Abstract
High-fidelity models are required for technical mastery of bronchoscopic procedures in the fields of anaesthesia, intensive care, surgery and respiratory medicine. Our group has created a three-dimensional (3D) airway model prototype to emulate physiological and pathological movement. Developed from the concepts of our previously described 3D printed paediatric trachea for airway management training, this model produces movements created by injection of air or saline through a side Luer Lock port. The anaesthesia and intensive care applications of the model could include bronchoscopic navigation through narrow pathologies and simulated bleeding tumours. It also has the potential to be used to practice placement of a double-lumen tube and broncho-alveolar lavage among other procedures. For surgical training, the model has high tissue realism and allows for rigid bronchoscopy. The novel and high-fidelity 3D printed airway model with dynamic pathologies represents capability to provide both generic and patient-specific advancement for all modes of anatomical representation. The prototype illustrates the potential of combining the fields of industrial design with clinical anaesthesia.
Introduction
Three-dimensional (3D) printing is a burgeoning technology that is being used in a vast range of applications in the field of anaesthesia.1,2 Three-dimensional printing has been used to aid preoperative airway evaluation and planning in patients with potentially difficult airways.3,4
To enable rapid and safe acquisition of flexible and rigid bronchoscopic proficiency in the fields of anaesthesia, intensive care, respiratory medicine, otolaryngology and thoracic surgery, proceduralists require high-fidelity models with tactile feedback to optimise their learning experience. Fibreoptic intubation training is usually taught using manikins with normal anatomy, non-anatomical models to improve dexterity, or anaesthetised patients commonly having normal anatomy. 5 Virtual reality trainers are available with pathological anatomy. However, the proceduralists do not receive tactile feedback. 6
We have previously published the use of a 3D printed paediatric trachea for use in airway management training. 7 By further refining our existing technology, we have produced a 3D printed adult trachea incorporating the ability to emulate dynamic pathologies. Adaptation of this has the potential to simulate normal cardiorespiratory movement within the airways. We report the application of pneumatic and fluid mechanics in creating dynamic movements within a 3D model, thereby enhancing visual realism and creating tactile feedback in the bronchoscopic environment.
Our 3D printed multi-property simulative airway model: Conception and development
The prototype was developed with Victoria University School of Design (Wellington, New Zealand) utilising open-source computed tomography from an anonymous adult patient. The base mesh was generated in 3D Slicer using segmentation tools from the ‘Chest Imaging Platform’ software extension followed by native tools in the ‘Segment Editor’. ZBrush (Maxon, Bad Homburg, Germany) was used to clean up and enhance the resolution of the mesh. Pathological features were modelled in Zbrush and Houdini (Side Effects Software Inc, Toronto, Canada), whereas Rhinoceros 3D (Robert McNeel and Associates, Seattle, WA, USA) was used to design fixtures for the dynamic parts and stents to help remove support material. The meshes were repaired in Netfabb (Autodesk, San Rafael, CA, USA) and then exported as 3D printable files.
The current model is printed using a high-resolution polymer on the Stratasys J850 3D printer (Rehovot, Israel). The printing materials digitally mix, allowing colour gradient and heterogeneous material qualities. This confers varying degrees of hardness from extremely soft to hard in the appropriate areas of the trachea.
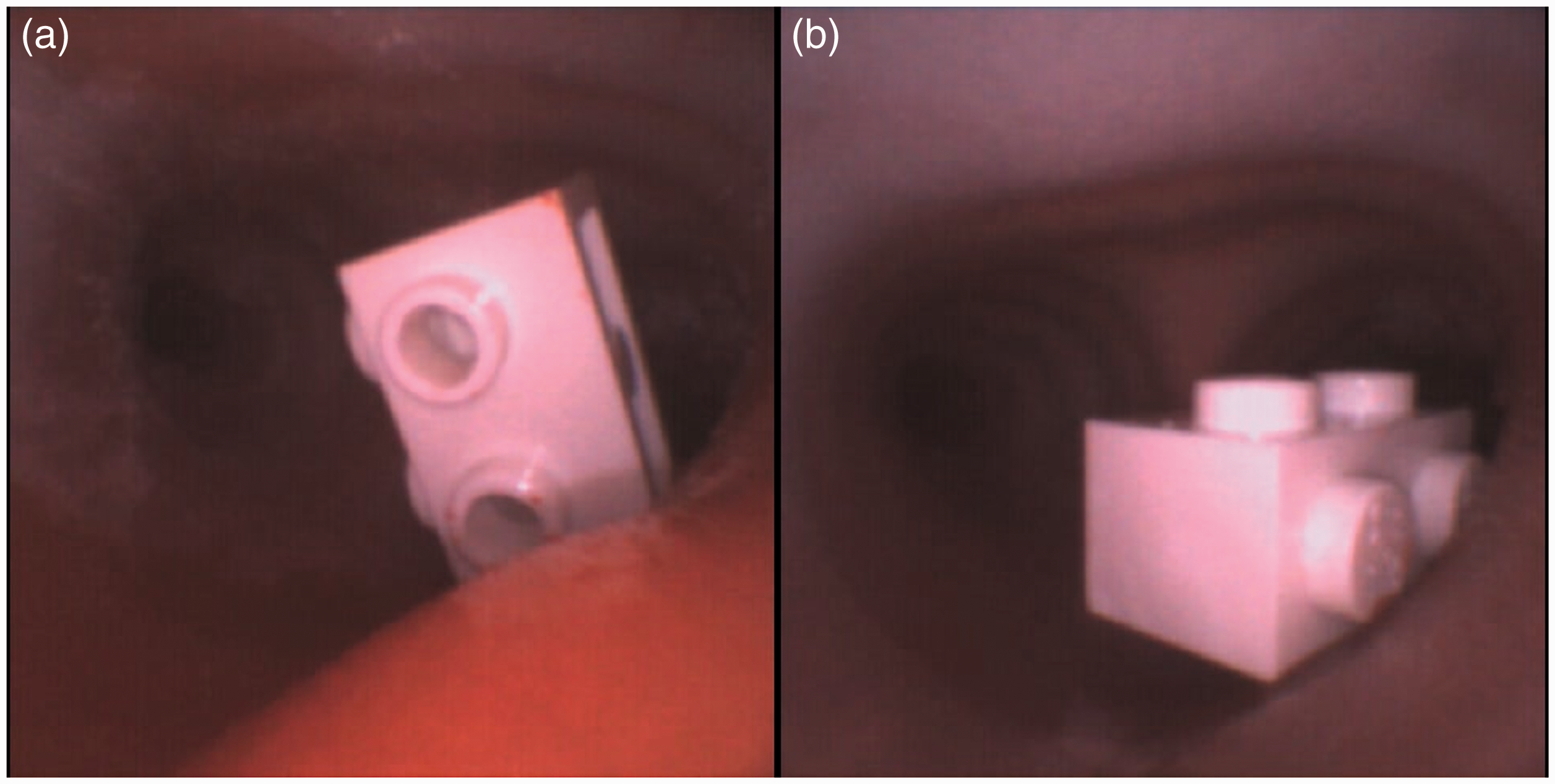
In addition to producing high-fidelity tissue properties, the ability for the model to emulate pathology was the main aim of its conception. By incorporating a channelled system, liquid or air can be injected via a Luer Lock connection port into the 3D tracheal model (Figures 1 and 2).
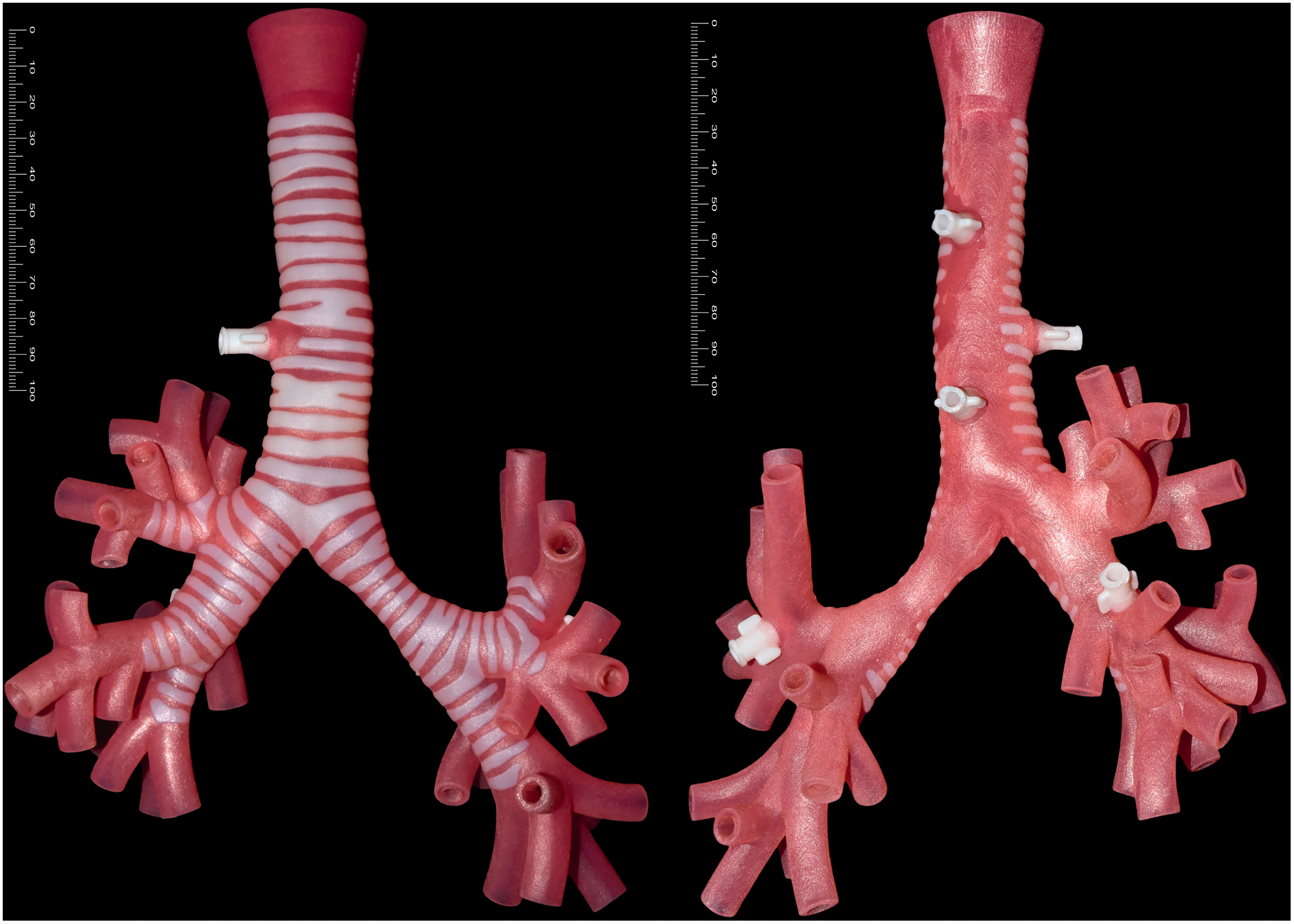
External view: three-dimensional (3D) printed high-fidelity adult trachea created in Wellington, New Zealand (scale reference in millimetres).
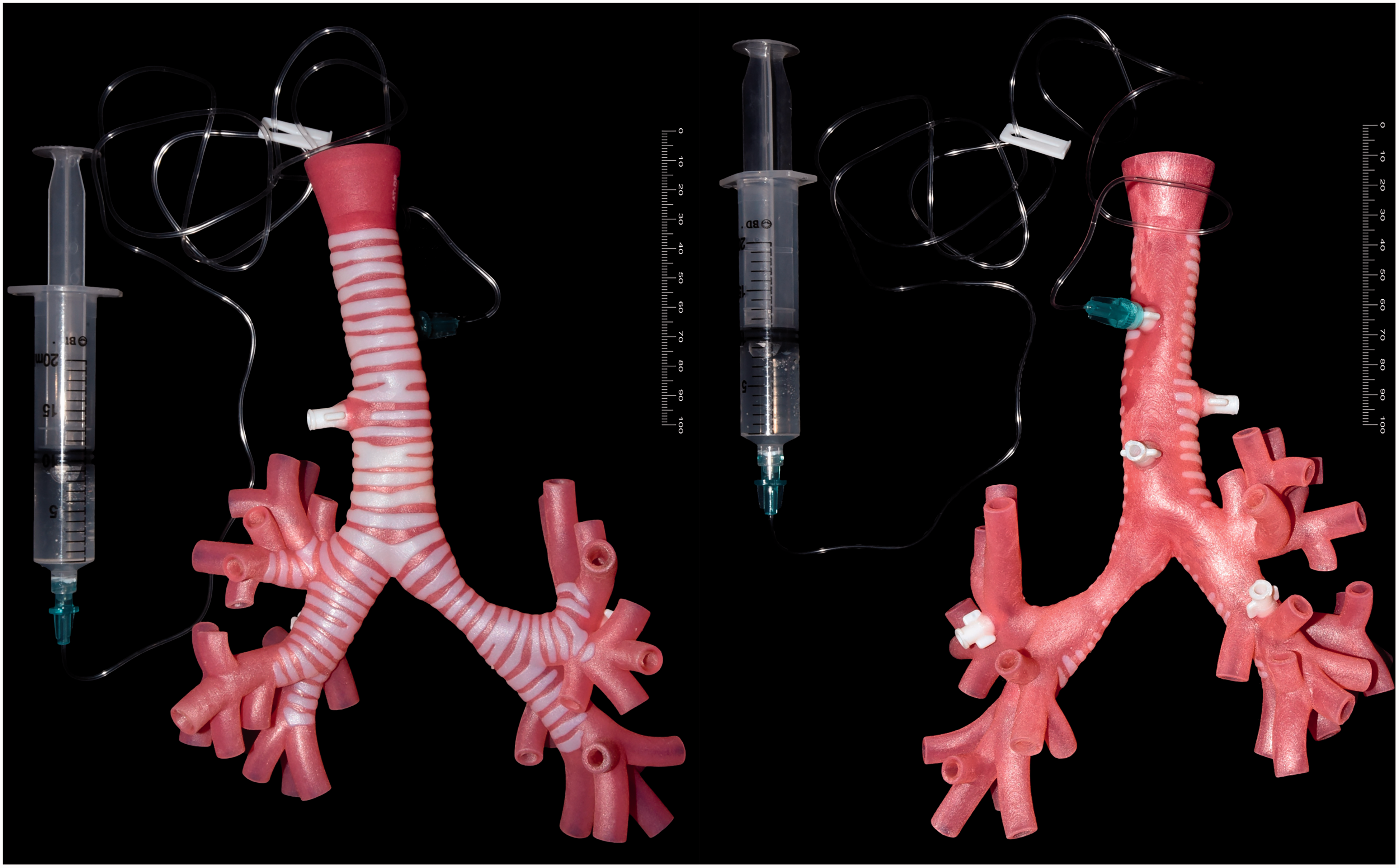
External view: 3D model with integrated Luer Lock side port attachment to syringe with water.
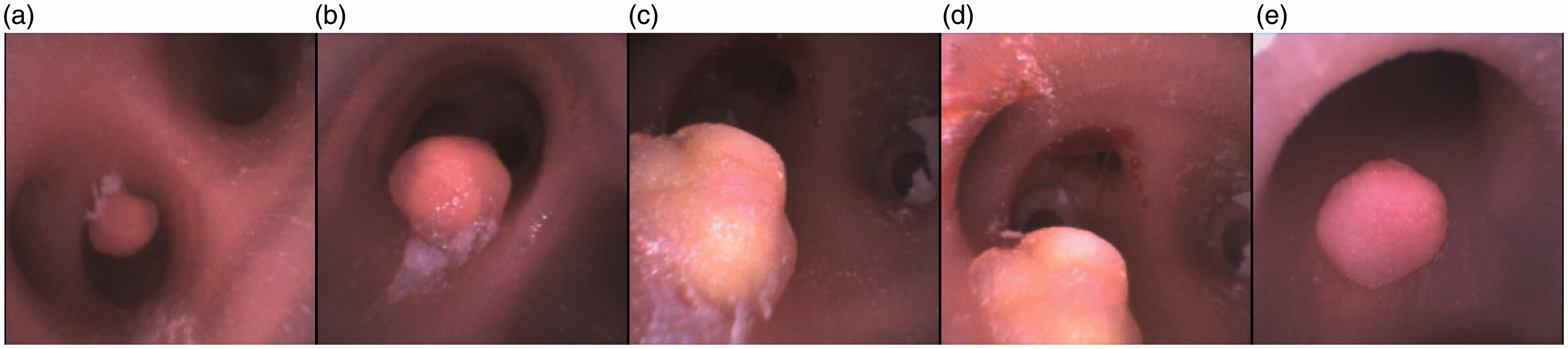
The volume of air or saline injected via the port determines the expansion of the pathology. In our prototype, we were able to emulate dynamic lesions, including cystic lesions, tracheal narrowing, tracheal or bronchial stenosis, haemangioma and adenoid cystic carcinoma. Figures 3 and 4 demonstrate the gradual enlargement of structures under direct and bronchoscopic view with increasing volumes of saline injected via the Luer Lock port.

Enlargement of a tracheal cyst under direct view with (a) 0 ml, (b) 1 ml, (c) 2 ml, (d) 3 ml, and (e) 4 ml of saline injected via the Luer Lock port, and (e) near complete obstruction of the trachea.
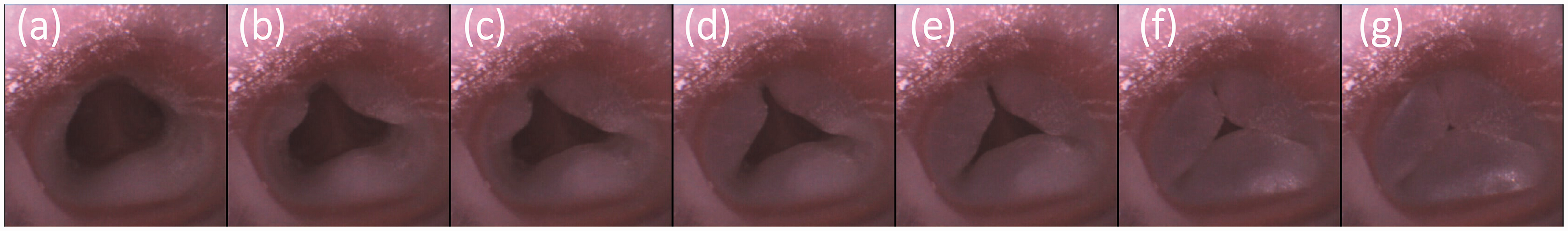
(a)–(g) Increasing severity of tracheal narrowing following gradual injection of saline. Endoscopic view with an Ambu® flexible endoscope (aScope 4 Broncho Slim 3.8/1.2, Ballerup, Denmark).
Applications for clinical simulation
Our 3D model has several simulation applications (Figure 5). In anaesthesia, the model may be used in the training of lung isolation techniques. It may also be utilised to simulate bleeding from the bronchial tree and include pathologies which may make intubation difficult such as tracheal stenosis. Combined ear, nose and throat and anaesthesia shared airway procedures can also be simulated. For rare pathologies, a pre-printed 3D model could be custom-made to aid perioperative planning. The model is also suited for practising broncho-alveolar lavage techniques.
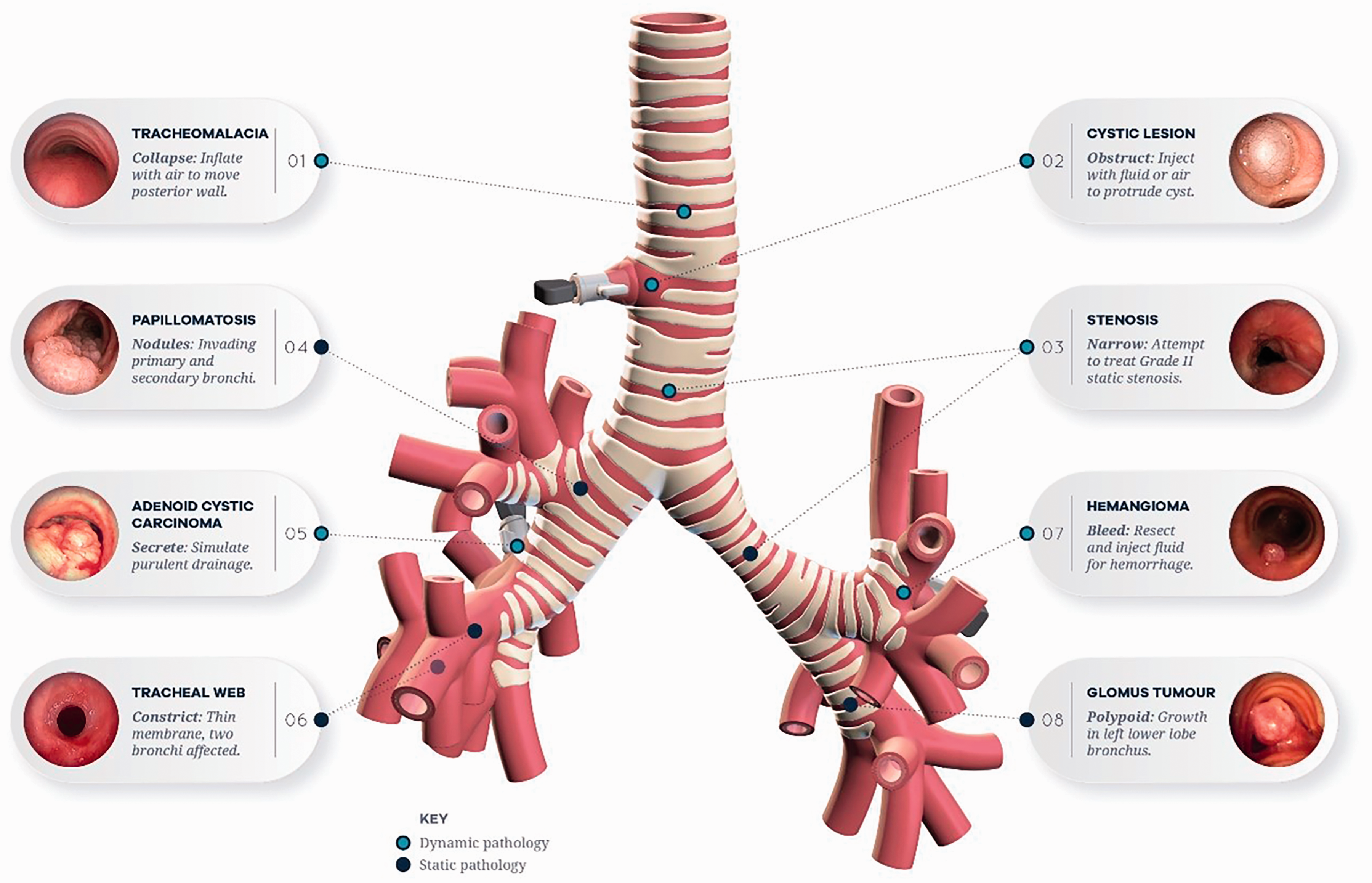
Overview of the features for the 3D printed high-fidelity trachea.
Massive bronchial bleeding simulation: Practise for bronchoscopic visualisation and deployment of double-lumen tubes
Massive bronchial bleeding events are rare and are with high mortality, whereas minor bleeding can significantly obscure the bronchoscopic view, making the procedure difficult to perform. 8 Significant bleeding requires immediate interventions such as placement of a bronchial blocker or double-lumen tube. 9 Our model can be used to emulate the emergency scenario of acute bronchial bleeding. A small perforation is incorporated within the centre of the bronchial tumour. Red-dyed saline is injected via the side port. Blood pulsations are simulated by intermittent injection of the syringe in synchronised cadence with the heart rate. The size of the perforation made to the cystic lesion determines the rate of bleeding and thus its difficulty in bronchoscopic view and management (Figure 6).
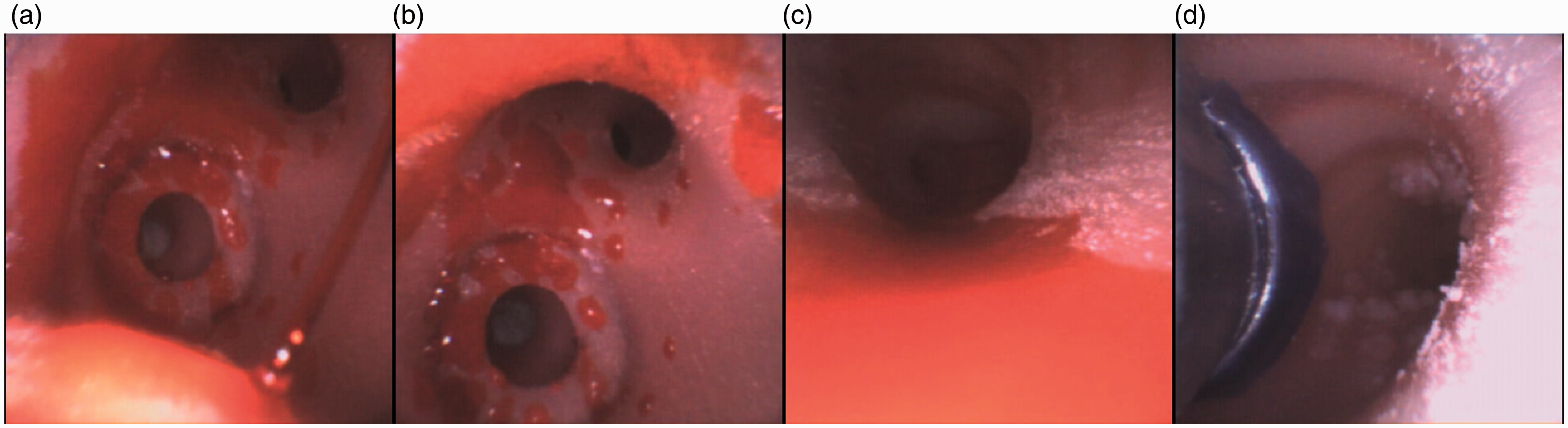
(a)–(c) Bronchoscopic view with an Ambu® flexible endoscope (aScope 4 Broncho Slim 3.8/1.2, Ballerup, Denmark) of acute bleeding from the emulated cystic lesion in the 3D model and (d) Placement of a double-lumen tube above the level of bronchial ‘bleeding’.
Broncho-alveolar lavage and surgical training with high-fidelity fixed lesions
Broncho-alveolar lavage involves instillation of sterile saline into a lung segment followed by suction and collection of the fluid for analysis. It plays an important role in the diagnostic evaluation and management of lung diseases. 10 The procedure is usually performed using a flexible bronchoscope. In our simulated scenario, artificial mucus can be injected to a segmental bronchus and then suctioned for sampling.
For surgical training, these models could be used to teach rigid and flexible bronchoscopic techniques, including the removal of foreign bodies, tissue resection and stent deployment.
We have incorporated high-fidelity fixed lesions, including papillomatosis (Figure 7), tracheal stenosis (Figure 8) glomus tumour, adenocystic carcinoma, and haemangioma (Figure 9).
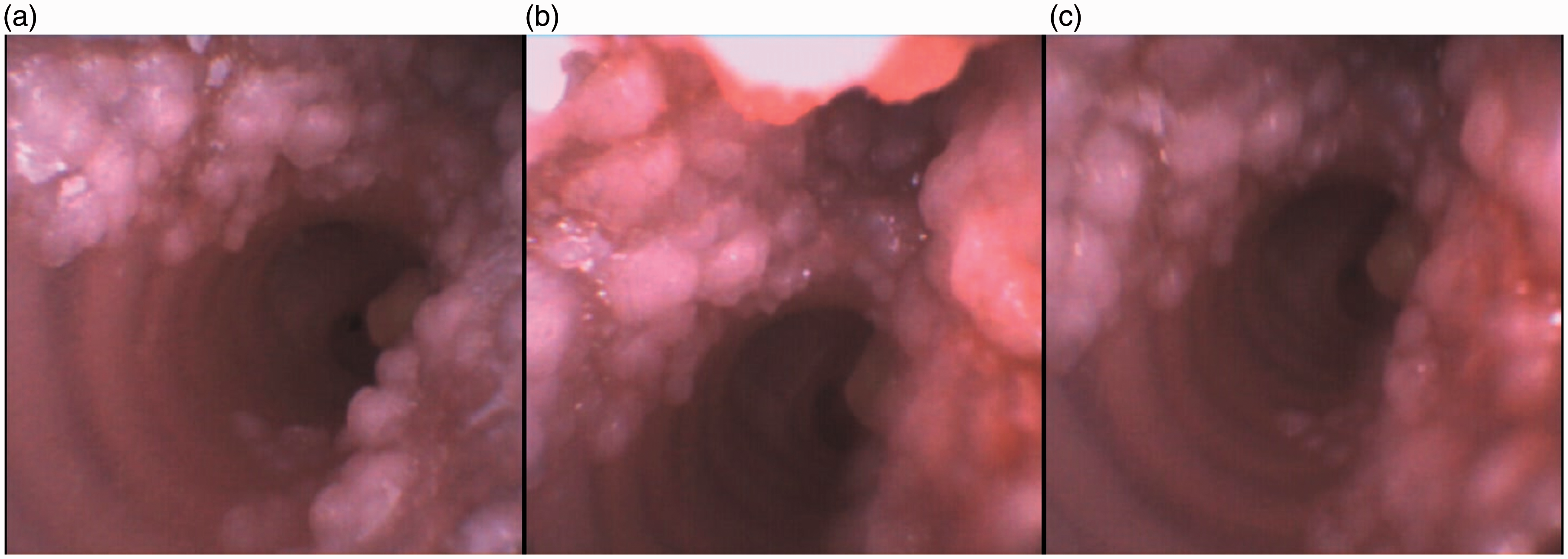
(a)–(c) Views of papillomatosis with bronchoscopic view with an Ambu® flexible endoscope (aScope 4 Broncho Slim 3.8/1.2, Ballerup, Denmark) in the right main bronchus.
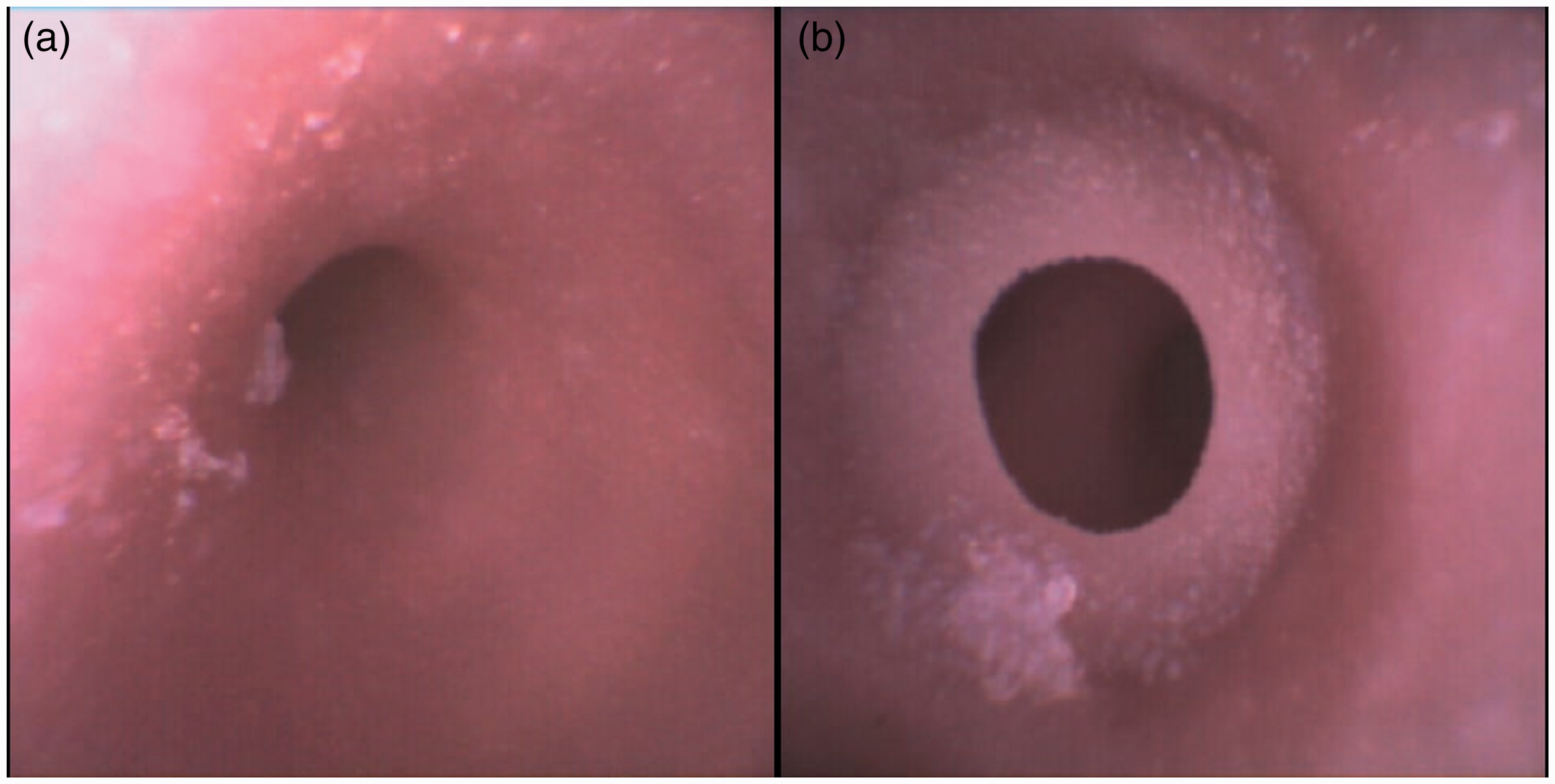
(a) Tracheal stenosis and (b) tracheal web viewed using an Ambu® flexible endoscope (aScope 4 Broncho Slim 3.8/1.2, Ballerup, Denmark).
(a) and (b) Glomus tumour, (c) and (d) adenocystic carcinoma and (e) haemangioma viewed using an Ambu® flexible endoscope (aScope 4 Broncho Slim 3.8/1.2, Ballerup, Denmark).
Foreign bodies can be lodged within the 3D printed model airway. This can be used as a tool to simulate foreign body retrieval (Figure 10). Future adaptation to include respiratory movements would bring a new level of realism into the time critical and potentially difficult simulation scenario.
(a) and (b) Different views of foreign body lodged in the right main bronchus viewed using an Ambu® flexible endoscope (aScope 4 Broncho Slim 3.8/1.2, Ballerup, Denmark).
Limitations and future developments
High-fidelity 3D printing requires a specialised printer and materials associated with a high initial setup cost. Through collaboration (in our case, with the Victoria University School of Design), we foresee the potential of making it an economically viable option for producing high-quality simulation models. The impact of the global pandemic has put a strain on the supply chain in manufacturing and shipping. Therefore, in-house production of teaching models with the benefit of customisation may become the new normal for individual institutions.
Material longevity is another longer-term limiting factor in our 3D model. In order to produce tissue realism, the highly flexible polymer material can fatigue and is at risk of breakage with excessive force.
The dynamic 3D printed airway model is a prototype. Although functional as a simulation tool in its current state, we should emphasise that it is still early in its evolutionary process and is not yet ready for commercial distribution or personal creation. Although we have gained feedback from local specialists, the efficacy of the model as a training aid still requires further validation.
We endeavour to develop and test its application further in the near future by gaining feedback from a greater audience of anaesthetists, intensive care specialists, ear, nose and throat specialists and respiratory medicine specialists through its use in simulation scenarios.
Conclusion
Our prototype model produces dynamic movements of lesions within an anatomical model. This allows high-fidelity simulation and tactile feedback for respiratory, surgical and anaesthetic airway training. The versatility of 3D printing allows for customisation and in-house production of such models. Its development is an illustration of the collaboration between the fields of industrial design and medicine. It showcases the potential end product of combining design expertise in 3D printing of multi-property materials with the application of medicine. We hope this article will be an example to inspire others to seek cross-disciplinary collaboration in furthering scientific research.
Footnotes
Author Contributions
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors received financial support from MedTech CoRE and the Campbell Barrett trust for this project.
