Abstract
Effective pain control after major surgery in neonates presents many challenges. Parenteral opioids (and co-analgesics) are often used but inadequate analgesia and oversedation are not uncommon. Although continuous thoracic epidural analgesia is highly effective and opioid-sparing, its associated risks and the need for staff with specialised skills and/or neonatal intensive care unit staff buy-in may preclude this option even in many academic centres. We present the case of a six-day-old infant who underwent upper abdominal surgery and received intermittent morphine doses via a tunnelled caudal epidural catheter, which provided satisfactory analgesia and facilitated early extubation.
Providing postoperative analgesia using intravenous opioids while minimising opioid-induced respiratory depression can be challenging in neonates, as pain assessment is relatively difficult and apnoea is common in this population. Multiple observational studies have shown that neuraxial analgesia improves postoperative respiratory function and facilitates earlier extubation in infants and children.1 –8 Unfortunately, in neonates, placing an epidural catheter at the thoracic level, or threading it to the thoracic level via a lumbar or caudal entry 8 requires skills that not all paediatric anaesthetists possess. Furthermore, many neonatal intensive care units (NICUs), ours included, do not permit continuous epidural local anaesthetic (LA) infusion. In contrast, caudal block is commonly performed by all paediatric anaesthetists. Caudal morphine has a long duration of action and requires no continuous infusion.
We present the case of a neonate who received a caudal epidural catheter for intermittent morphine doses for post-laparotomy analgesia. This report complies with the Declaration of Helsinki, has parental consent, and was approved by the Queen’s University research ethics board (approval no. 6030182).
Case report
A six-day-old 2.6 kg infant girl, born at 38.5 weeks, had a type IV congenital hiatal hernia, a small patent ductus arteriosus, unilateral renal agenesis and dysmorphic features (hypertelorism, under-folded bilateral helices, overlapping toes, flat nasal bridge, scalp port-wine stain). She underwent open repair of the hiatal hernia and partial fundoplication via a left subcostal incision. As there is no specific policy/guideline for postoperative pain management in our NICU, the attending paediatric anaesthetist, neonatologist, paediatric surgeon and NICU charge nurse agreed preoperatively on the placement of a caudal catheter for intermittent morphine administration as the postoperative analgesia strategy. This decision was based on the literature and on predicting that regional analgesia would have less respiratory depressant effect and better analgesia than intravenous opioids in this syndromic baby. General anaesthesia was induced with propofol and rocuronium, followed by uneventful tracheal intubation with a size 3.0 cuffed endotracheal tube. General anaesthesia was maintained with sevoflurane (minimum alveolar concentration ∼0.7) and fentanyl infusion of 2–4 μg/kg/hour tapering to 1 μg/kg/hour in the final 45 minutes. Surgical time was 2.5 hours and the procedure was completed uneventfully. The surgeon infiltrated the subcostal wound with plain bupivacaine 0.25% 2.5 ml prior to skin closure.
On completion of the surgery, an 18-gauge cannula was inserted into the caudal epidural space via the sacral hiatus, through which a 20-gauge epidural catheter was threaded to approximately 2 cm measured at skin level. The catheter was then tunnelled subcutaneously to exit at the buttock (Figure 1). Isotonic preservative-free morphine LP (Sandoz Canada, Boucherville, Quebec, Canada) 100 μg (38 μg/kg) diluted with normal saline to 2 ml was given via the catheter. Paracetamol 80 mg was administered rectally.
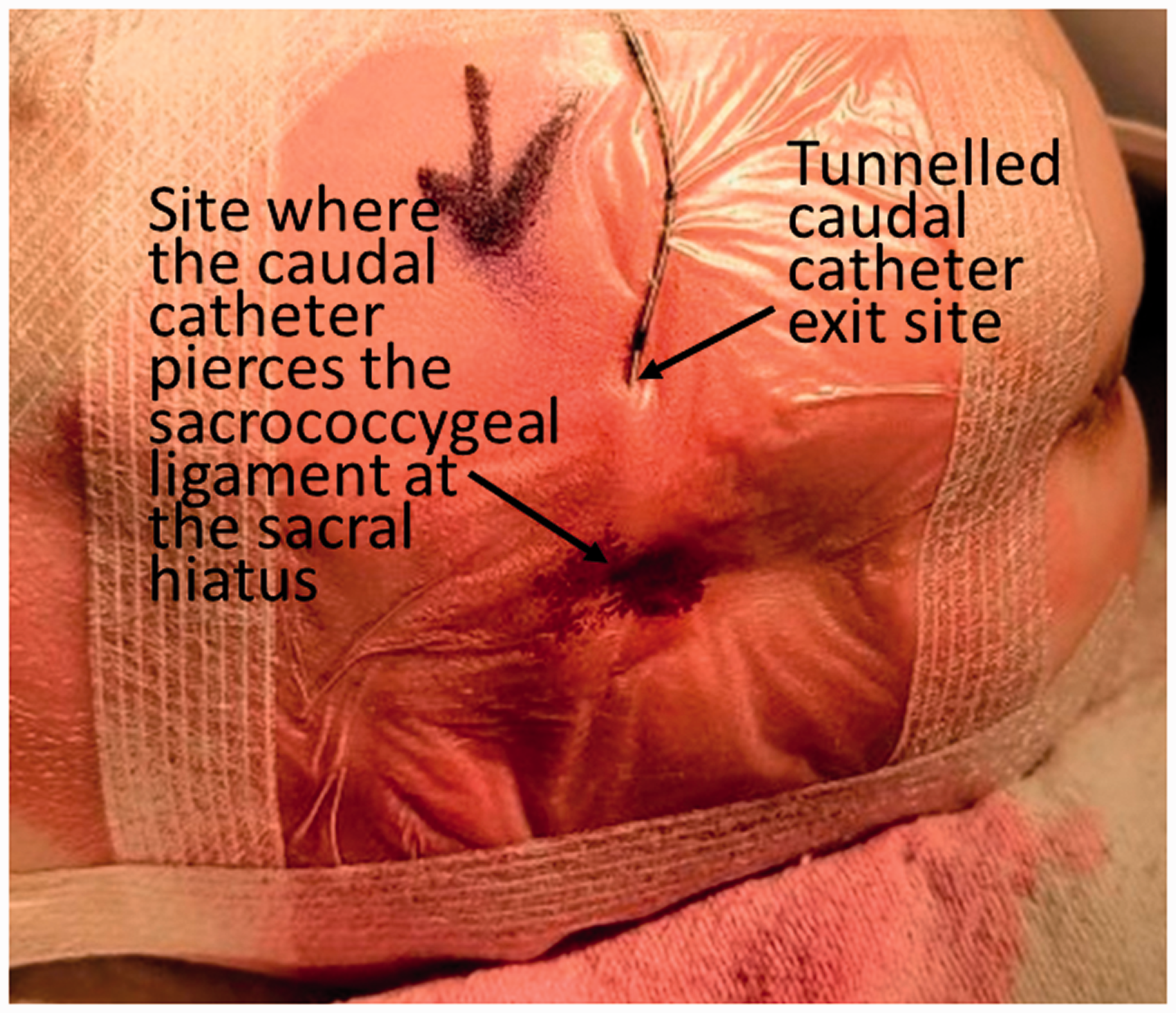
With the neonate in a lateral position, a 20-gauge epidural catheter was sited caudally and tunnelled subcutaneously with the exit site at the buttock.
Postoperatively, the baby, still intubated, was transported to the NICU at which point her fentanyl infusion was stopped. While intubated the neonate had transcutaneous carbon dioxide monitoring. Her trachea was extubated two hours later, and she did not require further respiratory support or supplemental oxygen. Throughout the neonate’s NICU stay she remained on continuous heart rate, respiratory rate and oximetry monitoring. She did not have an arterial line and her blood pressure was monitored with a cuff every one to four hours for the first 72 hours postoperatively and every eight hours afterwards. In addition, premature infant pain profile (PIPP) 9 scores were recorded hourly for the first 12 hours and every three hours thereafter.
Twelve hours after the first epidural morphine dose, the baby became irritable, and had PIPP scores of 7 to 8, for which she was given intravenous fentanyl 1.5 μg. She settled for the ensuing 12 hours (PIPP score 4). Twenty-four hours after the first epidural morphine bolus, when she became unsettled again, with PIPP scores of 4 to 7, caudal morphine 100 μg in 2 ml was given by the on-call paediatric anaesthetist. Subsequently, she remained stable, with PIPP scores lower than 4. She received two more doses of epidural morphine by the on-call paediatric anaesthetist at 12-hour intervals, and the catheter was removed 48 hours postoperatively. No respiratory depression, urinary retention, or pruritis was observed. She was given paracetamol for the first 48 hours postoperatively, and as necessary thereafter. She did not require systemic opioids after the last caudal injection. Paracetamol was discontinued on postoperative day seven, when she was advanced to full enteral feeds.
Discussion
Pain after major surgery in neonates can lead to chest splinting, cardiorespiratory compromise, metabolic sequelae and, in some, intraventricular haemorrhage. Pain is potentially neurotoxic to the developing brain, with long-term sequelae. 10 Intravenous opioids are commonly used in NICUs for postoperative pain. Both pain and excessive opioids delay tracheal extubation and increase the risk of reintubation. With intravenous opioids, after major neonatal surgery, it is uncommon to extubate the trachea before six to 24 hours postoperatively.
Neuraxial block is opioid-sparing and offers superior analgesia after thoracic and abdominal surgery in infants and children.1 –8 However, there is resistance to epidural analgesia in many NICUs. The reasons include: (a) there are no randomised controlled trials comparing neuraxial versus intravenous opioid analgesia for postoperative pain and respiratory depression in neonates; (b) lumbar and thoracic epidural catheterisation in neonates is technically challenging, 8 and not without risks of spinal cord injury, local anaesthetic systemic toxicity (LAST) and infection; (c) while ultrasound, electrocardiographic guidance, or electrical stimulation may facilitate the positioning of the epidural catheter tip at the desired thoracic level from a lumbar or caudal entry,8,11,12 one third of ‘blindly’ inserted caudal-to-thoracic catheters fail;12,13 (d) LAST in neonates may be difficult to recognise; (e) hypotension, which is of concern in neonates (especially those with certain cardiac anomalies), can occur; (f) afterhours, paediatric anaesthetic support for trouble-shooting epidural issues and pain may be inconsistent; (g) there are no standard management protocols for neonatal neuraxial analgesia; and (h) NICU staff are already proficient with managing intravenous opioid analgesia.
Our NICU is one of five Level 3 tertiary care centres in Ontario, Canada. We have access to some paediatric subspecialties, including paediatric general surgery, but do not have access to subspecialties such as cardiac, otolaryngology or urology, and we do not have a paediatric surgical fellowship programme. In our NICU (24 beds; 400 admissions/year), there are low volumes of major thoracic and abdominal surgery, making it difficult to maintain the expertise needed to sustain an epidural programme. Even in large NICUs (e.g. Toronto, Ottawa, Ontario; Halifax, Nova Scotia; Edmonton, Calgary, Alberta; Columbus, Ohio), epidural analgesia is rarely (if ever) used. Requests for outside assistance in acute pain management in our NICU are rare and are directed to the on-call paediatric anaesthetist. The NICU bedside team (nurse and physiotherapist) are well trained and receive regular training and skill maintenance. Our NICU uses sophisticated equipment (transcutaneous and end-tidal carbon dioxide monitoring, oxygen saturation, and continuous monitoring from a central/control room for heart rate, respiratory rate, apnoea alarm, blood pressure), and provides a low (up to 1:1) nurse-to-patient care ratio. These safety measures are important to prevent complications, including respiratory depression. In addition, we carry out frequent bedside multidisciplinary (nurse, physiotherapist, neonatologist and, as appropriate, surgeon and anaesthetist) rounds on all patients.
Injection of LA into the caudal space is technically simple, is standard paediatric anaesthesia practice, and is suitable for pelvic and lower abdominal surgery. For thoracic and upper abdominal surgeries, however, LA administered caudally requires large volumes, risking LAST, hypotension and decreased analgesia because of the use of dilute LA.
By comparison, caudal morphine does not cause LAST, hypotension or motor block. Its prolonged analgesic effect obviates the need for continuous infusion. Still, to extend postoperative analgesia, a catheter is needed. Siting a catheter in the caudal space (as opposed to a thoracolumbar epidural catheter via a caudal entry) for repeated morphine dosing has been described in children, 14 but apparently not in newborns. Our goal was to provide prolonged analgesia with the least maintenance and care burden on NICU staff. We were able to arrive at a multidisciplinary consensus that intermittent caudal morphine was possibly superior to intravenous opioids, with better analgesia and less respiratory depression in this neonate with a possible syndrome. The agreement was that our paediatric anaesthesia team would oversee the management of the caudal catheter and the patient’s analgesia, the catheter would be capped, and that no infusion pump would be used.
Morphine in the caudal space is carried rostrally in the cerebrospinal fluid, and provides effective analgesia following thoracoabdominal surgery. Breakthrough pain from coughing and excessive handling can occur, but analgesia is excellent in a quietly breathing neonate. The analgesic and respiratory effects of caudal morphine in neonates are largely unknown and could only be extrapolated from data in infants and older children. In 138 children, 15 of whom were less than one month old and another 27 between one and six months of age, high lumbar or low thoracic morphine 70 μg/kg provided effective analgesia, with 86% of patients extubated within two hours postoperatively and 75% of patients not requiring supplemental analgesia for the first ten hours. 15 There were 11 cases of respiratory depression (the youngest was approximately seven weeks old). 15 In 500 children, of whom the majority were less than five years old and 23 were three months old or less, caudal morphine 30 μg/kg and 40 μg/kg provided analgesia for 15 ± 5.5 hours and 13.9 ± 5.3 hours, respectively, with no respiratory depression. 16 Use of single-dose morphine of 60 μg/kg caudally in 113 paediatric patients (aged two months to 15 years, weight 3.4–98 kg) resulted in excellent analgesia in most of them during the first 24 hours. 17 None developed respiratory depression. 17 Several studies in older children (one to 12 years of age) also showed excellent analgesia, with rare respiratory depression (only requiring naloxone when morphine 70 μg/kg was used).14,18 –21
In our syndromic neonate, we were concerned about the risk of opioid-induced respiratory depression, whether administered intravenously or epidurally, after major surgery. As cited earlier,14 –21 caudal morphine 100 μg/kg provides the longest analgesia, but with an increased risk of respiratory depression, whereas lower doses also provide analgesia and with a low risk of respiratory depression.14 –21 Having a catheter in situ allowed us to maintain analgesia while minimising respiratory depression through repeated injections of smaller doses of morphine. This case was similar to a previously reported case in our NICU, in which tracheomalacia after tracheo-oesophageal repair caused near-asphyxiation during crying episodes, which was managed with repeated caudal epidural morphine 40 μg/kg injections over several days. 22 The lesson learned from that case led us to site a catheter in this case.
Of note, there are potential catheter-related risks with this technique. Fortunately, meningitis, epidural abscess or sepsis are rare, 23 and subcutaneous tunnelling of the catheter can further reduce the risk, 24 as can an aseptic technique and keeping the catheter in situ for no more than two to three days.
Conclusion
Siting a catheter for repeated morphine dosing into the caudal epidural space for postoperative analgesia in neonates has, to the best of our knowledge, previously not been reported. In our case, caudal injection of morphine 38 μg/kg every 12–24 hours provided satisfactory analgesia after upper abdominal surgery. Based on the literature on infants and older children, it may have a very low risk of respiratory depression. Even so, low nurse-to-patient ratios and frequent vital signs monitoring (including pulse oximetry) together with PIPP monitoring are standard of care as neonates are prone to apnoea, regardless of the route of opioid administration. The respiratory depressant effect of caudal morphine is insidious, and continuous transcutaneous carbon dioxide monitoring would detect early onset. Naloxone (0.01–0.02/mg/kg/dose; repeated every two to three minutes as necessary) must be immediately available.
The advantages of regional analgesia have been well documented in adults. Similarly, caudal morphine leads to early extubation and minimal supplemental analgesic requirements in small children. 17 Nevertheless, the relative risk:benefit ratio for regional versus intravenous opioids in neonates is unknown. Without robust evidence to show the superiority of regional analgesia in neonates, a culture change of switching from intravenous opioids for postoperative pain management in many large NICUs is difficult. If change should occur, it will require close collaboration between NICU staff, and enough paediatric anaesthetists who are proficient with neonatal neuraxial analgesia and who can provide 24-hour support. Paediatric anaesthetists appreciate that the technical challenges and problems associated with caudal-to-thoracic catheters8,11 –13 are largely avoided when the catheter tip stays within the caudal space. Respiratory depression is a potential risk, and requires careful consideration and appropriate supportive care; in this case with a lower dosage there was no respiratory depression. Our case illustrates that there is a middle ground on which neonates can enjoy the benefits of neuraxial analgesia based on an uncomplicated, less invasive, more reliable and possibly safer technique that all paediatric anaesthetists are comfortable with, and one that does not place undue additional burden on the NICU staff. Finally, while both caudal morphine and intravenous fentanyl are viable techniques in many Level three NICUs, it would be of interest to explore the advantages and disadvantages of both techniques in settings where resources are not as abundantly available.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
