Abstract
Amyotrophic lateral sclerosis, or motor neuron disease, is an uncommon progressive neurological disorder. Professionals working in the perioperative field may encounter patients with amyotrophic lateral sclerosis only rarely. The relevant published literature on amyotrophic lateral sclerosis is broad in scope, but a contemporary review focused on the perioperative period is absent. This structured narrative review seeks to provide a summary of the contemporary management of patients and then focuses on eliciting if there are perioperative management considerations specific to amyotrophic lateral sclerosis that can be optimised. A comprehensive structured narrative literature review, including grey literature searching, indicated worsening ventilatory failure is of prime concern but that patients may present with a broad range of neurological symptoms, and that cardiovascular and cognitive dysfunction specific to amyotrophic lateral sclerosis may exist and be occult. Exacerbation of neuromuscular weakness during the perioperative period is multifaceted and requires the application of a high standard of the core principles of surgical and anaesthetic management of neuromuscular disease. Standard perioperative approaches require rigorous attention and potential exists for significant alteration. There is a potential high risk of postoperative increased morbidity from neurological decline and mortality from pulmonary complications. A meticulous approach to planning preoperative assessment, shared decision-making, intraoperative and postoperative care is required.
Keywords
Introduction
Amyotrophic lateral sclerosis (ALS) is a progressive disorder of upper motor neurons (UMNs) and lower motor neurons (LMNs) 1 with a lifetime risk of approximately 2.1–2.9 per 1000. 2 It is an uncommon disease that presents complex challenges to all involved in patients’ perioperative care. The core feature is progressive loss of muscular strength leading to death a median 30 months from diagnosis, usually from ventilatory failure. 1 The often rapidly progressive and fatal nature of the disease means perioperative healthcare professionals infrequently manage patients with this condition. This review provides a contemporary summary of the general management of ALS to assist perioperative healthcare professionals in assessing patients and addresses a current gap in the published literature by consolidating and critiquing evidence specific to the management of ALS in the perioperative period.
Methods
The review was conducted with an electronic search of PubMed using ‘motor neurone disease’ OR ‘amyotrophic lateral sclerosis’ AND ‘an*esthesi*’ OR ‘an*estheti*’ OR ‘surgery’. Articles were reviewed by title, then abstract and potentially relevant publications underwent full text review. Reference lists were reviewed for additional literature. An initial synthesis structure was produced and then due to the diverse nature of the subject, supplementary subsection searches were undertaken replacing search terms with alternatives such as ‘tracheostomy’. Grey literature review was undertaken, including a review of websites of relevant professional and disease-specific organisations. The multidisciplinary team of authors added literature relevant from their own backgrounds in anaesthesia, perioperative medicine, intensive care, neuromuscular disorders, long-term ventilation and general surgery. The last electronic search was undertaken on 16 November 2021 and identified 154 relevant published and grey literature references for inclusion.
Background
Aetiology
The exact aetiology of ALS is uncertain. There is multifactorial interaction of genetics and environmental insults. 2 Insults to pre-existing neuronal susceptibility leads to neuronal injury and a cascade of cell death, specifically the degeneration of UMNs and LMNs in the motor cortex, brainstem nuclei and anterior horn cells of the spinal cord. 3
Epidemiology
The incidence of ALS varies globally; 2.6 women and 3.9 men per 100,000 people in a United Kingdom database developed ALS over 15 years. 4 The peak incidence was between ages 75 and 79 years. This is similar in the United States 5 and New Zealand 6 but regional variations exist that cannot currently be explained by demographic distributions.
Presentation
Neurodegeneration of motor neurons leads to progressive skeletal muscle weakness. Individual presentations vary and there may be purely upper or lower motor signs and symptoms. Damage of the corticospinal and corticobulbar tracts leads to UMN signs. Anterior horn cell damage leads to LMN signs. There is near equal presentation between upper limb, lower limb and bulbar onset disease. 5 Classic presentation is the insidious onset of mixed UMN and LMN symptoms in the limbs. Bulbar presentations include dysarthria, dysphagia and sialorrhoea. Less typical presentations include weight loss, respiratory failure, autonomic dysfunction and frontal lobe dysfunction.2,7,8 Patients may present in the perioperative period with a wide range of symptomatology.
Diagnosis
Diagnosis is made primarily on clinical grounds and the exclusion of other possible diagnoses with investigation. The Revised El Escorial criterion provides a framework, particularly for enrolment in research. It requires the presence of the following:
5
Signs of LMN degeneration by clinical, electrophysiological or neuropathological examination. Signs of UMN degeneration by clinical examination. Progressive spread of signs within a region or to other regions. Electrophysiological evidence of other disease processes that might explain the signs of LMN and/or UMN degenerations. Neuroimaging evidence of other disease processes that might explain the observed clinical and electrophysiological signs.
In the absence of:
There is no specific test for ALS and a degree of diagnostic uncertainty may remain.
Prognosis
The median time from diagnosis to death is two and a half to three years, 9 but around 20% of patients live for five to ten years 2 and 10% have a slow form of the disease with survival of ten years or longer.9,10 At first presentation 50% of patients have a functional vital capacity (FVC) less than 75% predicted. Faster progression is seen in bulbar onset phenotypes, older patients and those with more marked FVC reduction at diagnosis. 5 Studies may have an inclusion bias by tending to include patients with longer survival and less progressive disease. 1 Death is usually a result of respiratory failure, but sudden unexpected death occurs in 6%–12% of cases. Plausible causes include pulmonary emboli, cardiac causes and loss of brainstem respiratory reflex controls due to neuronal loss in the pre-Bötzinger nucleus. 1
Treatments
Multidisciplinary care
Key to care is a multidisciplinary team (MDT) approach incorporating professionals including surgeons, anaesthetists, intensivists, perioperative physicians, neurologists, respiratory physicians, gastroenterologists, occupational therapists, speech therapists, physiotherapists, specialised nurses, dietitians, psychologists, social workers, palliative care teams and others. Formal MDT approaches have reduced the risk of death at five years by 45%. 2 It can also improve quality of life for patients, reduce hospital admissions and shorten length of stay. 11 If locally available such specific ALS networks may be invaluable in planning perioperative care.
Pharmacological
Present treatments are mostly orientated on symptom management rather than treating the disease and are guided by consensus rather than high-grade evidence.
12
Riluzole is a sodium channel blocker with anti N-methyl-
Patients may be on a significant number of drugs aimed at treating symptoms.10,11 There are several therapies currently being investigated and interaction with research teams may be necessary. Some drugs may have significant implications if stopped perioperatively. 14 Individualised assessment is required and may require neurology involvement. Some potential management options are presented (Table 1).
Pharmacological treatments used in amyotrophic lateral sclerosis, their implications in the perioperative period and potential management options.
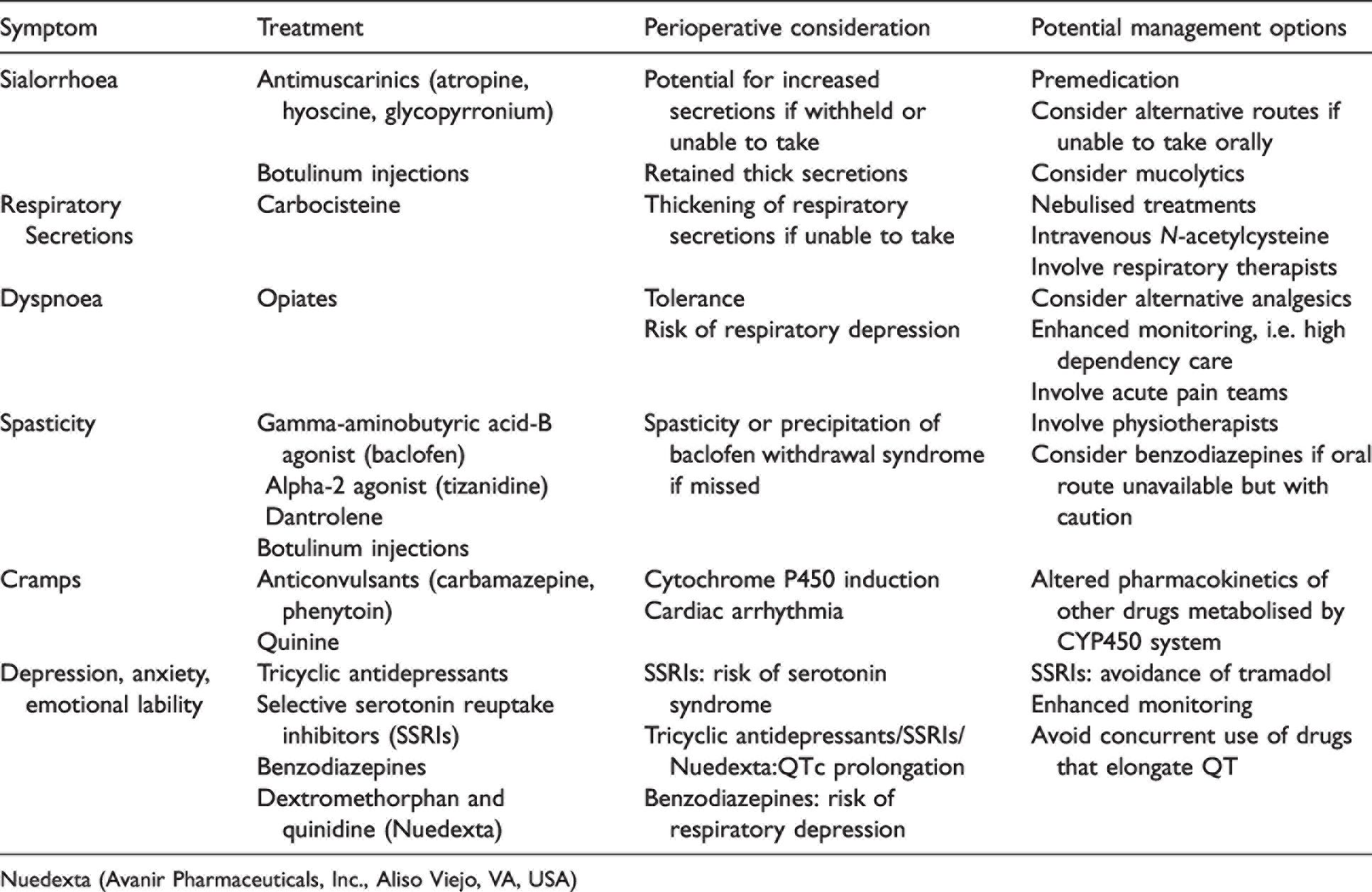
Nuedexta (Avanir Pharmaceuticals, Inc., Aliso Viejo, VA, USA)
Non-pharmacological
A range of non-pharmacological treatments are available that may need modification or even instigation in the perioperative period including non-invasive ventilation (NIV) and cough augmentation.
Non-invasive ventilation
Many patients may present established on NIV. Usage has increased over the past two decades but varies globally.15,16 Variable guidance exists but most suggest instigating NIV when FVC reaches less than 80% predicted if the patient has symptoms of respiratory or airway weakness, or when there is declining spirometry or maximal inspiratory pressure measurements. 17 The plausible physiological benefits of NIV include: (a) reversing chronic muscle fatigue by reducing work of breathing thereby improving cellular oxygenation and metabolism; (b) reversing hypercapnia and improving central respiratory drive; and (c) reversing atelectasis and improving lung compliance. 18 FVC measurements less than 50% predicted warns of impending ventilatory failure. 15
Despite its increasing use the evidence base for NIV has been limited. A 2017 Cochrane Review found only one high quality randomised controlled trial. This demonstrated a mean increased survival of 48 days. Subgroup analysis of patients with preserved bulbar function showed a survival benefit of 205 days. 19 Those with severe bulbar dysfunction had no measurable survival benefit. However, observational studies and cohort studies excluded in the Cochrane Review have suggested an association with prolonged survival. NIV can be problematic with high secretion burdens and bulbar dysfunction, including potential glottis closure. 10 Specialist support can facilitate the use of NIV in severe bulbar weakness, with emerging evidence that survival can be improved. 20
Cough augmentation
Inadequacy of secretion and mucus clearance can occur and potentially cause postoperative pulmonary complications. 15 Coughing is a complex sequence of inspiratory, expiratory and bulbar muscle control. Mechanical insufflation–exsufflation devices provide rapid positive pressure and then negative pressure to mimic a cough. A 2013 Cochrane Review failed to show superiority to manual cough assistance 20 but many patients report symptom relief from devices. 21 Guidance now advocates use when peak expiratory cough flow is less than 170 l/min. 16 The use of cough augmentation techniques is more challenging in those with severe bulbar weakness.
Procedural treatments
Patients with ALS can present for any form of elective surgery and for time-dependent operations such as emergency surgery,22–25 trauma26,27 and cancer surgery.28–32 ALS can coincide with pregnancy33–43 and increased respiratory requirements combined with ventilatory weakness can cause rapid decompensation as pregnancy progresses. Vaginal delivery is possible as uterine dynamics are unaffected. However, most published cases underwent early caesarean section to avoid precipitating respiratory failure.
A range of procedural interventions may be used specifically in the treatment of ALS. Examples include, nutritional supplementation via gastrostomy, airway surgery such as tracheostomy, and continence procedures such as suprapubic catheter insertion or colostomy formation. However, there is a paucity of information regarding surgical outcomes for patients with ALS. 44 Surgery carries a significant risk of precipitating respiratory failure and the potential to worsen functional decline. 45 Perioperative pulmonary complications may occur for many reasons; atelectasis, residual effects of anaesthetic agents, side-effects of analgesics, the inability to compensate for complications such as aspiration and respiratory infection, and potential neuromodulation by the stress response and inflammation. 46 The unexpected presentation of postoperative respiratory failure has been found to be undiagnosed ALS being unmasked.47,48
Airway surgery
As ventilatory failure progresses some patients, particularly those with upper airway dysfunction, may choose invasive ventilation via tracheostomy, or more rarely laryngectomy or laryngeal diversion. 49 Tracheostomy does not relieve aspiration risk and may worsen it. 50 Subglottic suction tubes, laryngectomy, or diversion surgery are alternative options that modify aspiration risk. Laryngectomy rarely appears to be used in Western countries 51 but is more common in Japan.52–54 There is limited evidence regarding the life-prolonging benefits of invasive ventilation in ALS.1,55 The rarity of invasive ventilation in ALS means professionals may not have encountered such situations before.
Most invasive ventilation is undertaken as part of an emergency admission and has associated long lengths of stay.55–59 Mean time to discharge from hospital was 18 weeks in the United Kingdom and 45% of patients died unexpectedly. 1 One-year survival in an elective cohort was 79%. 55 In another a median survival of 74 months was seen. 60 Reports exist of patients surviving ten years and 10% may survive five years or more. Invasive ventilation does not slow disease progression 15 and patients are at risk of going on to develop a locked-in state61,62 or a minimally conscious state. 63 Impact on caregivers can be substantial. 61 Regional differences in available support are factors that may need to be addressed. 56 Decision-making around withdrawal of ventilation should be considered before any procedure.
Diaphragmatic pacing
The surgical implantation of diaphragmatic pacing devices has been investigated in patients with ALS. This follows previous use in central hypoventilation syndromes, spinal cord injuries and brainstem lesions.15,64 A large trial was terminated after increased mortality in the pacing arm and concerns have been raised in other trials.65,66 Trial patients may present for surgery and consideration may need to be given to electrical interference from equipment such as diathermy.
Gastrostomy
Weight loss occurs due to dysphagia, muscle atrophy, poor appetite and a hypermetabolic state. 2 Malnutrition may contribute to disease progression and worsen quality of life. 67 As dysphagia progresses enteral feeding may be considered. The effect of percutaneous endoscopic gastrostomy (PEG) on survival remains debated.12,68,69 Placement before severe respiratory muscle dysfunction reduces the risk of complications and maximises potential benefit.69,70 Placement under sedation has been shown to be safe in mild respiratory failure.71,72
Continence
Urinary frequency and urgency are common. 70 Toileting can become more challenging and indwelling urinary catheters can be used 73 resulting in improved quality of life. For some a suprapubic catheter (SPC) is an option but there is limited evidence supporting SPC in ALS. Multifactorial constipation is common. 74 In rare circumstances patients may consider end colostomy formation. A ‘triple-ostomy’ for a patient with ALS has been described; a combined procedure in which a PEG, SPC and colostomy were undertaken, under a single anaesthetic. 75
Sialorrhoea
Surgery for severe sialorrhoea has been raised in some reviews,76,77 and case reports include neurectomies and salivary gland ligation in children with cerebral palsy. Thickened secretions post intervention can be problematic. Pharmacological management of sialorrhoea should be exhausted before considering surgery. 10
Preoperative assessment
A diagnosis of ALS may impact on preoperative planning, including general anaesthesia, sedation, and central and peripheral regional anaesthesia. Even accounting for these nuances, the exact balance of risks will need to be assessed on an individual basis accounting for patient preference and the procedure proposed. Precipitating ventilatory failure and requiring non-invasive or invasive support may be a primary concern to many patients and clinicians. Discussion with patients and meticulous planning is required to avoid later dilemmas.1,78
The predictability of postoperative pulmonary complication is poor. There is potential for progression to long-term invasive ventilation particularly in those with severe bulbar weakness following acute respiratory failure.79,80 Ideally patients will have discussions with professionals around advance care planning, mortality concerns and the potential impact on quality of life/care needs of long-term ventilation.
Shared decision-making
Preoperative assessment aims to understand and, if possible, reduce the risks of surgery and enable shared decision-making. In particular, shared decision-making in ALS has been proposed as ‘facilitating patient-centred decisions to suit the inevitable changes to patient health and lifestyle’. 81 The heterogeneity of patients will require a highly individualised approach. A comprehensive and collaborative approach to preoperative assessment, information sharing, patient optimisation, and planning for the perioperative period will need to be undertaken.
Caution in extrapolating likely disease progression and prognosis is required. Diagnostic uncertainty, the potential for treatable mimic conditions, and the need for repeat investigation and testing to confirm the diagnosis may be present in some patients. There may be a requirement for urgent multidisciplinary input to help guide treatment plans.
While time constraints exist for emergency and expedited surgery it must also be considered that the underlying disease is likely to be progressing and ventilatory parameters may worsen, increasing risk. FVC may decline at a rate of 3%–5% per month. 82 There may be a requirement to circumvent standard operating list waiting procedures.
A difficult balance between the need for surgery and the risk of precipitating decline will need to be worked through. 83 A lack of evidence around operative interventions for patients with ALS may cause divergent opinion between professionals or patients. However, at the same time the use of a wide MDT may produce novel ways of solving problems for patients with ALS. 84
Respiratory assessment
Respiratory dysfunction is a key area of concern in the perioperative period for patients with ALS. Assessment focuses on the ability to compensate for the increased respiratory work required in the perioperative period. Increased demand will come from the physiological stress of surgery and potential complications. All phenotypes of ALS eventually progress to respiratory muscle weakness. Denervation and respiratory muscle atrophy reduce FVC. 15
Central respiratory control may be impaired due to damage to descending motor neurons from the cortex. 18 Symptoms include exertional dyspnoea, orthopnoea and those of sleep disordered breathing 5 such as unrefreshing sleep and/or daytime somnolence. Headache on awakening is a later symptom and is highly suggestive of nocturnal hypoventilation. Sleep unmasks ventilatory insufficiency due to hypotonia of intercostal and accessory muscles and depression of central drive. 18 Bulbar dysfunction predisposes sufferers to upper airway obstruction, impaired secretion clearance and aspiration. 15
Careful history taking is required to elucidate symptoms of ventilatory failure but symptomatology does not correlate well with the degree of respiratory muscle weakness in ALS and formal testing is advisable. 18 Spirometry is the recommended mainstay for respiratory muscle testing with repeat recordings at three-monthly intervals. 15 Inspiratory muscle strength, blood gas analysis, chest radiography, and overnight oximetry can be helpful. A primarily restrictive picture will be seen on spirometry. 85 Measures of FVC and inspiratory pressures will correlate with disease progression and the likelihood of impending respiratory failure. 86
Different tests have different sensitivity and specificity for detecting aspects of respiratory failure. Parameters that correlate with disease severity and progression include FVC and maximal inspiratory pressure (MIP) measurements. Sniff nasal inspiratory pressures (SNIPs) are a sensitive measure of inspiratory muscle weakness. A facemask is helpful in patients with bulbar disease to record FVC. Supine FVC may detect diaphragmatic weakness, 87 nocturnal oximetry may detect early respiratory failure, and sniff pressures may be better for detecting the risk of hypercapnia. 70 If there are respiratory symptoms it is prudent to undertake further testing such as supine spirometry 15 even if erect vital capacity is within the normal range. Respiratory services involvement will be invaluable not only regarding trends in spirometry but for monitoring and advice in the postoperative period.
Cardiovascular assessment
Patients may have autonomic dysfunction including raised resting heart rates and orthostatic hypotension secondary to ALS. Historically, autonomic function, particularly of vagal nerves, was considered preserved. 88 More recent evidence has shown the presence of parasympathetic dysfunction90 and abnormal heart rate responses to blood pressure changes. 89 There is a possible link between those with bulbar disease and autonomic dysfunction. 91 Patients with ALS may have a loss of normal nocturnal heart rate variability, and despite normal nocturnal oximetry, appear to be at risk of sudden cardiac arrest and death.92–94
Substantial autonomic dysfunction has been unmasked by anaesthesia and trauma.95–97 Cardiovascular instability secondary to autonomic dysfunction may require perioperative invasive monitoring. Occult cardiovascular disease may exist due to functional limitation and a low threshold for investigation may be required, although cardiopulmonary exercise testing may be impossible. There is potential for QTc abnormalities from the disease itself as well as concurrent pharmacological treatments, 96 and electrocardiogram testing should be considered. If QTc abnormality is identified drugs causing prolongation should be avoided.
Cognitive assessment
While historically considered a pure motor pathology, it has become apparent there is a risk of cognitive dysfunction. 11 Many patients have identifiable deficits in executive cognitive function and 5% can be identified to have diagnosable frontotemporal dementia. 98 Symptoms of frontotemporal dementia such as emotional blunting, loss of insight and behaviour changes can make capacity assessments challenging. 98 Careful assessment of cognition and capacity to consent may be required using standard approaches.
Neurological assessment
Formal documentation of a patient’s neurological state preoperatively will enable comparison if there are any postoperative concerns regarding a change in neurology. 99 This also enables discussion if a regional or neuraxial technique is to be chosen regarding the potential risks of side-effects or complications for that patient. A subsequent neuropathy in a previously well-functioning nerve may cause profound loss of function for a patient with ALS.
Estimating disease progression may be difficult for the non-specialist. Functional assessment scales can be used. Perioperative professionals should be aware that specific ALS functional rating scales exist such as the ALS-FRS.100 They can be used objectively to monitor the disease over time and may help non-specialists understand an individual’s current function and potential trajectory of decline.
Intraoperative considerations
General anaesthesia
General anaesthesia may be necessitated by the nature of the surgery or the patient’s wishes. However, risks of postoperative pulmonary complications are potentially high from the disease when combined with superadded atelectasis, retained secretions, aspiration, residual neuromuscular blockade, restricted ventilation from pain, and pharmacological respiratory depression. Optimising positioning may enable maximal respiratory function and may involve discussion as to whether operations can be undertaken in non-standard positions. 36 Avoidance of invasive ventilation and anything that may worsen neuromuscular weakness will reduce these risks but may not be possible.
Neuromuscular considerations
If tracheal intubation is required, then careful assessment and planning will need to be undertaken. Patients with ALS may have an increased aspiration risk secondary to bulbar weakness causing dysphagia and secretion pooling that will be worsened by any sedation or general anaesthesia. 101 If general anaesthesia with airway intervention is required, it may be necessary to use agents to dry secretions 102 or reduce gastric pH.
Choice of airway management will affect the pharmacological considerations to be made, which are considerable and follow the basic principles of management of neuromuscular disease and anaesthesia such as avoidance of depolarising agents. Some specific potential concerns in ALS relate to possible increased sensitivity to non-depolarising agents, inadequacy of reversal with sugammadex and issues with monitoring devices.
Depolarising neuromuscular blocking drugs such as suxamethonium are contraindicated in neuromuscular disorders including ALS. Denervated, dystrophic, or metabolically altered muscle tissue risks the presence of increased extra-junctional nicotinic receptors or a proliferation of fetal gamma type nicotinic receptors. 103 Administration of depolarising neuromuscular blocking drugs can lead to lethal hyperkalaemia, rhabdomyolysis, dystonic reactions with masseter spasm, and difficulty in intubation.104,105 Caution should also be exercised in patients without a formal diagnosis but signs or symptoms of neuromuscular weakness. If the diagnosis is uncertain there may be a risk of malignant hyperthermia from other neurological diseases.101,106
Specific potential concerns have been raised over increased sensitivity and prolonged effects of non-depolarising neuromuscular blocking drugs in ALS. 107 In ALS, sensitivities to rocuronium,28,108 vecuronium 109 and cisatracurium 110 have been reported, and augment extrapolations from other neuromuscular diseases. 103 Avoiding neuromuscular blocking drugs if possible is recommended. 111 If they cannot be avoided, then reduction in dose and titration to a neuromuscular monitoring system is essential.
However, neuromuscular monitoring devices use a reference twitch magnitude from a healthy population range. 112 For patients with ALS, with a likely reduced twitch amplitude, calibrating devices may be preferred.113,114 Concern has been raised about discordance between train-of-four counts and levels of neuromuscular weakness in patients with ALS.108,115 In these cases, despite apparent complete reversal of paralysis, patients’ ventilatory parameters remained inadequate to extubate.
Reversal of neuromuscular blockade may be required to reduce postoperative pulmonary complications. Anticholinesterases such as neostigmine may be inadequate in patients with residual deep block. 116 Sugammadex has emerged in recent years as a potentially more effective and reliable method of reversing neuromuscular blockade from steroidal neuromuscular blocking drugs. 117 although concern has been raised regarding inadequate reversal 118 and even recurarisation 31 in patients with ALS, sugammadex has been used to good effect.115,119 Meticulous monitoring and titration of reversal agents seems prudent.
Alternatives to neuromuscular blockade to obtund laryngeal reflexes include the use of total intravenous anaesthesia such as propofol and remifentanil75,82,121,122 or topicalisation of the airway with local anaesthetic.38,64,102 Total intravenous anaesthetic techniques allow for the avoidance of volatile agents with their respective risk of worsening neuromuscular weakness.117,118,121,123 The use of ultra-short-acting agents such as remifentanil may help to reduce the incidence of respiratory depression secondary to residual opiates in the postoperative period, while ensuring adequate intraoperative analgesia.24,64,122,124,125 The immediate post-extubation use of NIV, in conjunction with total intravenous anaesthesia, has been successful in ALS patients with preoperative FVCs less than 50% predicted. 121
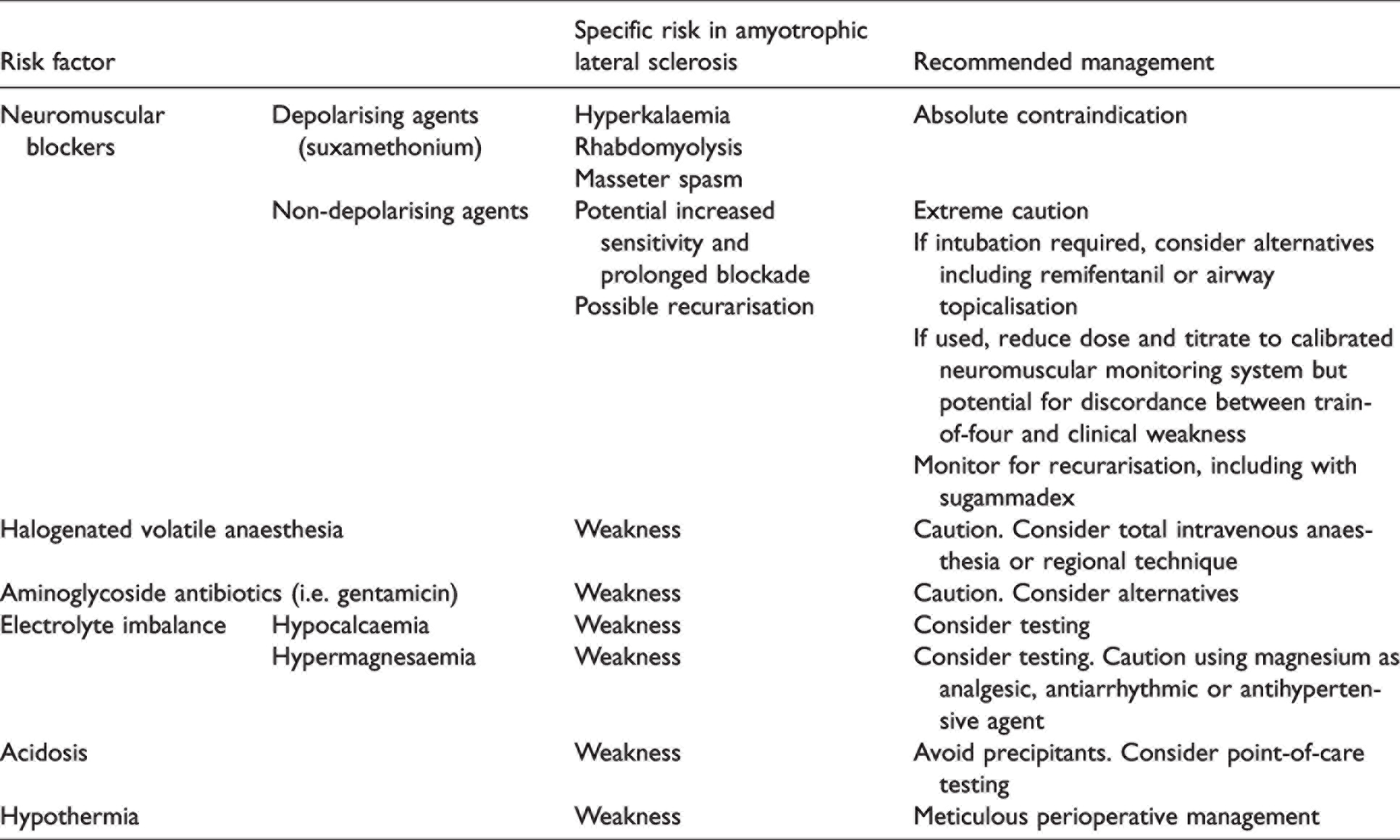
Patients are at risk of hypothermia both from general and regional anaesthesia because of an increased proportion of immobile or atrophied muscles. 106 Other factors not specific to ALS can cause postoperative residual weakness such as pharmacological precipitants and electrolyte imbalance. These are summarised and presented with management recommendations (see Table 2).
Risk factors for perioperative neuromuscular weakness and author management recommendations.
Sedation
Conscious sedation may be appropriate for some interventions.126–128 Carefully targeted it may be possible to maintain spontaneous ventilation and avoid airway intervention which may be of benefit in ALS. It is commonly used in ALS for PEG placement.128,129 It may be possible to use the patient’s own NIV or supplementary NIV during the procedure.76,130 However, a nasal interface or special mask will be required. In combination with regional anaesthesia operations normally undertaken under general anaesthesia may be achievable under conscious sedation. 127
For non-anaesthetists, who may otherwise give sedation, such as gastroenterologists or dentists, extreme caution and the involvement of an anaesthetist is recommended. 131 If undertaking sedation, the ability of the patient with ALS to communicate that sedation and analgesia are inadequate needs to be considered. In a case of cataract surgery for a patient with advanced ALS who had a tracheostomy the team opted for general anaesthesia rather than sedation due to difficulties in communication. 132
Regional anaesthesia
Peripheral and neuraxial regional anaesthesia, in isolation or combination with sedation or general anaesthesia, may reduce the risk of postoperative pulmonary complication. 133 However, there are longstanding concerns over the use of regional anaesthesia with coexisting neurological disease.134,13 Evidence is low quality and based on expert consensus and case studies and there is a need to consider risk and benefit on an individual basis. 99 Evidence specific to ALS is generally lacking. Even if general anaesthesia is required the use of supplementary techniques may enable a reduction in systemic analgesic use perioperatively and thus a possible reduction in the risk of respiratory depression.
The ‘double crush’ phenomenon 136 describes an increased risk of secondary neuronal insult in patients with pre-existing neurological conditions. 137 However, there is still discordance in expert opinion regarding causation or even existence of the phenomenon. 138 Because of the low prevalence of ALS, and then even lower prevalence of patients undergoing regional anaesthesia, it is unknown as to the exact risks in ALS. Plausible risks lie in nerve damage via direct trauma, drug-induced neurotoxicity or localised ischaemia, and precipitating respiratory depression via procedural complications or blockade of respiratory muscles. 139 Careful selection of drug concentrations and doses should be considered. While concentrations and doses in normal practice are unlikely to be directly neurotoxic to healthy nerves there is a lack of evidence of this in ALS. 99
Concern around spinal anaesthesia in central neurological disease has existed for some time.140,141 Available series unfortunately are combined cohorts of neurological diseases and contain only small numbers of patients with ALS. 137 Reports of the use of spinal anaesthesia without complications exist for patients with ALS.44,122 Specific caution will be needed to avoid a block height that impedes respiratory function. Despite concern, complication rates appear low in central neurological disease. The use of epidural anaesthesia has been more commonly published for patients with ALS142–144 including concurrent sedation127,145,146 or the intraoperative use of NIV. 22 While most epidural blocks seem to have little effect on respiratory function in well patients 147 this may be less well tolerated in patients with ALS. High quality evidence of the specific risks or safety of central neuraxial anaesthesia in ALS is lacking.
The use of peripheral regional anaesthesia may be an alternative including the use of blocks in novel fashions. Gastrostomy and breast surgery for patients with ALS have been managed with thoracic paravertebral blocks.30,148–151 Care will need to be undertaken with any block that may impact on respiratory function or risk pneumothorax. Preoperative neurological status should be documented to enable comparison if there is concern over postoperative neurological dysfunction. 135 Plans should be clearly made for what should be done if a regional or neuraxial approach becomes inadequate at any stage.
Postoperative care
Postoperative plans will need to be considered from the outset. The need for inpatient admission and higher levels of care will be significant for many patients with ALS. 152 Exact needs will be dependent on the patient’s individual respiratory parameters and functional state, the nature of the surgery being undertaken, anaesthetic and analgesic requirements, and the nuances of the individual hospital. Specific considerations include the management of residual neuromuscular weakness, non-invasive and invasive respiratory support, autonomic instability monitoring and the management of a potentially non-standard analgesic regimen. A low threshold for high dependency care would address many of these issues but is dependent on the local healthcare systems arrangements.
Postoperative pulmonary complications
Patients are at high risk of postoperative pulmonary complications and many aspects of postoperative care planning will be based around mitigating and managing this. The combination of a restrictive disease combined with the inability to clear secretions and decreased peak expiratory cough flow can lead to intrapulmonary shunts, hypoxia and respiratory muscle fatigue, with subsequent respiratory failure. 105 Patients with ALS who have had any surgery, anaesthetic technique, or analgesic plan that risks impacting ventilatory function may require high dependency care. Patients remaining ventilated postoperatively have significant risks of difficulty weaning from the ventilator. 111
If patients require NIV preoperatively, they may require more complex NIV and/or higher levels of support postoperatively.55,122 Returning patients to their baseline NIV as soon as possible will be a minimum requirement. Invasive arterial monitoring will allow monitoring of ventilation until the patient is re-established to their baseline. The titration of oxygen therapy to a strict saturation range may be needed to reduce the risk of hypercapnic failure.
Post-procedural analgesia
Analgesia needs to be optimal in ALS to enable patients to cough and mobilise. The use of regional analgesia may reduce the requirement for systemic drugs. When selecting systemic analgesics preference for a multimodal analgesic regimen is advisable to reduce the use of respiratory depressants. Subcutaneous ketamine infusions, 75 lidocaine infusions 153 and patient-controlled analgesic devices have been used to good effect. A Cochrane Review found no specific research for analgesic treatments in ALS. 154 A balance between comfort and safety will be required and may require high dependency care. Individuals with ALS are vulnerable to frontotemporal dementia, which may be undiagnosed, and while no specific literature could be found there is hypothetically an increased risk of postoperative delirium. The increased nursing levels in high dependency care may be beneficial in managing the risks associated with this.
Multidisciplinary care
Pre-planned multidisciplinary postoperative care is advised in ALS. This enables the optimal use of the MDT. For example, respiratory physiotherapists being able to optimise patients’ cough technique rapidly and the use of cough-assist devices.15,18 Guidance from home ventilation specialists will enable patients to use their own devices and optimise support while in hospital. Input from dietitians and speech and language therapists will be required to optimise nutrition, change to gastrostomy feeding, or alter calorific requirements. The involvement of physiotherapists and occupational therapists will enable patients’ mobility and ongoing care needs from the offset. Pharmacists will optimise pharmacological treatments for the patients and help reduce interactions and side-effects that may increase morbidity. Involvement preoperatively will optimise these processes and include discharge planning. Patients may have significant changes in care needs even with the simplest of operative interventions.
Conclusions
The perioperative care of patients with ALS is complex. It impacts on every part of perioperative care and the risk of morbidity and mortality is significant. The baseline prevalence of the condition and subsequent rarity of patients presenting for surgery may mean this is an uncommon event. This review summarises the extent of what is currently known and the uncertainties that exist. Unfortunately, research quality is low, and evidence is predominantly based on case reports and expert consensus.
Specific considerations in ALS are highlighted such as specific surgical treatments and preoperative, intraoperative and postoperative management considerations. This review highlights the complexity in many areas of the perioperative period, but that management is based predominantly on first principles in perioperative neuromuscular disease management. There is a critical need for multidisciplinary involvement and shared decision-making to achieve this. A comprehensive approach is required to managing respiratory, cardiovascular and neuromuscular risk in the perioperative period for patients with ALS. The potential for new therapeutics, interventions and novel perioperative management approaches to support people with ALS may mean more patients present for surgery in the future.
Footnotes
Author Contribution(s)
Acknowledgements
The authors would like to thank Elizabeth Househam, Consultant Neurologist and Neuromuscular Specialist, Derriford Hospital, Plymouth, UK, for her time in reviewing a draft of this manuscript.
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
