Abstract
This multicentre, retrospective medical record audit evaluated opioid analgesia prescribing within a Victorian metropolitan public hospital network. The study included all surgical patients discharged between January 2012 and December 2020 with one or more discharge prescriptions from three metropolitan hospitals (
Total opioid prescribing (mean OMEDD) peaked in 2013. Between 2017 and 2020 there was a trend towards prescribing fewer opioids on discharge. Over the study period, there was decreasing prescription of codeine and increasing prescription of oxycodone and tapentadol. The proportion of patients prescribed slow-release opioids increased in the earlier years of the study, reaching a peak of 20.6% in 2017. Since 2017 there has been a rapid reduction in the prescription of slow-release opioids.
Subanalysis was undertaken to evaluate key changes in the opioid prescribing landscape in the health network. The removal of default opioid pack sizes in the electronic medication management system (December 2014) and the release of the Faculty of Pain Medicine–Australian and New Zealand College of Anaesthetists’ statement regarding the use of opioid analgesics in patients with chronic non-cancer pain (March 2018) were associated with significant reductions in mean OMEDD prescribed on discharge (136 mg vs 122 mg and 120 mg vs 85.4 mg, respectively,
In conclusion, the quantity of opioids prescribed on discharge in this patient group peaked in 2013 and has been decreasing since.
Introduction
The last decade has seen considerable interest in the harms of prescription opioids in many developed countries including the United States of America (USA), Canada and Australia.1 –3 While the intention of prescribing opioids is to reduce pain and suffering after surgery or for other painful conditions, adverse outcomes frequently occur, and every day in Australia approximately three people die and 150 people are hospitalised due to opioid-related toxicity.3 –5 Adverse outcomes from opioid use are considerably higher for slow-release opioids compared with immediate-release formulations. 1 Prescribing slow-release opioids in acute non-cancer pain remains commonplace, despite revised pain management advice to the contrary from professional bodies.1,6
Prescription-opioid misuse has been recognised at international, national and institutional levels.1,4,5,7 Opioid-related deaths are the number one cause of unintentional medication-related deaths in Australia, with more than half the result of prescription opioids. 5 Deaths related to oxycodone, morphine and codeine increased by 102% between 2006 and 2017, and deaths involving fentanyl, pethidine and tramadol increased by 1,000%. 5 In Australia in 2016 and 2017, 15.4 million opioid prescriptions were dispensed under the Pharmaceutical Benefits Scheme (PBS) to 3.1 million people, of which oxycodone was the most common (5.7 million prescriptions dispensed to 1.3 million people), followed by codeine (3.7 million prescriptions dispensed to 1.7 million people) and tramadol (2.7 million prescriptions dispensed to 600,000 people). 4 While opioid prescribing in the community increased between the 2013/14 reporting period and 2016/17 reporting period per the Third Australian Atlas of Healthcare Variation, this trend appears to have reversed in new data looking at the 2016/17 and 2020/21 reporting periods. 8 Regardless, initiation of opioids in hospital surgical settings has been identified as a key risk for ongoing use.3,9,10
A systematic review identified that the prescription of opioids on discharge from hospital presents an important opportunity for intervention. 10 A 2015 study found 17% of opioid-naïve patients prescribed opioids on discharge from emergency departments continued to use opioids 1 year later, and a 2012 study of post-surgery patients showed patients prescribed opioids on discharge from surgical care are 44% more likely to be taking opioids 1 year later than those discharged without opioids.11,12 A 2018 study commissioned by the Society of Hospital Pharmacists of Australia found 70% of hospital respondents reported opioids being prescribed on discharge from hospital even when not required in the 48 h prior to discharge, ‘just in case’. 13 The initial dose and quantity of opioids prescribed at discharge have been identified as risk factors for long-term use and predictors of future opioid misuse.2,9,14 –16 Policies establishing opioid discharge quantities vary significantly across Australian hospitals despite evidence showing the link between prescribed quantities and risk of long-term opioid use.2,9,14 –16 Over-prescribing opioids also results in a large pool of unused opioid available for later use by the patient or diversion to others in the community.2,4
Within a 1,122-bed public hospital network in Victoria, Australia, local audits show that there has been progress in improving prescribing practices through strategies including provision of pain medicine education to medical students, junior medical, senior medical and nursing staff. This network covers most surgical specialities except cardiac and neurosurgery. Further, the network has a well-established acute pain service which provides advisory support to clinical teams, monitors internal prescribing patterns, supports governance of opioid prescribing and promotes research and local improvement activities. The introduction of an electronic medication management system (eMMS) (Cerner Powerchart®) enabled further system-based interventions aimed at influencing opioid prescribing practices and an example of this improvement work was the removal of default PBS or pack size quantity (for non-PBS medications) for opioid discharge prescriptions. A small, unpublished audit conducted following this change found a 50% reduction in the use of PBS/full pack size quantities (
There have also been significant changes in the opioid prescribing landscape outside of the hospital network that may have impacted opioid prescribing practices. The release of the Faculty of Pain Medicine–Australian and New Zealand College of Anaesthetists’ (FPM-ANZCA) statement regarding the use of opioid analgesics in patients with chronic non-cancer pain challenged established practices of anaesthetists and other medical and surgical specialists using opioid analgesia and has contributed to a reduction in the dispensing of slow-release opioids on discharge.6,17 Following a successful real-time prescription monitoring pilot in Tasmania, where a reduction in the quantity of opioids prescribed on hospital discharge was observed, Victoria’s real-time prescription monitoring system (SafeScript) was released in April 2019.18,19 In April 2020 it became mandatory to check SafeScript in Victoria before writing or dispensing a prescription for a medicine monitored through the system in all settings except hospitals.18,19
The primary aim of this study was to investigate surgical opioid prescribing trends between 2012 and 2020 at the hospital network, and in particular, the choice of opioid and use of slow-release preparations. The secondary aims were to determine whether the removal of default opioid PBS and pack size quantities in eMMS, the release of the FPM-ANZCA statement regarding the use of opioid analgesics in patients with chronic non-cancer pain, and the introduction of SafeScript were associated with a reduction in the quantity of opioids prescribed on discharge from surgical units in the hospital network.
Materials and methods
A retrospective medical record audit utilising electronic prescribing records from the eMMS system at three metropolitan hospitals within the same hospital network was undertaken. All patients discharged from any surgical unit who were prescribed one or more medications on discharge were included. Patients were included if they had any medication prescribed on discharge, even if the medication was unrelated to analgesia. In situations where the same patient had two independent encounters during the study period, each encounter was included separately. Details of the discharge prescriptions including medication, formulation and quantity were obtained from the eMMS. Patient demographic details were obtained from the hospital administrative database. Data collection was undertaken by the lead author with independent manual validation of 50 records undertaken by other researchers to confirm accuracy. All data was stored and analysed in a spreadsheet (Microsoft Excel®) with subsequent statistical analysis occurring in R® version 3.6.3 or Microsoft Excel®. For each patient encounter, the total quantity of opioids prescribed on discharge was calculated and converted to oral morphine equivalent daily dose (OMEDD) using the FPM-ANZCA Opioid Dose Equivalence Calculation Table. 20
Prescribing trends were assessed on an annual basis (number of opioid types prescribed on discharge, formulation, specific opioid and OMEDD). Evaluation of opioid prescribing changes associated with the removal of default pack size in the eMMS was undertaken by comparing three years before (January 2012–December 2014) and post change (January 2015–December 2017). Evaluation of the effect of the release of the FPM-ANZCA Position Statement was undertaken using the same method with data from two years before and after (April 2016–March 2020) and for SafeScript six months before and after (October 2019–September 2020), respectively. The periods for the above comparisons were determined before data collection and selected to maximise the potential sample sizes. Statistical significance was determined via chi-square test for categorical variables and two-tailed t-test for continuous variables.
For the purposes of calculating the OMEDD, patients prescribed sublingual fentanyl, methadone and sublingual buprenorphine formulations (unable to undertake OMEDD conversion), liquid opioids (variable methods of documenting prescribed quantities in eMMS) and patients with ambiguous quantities on discharge (for example ‘box’) were excluded. For analysis and calculations unrelated to OMEDD, these orders were included. Fentanyl and buprenorphine patches were considered slow-release formulations.
Results
Between January 2012 and December 2020, a total of 117,989 surgical patients were prescribed one or more medicines on discharge, of which 77,278 were prescribed one or more opioids (Table 1). For the purposes of calculating OMEDD, 1,052 patient encounters were excluded (liquid formulation: 832; no OMEDD conversion available: 116; ambiguous quantity: 104). For other analyses, these 1,052 patient encounters were included.
Patient demographics and opioid prescription details (surgical discharges between January 2012 and December 2020).
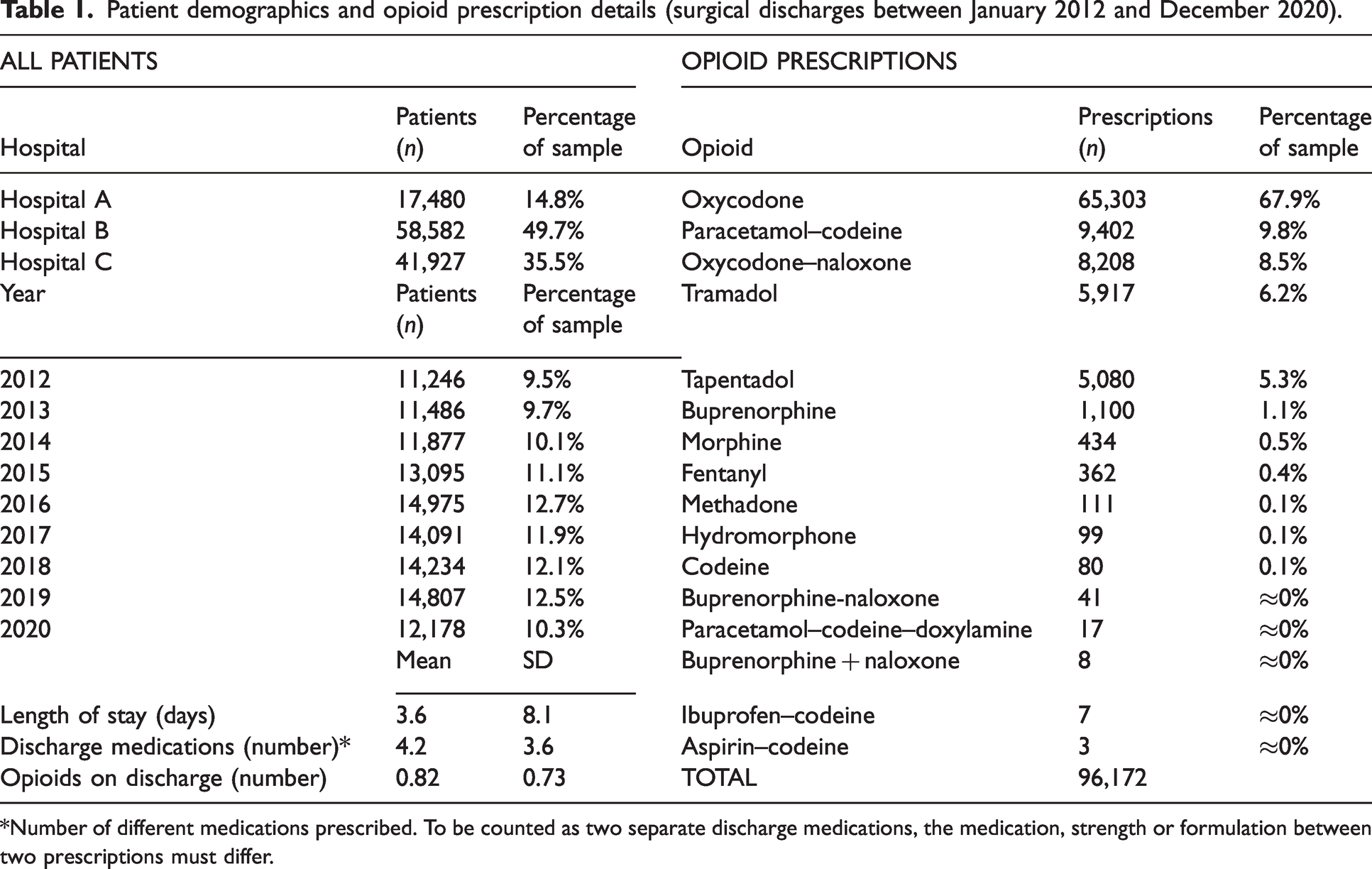
*Number of different medications prescribed. To be counted as two separate discharge medications, the medication, strength or formulation between two prescriptions must differ.
Between 2017 and 2020 there has been a trend towards prescribing fewer opioids on discharge (Figure 1). Over the study period, there was decreasing prescription of codeine and increasing prescription of oxycodone (as either an individual medication or in combination with naloxone) and tapentadol (Figure 2). The proportion of patients prescribed slow-release opioids increased in the earlier years of the study, reaching a peak of 20.6% in 2017 (Figure 3). Since 2017 there has been a rapid reduction in the prescription of slow-release opioids, with each year between 2018 and 2020 showing a lower proportion of patients prescribed one or more slow-release opioids than the preceding year (
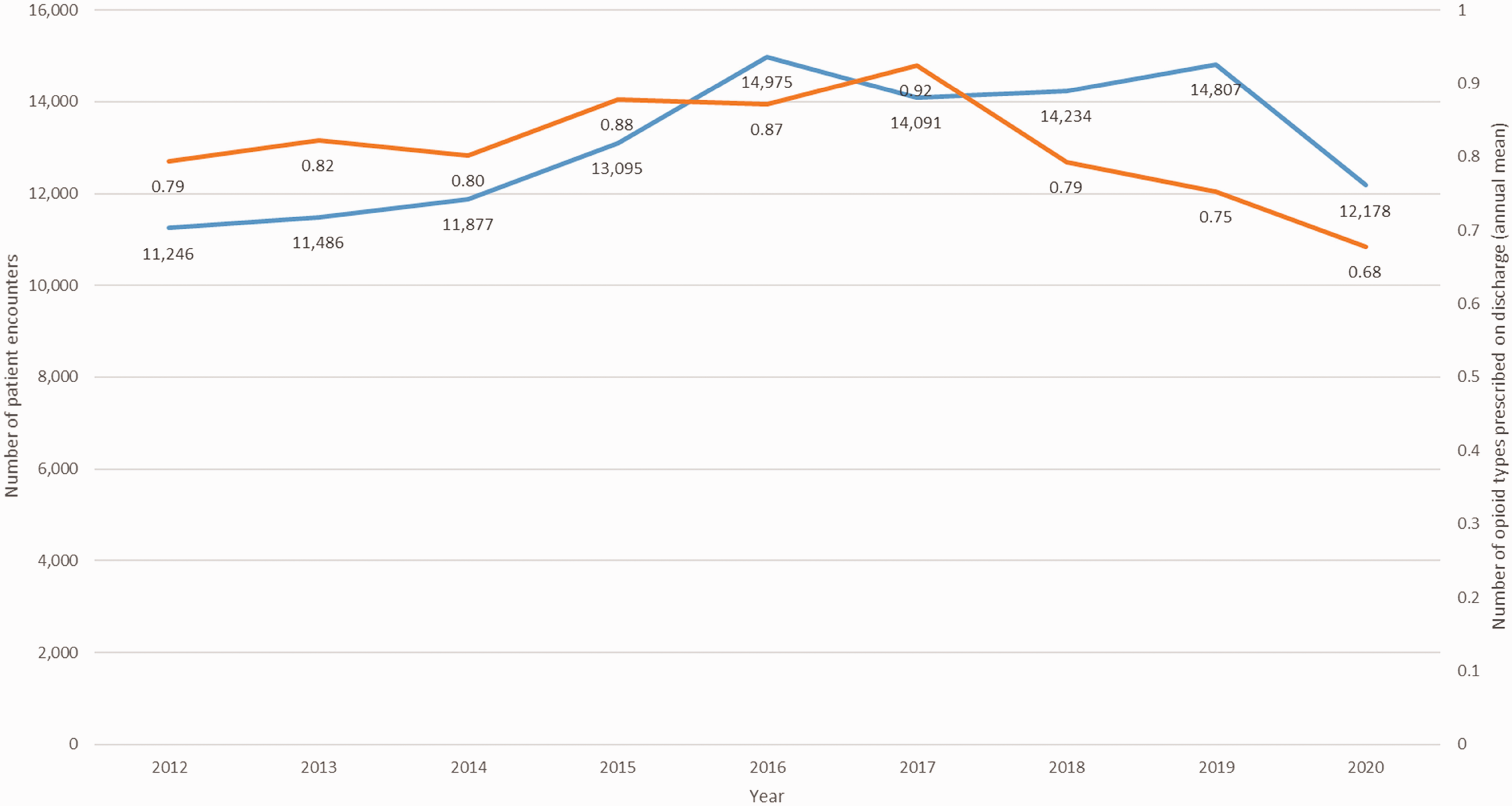
Annual number of patient encounters included in the study and mean number of different opioid types prescribed on discharge per encounter.
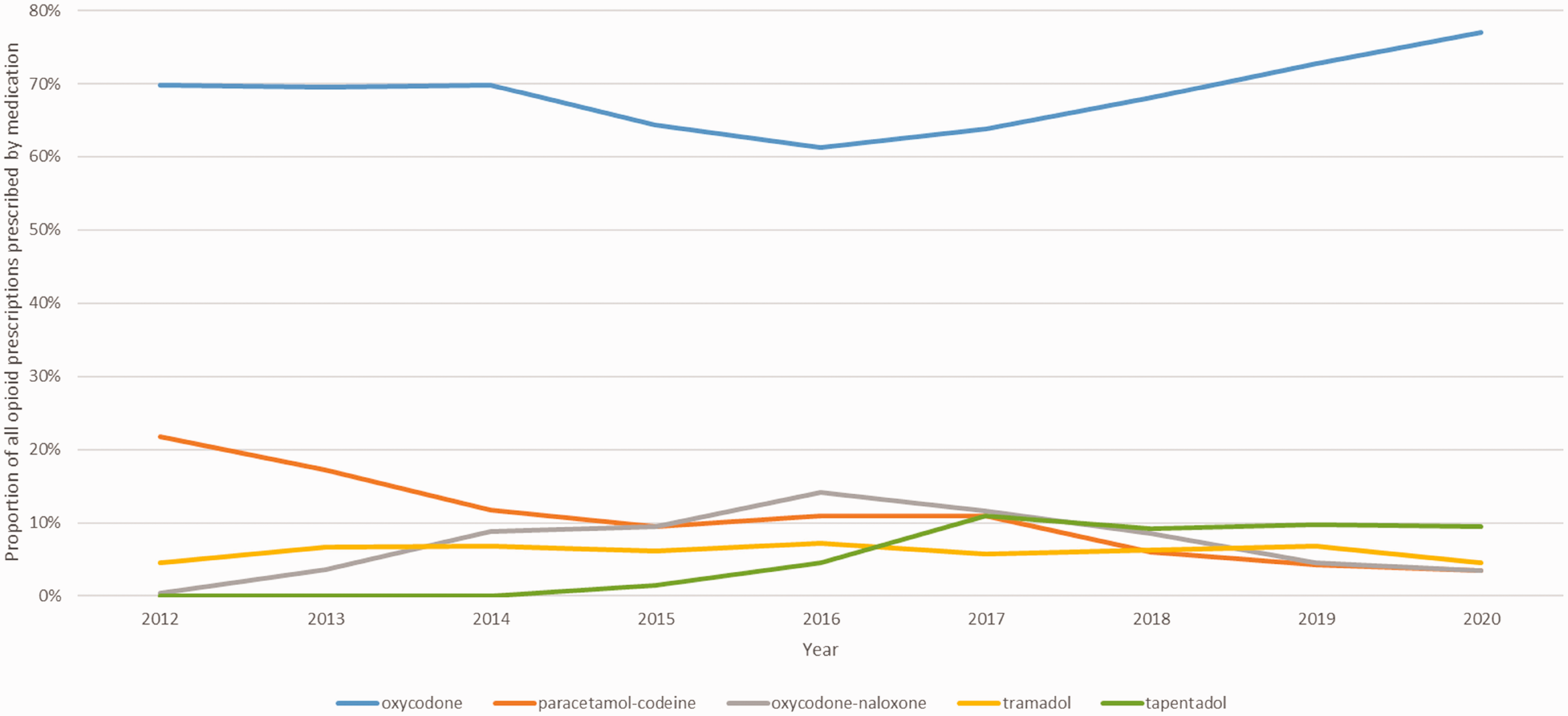
Annual proportion of all surgical opioid prescriptions prescribed by medication for the five most commonly prescribed opioids during the study period.
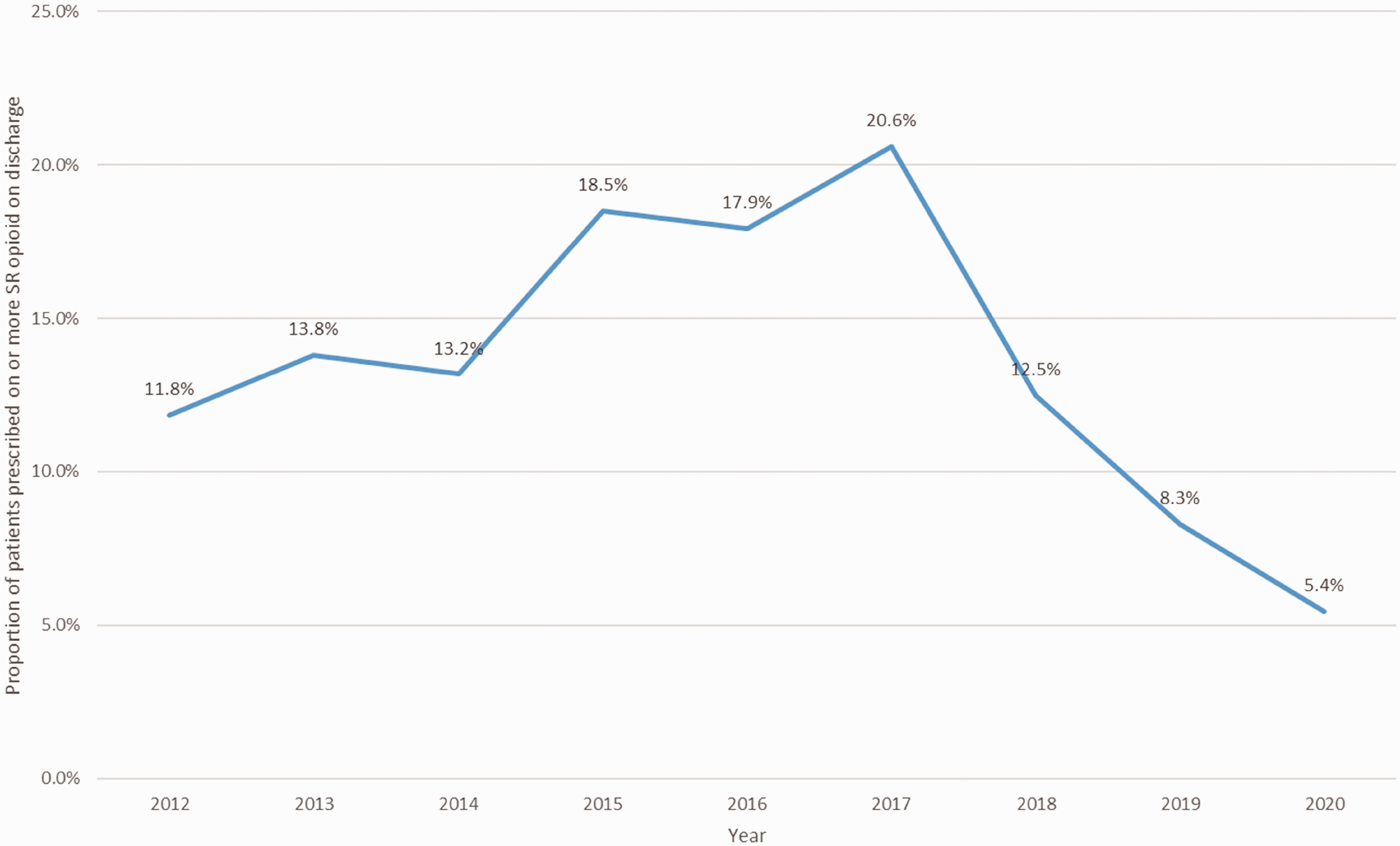
Annual proportion of surgical patients prescribed one or more slow-release opioids on discharge.
Since 2014 there has been a year-on-year decrease in the quantity of opioids prescribed (converted to OMEDD) on discharge (Figure 4). This decrease, when each year was compared with the preceding year, was found to be statistically significant for years 2015–2020 (all
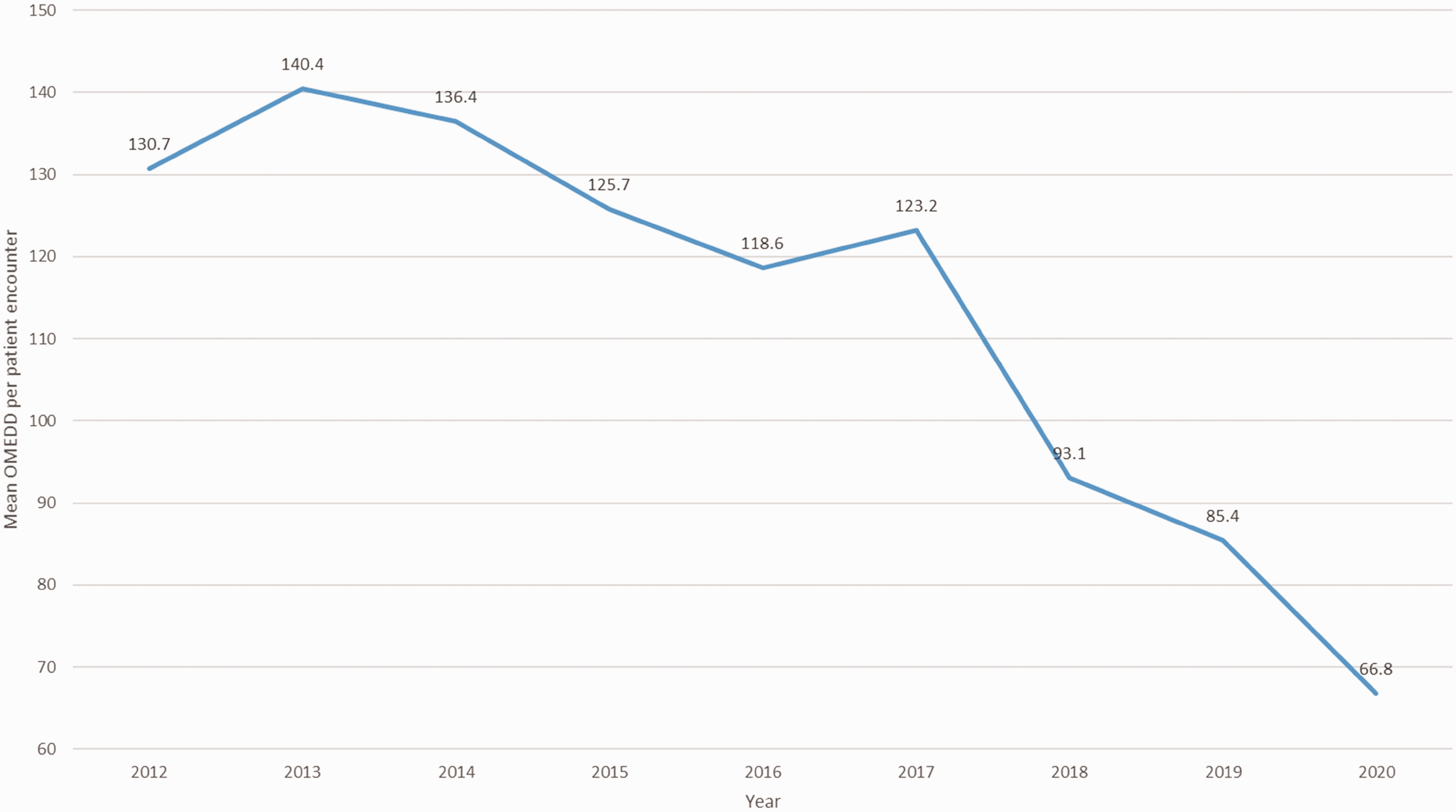
Annual surgical discharge prescription oral morphine equivalent daily dose (OMEDD, mean per patient encounter).
Electronic medication management system pack size subanalysis.
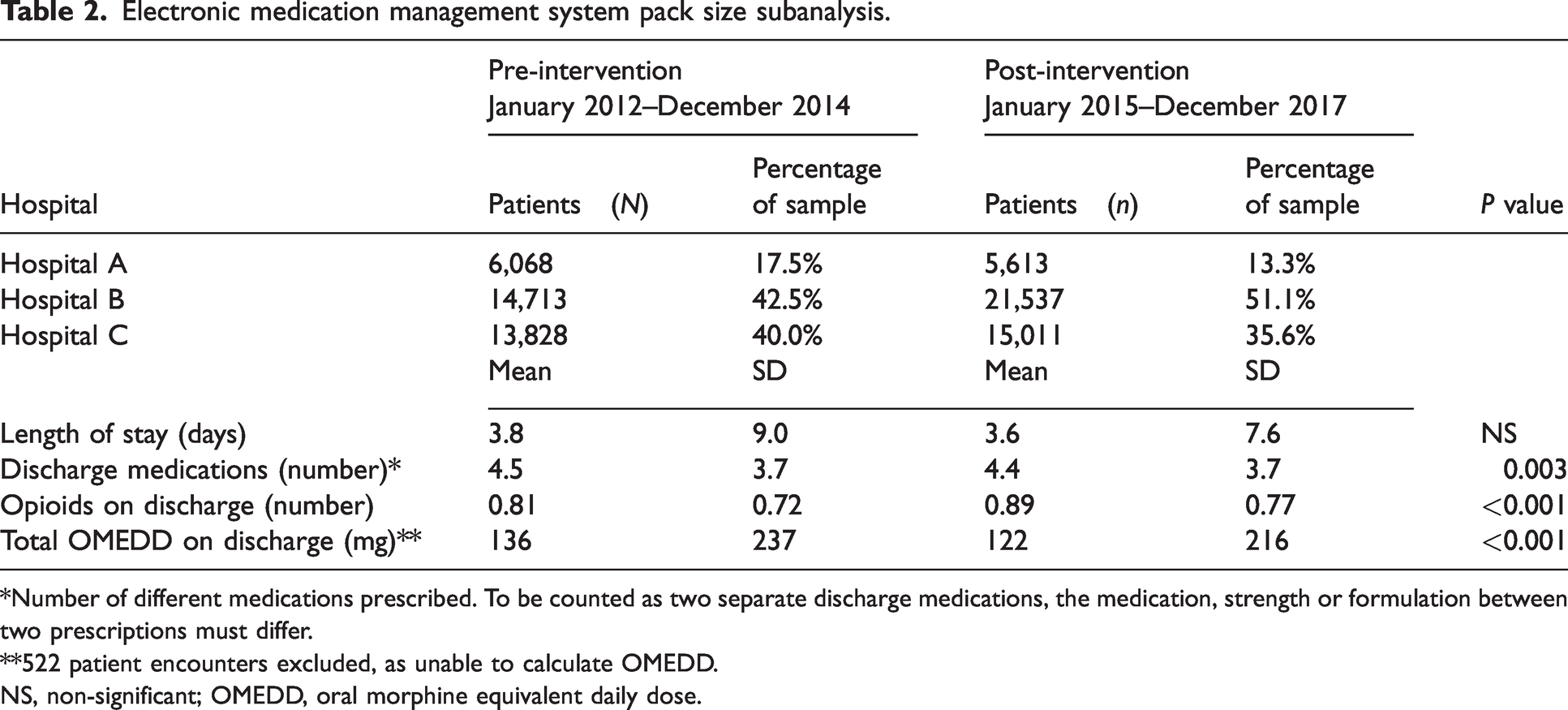
*Number of different medications prescribed. To be counted as two separate discharge medications, the medication, strength or formulation between two prescriptions must differ.
**522 patient encounters excluded, as unable to calculate OMEDD.
NS, non-significant; OMEDD, oral morphine equivalent daily dose.
Faculty of Pain Medicine–Australian and New Zealand College of Anaesthetists position statement subanalysis.
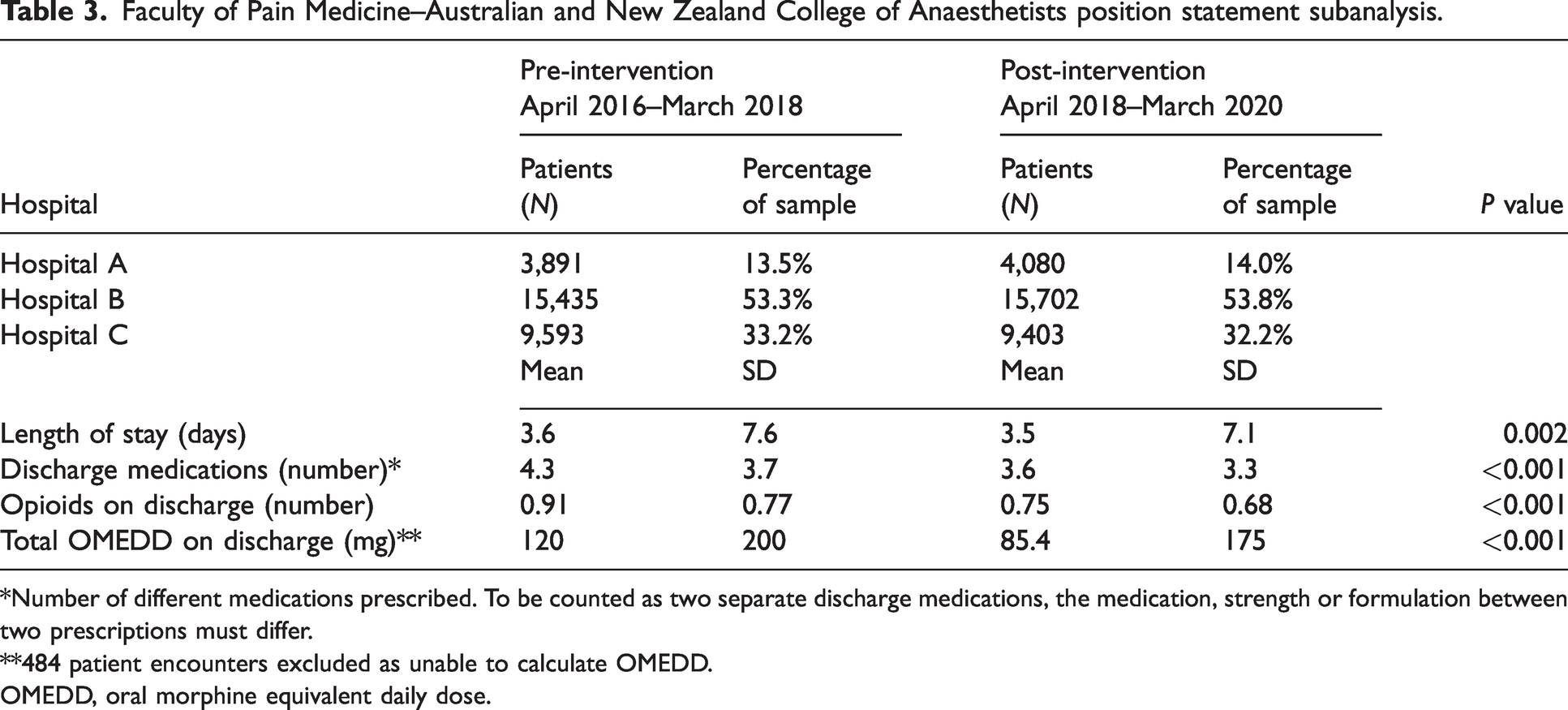
*Number of different medications prescribed. To be counted as two separate discharge medications, the medication, strength or formulation between two prescriptions must differ.
**484 patient encounters excluded as unable to calculate OMEDD.
OMEDD, oral morphine equivalent daily dose.
Real-time prescription monitoring (SafeScript) subanalysis.
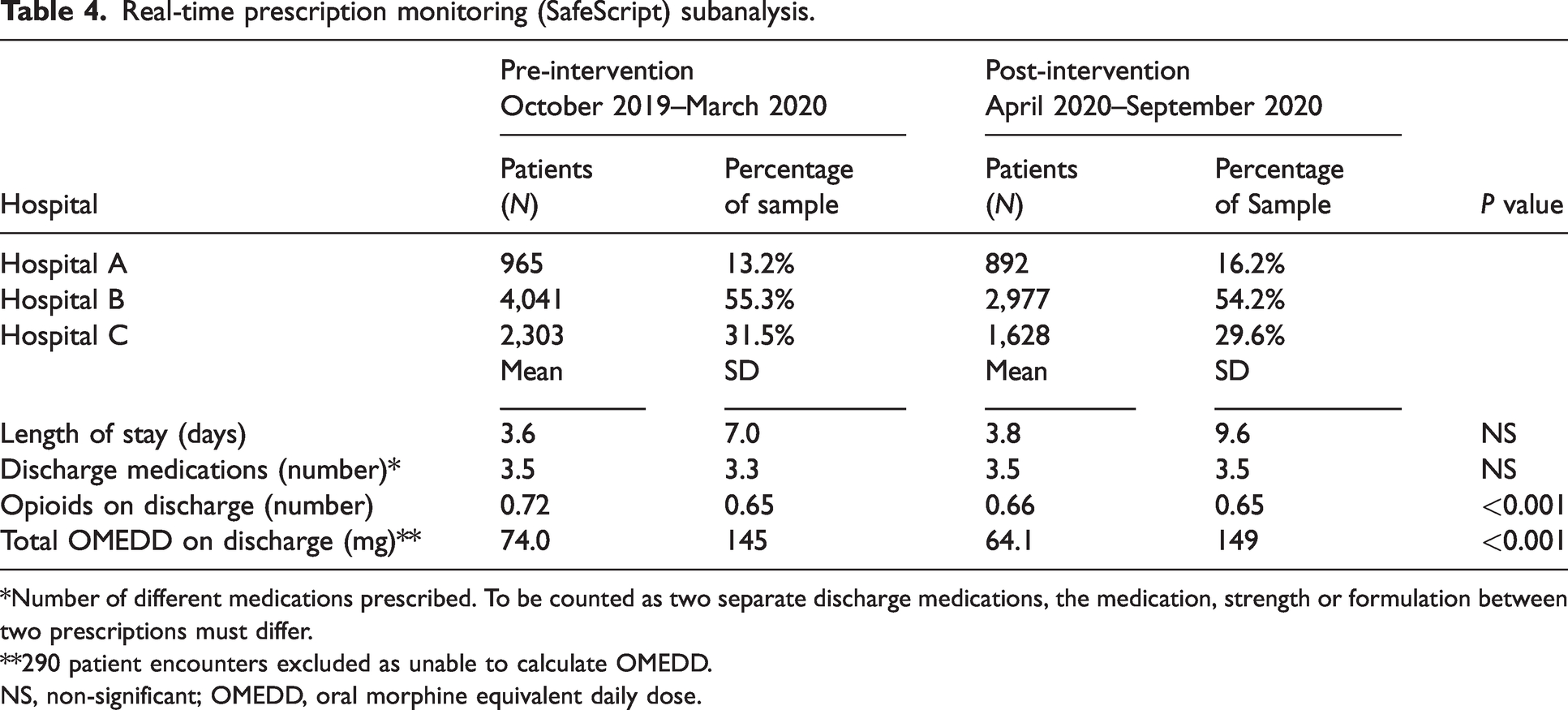
*Number of different medications prescribed. To be counted as two separate discharge medications, the medication, strength or formulation between two prescriptions must differ.
**290 patient encounters excluded as unable to calculate OMEDD.
NS, non-significant; OMEDD, oral morphine equivalent daily dose.
Removal of default eMMS opioid discharge prescribing quantities was associated with a decreased quantity of opioid prescribed (mean OMEDD: 136 mg vs 122 mg,
Both the release of the FPM-ANZCA statement and the introduction of SafeScript were associated with a decreased quantity of opioids prescribed (mean OMEDD 120 mg vs 85.4 mg,
Discussion
This retrospective audit analysed nearly 118,000 surgical discharges and found significant changes in prescribing behaviour at the hospital network which aligned with internal improvement strategies and external changes in the opioid prescribing landscape. There was a general trend at the hospital network over the study period towards reduced quantity of opioid prescribed on discharge and a movement away from codeine and towards oxycodone and tapentadol. Interestingly, this trend was mirrored in a recent report examining dispensing of opioid analgesia in the community across Australia between the 2016/17 and 2020/21 reporting period 8 . This report found a reduction in opioid medicine dispensing rates and overall volume of opioid medicines dispensed, reversing the trend for the reporting period between 2013/14 and 2016/17 in the Third Australian Atlas of Healthcare Variation. 8
The introduction of compulsory manual opioid discharge prescribing quantities in eMMS rather than defaulting to a full pack was a significant internal improvement strategy. A full pack quantity often far exceeded the requirements for the type of surgery and opioid consumption before discharge. 3 While the total OMEDD reduced in this period, there was an increase in the number of different types of opioids prescribed on discharge. This could have been related to the underlying trend in the early 2000s towards the practice of adding slow-release opioids to immediate-release opioids for surgical patients, which was later reversed.
For many anaesthetists and pain specialists, the release of the FPM-ANZCA statement was a considerable catalyst for change. Before the release of Oxycontin® in 1996, it was uncommon to prescribe slow-release opioids for acute pain management, but due to aggressive promotion of this drug in the early 2000s, it became routine practice, even for procedures considered less painful such as superficial and laparoscopic surgery. 21 Unfortunately, this well-intentioned practice in pursuit of the elusive goal of effective pain management for all, resulted in unexpected injury for many and increases in the number of unintentional drug-induced deaths in Australia. 22 As a result of the FPM-ANZCA statement, the health network embarked on an educational campaign and updated practice guidelines to discourage the use of slow-release opioids in acute pain management. These interventions were associated with a reduction in the quantity of opioids prescribed, and the proportion of slow-release opioids prescribed on discharge from surgical units in the hospital network (Figure 3). It is important to note that while the pendulum has swung back to a more opioid-phobic position, this may not deliver high-quality care for individuals and it may not represent more appropriate use of analgesia. 22
Real-time prescription monitoring is now commonplace in many developed countries such as the USA and Canada. Although many states in Australia have now adopted a form of real-time prescription monitoring, the implementation is not uniform or universal.18,19 In Victoria, use of the prescription monitoring system by hospital prescribers remains voluntary, and internal auditing has found uptake to be inconsistent across the health network. For this reason, we theorise that the statistically significant reduction in OMEDD and slow-release opioid prescribing in the SafeScript subanalysis may potentially be related to ongoing cultural changes in response to the FPM-ANZCA statement.
When interpreting the study results, it is important to draw attention to two significant changes which may have influenced surgical prescribing in the health network. The first being the transition to a new hospital at the largest site in 2017. Although no new surgical specialties were introduced, this move coincided with an expansion in the number of operating theatres from 7 to 13, the recruitment of new staff and increased surgical activity. Indeed, this may be one of the factors behind the stabilisation of OMEDD prescribed on discharge during this year (Figure 4). The second factor was the COVID-19 pandemic, which resulted in reduced surgical activity at the hospital network during 2020. This reduction would not have been consistent across all surgical procedures, with emergency and urgent surgeries continuing while minor and elective procedures were deferred. For this reason, the overall complexity of patients during 2020 is likely to be higher than earlier years. Despite this, total opioid prescribing and the use of slow-release opioids during 2020 continued to decline in line with preceding years.
A further factor to consider when interpreting this study is that it is based on prescription data only and may not reflect what was dispensed or taken by patients. Jivraj et al. recently published a population-based, retrospective cohort study investigating postoperative opioid use in 900,989 opioid-naïve patients who underwent surgery between 2013 and 2019 in Canada.23 The primary outcomes of the study were based on prescription-dispensing data but remained generally consistent with the prescribing-based study described above.23 Jivraj et al. identified a reduction in the overall volume of opioid use in Canada towards the later part of the study period, with the proportion of patients receiving opioids and the mean morphine milligram equivalent peaking in 2016. 22 Although Jivrari et al. also found a trend towards decreased codeine use over time, this was being replaced with increasing use of hydromorphone and morphine (in contrast to oxycodone and tapentadol in this study, Figure 2).23
The primary strength of this study was the large sample size, enabled in part by early adoption of eMMS for discharge prescriptions within the hospital network. Although the exclusion of some patients from OMEDD calculations may be considered a limitation of this study, the total number of patients excluded was small (<1%) and therefore unlikely to affect the overall findings. We did not investigate the effects of non-opioid analgesia over this period, and further research is required to quantify the use of multimodal prescribing and the use of gabapentanoids over this period. Finally, the generalisability of this study is limited by its restriction to public hospitals in a specific geographical area and the exclusion of obstetric patients, gynaecology patients or patients from other non-surgical specialties. Further research including non-surgical specialties, private hospitals and those from more diverse areas such as rural and remote locations is required.
This study also has some additional limitation related to the automated data collection through electronic medical record reports. Limited baseline characteristics were available for patients other than simple demographic data which may have added value to generalisability of the results. Lack of subanalysis by type of surgery was another limitation which may have yielded more valuable results for other hospital networks with a different surgical caseload. Data were handled as independent for the purposes of analysis in this study but in reality, it is likely that some patients may have had one or more encounters during the study period. Matching was not possible due to the lack of unique identifiers in the data set (each hospital in the study utilised a different patient identification number, preventing accurate data matching between hospitals). Lastly, there was overlap between the different events specifically analysed in this study and, due to time constraints, there was no secondary subanalysis with narrower timeframes to account for this, which may have affected the primary outcomes.
This study demonstrated the importance of internal improvement strategies, including system optimisations (eMMS changes, real-time prescription monitoring) and professional advocacy from clinical leaders (FPM-ANZCA statement) and responsiveness to external changes in the opioid prescribing landscape to ensure appropriate opioid prescribing on discharge from hospital. Further planned work to improve prescribing practices in relation to opioids in the hospital network includes the implementation of a multidisciplinary analgesic stewardship service. This will be a dedicated service consisting of medical, nursing and pharmacy staff providing policy review, education and advocacy roles. This service will also be responsible for auditing and monitoring inpatient and discharge analgesic prescribing. It is anticipated that the introduction of the analgesic stewardship service at the health network will further impact these outcomes.
