of the left wrist was used throughout the study. Day‐to‐day positions of measurements were reproduced by recording the scanning position in relation to the angle and distance relative to the orbitomeatal line (temporal artery) and to the distal volar crest of the wrist (radial artery) (15, 16). Blood pressure and heart rate were measured with an automatic inflatable arm cuff (Tonoprint).
Cluster attack frequency was scored on a daily basis; each attack was carefully described regarding time of occurrence, duration, severity, location, associated autonomic symptoms, along with the use of abortive therapies. Each patient assessed the severity of their pain using a graded scale from 0 to 10 (1 representing a pre‐pain, including a feeling of pressure, 5 a moderate pain, and 10 the worst possible pain).
Interval headache (including 5‐ISMN‐induced headache) was scored on a similar rating scale from to 0–10 (17). Headache characteristics (pain location, quality), its aggravation by physical activity and the presence of accompanying symptoms (nausea/vomiting, phono‐/photophobia) were simultaneously recorded.
Procedure
The subjects arrived in the laboratory at 09.00. After 30 min of rest in the supine position, baseline values of V mean, diameters of the temporal and radial arteries, blood pressure and heart rate were recorded, followed by an oral administration of 30 mg 5‐ISMN or placebo. Afterwards the subjects were discharged and asked to return to the laboratory for repeated measurements – preceded by 30 min of supine rest – the next day, and after 3 and 7 days at 11.00 (i.e. at time of peak plasma concentration, 4 hours after a morning dose at 07.00 (11)). 5‐ISMN or placebo was administered for 4 weeks by the subjects themselves at home at 07.00, 15.00 and 22.00. A prerequisite for the haemodynamic measurements was that the patients had not had an attack (with or without the use of abortive therapy) in close temporal relation to these. In case it did happen, the measurements were consequently postponed for at least 3 hours. During each period a questionnaire concerning cluster headache attacks and a questionnaire concerning interval headache (at 07.00, 15.00, 18.00 and 22.00) and the use of acute therapy was filled out every day at home. If the severity of cluster headache attacks or interval headache became unacceptable, sumatriptan/oxygen and paracetamol/acetyl‐salicylic acid, respectively, were permitted and offered to each subject for later use. At each visit, compliance was tested by capsule count and review of diary recordings.
Statistical analysis
The haemodynamic data
The arterial responses were calculated in percentage of baseline and expressed as mean and standard error of the mean (±
The headache data
Cluster headache attacks were evaluated as frequency of attacks and number of treated attacks, as duration and severity of cluster attacks are considered unreliable parameters if acute treatment is permitted and effective (10).
Interval headache scores were evaluated as maximal headache scores and statistically evaluated and described using median values. The curves, however, are presented using mean values in order to illustrate more clearly the changes over time.
Differences in summed cluster attack frequency, summed treated cluster attacks and headache score (total summed maximal headache score) between the 5‐ISMN period and placebo period were analysed using Wilcoxon test for paired‐data. Differences in cluster attack frequency (sum/week), treated cluster attacks (sum/week) and interval headache response over time (sum of maximal headache scores/week) during 5‐ISMN administration and placebo were analysed separately using a non‐parametric two‐way analysis of variance, Friedman's test. Significant changes from baseline were located by a multi‐comparison procedure (Dunn's range test) (19).
Statgrafics 3.0 was used throughout for statistical analysis. For all analyses, a P‐value < 0.05 was considered statistically significant.
Results
Four of nine patients withdrew within 24 h from start of the 5‐ISMN period because of adverse effects: hypotension (one subject), severe interval headache with a median of 5 (three subjects). Data from these subjects were excluded from statistical analysis.
Haemodynamic values at each investigative time‐point are presented in Table 2 (absolute values). No systematic side‐to‐side differences of pre‐treatment values between symptomatic (SS) and non‐symptomatic (NSS) side of the middle cerebral arteries or temporal arteries were found (P > 0.05). None of the baseline values of any of the parameters differed significantly between the two study periods (P > 0.05).
Haemodynamics (mean (absolute values),


MCA, middle cerebral artery; TA, temporal artery; RA, radial artery; V mean, blood velocity; SS, symptomatic side; NS, non‐symptomatic side; HR, heart rate; BP, sys, systolic blood pressure; BP, dia, diastolic blood pressure;
The middle cerebral arteries
5‐ISMN did not change the velocity (V mean) compared to placebo (P = 0.25 (SS); P = 0.77 (NSS)), neither were changes over time observed when each period was analysed separately (5‐ISMN: P = 0.29 (SS), P = 0.59 (NSS); placebo: P = 0.82 (SS), P = 0.22 (NSS)). During 5‐ISMN, however, a trend towards a decrease in V mean at 24 h was observed on both sides (91 ± 6% of baseline (SS), 93 ± 7% of baseline (NSS)) but it did not reach statistical significance (P > 0.05) (Fig. 1).

The middle cerebral artery. Changes of blood velocity responses of the middle cerebral artery during treatment with 5‐ISMN 30 mg × 3 or placebo for 7 days (mean (% of baseline)). No significant changes over time were observed. Complete tolerance may, however, have developed within 24 h of the first dose. •=symptomatic side/5‐ISMN; ▪=non‐symptomatic side/5‐ISMN; ○=symptomatic side/placebo; □=non‐symptomatic side/placebo.
The temporal arteries
5‐ISMN significantly dilated the artery on the symptomatic side compared to placebo (P = 0.002), whereas no difference between periods was observed on the non‐symptomatic side (P = 0.87).
Symptomatic side
At 24 h after administration of 5‐ISMN the artery was dilated to 118 ± 6% of baseline (P < 0.05). After 4 days an insignificant attenuation of the dilatory effect was observed compared to peak value at 24 h (P > 0.05, range test) and the artery was still significantly dilated to 114 ± 3% of baseline (P < 0.05). After 8 days the diameter was not significantly different from baseline values. No significant changes over time were observed during placebo (P = 0.39).
Non‐symptomatic side
No significant changes over time were observed during 5‐ISMN (P = 0.32); however, numerically the artery was dilated at all time‐points during this period to 116 ± 5, 117 ± 10, 113 ± 9% of baseline after 1, 4 and 8 days, respectively. During placebo the artery was dilated to 125 ± 7% of baseline at 24 h (P < 0.05). This effect was no longer significant after 4 days or later (P > 0.05) (Fig. 2).

The temporal artery. Changes of the diameter of the temporal artery during treatment with 5‐ISMN 30 mg × 3 or placebo for 7 days (mean (% of baseline)). Tolerance was complete after 7 days on the symptomatic side while changes over time were not significant on the non‐symptomatic side. •=symptomatic side/5‐ISMN; ▪=non‐symptomatic side/5‐ISMN; ○=symptomatic side/placebo; □=non‐symptomatic side/placebo.
The radial artery
5‐ISMN changed the response compared to placebo (P = 0.0001). At 24 h the artery was dilated to 123 ± 4% of baseline (P < 0.05). No further significant changes in diameter were observed after 4 and 8 days compared to peak value at 24 h (P > 0.05, range test) and the artery was still significantly dilated compared to baseline (P < 0.05). No significant changes over time were observed during placebo (P = 0.07) (Fig. 3).

The radial artery. Changes of the diameter of the radial artery during treatment with 5‐ISMN 30 mg × 3 or placebo for 7 days (mean (% of baseline)). Tolerance had not developed after 7 days. •=5‐ISMN; ○=placebo.
Heart rate and blood pressure (BP)
Changes in heart rate (P = 0.0009) and diastolic BP (P = 0.02) were observed during 5‐ISMN compared to placebo, while no significant difference between periods was detected in systolic BP (P = 0.48). No significant changes over time were observed in any of the parameters during placebo (P = 0.14, heart rate; P = 0.60, systolic BP; P = 0.19, diastolic BP) or in heart rate (P = 0.15) and in systolic BP (P = 0.24) during 5‐ISMN. Diastolic BP, however, had decreased to 90 ± 2% of baseline (P < 0.05) at 24 h during 5‐ISMN. This effect was no longer significant after 4 days or later (P > 0.05).
Cluster headache
The clinical features of cluster attacks (including the presence of autonomic symptoms) during 5‐ISMN were identical to those during placebo and to those outside study periods. No significant differences in attack frequency (P = 0.50, Wilcoxon) or number of treated attacks (P = 0.35, Wilcoxon) were observed during 5‐ISMN compared to placebo. Thus, mean frequency of attacks and number of treated attacks during 5‐ISMN was 56 (range 19–118) and 47 (mean, range 2–118), respectively, compared to 48 (range 30–71) and 37 (mean, range 11–73), respectively, during placebo. Significant changes over time were neither observed in attack frequency (summed attacks/week) nor in number of treated attacks (summed treated attacks/week) during either period (Table 3, Fig. 4).
Changes over time of summed cluster attacks, treated attacks and maximal interval headache score
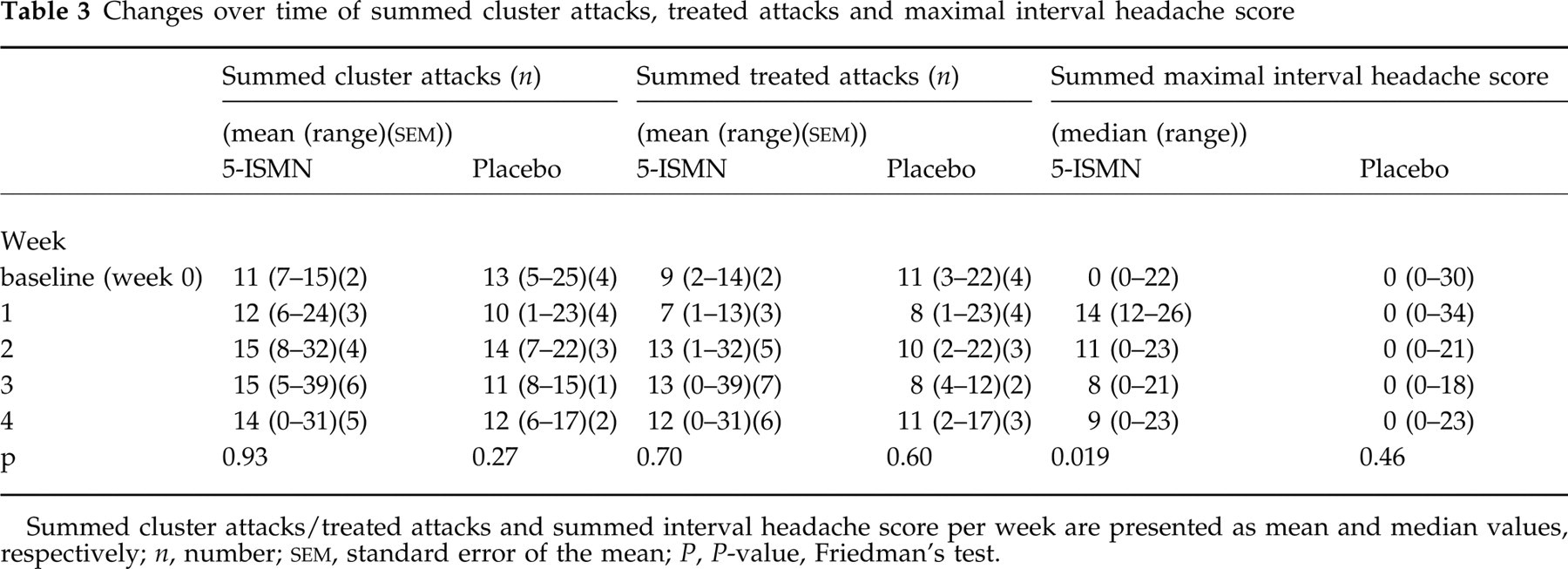
Summed cluster attacks/treated attacks and summed interval headache score per week are presented as mean and median values, respectively; n, number;

Cluster attack frequency and number of treated attacks. Changes in attack frequency and use of acute therapy during treatment with 5‐ISMN 30 mg x 3 or placebo for 28 days (mean). The use of acute therapy is an indirect estimation of severity and duration of attacks, both of which are unreliable parameters when acute therapy is permitted. No differences between periods in attack frequency and use of abortive therapy were observed. •=cluster attacks (n)/5‐ISMN; ▪=treated attacks (n)/5‐ISMN; ○=cluster attacks (n)/placebo; □=treated attacks (n)/placebo.
Interval headache
All subjects described the nitrate‐induced headache as a pressing bilateral pain of frontal location. The patients were able to differentiate the experienced headache from the in‐between attacks — slight discomfort, described by some patients as an ache/light pressure behind the eye on the symptomatic side. No accompanying symptoms were observed nor was the headache aggravated by routine physical activity. No significant difference in total headache score was observed during 5‐ISMN compared to placebo (P = 0.36, Wilcoxon). Thus, median summed maximal headache score was 42 (range 12–93) during 5‐ISMN compared to 0 (range 0–80) during placebo (a median of 0 was observed during placebo as three patients experienced no headache during this period). Headache (sum of maximal headache score/week) changed significantly over time during 5‐ISMN, while no significant changes over time were observed during placebo (Table 3). The patients thus experienced more headache during the first week of 5‐ISMN administration compared to the following 3 weeks. Significant changes from baseline could, however, not be located (P > 0.05, range test). Headache (maximal headache score/day) changed significantly over time during the first week of 5‐ISMN administration (P = 0.037, Friedman)(Fig. 5). Thus, headache gradually attenuated from a median of 4 (range 2–4) on the first day to a median of 2 (range 1–5) on the fourth day. On the eighth day headache had attenuated to a median of 1 (range 0–3). Significant changes from baseline could, however, not be located (P > 0.05, range test). Three subjects, of whom two usually suffered from interval headache, experienced a continued headache during the rest of the 5‐ISMN period, with a median daily headache score alternating between 0 and 2.

Interval headache. Changes in interval headache (maximal headache score) during treatment with 5‐ISMN 30 mg × 3 or placebo for 28 days (mean). Headache was most severe during the first days of 5‐ISMN therapy; thereafter headache gradually attenuated and tolerance to headache had developed at the end of the first week of therapy. Mean values are presented in order to illustrate more clearly the changes over time. Median values are, however, used for statistical analyses and description. •=5‐ISMN; ○=placebo.
Discussion
Background
Fifty years ago Bayard Horton advanced the hypothesis that histamine played a major role in the pathophysiological mechanisms of cluster headache and proposed histamine desensitization as the treatment of choice (20). Diamond subsequently used this method of treatment for those chronic cluster patients who were unresponsive to standard therapies (21). Recently it has been shown that histamine causes headache via its stimulation of endogenous NO production (22). The refractory time is the period following both spontaneous and GTN‐provoked attacks (2, 23), during which GTN cannot provoke an attack, and repeated GTN exposures seem to lengthen the refractory period (23). During continuous use of nitrates, tolerance occurs both to their vascular effects and to the induction of headache (7, 8). The present study was undertaken in order to determine the effect of nitrate tolerance on cluster headache. We intended to analyse whether induction of nitrate tolerance could be used as a novel therapeutic principle in patients with severe chronic cluster headache and to consolidate knowledge about the mechanisms underlying the attacks by comparing the development of tolerance in blood vessels to headache.
Study limitations
The apparent limitation of the present study is the small sample size, which only allows large differences between treatments to be detected. Several factors limited the recruitment of patients to the present study. Chronic cluster headache is a very rare condition (24) and out of the possible candidates many were reluctant to give up prophylactic treatment. Many of the included patients could not tolerate the GTN‐induced headache, which furthermore was aggravated in those patients who had to travel long distances four times per week for laboratory measurements. Finally, the unavoidable use of attack therapy was a confounding factor. Despite these limitations a number of important conclusions can, however, be made and parallels drawn to our previous studies of nitrate tolerance in healthy subjects (8, 25)
Nitrate tolerance is not a useful treatment
We hypothesized that the patients during the initial phase of 5‐ISMN administration would experience side‐effects such as interval headache and more cluster headache attacks but that the condition would normalize and eventually improve compared to the pre‐treatment period in parallel with the development of nitrate tolerance. Indeed, interval headache was most severe during the first 2 days of 5‐ISMN administration, followed by a gradual attenuation, until a state of tolerance to headache had developed at the end of the first week. The response was not significant but – estimated from area under the headache curve (Fig. 5) – roughly identical to the response observed in healthy subjects in whom tolerance to headache developed after 5 days (8). Neither was total summed interval headache significantly higher during 5‐ISMN compared to placebo. Likely explanations for the lack of significance of these parameters are the small sample size, which was further reduced by the withdrawal of three subjects due to severe interval headache, and the fact that the study population – due to limitation of subject availability – unfortunately had to comprise two patients who also suffered from tension‐type headache.
Following GTN a headache similar to the patient's spontaneous cluster headache usually develops after a latency period of 30–50 min (2) but, presumably due to differences in pharmacokinetics, none of the patients in the present study experienced an attack in close temporal relation to the first dose of 5‐ISMN. Alternatively, the patients were still benefiting from the effect of sumatriptan used during nocturnal attacks or were in a refractory period at the time of 5‐ISMN administration. It has been hypothesized that the refractory period is caused by a transient insensitivity of receptors for some substances involved in the attacks (26). This mechanism may have provided protection against an accumulation of attacks during the initial phase of 5‐ISMN administration, which, however, was not succeeded by a reduction in attack frequency.
The vascular adaptation to 5‐ISMN
In a previous study in healthy subjects, nitrate tolerance was complete within 24 h in the middle cerebral artery while superficial temporal and radial artery tolerance was only partial and developed more slowly. Thus, an attenuation of the vasodilatory effect compared to peak values was observed after 7 days but the arteries were still dilated compared to baseline (25). The present study showed in large part the same in patients with chronic cluster headache but not all parameters were significant, probably due to a small sample size, large variance of individual responses, and infrequent measurements. For example, the response of the middle cerebral artery did not change significantly over time during 5‐ISMN, probably due to the fact that no measurements were done at the time of maximal dilatation. The lack of response at 24 h probably reflected that tolerance had already developed.
It is obvious from the sum of results that cluster headache patients respond to nitrates in a fashion roughly similar to normal individuals. Thus, both the direct response to NO and the development of tolerance was normal.
Nitrate tolerance in different sections of the vascular bed, its mechanisms and relevance to nitrate‐induced headache
The underlying mechanisms responsible for nitrate tolerance probably are multifactorial and may include neurohormonal counter‐regulatory mechanisms (27, 28), intravascular volume expansion (29), or properties intrinsic to the vasculature. Such intrinsic abnormalities may include desensitization (down‐regulation) of the target enzyme guanylyl cyclase (30), decreased biotransformation of organic nitrates to NO (31), enhanced vascular superoxide anion production, which degrades NO (32), and enhanced propensity for vasoconstriction secondary to increased endothelin expression within the vascular smooth muscle (33).
The time course of tolerance appearance varies widely in different sections of the vascular bed (34). This may account for the heterogenous response observed in the present study. In previous studies the response to GTN was more pronounced in the temporal artery than in the radial artery (35) and more prolonged in intracranial than in extracranial arteries (36). Furthermore, venous tissue possesses a lower basal release of NO and an increased sensitivity to nitrovasodilators compared to the arterial side (30). Assuming that tolerance follows reduced metabolic conversion of nitrates to NO in combination with down‐regulation of guanylate cyclase, tolerance may initially be detected in the vascular segments with the lowest basal NO production/highest sensitivity to NO, i.e. the veins (30, 37). Interestingly, the rate of tolerance appearance in veins is identical to the rate we observed in intracranial arteries, which furthermore appeared earlier compared to the rates observed in the temporal and radial arteries. Thus, based on the direct response to NO as well as the speed of tolerance development, cerebral arteries are particularly sensitive to NO. Tolerance to nitrate‐induced headache developed in close temporal association to tolerance of the temporal artery.
Pathogenesis of spontaneous and provoked attacks of cluster headache
Spontaneous and GTN‐induced cluster headache attacks are associated with dilatation of intra‐ and extracranial arteries. Dilatation of the affected temporal artery has been demonstrated by several studies, using different examination techniques, including clinical observation of swelling (20), photoplethysmografi (38) and high frequency ultrasound (39). Dahl et al. (12), using transcranial Doppler, demonstrated that spontaneous and GTN‐provoked attacks were accompanied by a bilateral decrease in middle cerebral artery flow velocities that was more pronounced on the symptomatic side. Interestingly, provoked attacks appeared to begin when the vasodilatory effect of GTN was receding, implying that vasodilatation alone is not the only cause of pain (12) or perhaps merely an initiating event. This line of thought may explain why the induction of vascular tolerance turned out to be unsuccessful in the treatment of cluster headache.
NO is, however, capable of generating pain in several ways other than vasodilatation. NO is a noxious molecule which may sensitize perivascular sensory nerve endings via the formation of free radicals or via the initiation of perivascular neurogenic inflammation (40). Increased levels of calcitonin gene‐related peptide (CGRP) have been observed at the peak of GTN‐provoked attacks (41), probably due to NO‐induced release from sensory nerve terminals (41–43). Finally, via interaction with NMDA‐receptors in the central nervous system, NO may give rise to hyperalgesia (40). The possibility exists that these mechanisms of which the time course of tolerance appearance is unknown, may play a more important role compared to vasodilatation in relation to pain in cluster headache.
