Abstract
A 77-year-old lady with a laparoscopic adjustable gastric band (LAGB), implanted 12 years earlier for obesity, developed an unusual but almost fatal complication, characterised by dysphonia and stridor within minutes and a tensely swollen anterior neck. The condition mimicked haemorrhage into the subcutaneous tissues of the neck, and the airway was secured with an awake fibreoptic intubation. Subsequent computed tomography imaging of the abdomen and chest revealed megaoesophagus with dilatation up to 7 cm, proximal to the gastric band. The band was then deflated percutaneously with immediate resolution of the neck swelling. In patients with LAGB in situ, fluid should be removed from the band for any suspected device-associated symptoms such as severe vomiting, severe gastro-oesophageal reflux or neck swelling and respiratory distress. When there is radiological evidence of gross oesophageal dilatation or a high likelihood of an obstructive pathology at the level of the band, a careful nasogastric tube decompression may also resolve reflux, neck swelling or respiratory distress. A complication associated with LAGB should be considered in a patient with unexplained neck swelling or other features of airway obstruction who has an LAGB in situ.
Introduction
The popularity of laparoscopic adjustable gastric banding (LAGB) as a treatment for morbid obesity has declined over the last decade in view of the high failure rates and complications. 1 However, patients with these devices are still likely to be encountered in the emergency and perioperative settings. We report a patient who presented with airway compromise secondary to megaoesophagus and tracheal compression, a rare complication of LAGB. The purpose of this report is to outline the importance of recognising this life-threatening complication in the context of LAGB, which may resolve with careful decompression of the stomach with a nasogastric tube and fluid removal from the band by deflating the subcutaneous port. This patient gave written consent to publish this report.
Case report and management
A 77-year-old woman was visiting her husband in hospital when she complained of sudden dysphagia and severe dyspnoea after consuming soup. Within minutes, she developed a hoarse voice and stridor, and was immediately transferred to the emergency department. She had a background history of hypertension and single-vessel coronary artery disease. She had undergone laparoscopic insertion of an adjustable gastric band 12 years earlier for treatment of obesity. The patient experienced occasional swelling on the right side of her mid neck, explained to her as an oesophageal pouch by her bariatric surgical team.
At the onset of her symptoms, her oxygen saturation was 92% on room air, improving to 94%–95% on 6 l/min oxygen via a low-flow simple facemask. She was noted to be tachycardic (110/min) and tachypnoeic (22/min). Once the patient was examined in the emergency department, an urgent call was made to the on-call anaesthesia and ear, nose and throat (ENT) teams. It was now evident that her entire anterior neck had become tensely swollen over 15–20 minutes, and a presumptive diagnosis of spontaneous bleeding into the soft tissues of the neck was made.
A portable anteroposterior chest X-ray showed ‘some gaseous distension of the oesophagus and some fluid in the distal oesophagus’. Although the gastric band was evident, the rotation of the patient along with accentuation of the thoracic kyphosis caused significant distortion of the mediastinal structures, and hence megaoesophagus was not apparent (see Figure 1). As the patient was in respiratory distress with imminent airway obstruction, securing the airway and preventing aspiration was prioritised. She was therefore transferred to the operating theatre for airway management and further evaluation. An awake intubation approach was chosen in view of the uncertainty of the cause of airway obstruction and the likelihood of complete airway collapse with unknown lower airway pathology following neuromuscular blockade. The ENT surgeons were present in theatre for the backup plan of a surgical airway.
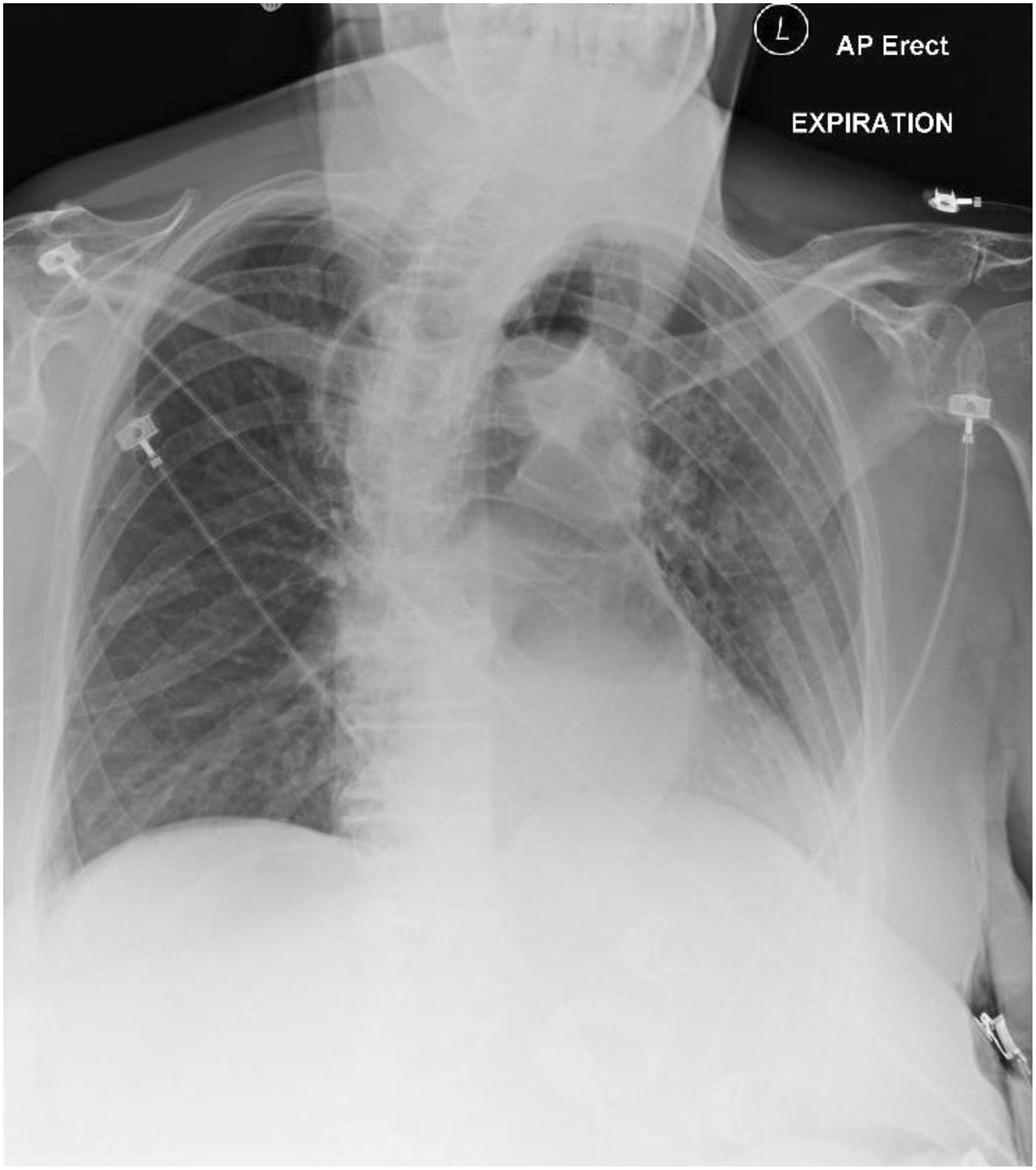
Chest X-ray anteroposterior view showing some gaseous distension of the oesophagus and some fluid in the distal oesophagus. Megaoesophagus was not evident/apparent.
Intravenous glycopyrrolate (200 μg) was used as an antisialagogue. Airway topicalisation was achieved using a proprietary preparation of phenylephrine hydrochloride 0.5% and lidocaine hydrochloride 5% (CoPhenylcaine
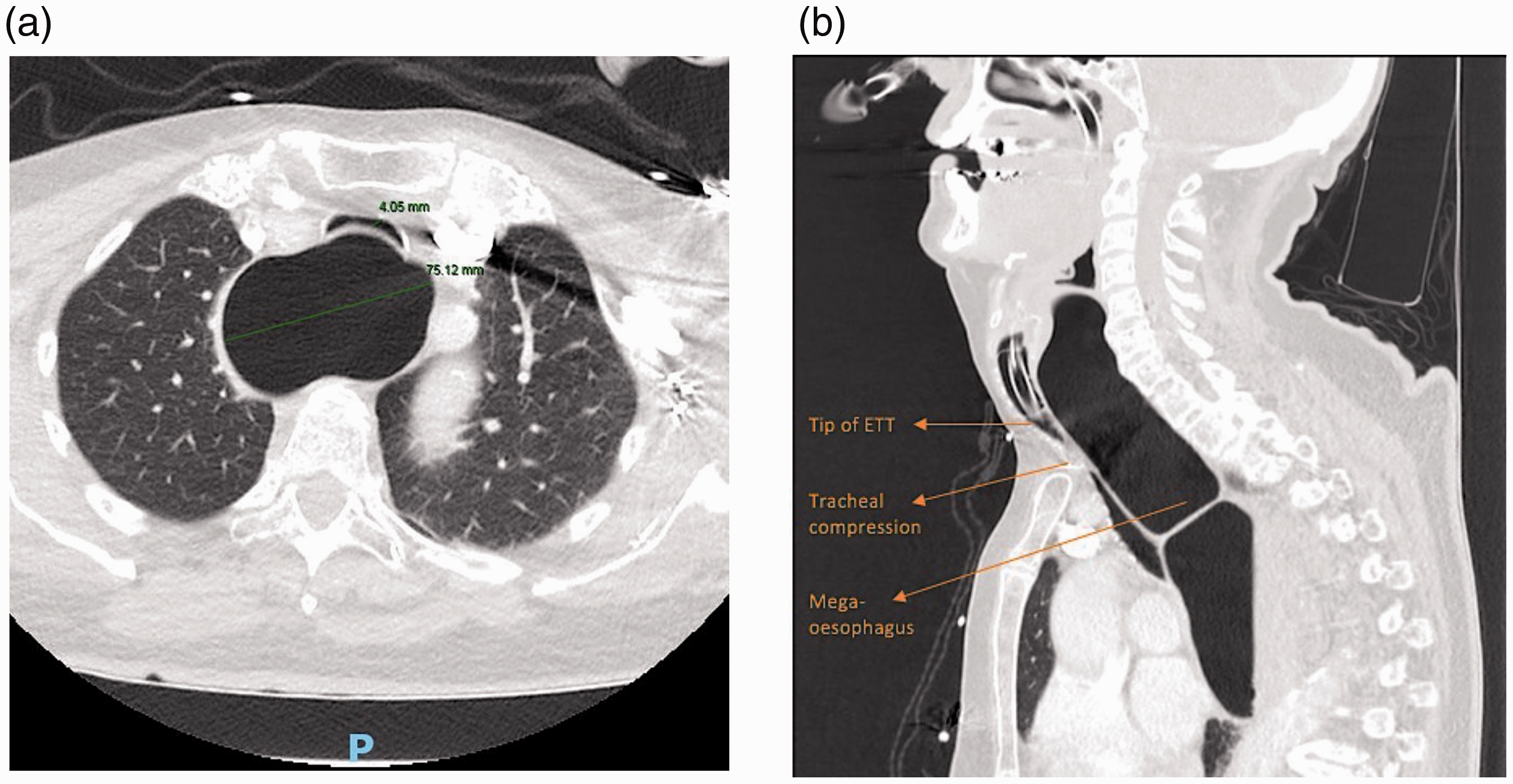
Computed tomography scan of the chest. (a) Sagittal and (b) coronal views showing gross dilatation of the entire oesophagus to the gastric band, and tracheal compression (4 mm) above the level of carina.
She was transferred to the intensive care unit for elective ventilation in view of anticipated airway oedema and to facilitate gastroscopy the following morning. Absence of an oesophageal pouch was confirmed endoscopically, and a diagnosis of megaoesophagus was ascertained. The patient had an uncomplicated extubation after 14 hours of ventilatory support. During step-down to the high dependency unit, she developed chest pain and was diagnosed with non-ST elevation myocardial infarction and underwent percutaneous coronary intervention with deployment of two stents. We couldn’t ascertain whether the cardiac event was incidental or related to the trauma of the complication. She made an uneventful recovery and was discharged with follow-up advice for band removal from her bariatric surgical provider. The anaesthetist involved in the case management (RL) educated the patient and organised an alert letter highlighting the importance of band deflation for future emergencies.
In the next three years, the patient gained weight during a time of personal grieving, and she wanted the band to be reinflated in an attempt to reduce weight, given her increasing cardiac morbidity due to ischaemic heart disease and obstructive sleep apnoea. Investigations including a CT scan of the abdomen and barium swallow ruled out complications such as band slippage. It was then carefully inflated with fluid by her bariatric surgical provider in line with the patient’s wishes. Three years following this, the patient re-presented to our hospital with hoarseness of voice, stridor and acute airway obstruction. The patient had retained the previously issued alert letter, and based on that instruction, the band was deflated immediately in the emergency department, with swift resolution of her airway symptoms. The band remains in place in a deflated state, as it was noted by her surgical provider to be in a good condition.
Discussion
An LAGB procedure involves placing an adjustable low-pressure silicone band around the upper gastric area (see Figure 3) below the gastro-oesophageal junction. 2 The stoma of the band can be inflated or deflated by saline injection or aspiration via subcutaneous port fixed to the anterior rectus sheath at the left upper quadrant of the abdomen. 3 The main mechanism of action is creating a small proximal gastric pouch which will restrict portion size. Satiety stimulation has also been suggested as a probable mechanism of action. 2 Oesophageal motility disorders are common after LAGB insertion, and manifest as fluid and food accumulation in the gastric pouch, regurgitation, and dysphagia.2,4 LAGB can impair the relaxation of the lower oesophageal sphincter and can also affect oesophageal peristalsis and can potentially lead to oesophageal dilatation.5,6 Oesophageal dilatation is further attributed to a combination of overeating, an overinflated band, an underlying motility disorder, and psychological factors. 7 On rare occasions, aperistalsis and severe dilatation can lead to megaoesophagus or pseudoachalasia, characterised by either a 100% increase from baseline diameter or an enlargement >50 mm in diameter.5,8 This mimics major achalasia cardia–like dilatation, and warrants band removal. 2 Notably, the normal oesophageal diameter is around ≤35 mm. 2 Megaoesophagus is suspected when patients report severe symptoms of dysphagia, vomiting, regurgitation and gastro-oesophageal reflux.5,9 A prospective clinical trial across 167 LAGB procedures from our institution revealed disordered oesophageal motility in 68% of patients, oesophageal dilatation in 25% (with a mean diameter of 47 mm), and megaoesophagus requiring band removal in six patients. 2 Megaoesophagus can present anywhere from three to ten years after LAGB insertion, 2 and thus these presentations can be encountered even now in patients with existing LAGBs. Band removal is recommended once megaoesophagus is diagnosed to prevent any irreversible motility disorders of the oesophagus.7,9 It is possible that the occasional mid-neck swelling in our patient that was described as a pouch could be a result of longstanding band overinflation and/or overeating. 10
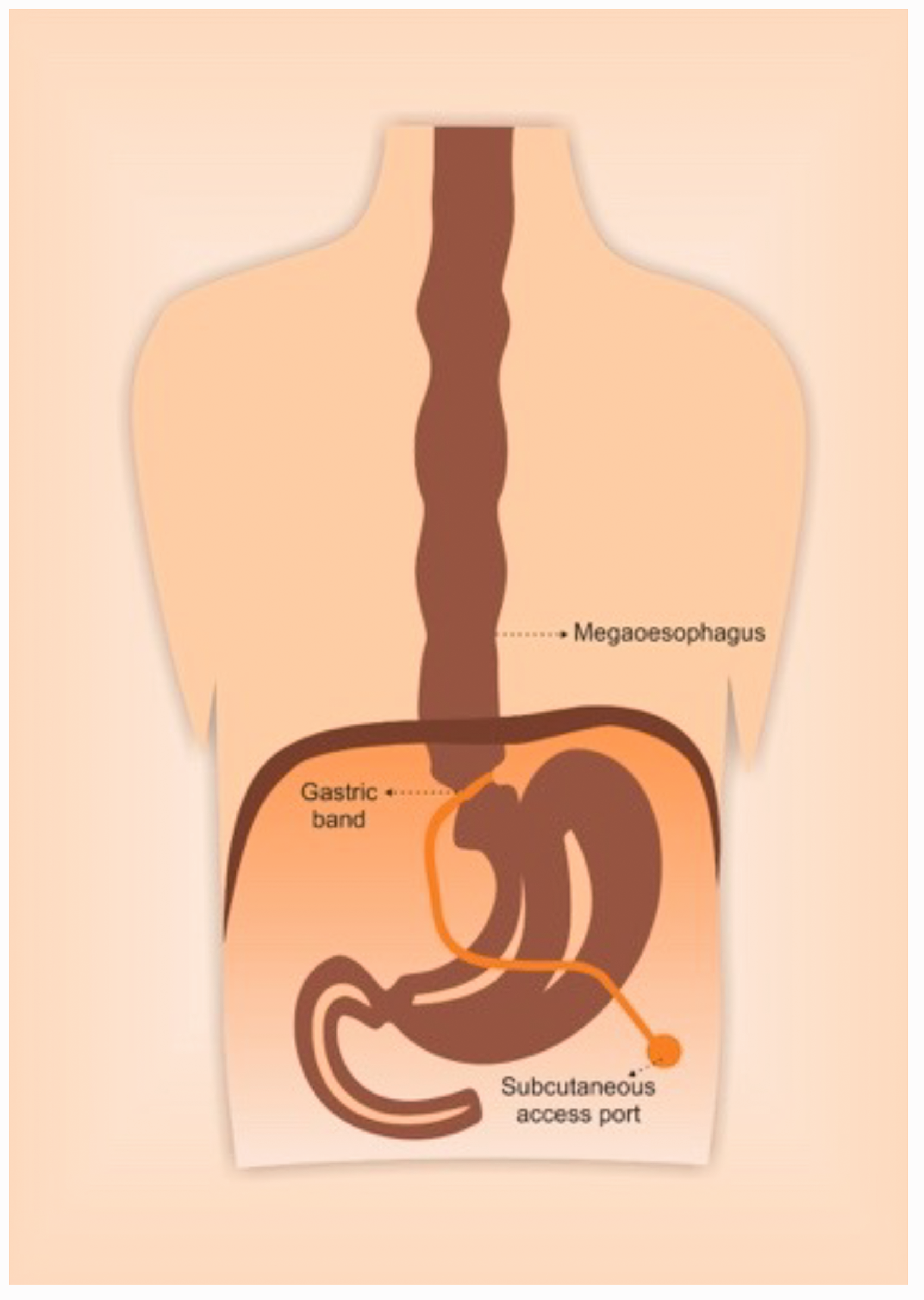
Schematic illustration of the adjustable gastric band in situ, the subcutaneous access port and the distended oesophagus (megaoesophagus).
Two published cases have reported acute airway compromise secondary to megaoesophagus related to LAGBs.7,11 The first report described acute respiratory distress within 24 hours of an abdominoplasty in a 39-year-old female patient. 11 A CT scan was performed to rule out pulmonary embolism, and it revealed severe oesophageal dilatation up to 47 mm in diameter. The patient’s symptoms improved immediately following the band release. The second case was a 43-year-old female who presented to the emergency department with respiratory distress and inspiratory stridor. 7 The megaoesophagus was evident in both chest X-ray and CT scan, and the symptoms resolved with band deflation. In our case, although the pre-existing LAGB was known and the oesophagus was dilated up to 75 mm (CT scan finding), the chest X-ray was inconclusive of megaoesophagus. Conceivably, an earlier port deflation would have probably improved the symptoms in our patient. Bariatric surgery expert opinion suggests that fluid should be removed from the band, and bariatric surgery team liaison should occur for any suspicion as to the role of an LAGB in a clinical scenario. 3 The perioperative indications for fluid removal from the band include nausea, vomiting, regurgitation and bowel obstruction. 3 The techniques of fluid removal from the band are described in detail elsewhere, and readers are encouraged to refer to this. 3 Indeed, band emptying and removal are also the definitive management for recurrent aspiration as a late complication associated with LAGBs. 12
There are a few cases of airway obstruction from megaoesophagus in the setting of achalasia cardia reported in the literature.13,14 In those, oesophageal decompression using a nasogastric tube has been shown to resolve symptoms immediately.13,14 A careful decompression of the oesophagus using a nasogastric tube insertion would also reduce the risk of oesophageal rupture. In our case, as the megaoesophagus was not evident until CT imaging, neither nasogastric decompression nor band deflation were considered.
LAGB is currently offered for select subgroups, such as the young, and in planned pregnancies in those with binge-eating patterns. Our report highlights megaoesophagus following LAGB as a potential life-threatening cause of airway compromise, and it should be considered as a differential diagnosis in presentations of respiratory distress in patients with implanted LAGBs. If the patient’s history of LAGB is unclear, the subcutaneous access port in the left upper quadrant of the abdomen may be palpated, apart from recognising previous trocar scars in suspected patients. After weight regain, it may be difficult to locate/palpate the band. A plain abdominal X-ray will reveal the port, tubing and band very well.
In cases with a known LAGB in situ, even if the diagnosis of megaoesophagus is not evident from bedside radiology, port deflation should be attempted to resolve symptoms. 3 In our opinion, there would need to be radiological evidence or a high likelihood of an obstructive pathology at the level of the band before a blind insertion of a nasogastric tube. Before attempting a blind insertion, knowledge of the level of the obstruction from the band will guide careful advancement of the nasogastric tube to that level, for instance 40 versus 20 cm from the nostrils. Nasogastric tubes are intended for the highly distensible normal stomach that can hold up to 1600 ml of air with minimal alteration in intragastric pressure. 15 Intestinal perforation has been reported with nasogastric tube insertion in the setting of gastric bypass, attributed to the small gastric pouch before the anastomosis. 16
In conclusion, we report a case of megaoesophagus secondary to a complication of LAGB presenting as neck swelling and airway compromise. The megaoesophagus was not evident in the emergency department, and the airway was secured with an awake fibreoptic intubation. A subsequent CT scan revealed megaoesophagus, and the band was then deflated under ultrasound guidance with immediate resolution of neck swelling. In cases of neck swelling or acute airway obstruction in patients with a history of LAGB, band deflation should be attempted to resolve symptoms. If there is radiological evidence or a high suspicion of an obstruction at the level of the band, a careful nasogastric tube placement just above the level of the obstruction may aid in decompressing the oesophagus and resolve symptoms.
Footnotes
Author Contribution(s)
Declaration of conflicting interests
The authors have no conflicts of interests to declare.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
