Abstract
The laryngeal mask airway, the LMA® Gastro™ (Teleflex Medical, Athlone, Ireland), is a novel airway device which permits upper gastrointestinal (GI) endoscopy to be performed via a dedicated large calibre oesophageal lumen. It has been validated in adult studies, but to our knowledge, there have been no data published regarding its use in a paediatric population. Following a brief education programme, the LMA Gastro was introduced on a trial basis at our institution and made freely available. Over a four-month period, our pre-existing endoscopy audit framework captured data on a total of 55 patients who had the LMA Gastro device chosen as the primary airway for elective upper GI endoscopy. These data were collected prospectively by the treating anaesthetist and included a range of airway and endoscopy outcomes. Of the 55 patients, the LMA Gastro provided an adequate airway in 52 (94.5%). Forty-six (88.5%) were sited on first attempt, and 50 (96.2%) insertions were rated ‘easy’ by the anaesthetist. Aside from three insertion failures, there were no airway events. The endoscopy success rate was 100% in the 52 patients who had an LMA Gastro airway successfully inserted. First-pass oesophageal access was achieved in 51 (98%) cases, and 100% of insertions were rated ‘easy’ by the gastroenterologist. Despite our relative inexperience with it, the device had a high success rate for airway maintenance and oesophageal access in our paediatric patient population. In our institution, the LMA Gastro provided a useful airway option in older children undergoing elective upper GI endoscopy.
Introduction
The laryngeal mask airway, the LMA® Gastro™ (Teleflex Medical, Athlone, Ireland; see Figure 1), is a novel airway device designed to facilitate upper gastrointestinal (GI) endoscopy via a dedicated lumen while maintaining airway patency via a supraglottic cuff. It is a silicone-based, cuffed, second-generation supraglottic airway device (SAD), with a large oesophageal port to allow passage of an endoscope. There have been several recent publications regarding its utility in the adult population, 1 including its use in high-risk patients and in advanced endoscopic procedures. 2 , 3 Thus far, to our knowledge, there have been no data published regarding its use in the paediatric population.
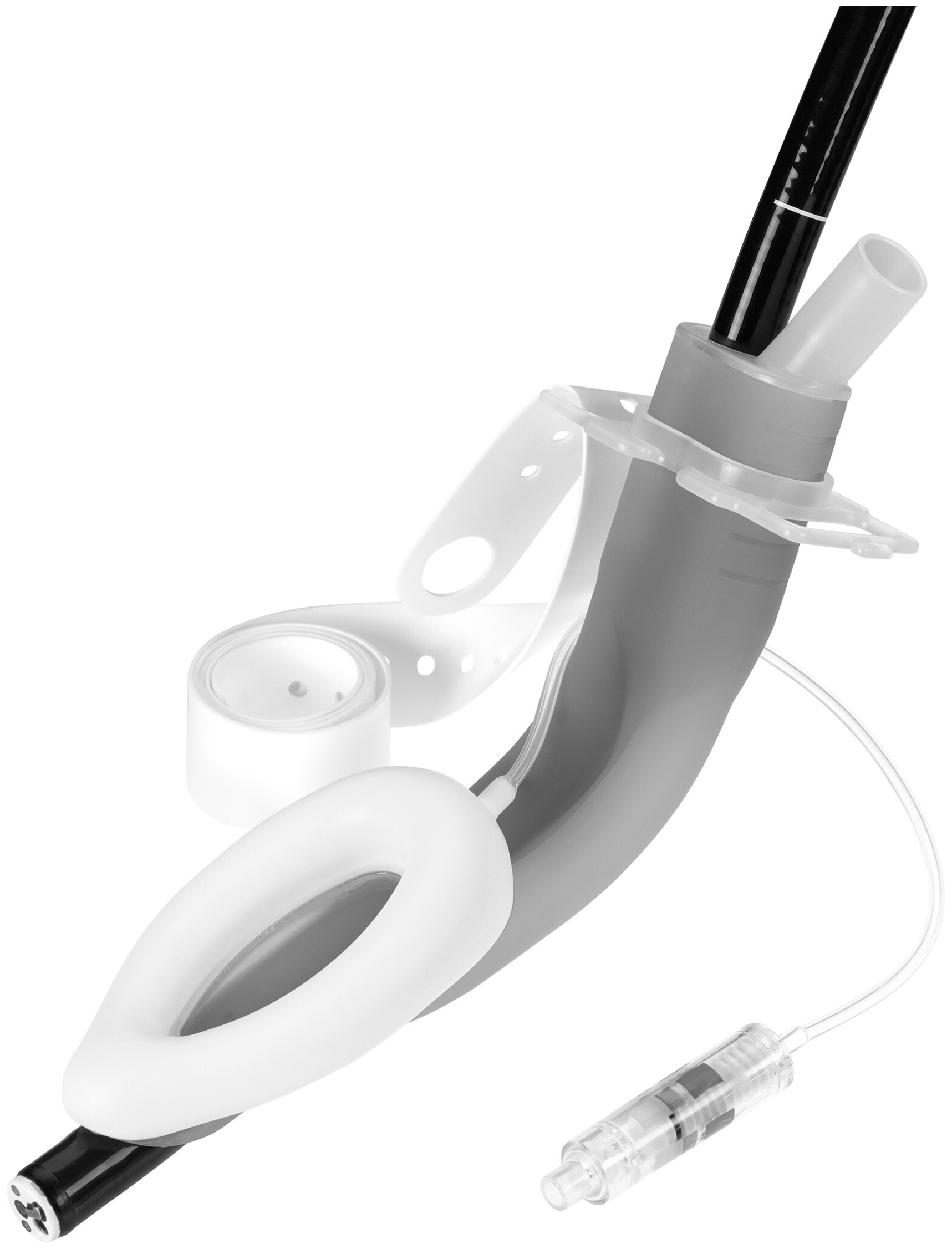
Laryngeal mask airway, LMA®Gastro™ (Teleflex Medical, Athlone, Ireland). Image supplied by Teleflex with permission to reproduce.
Paediatric upper GI endoscopy is a relatively common procedure which can create a dilemma regarding airway management. 4 In our institution, practice is highly variable amongst consultant paediatric anaesthetists. Most prefer endotracheal intubation due to perceived safety and ease with which a gastroenterologist can pass an endoscope. Others utilise standard SADs or bite blocks. Choice of airway device is individualised for each patient in consultation with the gastroenterologist.
The Women’s and Children’s Hospital (WCH) is the tertiary paediatric referral centre for South Australia, the Northern Territory, as well as areas of rural New South Wales and Victoria. It is the only public institution in this area where paediatric upper GI endoscopy is routinely performed. Given the relatively large number of patients undergoing these procedures, as well as the dilemma regarding optimal airway management, we thought it would be interesting to trial the currently available size 3 and size 4 LMA Gastro devices in our population.
The aim of this study was to assess the performance of the device from both the anaesthetists’ and gastroenterologists’ perspective, evaluating ease of use, technical success and complications in both domains.
Methods
An endoscopy audit framework including local ethics approval was already in place (WCH Audit 1095A/06/2022) when the WCH Product Advice and Evaluation Advisory Group approved a limited trial of the LMA Gastro devices for elective endoscopy procedures. All data were collected prospectively during a four-month period from July 2019 until October 2019. Thirty devices were provided by Teleflex, and a further 30 were purchased using the hospital equipment fund. Initially, only size 3 airways were available, with size 4 becoming available towards the end of the trial. In addition to the devices, Teleflex provided on-site clinical education to anaesthetists, endoscopists and nursing staff regarding the correct use of the devices prior to their introduction. Information sheets detailing the recommended insertion technique were also provided.
Patient characteristics
Data were collected for all paediatric patients (age <18 years) undergoing elective upper GI endoscopy during the study period. Data were not collected for patients who underwent emergency procedures.
Study procedures
Patient care was entirely at the discretion of the treating gastroenterologist and anaesthetist. Following the initial education session, the LMA Gastro devices were made freely available for the anaesthetists to use, should they elect to do so. The primary anaesthetist was asked to complete a standardised paper audit form prospectively. Data were then transferred to a Microsoft Access database and analysed using Microsoft Excel by the authors.
LMA Gastro insertion
The method described is that taught as part of the LMA Gastro education, although a standardised insertion technique was not enforced:
The endoscope channel is prepared using a generous amount of medical-grade silicone spray to lubricate it for endoscope insertion. The cuff is fully deflated, and the syringe is disconnected from the inflation port. Water-based lubricant is applied to the convex surface of the device just prior to insertion. Holding the integrated bite block, the tip of the mask is gently pressed against the hard palate and advanced behind the tongue. The device is then guided along the hard palate to the soft palate, down the posterior pharyngeal wall until resistance is felt as it seats in the hypopharynx. The cuff is inflated using the inflation port. Effective insertion and ventilation are confirmed by the anaesthetist.
Outcomes assessed
Table 1 lists the data collected in our audit. A free comments section was also included to provide further detail on any events or issues encountered.
Audit data collected.

aDefined as occurring where an unplanned intervention was required to remedy a problem.
bGraded as easy or difficult by operator. IV: intravenous.
Results
A total of 108 patients underwent elective upper GI endoscopy during the four-month study period. Of these, 55 (50.9%) received the LMA Gastro device. The 53 (49.1%) who received another airway device are excluded from further detailed analysis here (endotracheal tube (ETT)=47, bite block=4, standard LMA=2).
Patient characteristics
Table 2 presents the characteristics of the audit participants, while Table 3 presents the procedural characteristics. Figures 2 and 3 show the age and weight of the 41 patients who received the size 3 device and the 14 patients who received the size 4 airway.
Baseline demographic and clinical characteristics of audit participants.

Results are presented as number (percentage) or median (range) for continuous data.
Procedural characteristics.
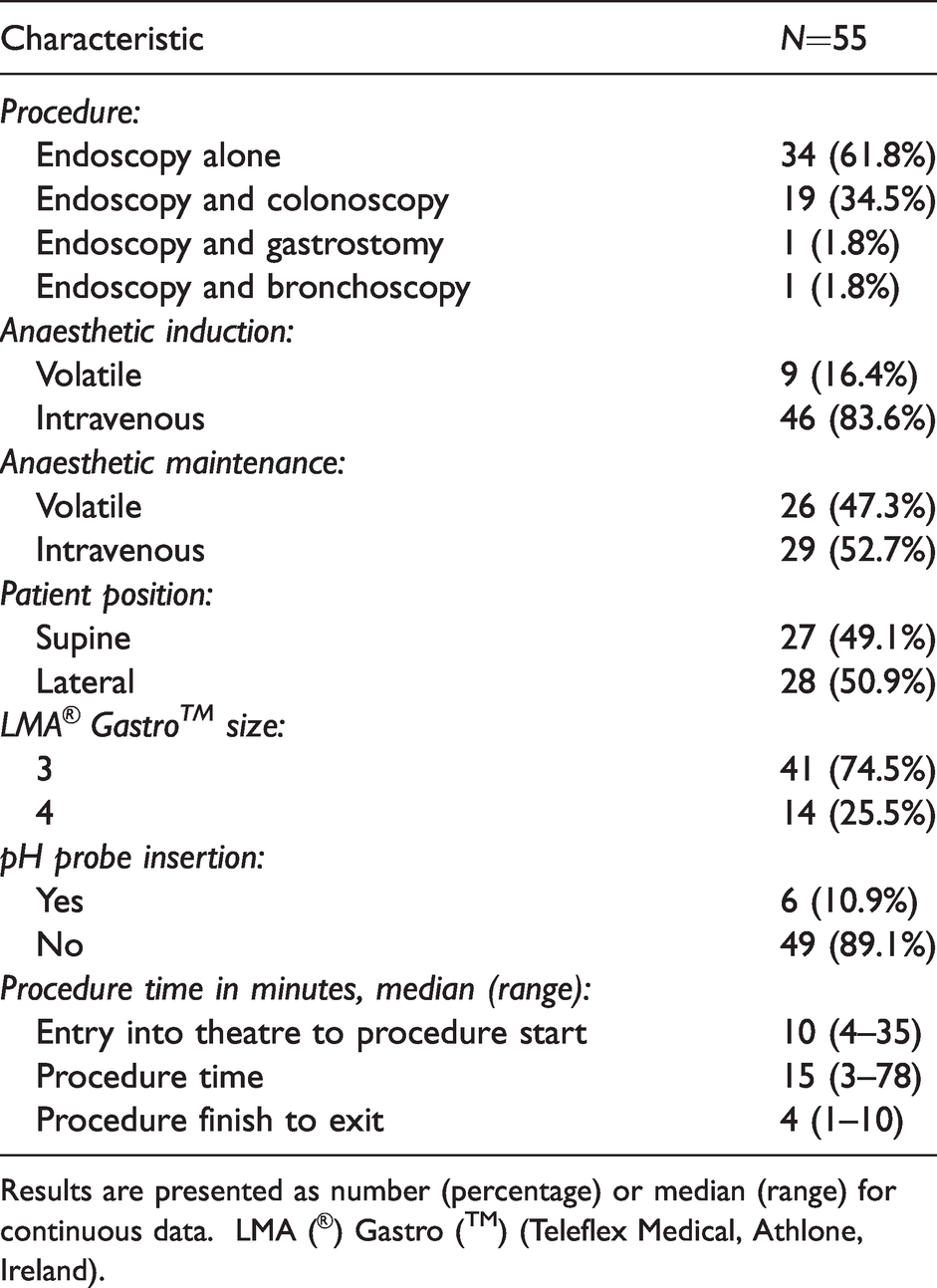
Results are presented as number (percentage) or median (range) for continuous data. LMA (®) Gastro (™) (Teleflex Medical, Athlone, Ireland).
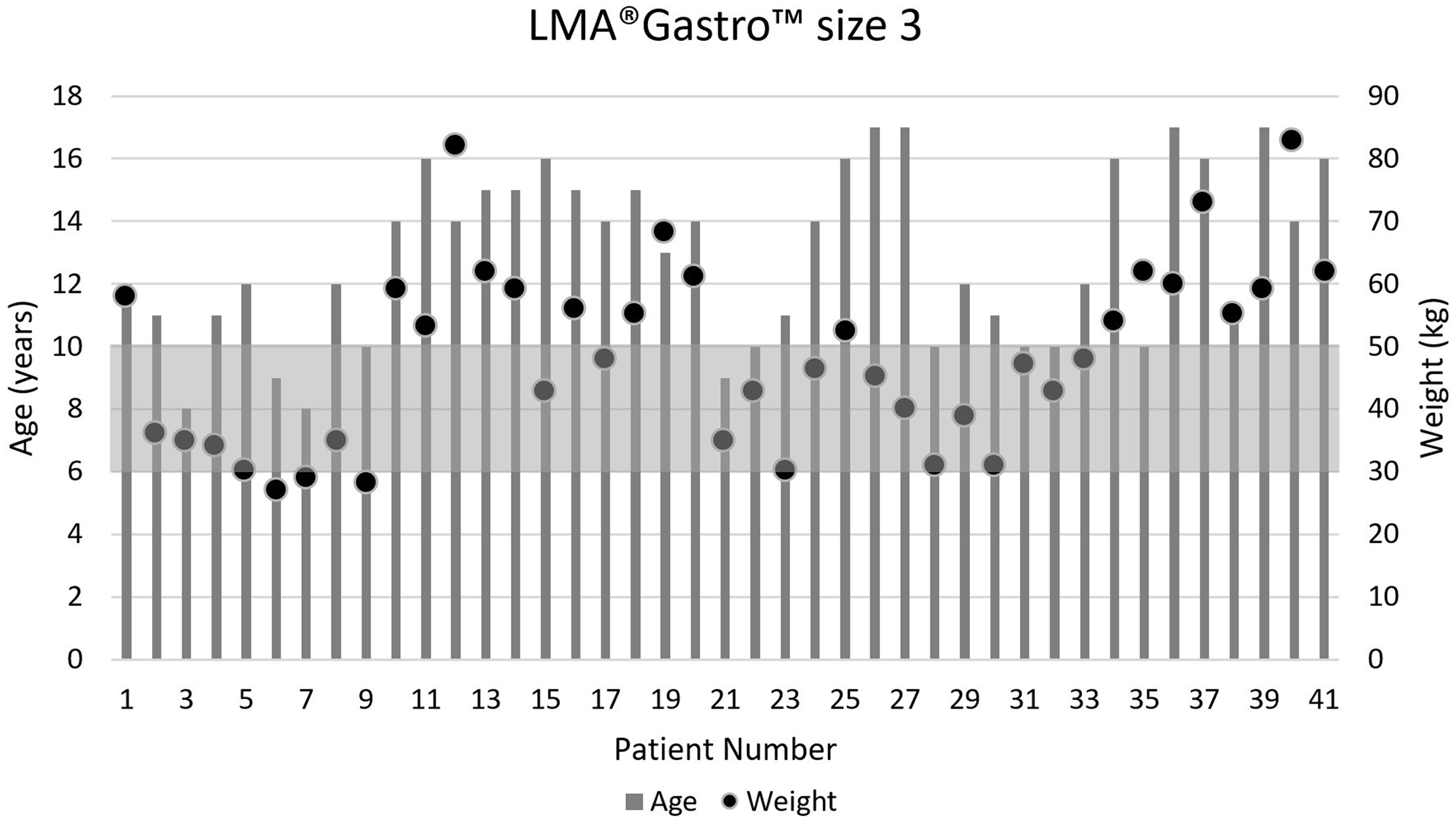
Age and weight of the patients who received the size 3 LMA® Gastro™ (Teleflex Medical, Athlone, Ireland). Shaded area represents the recommended weight range for the device.
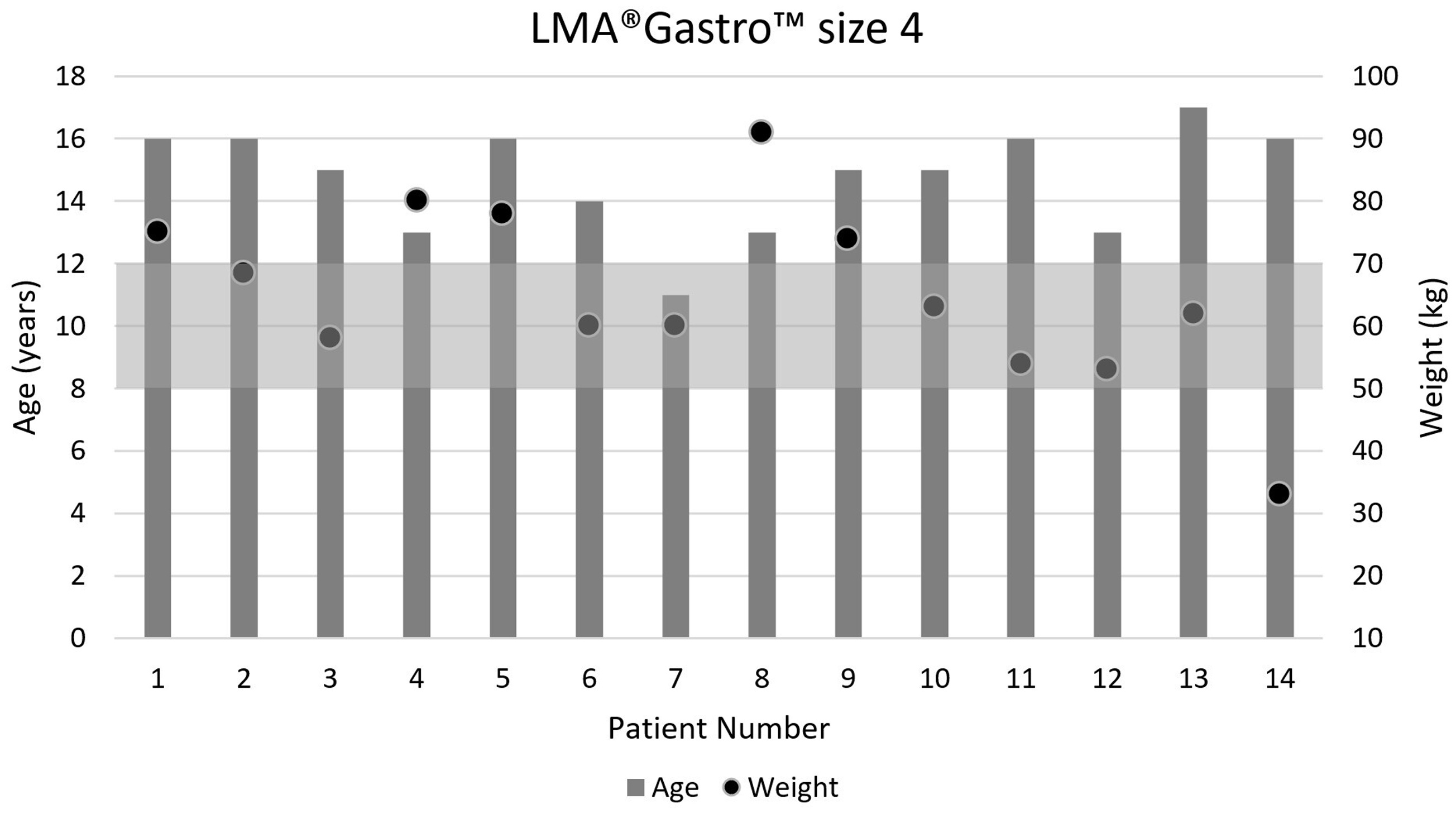
Age and weight of the patients who received the size 4 LMA® Gastro™ (Teleflex Medical, Athlone, Ireland). Shaded area represents the recommended weight range for the device.
Operator characteristics
Of the airway insertions, 37 (67.3%) were performed by specialist anaesthetists and 18 (32.7%) by trainees. Twenty-five different anaesthetists participated in elective upper GI endoscopy during the study period, with 22 of these using the LMA Gastro device at some point. Twenty-five (45.5%) of the endoscopy procedures were performed by specialists and 30 (54.5%) by trainees.
LMA Gastro airway outcomes
Of the 55 patients who received the LMA Gastro as the primary airway device, an adequate airway was achieved in 52 (94.5%) cases. Therefore, there were three cases where the LMA Gastro failed to achieve a satisfactory airway. On two occasions, the operator had difficulty inserting the device, and on the third occasion, ventilation was deemed to be inadequate with the device in situ. On all three occasions, the LMA Gastro device was removed and replaced with an ETT without further incident. All failures involved the size 3 device and are represented in Figure 2 by patient 14 (aged 15 years, 59 kg, female), patient 20 (aged 14 years, 61 kg, female) and patient 21 (aged nine years, 35 kg, female). Of note, two of these three failures were outside the recommended weight range for a size 3 device and occurred when there were no size 4 devices available. All three failed insertions were by specialist anaesthetists.
Forty-six (83.6%) devices were successfully sited on the first attempt, and the remaining six (10.9%) devices were sited on the second attempt. Fifty (90.9%) insertions were rated as ‘easy’ by the treating anaesthetist. Aside from the three instances where the LMA Gastro device was changed for an ETT, there were no airway events recorded, and specifically no episodes of laryngospasm or desaturation were documented.
LMA Gastro endoscopy outcomes
Oesophageal access was achieved in 100% of cases and rated ‘easy’ by the gastroenterologist on all 52 occasions that the LMA Gastro device was used as a conduit. First-pass success was achieved in 51 (98.0%) cases, with a single case requiring three passes of the endoscope by a trainee.
pH probes
A pH probe was inserted in six patients. The anaesthetist described removing the SAD for the pH probe insertion in three of these patients. In the other three patients, no comment was made about the method of pH probe insertion. Where the device was described as being removed for pH probe insertion, it was reinserted immediately after pH probe placement on two occasions and left out on the other occasion, with no further comment made about ongoing airway management.
To explore the issue of pH probes further, we looked at the proportion of patients who had a pH probe inserted in the 53 patients who did not have an LMA Gastro as their airway device. There were 24 patients aged eight years or older who did not have an LMA Gastro, of which the majority (80%) were intubated. Of these, 8/24 (33%) had a pH probe inserted compared to 6/55 (11%) in the LMA Gastro group.
The median total time in the operating theatre for patients having the LMA Gastro device was 28 minutes. The median time for patients aged eight years and older in this audit cohort who had another airway device was 31 minutes.
Comments and lessons learned
The LMA Gastro devices are necessarily bulky compared to traditional supraglottic airways. Based upon the comments we received, some anaesthetists found that a deeper plane of anaesthesia was required to facilitate placement compared to a standard SAD. This was the most common reason cited in the few cases where multiple attempts at placement were required. It was also noted that it was significantly easier to insert the device in the left lateral position and with the cuff deflated, as per the manufacturer’s instructions (although these instructions were not universally followed).
We discovered that it was also easier for the gastroenterologist to perform the endoscopy with the patient in the left lateral position, as this rotates the airway channel of the device down and out of the way of the oesophageal port. This resulted in a change in practice when using the LMA Gastro device in our institution, as prior to this time, almost all paediatric endoscopies were performed with the patient in the supine position. This change may have influenced clinical practice overall, as further inspection of the audit data revealed seven instances where endoscopy was performed in the lateral position in patients who had other airway devices.
We did not receive any information in the free comments section regarding the clinical decision-making around airway device or size.
Discussion
Our main finding was that the LMA Gastro performed well in our paediatric population regarding airway outcomes, and very well in the endoscopy outcomes. Oesophageal access via the device was universally successful and straightforward. Despite our relative inexperience with the LMA Gastro airway, we had a high insertion success rate, and the time spent in the operating theatre was not increased. We postulate that with more experience using the device, overall success rates are likely to improve from the 94.5% observed in this study, and may even approach those seen with the LMA® Gastro™ (Teleflex Medical, Athlone, Ireland) Classic airway device. 5 The first-pass success rate of 83.6% is already comparable to that of other similar devices in both adults and children. 1 ,5–9
It is interesting to note that two of the three patients who had failed insertion of the LMA Gastro were outside the recommended weight range for the chosen device, possibly due to unavailability of the larger size at the time. A retrospective study of LMA insertions in more than 11,000 children examined many risk factors associated with LMA failure. LMA size not matching the manufacturer’s recommendations was more frequently noted in LMA failure on univariate analysis. However, this association was not found in multivariate analysis. 10
pH probes were inserted in just 11% (6/55) of patients who received an LMA Gastro but in 33% (8/24) of patients of a comparable age who did not receive the LMA Gastro. It appears likely therefore that planned pH probe insertion influenced airway choice. In three patients, the anaesthetist described removing the LMA Gastro to insert the pH probe, and in three patients no information was recorded.
Limitations
While our data were collected prospectively, it is pragmatic and observational in nature and does not permit a direct comparison of efficacy and safety of the device with other airway management options for upper GI endoscopy. We also did not collect data to assess the incidence of sore throat or blood on the device after removal in patients with the LMA Gastro. Further studies with larger numbers would be required to answer such questions. No emergency cases were included amongst the audited patients, and it is highly likely that anaesthetists used their usual clinical judgement to exclude patients for whom endotracheal intubation was deemed to be a more suitable option. We are unable to provide any detailed information regarding how any particular airway device or size was chosen.
The LMA Gastro device is currently available in sizes 3, 4 and 5, and therefore is not recommended for patients weighing <30 kg. That said, three patients in our dataset weighed <30 kg, the lightest being 27 kg and nine years of age.
Conclusions
At our institution, the LMA Gastro is a viable airway option for children >30 kg undergoing upper GI endoscopy and provides excellent conditions for endoscope insertion.
Footnotes
Author Contribution(s)
Acknowledgements
The authors would like to acknowledge the staff members of the Gastroenterology Department at the Women’s and Children’s Hospital.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Thirty LMA Gastro devices were made available for use by Teleflex and they provided some education on how to use the devices; Teleflex had no other involvement in the study and the results have not been made available to them prior to publication.
