Abstract
Prevention of arterial oxygen desaturation during anaesthesia with high-flow nasal oxygen (HFNO) has gained greater acceptance for a widening range of procedures. However, during HFNO use there remains the potential for development of significant anaesthesia-associated apnoea or hypoventilation and the possibility of hypercarbia, with harmful cardiovascular or neurological sequelae. The aim of this study was to determine whether any HFNO-related hypercarbia adverse incidents had been reported on webAIRS, an online database of adverse anaesthesia-related incidents. Two relevant reports were identified of complications due to marked hypercarbia during HFNO use to maintain oxygenation. In both reports, HFNO and total intravenous anaesthesia were used during endoscopic procedures through the upper airway. In both, the extent of hypoventilation went undetected during HFNO use. An ensuing cardiac arrest was reported in one report, ascribed to acute hypercarbia-induced exacerbation of the patient’s pre-existing pulmonary hypertension. In the other report, hypercarbia led to a prolonged duration of decreased level of consciousness post procedure, requiring ventilatory support. During the search, an additional 11 reports of postoperative hypercarbia-associated sedation were identified, unrelated to HFNO. In these additional reports an extended duration of severe acute hypercarbia led to sedation or loss of consciousness, consistent with the known effects of hypercarbia on consciousness. These 13 reports highlight the potential dangers of unrecognised and untreated hypercarbia, even if adequate oxygenation is maintained.
Introduction
A ventilatory effect termed THRIVE (Transnasal Humidified Rapid-Insufflation Ventilatory Exchange) was described in 2015 and suggested to arise largely from the use of high-flow nasal oxygen (HFNO). 1 This was reported to slow the progression of hypercarbia, which has limited the duration of anaesthesia-induced apnoea. 1 Subsequent studies have failed to demonstrate a moderation in hypercarbic rise with HFNO use during apnoea,2,3 and a finding of oxygen desaturation prevention only in low-risk patients when compared with low-flow nasal oxygen (LFNO). 4
HFNO has been extensively reported for use during gastroenterological endoscopy 4 and bronchoscopy. 5 In these instances, the primary aim of HFNO is to sustain oxygenation while simultaneously allowing ease of proceduralist access through the upper airway with a limitation of ventilatory movements. 6 A systematic review published in 2022 of HFNO use in gastroenterological endoscopy found reduced rates of hypoxaemic events (SpO2 < 92%) and need for simple airway intervention of jaw thrust or increase in oxygen flow compared with LFNO, but only in patients at low-risk of hypoxaemia. 4 Patients at high risk of hypoxaemia, such as the morbidly obese, obtain no additional benefit from HFNO.4,5,7
Prevention of desaturation during procedural sedation by increasing the fraction of inspired O2 (FiO2) is a logical approach. Desaturation events are nonetheless reported to be very common. For example, during gastroenterological procedures, episodes of SpO2 < 92% for greater than 15 s are reported in up to 21% of patients receiving HFNO and 33% receiving LFNO. 8 Increasing the FiO2 brings into play two issues related to hypoventilation detection or complications. The first being that only modest increases in FiO2 can readily maintain SpO2 > 90% in the presence of substantiative declines in alveolar ventilation. 9 This means that when relying on SpO2, the presence of an emerging obstruction or medication-induced hypoventilation might not be reflected by a pre-warning fall in SpO2. A progression to complete obstruction of the airway or drug-induced apnoea, in the absence of oxygen delivery to the alveolus, might instead present as a precipitous fall in SpO2. 10 The second relates to the direct relationship between alveolar PCO2 and PO2 (PACO2, PAO2), wherein as PACO2 increases, the PAO2 falls.9,11 This mathematical relationship can be estimated using the alveolar gas equation, which pertains to a perfect theoretical circumstance at equilibrium without contribution from venous admixture (shunt).12,13 This allows for the estimated lowering in PAO2 to be calculated when this results not from insufficient oxygen delivery to the alveolus to meet demands, but rather from a rise in the PACO2. For example, when breathing room air, an increase in the PaCO2 to 72 mmHg will reduce the SpO2 to about 90% (arterial oxygen partial pressure, PaO2 about 60 mmHg). However, with only a modest increase in FiO2 to 0.3, as obtainable with LFNO, 10 the alveolar gas equation calculated theoretical estimate for a breach below SpO2 90% now climbs to a potential causative PaCO2 of 120 mmHg.
Given the above, it is possible procedures conducted with oxygenation maintained by HFNO in the presence of prolonged apnoea or hypoventilation associated with anaesthesia could present as unintended or unrecognised cases of severe hypercarbia. The purpose of this study was to interrogate the webAIRS database for reports of adverse clinical incidents occurring due to hypercarbia when HFNO is utilised during procedural anaesthesia. A secondary aim was to identify other incidents, unrelated to HFNO use, in which hypercarbia had resulted in sedation or unconsciousness in the postoperative period.
Methods
WebAIRS is a voluntary internet-based anaesthesia incident database established in 2009 for anaesthetists in Australia and New Zealand to assist in identifying patient safety issues and guide safer practice. The ethics approval process for the webAIRS data collection methodology has previously been published. 14 It is briefly repeated here: webAIRS data collection complies with the current ethics requirements for the collection of de-identified quality assurance data in Australia, as outlined by the National Health and Medical Research Council (NHMRC). 15 In order to ensure data collection meets the NHMRC requirements ethics approval was been sought and obtained from two hospitals in Australia: Royal Brisbane and Women’s Hospital Human Research Ethics Committee (HREC/11/QRBW/311) and Nepean Blue Mountains Local Health District (HREC/12/NEPEAN/18). Ethics approval in New Zealand was obtained from the Health and Disability Ethics Committee, (MEC/09/17/EXP). In addition, reporters at each site had to comply with local institutional approval requirements. The reports are confidential and protected by qualified privilege in both Australia and New Zealand.
A search of the webAIRS database was conducted by the authors (GP, MC, YE) in December 2022. The word search criteria included ‘THRIVE’, ‘HFNO’, ‘Optiflow’, ‘HFNP’, ‘hyperca’ (to match hypercarbia, hypercarbic, hypercapnia and hypercapnic), ‘high CO2’, or ‘acido’ (to match acidotic or acidosis). The narrative for each report was then assessed by the searching authors. Reports for which there was agreement by all the searching authors of a likely association were included for the purpose of the study.
Results
The webAIRS database yielded 493 reports using the word search criteria. After a review of the report narratives by the researchers and agreement of a likely association, two reports of HFNO use during anaesthesia with the development of hypercarbia and an adverse incident were identified. The details of these reports are summarised in Table 1.
Anaesthesia and high-flow nasal oxygen associated incidents.
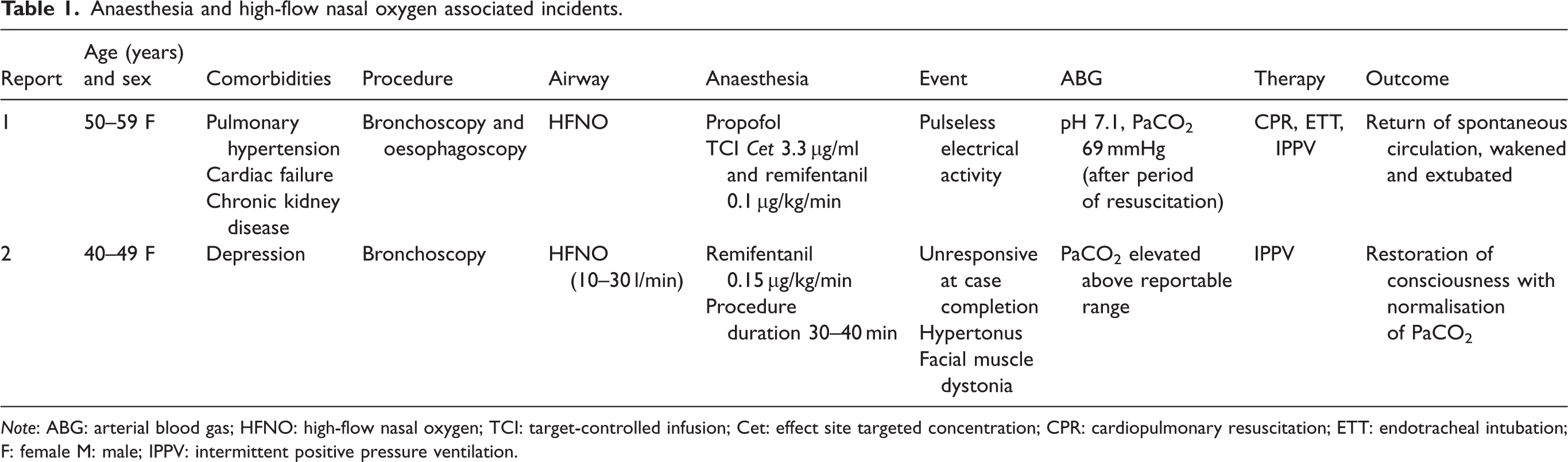
Note: ABG: arterial blood gas; HFNO: high-flow nasal oxygen; TCI: target-controlled infusion; Cet: effect site targeted concentration; CPR: cardiopulmonary resuscitation; ETT: endotracheal intubation; F: female M: male; IPPV: intermittent positive pressure ventilation.
Case 1 was attributed by the reporter to hypercarbia leading to an exacerbation of pulmonary hypertension and cardiac arrest. The patient, in her fifties with multiple comorbidities, was undergoing bronchoscopy and oesophagoscopy. Preoperative echocardiogram demonstrated impaired right ventricular function and right ventricular systolic pressure (RVSP) of 52 mmHg (normal < 37 mmHg 16 ). Intravenous infusion of propofol to effect a site target concentration of 3.3 µg/ml and remifentanil at 0.05 µg/kg/min were administered to achieve deep sedation. SpO2 was maintained at 99% with HFNO (FiO2 and flow rate not detailed). During the procedure, remifentanil was increased to 0.1 µg/kg/min. Shortly thereafter the systolic blood pressure, as measured non-invasively, fell to 50 mmHg and rapidly progressed to pulseless electrical activity. A successful resuscitation was achieved with immediate endotracheal intubation, positive pressure ventilation, external chest compressions and intravenous adrenaline of 900 µg. The end-tidal CO2 (EtCO2) once intubation was achieved was 74 mmHg. A short time later a PaCO2 of 69 mmHg was recorded while the EtCO2 was in the ‘low 50 s’. The reporter noted ‘hence PaCO2 would have been close to 90s’, and ‘Hypoventilation was masked by the high flow nasal O2’. The procedure was abandoned.
Case 2 was of a decreased level of consciousness identified by the reporter as a complication of hypoventilation-induced hypercarbia. The patient, a female in her forties underwent an investigative bronchoscopy. Airway topicalisation with lignocaine and intravenous remifentanil infusion 0.1 µg/kg/min was provided for the procedure. Oxygenation was supported with THRIVE at 10–30 L/min. During the procedure an increase in remifentanil to 0.15 µg/kg/min was required. The report goes on to state, ‘Breathing efforts were noted to be slow. Given THRIVE was used, there was no capnography monitoring, but pulse oximetry confirmed adequate saturations throughout (99–100%)’. Procedure duration was 30–40 min and was followed by failure to wake 20 min after cessation of the remifentanil. The PaCO2 at this time was above the reportable upper limits for the arterial blood gas machine. The management plan was for tracheal intubation. However, during preparation for intubation the patient showed signs of response to pain and voice. Following this, ‘Respirations were supported by PSV (pressure support ventilation) via facemask and CO2 was 70 mmHg on a subsequent blood gas’. The patient continued to regain full responsiveness completely over the next few minutes.
There were 11 additional reports identified of decreased level of consciousness postoperatively, during which the patients were confirmed to have hypercarbia. These are summarised in Table 2. Ten of these patients had had successful removal of a formal airway after surgery followed by a decreased level of consciousness in the post anaesthesia care unit. Hypercarbia was detected during assessment or resuscitative intervention. Administration of naloxone was reported in seven patients, with none resulting in an observable response. In the majority, hypercarbia was managed with intubation, an extended period of positive pressure ventilation, and transfer to an intensive care unit. The reports contained statements of a clinical suspicion of hypercarbia-induced sedation, which included ‘good response to overnight ventilation’, ‘hypoventilation and subsequent hypercarbia causing patient to be obtunded’, ‘further hour of assisted ventilation until she awoke’, ‘on arrival in ICU she was starting to wake as CO2 decreased’, and ‘on low FiO2 until less drowsy and CO2 normalised’.
Postoperative hypercarbia-associated sedation incidents.
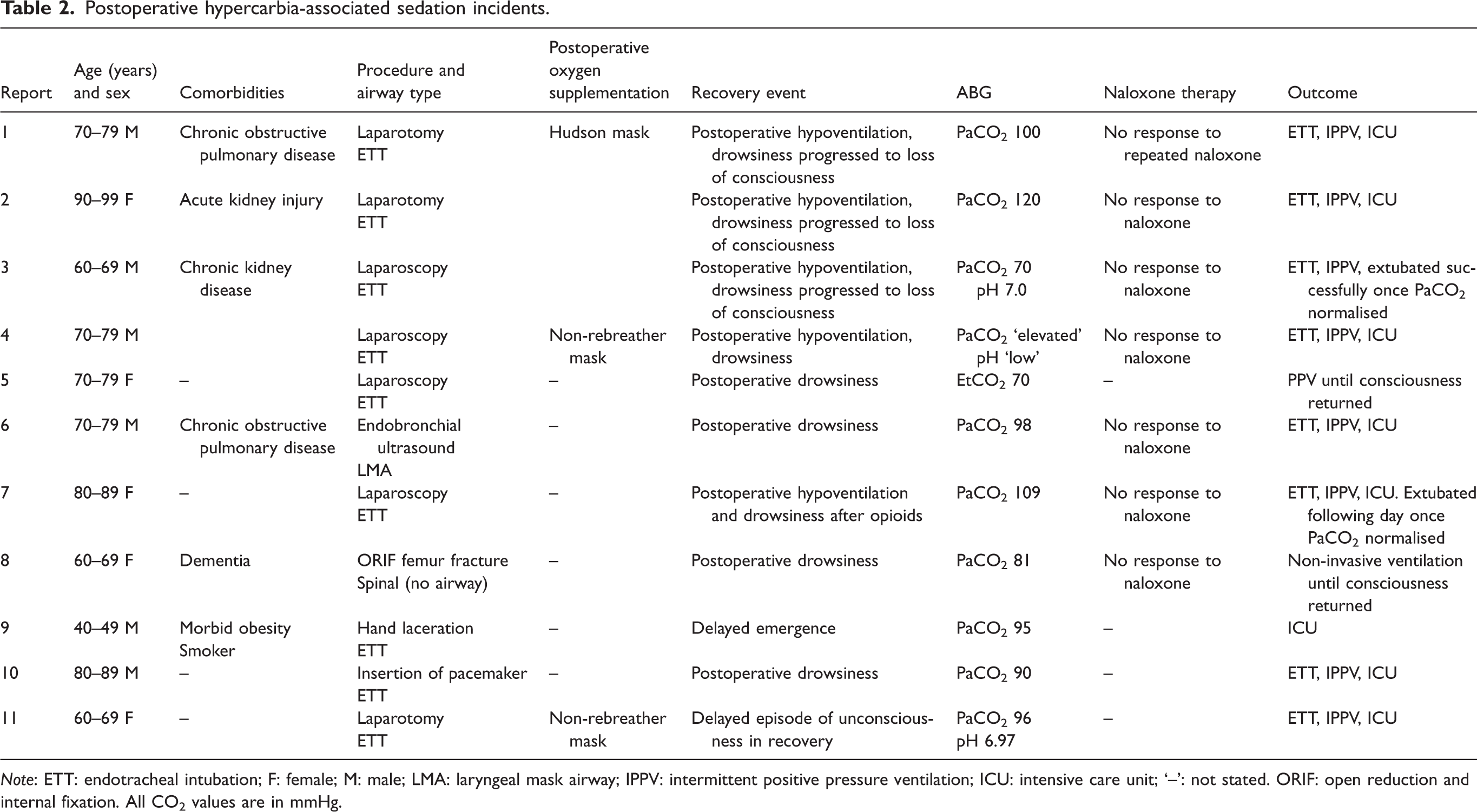
Note: ETT: endotracheal intubation; F: female; M: male; LMA: laryngeal mask airway; IPPV: intermittent positive pressure ventilation; ICU: intensive care unit; ‘–’: not stated. ORIF: open reduction and internal fixation. All CO2 values are in mmHg.
Discussion
The two anaesthesia incidents during HFNO use are a timely reminder to consider the potential for unrecognised hypercarbia and its sequelae during HFNO maintenance of oxygenation in anaesthetised or sedated patients. While we cannot determine the rate of adverse incidents using HFNO by interrogating a voluntary de-identified database, it is possible similar but perhaps less severe cases went unrecognised, or other cases of similar severity went unreported. In any event, the severity of the two incidents is sufficient to alert anaesthetists to the potential for hypercarbia during HFNO when it is associated with hypoventilation or apnoea. The incidents should prompt anaesthetists to consider alternative techniques depending on a patient’s premorbid condition or if the duration of the procedure may be such that the possibility of severe hypercarbia might go undetected.
Given the potential risks of unrecognised severe hypercarbia, it is opportune to review the physiology of apnoeic oxygenation, and the literature in support of its application. High FiO2 has long been known to prolong oxygenation in apnoea. 17 This results from a denitrogenating effect enhancing oxygen supply and a drawing effect of the mass movement of oxygen toward the alveoli as O2 is taken up by pulmonary blood in greater volumes than CO2 is released into the alveoli.17,18 The practice of apnoeic oxygenation was described in 1959 when a cuffed endotracheal tube with an open circle circuit flushed with 100% O2 at 8 L/min maintained SaO2 100% for 55 min of apnoea. 18 Reports of severe hypercarbia and fatal acidosis during apnoeic oxygenation tempered ongoing enthusiasm for its use during anaesthesia.1,17
The THRIVE study purported the hypercarbia of apnoeic oxygenation could be moderated with HFNO. 1 A median apnoea of 14 min without desaturation below SpO2 90% was reported in 25 patients with the use of humidified HFNO (Optiflow™ 70 l/min, FiO2 1.0). The rate of CO2 rise when measured using EtCO2 was 1.13 mmHg/min. This contrasted markedly with historical control values in apnoeic oxygenation of 2.6–3.4 mmHg/min – measured mostly as arterial partial pressure CO2 (PaCO2).18–21 An ‘insufflating’ effect of HFNO was proposed and the term THRIVE introduced. In a follow-on study the authors then concluded HFNO uniquely created vortices above the glottis which combined with cardiac oscillations to enable more effective elimination of CO2 from the alveoli. 22
In 2017 Gustafsson et al. aimed to recreate the conditions of the THRIVE study. 6 Similar to the THRIVE study, they found a median EtCO2 rise of 0.9 mmHg/min and no desaturations below SpO2 90% after 23 min of apnoea. However, when measured in arterial blood, the rise in PaCO2 at 1.8 mmHg/min was closer to historical values. The mean peak PaCO2 rise was 77 mmHg and pH nadir 7.2.
Randomised trials have since investigated the benefit of HFNO over alternatives for the rate of CO2 rise. Two were conducted in a paediatric population. Compared with no supplemental oxygen, HFNO did not alter the rate rise of transcutaneous CO2 but did expectedly double the apnoeic time. 23 When HFNO was compared with LFNO, neither a difference in the rate of rise in transcutaneous CO2 nor in apnoea time was seen. 3
A large randomised controlled trial in adults reported in 2022 further challenged the preferential use of HFNO to either maintain oxygenation or prevent hypercarbia. 2 This compared 125 patients with nasal oxygen flow rates of 2, 10 or 70 L/min over 15 min of apnoea. Importantly, two control groups were included: one with an endotracheal tube delivering 0.25 l/min FiO2 1.0, and the other HFNO at 70 l/min while airway patency was maintained with a laryngoscope. No significant difference in the rate of rise of PaCO2 was found between groups, and median values were 2.0–2.1 mmHg/min. Five percent of study participants desaturated below SpO2 92% with no significant difference between groups. The findings did not support the concept of HFNO-induced ventilation leading to additional CO2 elimination. 24 The authors concluded CO2 elimination was dependent on cardiac oscillations and not on nasal oxygen flow rate.
The cardiovascular effects of hypercarbia are predominantly mediated by sympathetic stimulation. 25 In the pulmonary vasculature, rises in PACO2 markedly increase pulmonary vascular resistance (PVR) and may be a greater determinant of pulmonary blood flow than oxygen. 26 Echocardiographic studies in healthy volunteers calculated the increase in RVSP as 0.6–1.0 mmHg for every 1 mmHg increase in PACO2 and being greater incrementally than the increase in RVSP seen with a fall in PAO2. 26 In a patient with pre-existing pulmonary hypertension, a rise in PVR resulting from hypercarbia can lead to rapid onset of acute right ventricular decompensation and cardiac arrest. 27
Elevations in PaCO2 can present neurologically as facial twitching, irritability, or gross myoclonic limb movements similar to those in Report 2.28,29 A sedative (narcotising) effect of hypercarbia has long been espoused. 10 A decreased level of consciousness only eventuates if hypercarbia results in (respiratory) acidosis. 30 This explains why patients with chronic obstructive pulmonary disease have been recorded with severely elevated PaCO2 of up to 175 mmHg, but as a result of chronically compensated pH changes maintain full consciousness.31–34
In 1958, Westlake reported on respiratory failure patients and concluded, ‘in general the critical level for loss of consciousness in carbon dioxide intoxication is at a pH of 7.1–7.14 and PCO2 of 120–130 mm(Hg)’. 35 Others have stated that abnormalities of mental state appear above PaCO2 90 mmHg or when pH falls below 7.25. 36 Experimental research has concluded that the resultant cerebrospinal fluid (CSF) pH is a greater determinate of CO2 sedation than arterial pH, 30 and the maximal change in CSF pH as PaCO2 alters can be delayed by up to 30 min. 37 These findings together may have implications in prolonged exposure to hypercarbia where, despite normalisation of PaCO2 and arterial pH with artificial hyperventilation, there may be ongoing sedation owing to the delay in CSF pH normalising.28,34,36 The mechanism by which acidosis leads to sedation has not been fully elucidated.38–40
Thus, it seems prudent to consider benefit and risk when utilising HFNO to support oxygenation during procedural anaesthesia when hypoventilation or apnoea results. The benefit is limiting the frequency in some patients of hypoxaemic events requiring remediation with simple airway manoeuvres. Risk can arise from failing to detect hypoventilation or apnoea and an insidious rise in PaCO2, leading to significant cardiovascular and neurological sequelae. In this circumstance the use of HFNO can lead to a presentation counter to expectation and be viewed as a ‘failure to thrive’.
The 11 incidents involving postoperative hypercarbia (without HFNO) and decreased level of consciousness are reminders of the interdependence of hypoventilation, hypercarbia, and sedation or loss of consciousness. In many of the cases, opioid-induced ventilatory impairment (OIVI) was suspected by the clinicians and naloxone administered. However, in none did naloxone lead to an improvement. This should not discourage the use of naloxone if OIVI is suspected. However, there may be a subset of cases in which CO2 narcosis has progressed so far that naloxone alone may be insufficient. The four cases of loss of consciousness are noteworthy for their extreme PaCO2 (100–120 mmHg) and pH (6.97–7.0) values being largely in the ranges reported by previous authors to be associated with loss of consciousness.35,36 The remaining seven cases of decreased level of consciousness, similarly recorded PaCO2 values (70–109 mmHg) in the range previously reported for such an occurrence, or else were potentially capable of inducing the pH values of concern.
In summary, we present 13 incidents, two involving HFNO and 11 involving postoperative narcosis, related to the development of hypercarbia. The two incidents involving HFNO are reminders of the risks of hypercarbia when using this technique, even if adequate oxygenation is maintained. The other 11 are a reminder of the potential severe consequences of postoperative hypercarbia.
Footnotes
Author Contribution(s)
Acknowledgements
The authors would again like to acknowledge the contribution of all members of the Australian and New Zealand Tripartite Anaesthetic Data Committee (ANZTADC), past and present, the administrative assistance provided by the Australian and New Zealand College of Anaesthetists staff, and the data analysis undertaken by Dr Heather Reynolds (ANZTADC). The authors would also like to thank the large number of anaesthetists reporting incidents to webAIRS in the interests of greater patient safety.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Yasmin Endlich is the current coordinator of the webAIRS publications group. Martin Culwick is the medical director of the Australian and New Zealand Tripartite Anaesthetic Data Committee. The other authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
