Abstract
Opioids are integral to multimodal analgesic regimens in children with moderate to severe acute pain. Throughout normal childhood there are marked changes in physiology, and social and psychological development that influence the perception and expression of pain, the pharmacology of opioids, and how they are used. A multidimensional pain assessment is key to guiding appropriate opioid prescribing. Most of the commonly used opioids in adults are used in children, with the increasing exception of codeine (as a result of regulatory change), and are generally well tolerated. Patient groups at increased risk of ventilatory impairment include neonates and those with obstructive sleep apnoea, severe neurodevelopmental conditions, trisomy 21, and severe epilepsy. Slow-release opioids are not recommended for general use, but may be used in select populations, for example, following scoliosis surgery, major trauma or burns. Prescribing and administration errors are a major issue in paediatrics generally; the potential consequences of opioid prescribing or administration errors are serious, particularly following hospital discharge. Opioids prescribed at discharge are frequently in excess of a child’s analgesic requirements; three to five days supply appears sufficient for the majority of common paediatric operations. Discharge opioid prescriptions have been linked to long-term opioid use in adolescents with risk factors. Misuse of prescription opioids by adolescents is also concerning, with prevalence estimates ranging from 1.1% to 20%. Caregivers have a tendency to underdose opioids in their children; caregiver education may improve appropriate administration. Caregivers must also be provided with instructions on safe storage and disposal of unused opioids.
Keywords
Introduction
Clinicians caring for children with acute pain bear a great responsibility for ensuring their comfort and safety. Opioids are integral to providing effective acute pain management in children, but are not without short-, and potentially, long-term adverse effects; thus a considered approach is required. A child is defined as a person from birth to 18 years of age. 1 The significant developmental changes occurring in the early years of life, particularly regarding neural pathways and social and psychological functioning which influence the perception and expression of pain, coupled with evolving pharmacokinetic and pharmacodynamic changes can make assessment and management of acute pain and the use of opioids in the paediatric patient challenging. When prescribing opioid analgesia in this context, a number of factors need to be considered. Some of these are the same as in adults, while others are specific to the younger age groups. This narrative review focuses on how opioids are prescribed for acute pain in children in hospital, as well as considerations for discharge opioid prescribing.
General considerations for opioid prescription in children
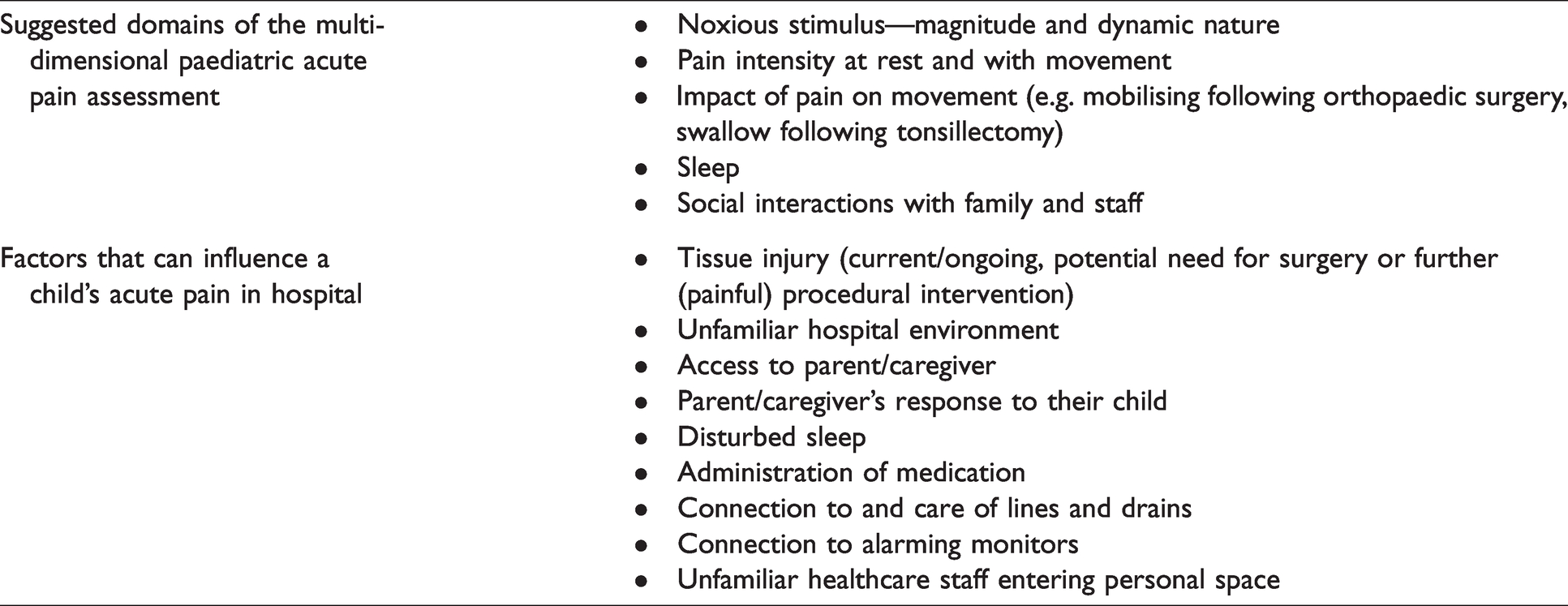
Opioids have been used for millennia to treat pain and, as in adults, are the cornerstone of multimodal analgesic regimens in children with moderate to severe acute pain. A pragmatic understanding of the sociopsychobiomedical model of pain is needed, beyond the pain score, to enable precision in opioid decision-making. This does not require a long and detailed analysis of the patient’s psychological state and social circumstances. Pain can be conceptualised as an output the brain produces when threat is perceived. Threat can be related to social, psychological and environmental factors in the acute care environment which are often distressing (Table 1). This contextual understanding informs a multidimensional pain assessment that encompasses evaluation of the magnitude and dynamic nature of the noxious stimulus (see further below: Modulation of the pain experience), and the patient’s function in different domains including movement, sleep and social interactions, which is necessary to manage opioids safely and effectively.
Sociopsychobiomedical assessment of the child with acute pain.
The various views held by the medical profession, and society in general, regarding opioids and pain considerably influence opioid use. The most prescient example of this is the so-called ‘opioid crisis’ which emanated from the USA. Since the early 2010s, legitimate concerns have been raised regarding increased deaths from prescription opioids,2,3 culminating in a public health emergency declaration in 2017 by the USA’s Department of Health and Human Services. 4 While the intent of actions undertaken to address this public health issue align with the ethical principle of non-maleficence, they have resulted in fear and stigmatisation around opioid prescribing and use. This risks inadequate pain relief for patients, ironically conflicting with the ethical principle this action seeks to achieve. Children are particularly vulnerable to being denied opioids as they are unable to advocate for themselves and are reliant on third parties (parents, caregivers, prescribers). For all the concern that has been raised with prescription opioids, we and other authors have found no convincing evidence that their appropriate prescription and administration in children for acute pain management leads to untoward deaths or later misuse.2,5 This does not negate the societal issue of prescription opioid misuse, and the role opioid prescribers have in addressing it. However, it does raise questions about strict restriction/avoidance of opioids as a strategy to address the opioid crisis, particularly if it then impedes patients accessing opioids appropriately for effective pain management.
Neonatal and infant pain neurophysiology
Until approximately 30 years ago, neonates and infants were commonly denied opioid analgesia for surgery and acute pain management.5,6 As our understanding of the physiology of pain in children has evolved, this practice has changed. We now know that spinal cord nociceptive tracts begin developing by seven weeks’ gestation and are functional by 25 weeks, with thalamic and cortical responses to noxious stimuli detectable by 26 weeks’ gestation. 7 Infants’ nociceptive systems are more excitable, and noxious stimuli induce more generalised reflex responses compared with adults. In addition, early pain experience results in structural nervous system changes at cortical and subcortical levels, which are then associated with changes to nociceptive function later in life; these consequences may be modulated by effective analgesia. 7
Relevant pharmacokinetics and pharmacodynamics
The study of pharmacokinetics details the relationship between medication dose and plasma concentration, while that for pharmacodynamics determines the relationship between plasma concentration and medication effect. The pharmacokinetic parameters of all medications, including opioids, changes substantially during normal childhood development, because of increasing body size and maturation of renal and metabolic drug clearance pathways. 8 Bioavailability influences dosing of oral medications and contributes to inter-drug variability when transitioning from parenteral to enteral dosing, while volume of distribution (Vd) and clearance are the key pharmacokinetic parameters that respectively determine the initial and maintenance dosing required to achieve a target drug concentration. Allometric scaling theory describes the relationship between an organism’s size, and its structure and function. It indicates that Vd is linearly related to body size, but clearance is not, a point that is poorly appreciated by the dose per kilogram approach commonly used in paediatric settings. This can lead to maintenance regimens which underdose, particularly in young children. The maturation of renal and metabolic drug clearance pathways is also a consideration in which immaturity reduces drug clearance. Maturation of glomerular filtration rate and clinically relevant enzymes for drug metabolism begins in utero, nearing completion by two years of age. 8 Further differences in physiology during the neonatal period can influence the concentration of unbound drug for a given dose: for example, reduced protein binding capacity results in increased unbound (free plasma) fraction of highly protein bound medications such as fentanyl and local anaesthetics.9,10 There is a lack of pharmacodynamic studies generally in children. In addition to their pharmacokinetic immaturity, it has been proposed that infants, particularly neonates, are pharmacodynamically immature and more sensitive to a given concentration of an opioid. However, this has not yet been adequately assessed. Regardless of how much pharmacokinetic and pharmacodynamic factors respectively contribute, neonates and infants remain a group susceptible to opioid-induced ventilatory impairment (OIVI).
Modulating the pain experience: When should an opioid be prescribed?
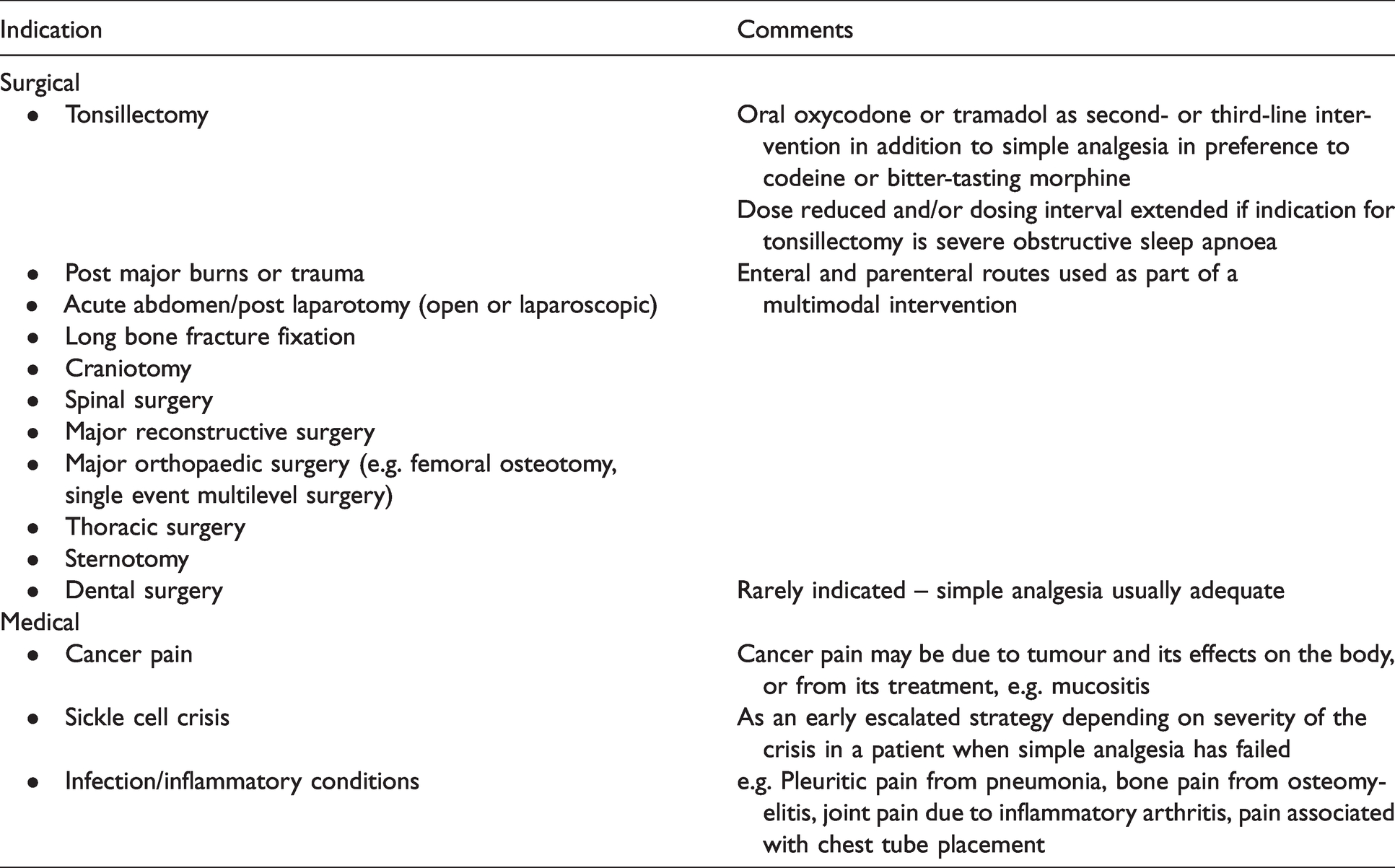
A generic response to this question is to prescribe opioids for moderate to severe acute pain as part of a multimodal analgesic regimen. However, this simple statement belies the complexity of its clinical application. Pain is defined as an ‘unpleasant sensory and emotional experience associated with, or resembling that associated with, actual or potential tissue damage’. 11 Physiologically, pain is mediated by dynamic networks of neurons that traverse multiple cortical and subcortical regions, and is not easily amenable to direct modulation. The principal effect of opioids is to reduce action potential transmission initiated by tissue damage through the nociceptive neurons of peripheral nerves and the spinal cord. This reduces the intensity of the threat signal reaching the brain and therefore can indirectly reduce the severity of pain. From this, one could consider that the severity of pain is correlated to the severity of a noxious stimulus, which to some extent is true. However, there is substantial variability in the severity of pain for a given noxious stimulus within individuals over time, and across populations. In addition, it is possible to experience pain in the absence of any damage. This is a reflection of the sociopsychobiomedical nature of pain. Clinically, it is prudent to assess the ‘actual or potential tissue damage’ occurring in a person’s body when considering when and how to prescribe opioids, because it is the physiological response to this tissue damage that opioids modulate peripherally. Considerations in this assessment include the nature and temporal relationship to pain of the inciting noxious event(s), the region of the body and extent of tissue damaged, and how much the damaged tissue is required to move. However, we have very little evidence to guide us on how to interpret this clinical information. There is a need to conduct well-powered studies to establish the correlation between common noxious insults from trauma and surgery, and the outcomes of pain and opioid use over time. Not only would these data provide an indication of different groups’ opioid requirements, they may help identify when a patient is not following the typical path, and needs either investigation for a complication, or optimisation of multimodal analgesia and non-pharmacological interventions. In the absence of these data, pragmatically, clinicians work from clinical experience. Table 2 provides a list of common paediatric scenarios where opioids would be considered.
Common indications for opioids in children with acute pain.
Which opioid to prescribe?
Most of the commonly used opioids in adults, including morphine, fentanyl and oxycodone, are also used in children of all ages, with codeine increasingly an exception (due to international regulatory changes in scheduling/prescription and newly applied contraindications in children). Opioids are generally well tolerated. However, paediatric recipients experience similar side-effects at similar frequencies to adults, with nausea, vomiting, sedation and pruritus the most commonly reported.11 –13 OIVI is a rare but serious adverse effect and comorbidities such as obstructive sleep apnoea (OSA),14 –16 severe neurodevelopmental conditions, trisomy 21 and severe epilepsy present an increased risk for this across all opioids. 14 Reducing the opioid dose by one-half to two-thirds in these high-risk groups has been recommended.
Morphine
Morphine is the most widely used and studied opioid in children. 15 Primarily a mu opioid receptor agonist, it has been shown to be effective and safe in children of all ages with accepted dosing regimens. More conservative dosing regimens are recommended in neonates and young infants, in whom morphine has been shown to have a reduced clearance rate of 10% to 20% compared with older children and adults, and an increased free fraction for a given dose, and in children with OSA who have been shown to have an increased risk of OIVI. 14 In addition to the intravenous route, it can be given orally, neuraxially, subcutaneously (in palliative care; uncommonly used in the hospital setting), or intramuscularly (the latter uncommonly employed in children in Australia due to the pain of injection). For postoperative pain relief in children under eight years of age, it is usually delivered as an infusion (10–40 μg/kg/h) with nurse-initiated boluses (10–20 μg/kg) given every ten to 30 minutes as needed. Neonates and young infants are possibly more sensitive to OIVI and a lower infusion rate (e.g. 5–20 μg/kg/h) and less frequent boluses have been suggested.7,14,15 Patient-controlled analgesia (PCA) can be used in children as young as five years old typically with a 5–10 minute lockout and no or low background infusion rate (e.g. 5–10 μg/kg/h). 17 Neuraxial morphine can be used in children and is typically administered intrathecally (2–5 μg/kg) or epidurally (30–50 μg/kg, maximum 3 mg). The risk of delayed respiratory depression, while low, is still present, and suitable monitoring postoperatively for 12 to 24 hours should be undertaken.
Morphine immediate-release formulations with the bitter taste of tablets and suspensions have been supplanted by the better tolerated oxycodone for enteral analgesia in many centres. However, it is still commonly used when oxycodone is unavailable or not tolerated, or for opioid rotation. It has a low oral bioavailability of approximately 30%, 16 and is typically prescribed as 0.25–0.5 mg/kg orally every three to four hours. Modified-release morphine granules (MS Contin suspension, Mundipharma, Sydney, NSW) 18 have been administered via enteral feeding tubes or sprinkled and disguised on food, and can be used following more painful operations and injuries (e.g. spinal fusion and massive burns) in children who do not take tablets, or who are too small for slow-release tablets which cannot be divided. Unfortunately, production of this formulation ceased early in 2021 and no other opioids have an equivalent slow-release granule formulation. In these select patients, immediate-release oral opioid solutions may be prescribed regularly to achieve a similar effect.
Oxycodone
Oxycodone is administered most commonly orally, although intravenous oxycodone use is increasing (off label), both for intraoperative and postoperative use. It has twice the oral bioavailability (∼60%) of morphine and is generally well tolerated by children of all ages (small tablet size and palatable butterscotch-flavoured liquid). 19 Buccal mucosal absorption has been shown to be effective in children under five years old. It is hepatically metabolised to noroxycodone (∼45%) and oxymorphone (∼18%); both are further metabolised to noroxymorphone. These latter two metabolites have demonstrated higher affinity for the mu receptor than oxycodone, but do not readily cross the blood–brain barrier and their contribution to analgesia is thought to be clinically insignificant.17,20 Hepatic clearance in neonates and young infants is reduced and is more variable compared with older children and adults. About 12% of oxycodone and its metabolites are excreted renally across all ages. While its elimination is slightly prolonged in renal failure, it is safe to use with consideration of an increased dosing interval in this setting.
Oxycodone’s intravenous potency has variably been reported as 1 to 1.5 times that of morphine. 17 Its faster onset of action compared with morphine (five versus ten to 15 minutes) and relative lack of active metabolites may offer advantages for perioperative use.17,21
Fentanyl
The parenteral opioid fentanyl is widely used in paediatrics but with little pharmacokinetic data. 19 It is administered primarily intravenously for paediatric acute pain management; however, the intranasal route is also commonly used, particularly in the emergency department and pre-hospital setting, with the transbuccal and transdermal routes used in palliative care. Fentanyl is highly lipid soluble, and while there are similar age-related changes in its pharmacokinetics to other opioids, throughout normal childhood development its Vd remains large and duration of action short following a single bolus injection, due to rapid redistribution to peripheral tissues. 19 Because fentanyl’s elimination half-life is similar to morphine, but its Vd is higher, fentanyl will take longer than morphine to be cleared from the body once the peripheral compartments have become saturated. It is unclear what loading dose is required to achieve this in individual children; however, its context-sensitive half-time progressively increases with the duration of the infusion. 15 Dose conversion of single bolus intravenous fentanyl compared with intravenous morphine is 100 μg: 7–10 mg,19,22 but this ratio is typically reduced when administered as an infusion by nurse-controlled analgesia (NCA) or PCA, e.g. 100 μg: 3.3 mg. 23 Fentanyl is metabolised by the hepatic enzyme CYP3A4 to inactive metabolites, and its clearance appears unaffected by chronic renal disease. 19 When administered by a continuous infusion, clearance may be highest in children aged six months to six years. 24
Administered as NCA or PCA, fentanyl has demonstrated safety and efficacy in paediatrics for acute pain management following neurosurgery, pectus excavatum surgery, urological surgery and for acute cancer pain management. 7 A review of prolonged opioid use in critically ill children found that opioid tolerance occurred more commonly with fentanyl than morphine. 25 A further study found that, in postoperative patients admitted to the paediatric intensive care unit, analgesic infusions of morphine were less likely to result in doubling the infusion dose over seven days or more, compared with fentanyl (odds ratio 0.48, 95% confidence interval 0.25–0.92). 26
Hydromorphone
Hydromorphone administered intravenously by NCA or PCA has been used in various paediatric settings including post scoliosis and pectus excavatum surgery, for cancer-related pain and less commonly as discharge analgesia via the oral route. 7 It appears to have similar efficacy and tolerability to morphine. 27 Typically, a parenteral dosing ratio for hydromorphone compared to morphine of 1 mg:5 mg is used. 22 It is more accessible than intravenous oxycodone in paediatric hospitals, and is sometimes used for opioid rotation if intravenous morphine and/or fentanyl has not achieved adequate analgesia. Its oral bioavailability is low (20%–25%). 28 Unfamiliarity with its dosing, and errors in both parenteral and enteral conversion, may be barriers to more widespread use. Oral, intranasal and epidural administration of hydromorphone in paediatric acute pain management is common in North America, 7 but its use via these routes in Australian and New Zealand practice is rare.
Codeine
Codeine has been used for decades as a ‘weak opioid’ for moderate to severe pain, particularly following adenotonsillectomy, with its previous over-the-counter ease of access. It has several metabolites and its analgesic action (but not its cough suppressant action) depends on its metabolism to morphine via the cytochrome P450 2D6 isoenzyme (CYP2D6).7,15,29 With over 100 CYP2D6 alleles identified, genetic variation results in different phenotypic activity grouped as: extensive metabolisers (one to two times relative activity; 77%–92% of the population), intermediate metabolisers (half of the relative activity) and poor metabolisers (no enzyme activity; 0–10% of the population with therefore no analgesic benefit over codeine).7,30 A variable percentage of people carry multiple copies of the normal (wild-type) gene, termed ultra rapid metabolisers (more than double relative activity; 0–29% of the population). Thus the analgesia and side-effects of codeine are unpredictable and deaths have occurred both in children following adenotonsillectomy 31 and in a breastfed neonate after maternal codeine ingestion. 32 Genotyping can be undertaken to categorise an individual’s CYP2D6 phenotype for metabolism of codeine; however, this is not in widespread clinical use. With the regulatory responses to the reported deaths and the availability of alternative medications, the use of codeine internationally and in Australia has reduced substantially in recent years. The Australian Therapeutic Goods Administration recommends that codeine not be used in children under 12 years of age, post-adenotonsillectomy in children aged 12–18 years, breastfeeding mothers, or people known to be ultra rapid metabolisers; 33 aligned with regulatory responses by the United States Food and Drug Administration and the European Medicines Agency.34 –36 Furthermore, the Therapeutic Goods Administration has up-scheduled codeine to prescription-only to limit access, 33 and the World Health Organization has removed codeine from its analgesic ladder for the treatment of pain in children. 37
Tramadol
Tramadol is an atypical opioid that can be given orally, intravenously or rectally. Its mu receptor agonist activity accounts for approximately one-third of its analgesic effect, with the remainder coming from noradrenaline and serotonin neuronal reuptake inhibition. 15 Tramadol can be used as an adjunct to conventional opioids, when there is neuropathic pain, or as the primary opioid in a multimodal analgesic regimen. It has been labelled along with codeine as ‘a weak opioid’ in the past, which reflects its scheduling as a prescription-only medication, instead of as a controlled drug. Despite this, its comparative potency is similar to conventional opioids, and it has a similar incidence of nausea and vomiting, sedation, constipation and pruritus. Adult studies have shown a lower risk of OIVI compared with conventional opioids at equianalgesic doses; however, paediatric studies have not been adequately powered to determine if this is the case in children, and OIVI occurs with overdose in both adults and children. 7
Tramadol is metabolised in the liver by CYP2D6 to O-desmethyltramadol (M1), an active metabolite with approximately 200 times the mu receptor affinity of the parent compound, purported to account for most of the medication’s opioid effects. 36 There is concern that ultra rapid metabolisers are at increased risk of OIVI and poor metabolisers will have reduced analgesic benefit from tramadol. While differences in CYP2D6 activity are known to influence a person’s response to tramadol, its influence on the risk of ventilatory impairment is unclear. 38 Responding to the theoretical concerns, and three deaths (all in children under six years of age) and nine cases of ventilatory impairment over almost 50 years of use, in 2017 the United States Food and Drug Administration applied the following to the tramadol drug label: a contraindication against its use in children younger than 12 years; a contraindication against its use in children under 18 years old following adenotonsillectomy; a warning against its use in children aged 12 to 18 years with OSA or severe lung disease that may increase the risk of serious breathing problems; and a strengthened warning to mothers that breastfeeding is not recommended when taking tramadol. 39 The reported fatalities occurred external to the USA (one in Australia) and involved the use of the highly concentrated tramadol drops (100 mg/ml), which were not developed for paediatric use and increase the risk of dosing error. The Society for Paediatric Anaesthesia in New Zealand and Australia (SPANZA) supports the cautious use of tramadol in paediatric patients, noting that ultra rapid metabolisers and children with sleep-disordered breathing may be at increased risk of ventilatory impairment with normal dosing (1–2 mg/kg). SPANZA recommends reduced dosing (0.5–1 mg/kg) and monitoring in hospital overnight for children who are administered tramadol following tonsillectomy (Table 3). 40 However the USA’s Society for Pediatric Anesthesia is more conservative, advising against the use of tramadol in children under 12 years and in those with OSA. 14 Caution is advised in patients with epilepsy as tramadol can lower the seizure threshold, and in patients on selective serotonin reuptake inhibitors due to a theoretical increased risk of serotonin syndrome, although this has rarely been reported. 7 In Australia, no low-concentration paediatric suspension is available; the oral immediate-release capsule is 50 mg, a dose typically suitable for larger children/adolescents, but the capsule’s granules can be dispersed in water. In New Zealand, a 10 mg/ml strawberry-flavoured elixir is readily available for paediatric use on the national compounding pharmacy compendium. 38
Typical opioid starting doses in children.
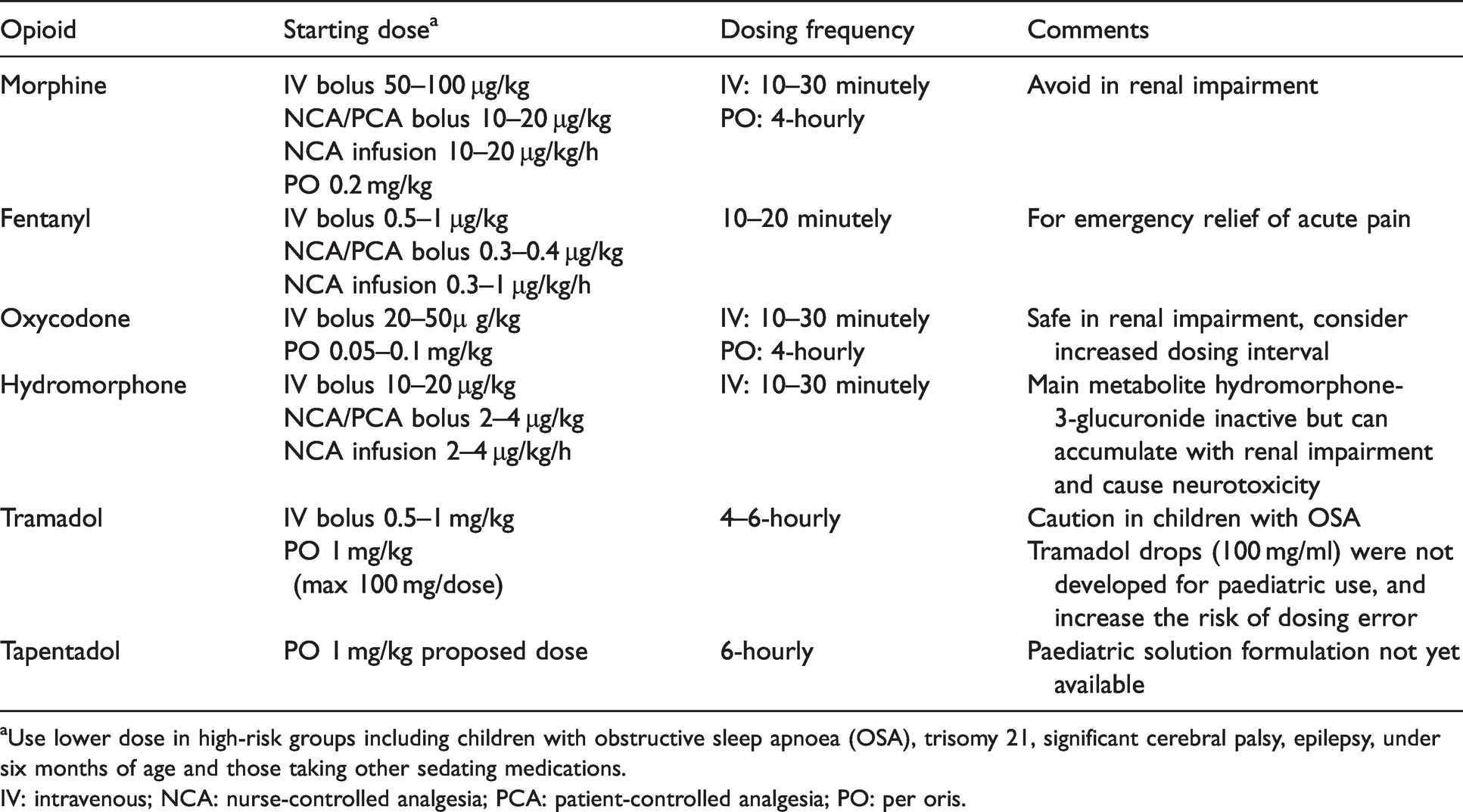
aUse lower dose in high-risk groups including children with obstructive sleep apnoea (OSA), trisomy 21, significant cerebral palsy, epilepsy, under six months of age and those taking other sedating medications.
IV: intravenous; NCA: nurse-controlled analgesia; PCA: patient-controlled analgesia; PO: per oris.
Tapentadol
Tapentadol is an atypical opioid with combined mu agonist and noradrenaline reuptake inhibitor activity. It is metabolised by glucuronidation in the liver, and has no active metabolites. 7 It can be given orally as immediate-release tablets up to every six hours or slow-release tablets 12-hourly. Immediate-release tapentadol appears to deliver similar analgesia to immediate-release oxycodone in a 5:1 dosing ratio, but with fewer gastrointestinal side-effects (nausea, vomiting and constipation). 41 Its mu receptor affinity is 50 times lower than morphine. It also has efficacy in the treatment of neuropathic pain. 42
No oral liquid formulation is available yet; however, pharmacokinetic studies in children aged two to 17 years undergoing various surgeries suggested a dose of 1 mg/kg is equivalent to an adult dose of 50–100 mg, with a median time to rescue analgesia of five to 6.3 hours.43,44 Nausea, vomiting and dizziness were the most common adverse effects. Studies of toxicity following accidental tapentadol ingestion in children under six years old have demonstrated no serious or minor adverse effects in the vast majority of cases, but sedation, confusion, coma and ventilatory impairment were reported. 45 The abuse potential appears to be similar to tramadol and less than for conventional opioids. 7
Opioid rotation
Opioid rotation originated in the management of cancer pain; 46 however, it is also used in the acute pain setting if a patient experiences poor analgesia despite increasing doses or intolerable side-effects with a particular opioid. 47 As patients develop tolerance to analgesic effects and side-effects at different rates, side-effects may begin to outweigh the analgesic benefit as doses escalate over time. 48 Opioid rotation to manage tolerance is based on the rationale that as different opioids have varying affinities for opioid receptor subtypes and differences in metabolism, the development of cross-tolerance between opioids is likely to be incomplete. 46 Opioid rotation may therefore improve analgesia and reduce side-effects. Equianalgesic dosing tables should be used only as a guide when performing an opioid rotation, particularly when opioid doses are high. 49 They are derived primarily from single dose studies in opioid-naive healthy adult volunteers 48 and therefore may be less applicable in unwell children who have received repeated opioid exposures. In addition these calculations may not reflect the variability of opioid pharmacokinetics and pharmacodynamics within and between individual children, or interactions between opioids and other medications. 22 Recommended practice is to decrease the calculated equianalgesic dose by 50% to reduce the risk of inadvertent overdose, and use a titratable regimen. 48 Each patient’s circumstances must be taken into consideration and may modify conversion dosing further; opioid rotation based on equianalgesic dosing tables alone carries a risk of toxicity and even fatality. 47 Children (and their caregivers) who have experienced difficulty managing their pain can quickly lose confidence in the pain management strategy when a change in their primary analgesic fails, and it can be challenging to regain their trust. Rotating to a dose that is commensurate with their opioid tolerance is therefore important, as is early and continued follow-up to ensure any need for significant dosage change occurs quickly.
Transitioning from intravenous to enteral route
Similar to the practice of opioid rotation, equianalgesic dosing tables can be utilised when transitioning a child from parenteral to enteral opioids. The most common method is to calculate the 24-hour opioid use and then convert this into morphine equivalents. This calculation should be considered in combination with contextual factors such as the duration of parenteral opioid administration, the nature of the noxious stimulus, and the anticipated trajectory of tissue healing (including ongoing sources of pain or painful interventions, activities the patient will be engaging in, whether regional analgesia has been used, and whether transition to an oral opioid will include an opioid rotation). This information can be used to inform an oral regimen that provides safe and adequate access to enteral opioids.
Slow-release opioids
Slow-release opioids are not registered for acute pain management by Australia’s Therapeutic Goods Administration or New Zealand’s Medsafe, and are not recommended for general use in acute pain management. 50 They may sometimes be useful in select populations with prolonged pain states on a temporary basis, 50 for example following scoliosis surgery, major trauma or burns. In these settings, in the authors’ experience, they may facilitate earlier transition from parenteral to enteral opioid administration. They are not suitable for patients with escalating or fluctuating acute pain because their longer time to peak effect (three to six hours) compared with immediate-release opioids (40–60 minutes), and longer time to reach steady state concentration, makes rapid titration difficult. 51 There is concern that with this reduced titratability, the risk of overdosing and OIVI is increased. Slow-release opioid initiation in naive children for acute pain is contentious, as it is in adults. 50 Importantly, if a slow-release opioid is started for acute pain in hospital and continued beyond discharge, a plan for the duration and follow-up needs to be documented and communicated to the primary care practitioner.
Special groups
Children with obesity
The World Health Organization defines overweight and obesity in zero- to five-year-olds as two and three standard deviations, respectively, above the median growth standard weight for length/height, 52 while for five–19 year olds overweight and obesity is defined as one and two standard deviations above the median growth reference body mass index for age. 53 Childhood obesity prevalence is increasing globally and there is the potential to cause opioid-related toxicity when a total body weight dosing regimen is used indiscriminately. Obese children have a higher absolute lean mass and fat mass than normal-weight children; however, the percentage of lean mass per kg of total body weight is lower, and the percentage of fat mass is higher in obese children than in non-obese children. 54 Obesity is a major risk factor for OSA, increasing the risk of OIVI and sedation 7 (see OSA section below). Vd and clearance are altered in obesity. These changes are medication specific and unpredictable, meaning a single size descriptor for medication dosing in obesity is not universally applicable. For example, Vd may be increased, unchanged or reduced for lipophilic medications and unchanged or reduced for hydrophilic medications. 55
Lean body mass, ideal body weight and total body weight have been recommended to calculate dosing of different opioids in obese children (Table 4). However, given the lack of readily accessible dosing charts and calculators, the complex pharmacokinetic equations proposed, the paucity of paediatric bariatric pharmacokinetic data, and differences between loading and maintenance dosing, they are not widely used.7,56 Proposed solutions include the use of a nomogram for ideal body weight and lean body mass estimation, 56 and the use of allometric theory to calculate normal fat mass for medication dosing. 38 The latter is an adaptable size descriptor that can be used in all people and is medication and pharmacokinetic parameter specific (Vd or clearance). It can range from fat-free mass to total body weight and estimates the pharmacokinetic effect of adipose tissue. However, it is still a cumbersome calculation that is not suited to a busy clinical environment. In the setting of these challenges, judicious initial dosing and titration to effect is paramount.
Dosing size descriptors for common opioids.
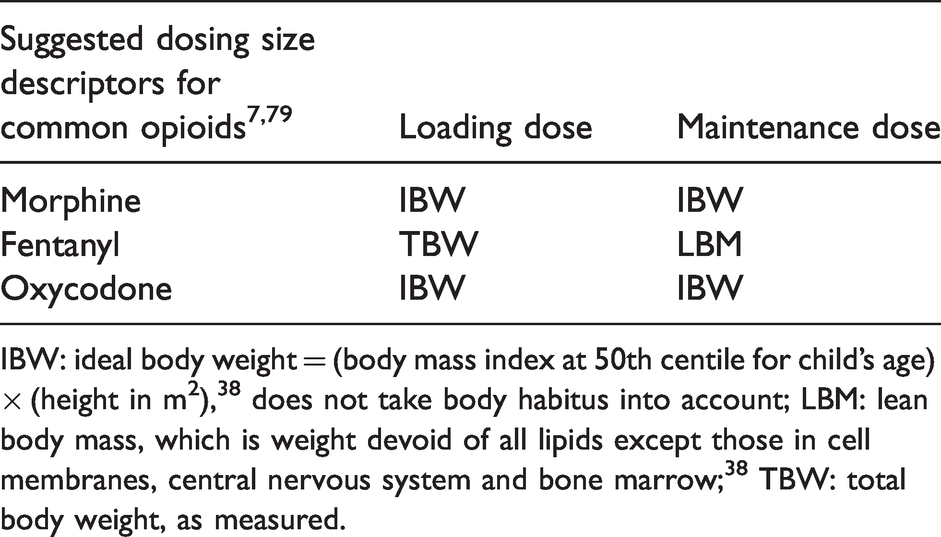
IBW: ideal body weight = (body mass index at 50th centile for child’s age) × (height in m2), 38 does not take body habitus into account; LBM: lean body mass, which is weight devoid of all lipids except those in cell membranes, central nervous system and bone marrow; 38 TBW: total body weight, as measured.
Children with obstructive sleep apnoea
Obstructive sleep apnoea, defined as partial or complete obstruction of the upper airway during sleep, is common in children, and is the main indication for children undergoing adenoidectomy with or without tonsillectomy. 57 Most children in Australia and New Zealand do not undergo pre-tonsillectomy polysomnography and only the very severe will be on continuous positive airway pressure (CPAP) or bilevel positive airway pressure (BiPAP) support. Children with OSA are more likely to have post-tonsillectomy respiratory complications including oxygen desaturations, obstructive episodes and apnoeic adverse events when compared with children without OSA.57,58 This has implications for postoperative disposition and monitoring, opioid dosing and discharge opioid prescribing. While CPAP or BiPAP can mitigate these risks, it is often poorly tolerated by children and difficult to introduce de novo postoperatively without sedation. Nasal high flow oxygen may be a promising alternative. 59 The patient with severe OSA should be nursed laterally and continuous postoperative oxygen saturation measurement should be the minimum monitoring for at least 24 hours or while the patient remains at increased risk of respiratory events.
Tonsillectomy is very painful and opioids remain an essential part of the analgesic strategy when simple analgesic therapy alone is inadequate. Children with OSA have reduced post-tonsillectomy morphine requirements by as much as 50%.60,61 This increased opioid sensitivity and concomitant risk of OIVI is thought to be a pharmacodynamic difference due to chronic intermittent hypoxaemia causing up-regulation of opioid receptors. 58 If these children are overweight or obese, a pharmacokinetic discrepancy between the expected and actual opioid plasma concentrations may also play a role. 57 The synergistic effect of the opioids combined with hypno-sedative medications used for general anaesthesia (both for tonsillectomy and other operations) is clinically relevant when loading and re-dosing postoperative opioids in children with OSA. Judicious titration is therefore warranted, particularly when managing these children for other procedures and in the outpatient environment. Multimodal analgesic regimens including paracetamol, non-steroidal anti-inflammatory drugs, regional anaesthesia where possible, alpha-2 agonists and ketamine can reduce or, in some circumstances, even eliminate the need for opioids in these patients. 62
The opioid-tolerant child or adolescent
Tolerance is defined as a predictable physiological decrease in the effect of a drug over time, so that a progressive increase in the amount of the drug is required to achieve the same effect. 63 Opioid tolerance in children and adolescents is less well studied than it is in adults, and its prevalence in the community is unclear. However, similar to adults, there are four main groups who may be taking opioids and become opioid tolerant: chronic non-malignant pain, chronic cancer pain, non-medical prescription opioid use (mostly adolescents), and acute or subacute opioid tolerance. 7 The latter group is typically acutely unwell inpatients who are receiving opioids by intravenous and/or enteral routes for multiple days, and may also be at risk of tolerance to other sedatives such as benzodiazepines. 64 Opioid-tolerant children and adolescents may be at risk of experiencing difficulty with their acute pain management. 7 Recommendations for strategies to manage opioid tolerance in children and adolescents with acute pain have been extrapolated primarily from the literature on multimodal analgesia in opioid-naive children with severe pain and the adult literature, and include the use of ketamine, alpha-2 delta ligands, alpha-2 agonists and/or local anaesthesia techniques (regional anaesthesia or systemic lidocaine infusion) as part of a multimodal analgesic approach.
Opioid-tolerant patients are at risk of withdrawal syndrome when opioids are weaned, and this may occur in children and adolescents who have been exposed to opioids as ward or intensive care unit inpatients. 65 In this setting, risk factors for opioid withdrawal include increasing duration of opioid therapy, particularly greater than five days, increasing cumulative dose of opioids and the use of synthetic opioids such as fentanyl.7,64 Children and adolescents at risk of opioid withdrawal should be assessed for it with the aid of a validated withdrawal tool such as the withdrawal assessment tool (WAT-1). 66 Of note, it can be challenging to assess these symptoms in children, particularly infants and toddlers who are hungry, sleep deprived, afraid, or septic, and withdrawal is a diagnosis of exclusion. When patients have been on moderate to high-dose opioids for a few days or more they may be managed with slowing the opioid wean (e.g. 10%–20% reduction of the total opioid dose every 48 hours),26,67 opioid rotation or the use of adjuvants such as clonidine if withdrawal occurs. 7
Discharge and follow-up
Discharging children on oral opioid medication after surgery is not without risk. Apart from the common side-effects described above there are some issues unique to paediatrics. These include appropriate prescribing, dispensing, caregiver education, and follow-up. General practitioners may also be less familiar with opioid prescribing in children than for their adult patients, necessitating clear communication.
Discharge opioid prescribing
Due to the frequent use of size descriptor dosing calculations, errors in prescribing are a major issue in paediatrics generally. A large multicentre USA case series found a prescribing error resulting in an increase of 10% or more of the expected maximum daily dose occurred in 2.7% of opioid prescriptions for children aged three years old or less. 68 Concerningly, the risk of a prescription error was highest in the most vulnerable, that is infants aged zero to two months (8.9%) and two to five months (5.7%). The issue of prescription error is compounded by administration errors confusing mg with ml or confusing different formulations (1 mg/ml with higher concentrations) and resultant accidental overdose by a caregiver at home.
Dispensing of discharge opioids and lack of appropriate aliquots for use in acute paediatric pain
The preferred discharge opioid for young children in many tertiary paediatric centres is oxycodone liquid (OxyNorm liquid, Mundipharma, Sydney, NSW). 69 In Australia, it is presented as 250 mg in 250 ml. This amount far exceeds the quantity required for small children discharged from hospital with moderate to severe acute pain, and can often result in a large quantity of unused opioid left in the home, which may then be subject to misuse and accident. Some private and most hospital pharmacies will dispense smaller (usually 20 mg) aliquots, but this is neither transparent, universal, nor cost effective for those pharmacies. Liquid morphine (Ordine oral solution, Mundipharma, Sydney, NSW) 70 is presented as 200 mg in 200 ml, also an excessively large amount to dispense.
Dosing and duration of opioid therapy
Careful dose titration prior to discharge in vulnerable groups at risk of ventilatory impairment such as ex-preterm infants, children with OSA, and children on long-term sedating medications is essential. Junior doctors prescribing opioids in this setting should have adequate supervision from senior clinicians. As discussed above, it is not always clear what size descriptor should be used for a given medication or dose in the obese child. There is no universally accepted opioid dosing guideline for obese children, and titrated empiric dosing to effect or side-effect with regular review is the mainstay for inpatients.71,72 Unfortunately, for day-stay patients this opportunity is not afforded, and caution or consideration for hospital admission is advised. Prescribing a lower dose with a longer dosing interval in groups at increased risk of ventilatory impairment is recommended.
Children are frequently prescribed opioids in excess of their analgesic requirements.7,14 For the majority of surgeries for which opioids are indicated, a limited number of doses for discharge (e.g. three to five days) is usually recommended, and it is essential that instruction to the caregiver is provided on how to administer the opioid as part of a multimodal regimen (i.e. use as needed following administration of simple analgesics), along with mandatory communication with general practitioners about the expected pain trajectory and need for follow-up. Staff prescribing discharge medications are often the most junior in public hospitals. It is therefore important to ensure they have adequate training about dosing, duration and formulation. Anaesthetists and pain services have a vital role to play in providing this training and supporting local governance. 73 Resources such as the Michigan Opioid Prescribing Engagement Network (OPEN) paediatric prescribing recommendations can help guide prescribers and primary care practitioners, but dosing should be adapted to local practices which differ in different regions. 74 A large-scale audit of usual postoperative trajectories is required to provide data to inform these resources and enable appropriate prescription for the norm, as well as to reduce the risk of inadequate analgesia for the outlier.
Risk of long-term opioid use and opioid misuse
Although uncommon, discharge opioid prescribing has been linked with long-term opioid use in adolescents with a history of chronic pain, substance use disorder and mental health conditions as risk factors. 7 Higher risk patients should ideally be identified preoperatively and followed up by a specialised pain service where available.
There are relatively few data on the misuse of prescription opioids in adolescents and young adults, and at times they can appear contradictory. Most of the literature on the misuse of prescription opioids in adolescents and young adults is North American data and suggest that misuse is common, with prevalence estimates ranging from 3.5% to 20%. 7 In Australia, community consensus data has reported a low rate (1.1%) of non-medical opioid use involving adolescents and young adults in the previous 12 months. 75 Excess opioids prescribed for family and friends are a major community source of opioids for non-medical use in adolescents. 76 Legitimate use of a prescription opioid before finishing high school was associated with a 33% increased risk of prescription opioid misuse on one or more occasions by the age of 23 years. 77 In contrast, medical use of prescription opioids without any history of non-medical use during adolescence was not associated with an increased risk of substance use disorder symptoms at 35 years of age. 76
Caregiver education
Caregivers tend to underdose their children with analgesia following discharge from hospital, often due to fear of medication side-effects. 78 Caregiver and patient education aimed at improving understanding of the multimodal analgesic plan and medication side-effects, in addition to the often forgotten non-pharmacological pain management strategies, may help improve pain management in the home. Follow-up with healthcare services should be actively encouraged and be accessible in the event of ongoing pain or analgesic issues. Caregivers must also be provided with instructions for the safe storage of opioids and how to dispose of unused medication.14,73 Unsafe storage of opioids and failure to dispose of excess opioids is common, and is a source of accidental poisoning in young children and poisoning from recreational use or intentional overdose in adolescents. 7
Follow-up
A fundamental component of opioid stewardship is ensuring adequate follow-up mechanisms exist both in hospital and after discharge. This is particularly important with the introduction of short-stay ward care and earlier discharge following the concepts of enhanced recovery after surgery pathways. Information, both verbal and written, provided to caregivers on discharge can be overwhelming. Confusion can occur easily around appropriate analgesic use including up and down titration, particularly in children who have undergone major surgery or who have comorbidities. A follow-up service for patients going home on complex analgesia including slow-release opioids establishes a point of contact for families, allows assessment of ongoing pain, side-effects of medications, and functional recovery, and permits experienced guidance for weaning plans to be carried out, ensuring medication safety. This is the role of the anaesthetist or the acute pain service clinicians including clinical nurse consultants and nurse practitioners, supported by the pharmacists.
Conclusions
Opioids are, and will continue to be, an essential part of acute pain management in children of all ages. The use of these potent analgesic agents begets responsibility, and the responsibility of safe and effective opioid use falls upon all clinicians who manage acute pain in children. Anaesthetists are uniquely positioned with knowledge, expertise and opportunity to show leadership in carrying this responsibility. Improved precision in the clinicians’ assessment, decision-making and prescribing, combined with systems that are informed by data and tailored to paediatrics will allow better and wider understanding of the complexities around opioid prescribing and access. This is integral to providing paediatric patients with optimal acute pain management.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
