Abstract
Correct intracuff pressure of endotracheal tubes and supraglottic airway devices is required to avoid complications such as sore throat, dysphagia and dysphonia, while maintaining an adequate airway seal. However, intracuff pressure monitoring of airway devices during general anaesthesia may not receive the attention it deserves. The aim of this survey was to investigate the current practice regarding intraoperative cuff pressure monitoring in hospitals across Australia and New Zealand. An online ten-question survey was disseminated by the Australian and New Zealand College of Anaesthetists Clinical Trials Network to a randomised selection of 1000 Australian and New Zealand College of Anaesthetists Fellows working in private and public hospitals of varying sizes. There were 305 respondents in total, but not all respondents answered all questions. In total, 67 of 304 respondents (22.0%) did not have access to a cuff pressure manometer at their main site of work, and of these, 30 (9.9%) expressed that they would like access to one in their daily practice. Of 288 respondents, 122 (40.0%) reported that they used cuff pressure monitoring as part of their routine practice, but 95 (33.0%) measured the cuff pressure at induction only. For supraglottic airway devices, only 44 of 250 respondents (17.6%) aimed for a cuff pressure of 40–60 cmH2O. Of 255 respondents, 101 (39.6%) aimed for a cuff pressure of 20–30 cmH2O for endotracheal tubes. These findings indicate that educational programmes are required to increase the availability and use of cuff pressure monitoring devices for both endotracheal tubes and supraglottic airway devices across Australia and New Zealand.
Keywords
Introduction
Both endotracheal tubes (ETTs) and supraglottic airway devices (SADs) are widely used airway devices in general anaesthesia, yet intracuff pressure (PINTRACUFF) is a monitoring parameter that may not receive the attention that it deserves in daily anaesthetic practice.1–10 In some countries, such as Canada, national guidelines recommend that cuff pressure manometers be immediately available in the operating theatre, but their use and availability is not mandatory in many institutions around the world. Normal occlusive cuff pressures should be 20–30 cmH2O for ETTs and 40–60 cmH2O for SADs to avoid complications,1–3,5,11–14 while maintaining an adequate airway seal.
Manual palpation of the cuff,6,8 listening to the disappearance of an audible air leak or injection of a standard volume of air into the cuff via a pilot balloon are common practices. However, in one study of monitoring PINTRACUFF by palpation, 40% of patients had a PINTRACUFF above the recommended range. 15 Many other studies support monitoring PINTRACUFF intraoperatively to reduce the incidence of a postoperative sore throat.8,16–23 Although cuff manometers can be used to guide the monitoring of cuff pressures, their use and availability is not mandatory in many institutions around the world. A meta-analysis of nine studies by Hockey et al. demonstrated that adjustment of PINTRACUFF guided by objective measurement as compared with subjective measurement or observation of the pressure value alone, has benefits in preventing adverse effects including voice hoarseness, cough, tissue damage, and aspiration. 24 A PINTRACUFF of greater than 50 mmHg (68 cmH2O) in an ETT for only 15 min can destroy mucosal columnar epithelium and will partially denude the basement membrane. 25 Similar adverse effects were demonstrated by mucosal pressure measurements in SADs by Brimacombe et al. 26
The aim of this survey was to investigate the current practice regarding intraoperative cuff pressure monitoring in hospitals across Australia and New Zealand and to elucidate attitudes behind this practice. The availability of cuff pressure manometers and preferred method of cuff pressure estimation was also investigated.
Materials and methods
Ethics approval
This electronic survey was approved as a Clinical Audit/Quality Assurance activity by the local institutional ethics committee (HREC/17/QRBW/243).
Survey design and implementation
This electronic survey was designed using a commercial internet-based service, SurveyMonkey Inc.® (San Mateo, CA, USA). It consisted of ten questions and was refined for simplicity, brevity, and to avoid ambiguity for respondents, while still providing comprehensive information regarding their workplace, practices and opinions or attitudes and pattern of use of cuff manometers intraoperatively. It was designed to be able to be completed in 5 min or less. Six anaesthetists reviewed the survey and minor changes were made to improve readability, avoid confusion, limit responder fatigue and increase response rates. A pilot survey was conducted in a metropolitan, tertiary referral hospital prior to dissemination by the Australian and New Zealand College of Anaesthetists Clinical Trials Network (ANZCA CTN) to a randomised selection of 1000 ANZCA Fellows working in hospitals across Australia and New Zealand. An invitation from ANZCA CTN to participate was sent via email. Participation was voluntary and all information was kept anonymous and confidential.
Statistical methods
All responses were downloaded from the internet-based survey provider into a Microsoft Excel (Microsoft, Redmond, WA, USA) spreadsheet for analysis. All data were analysed using number and percentage of responses using STATA 15® (StataCorp. 2017. Stata Statistical Software: Release 15. College Station, TX: StataCorp LLC).
Results
Of the 1000 anaesthetists in Australia and New Zealand invited by ANZCA CTN to participate, a total of 305 (30.5%) responded. Table 1 outlines characteristics relating to the survey participants’ main site of practice. There were participants from New Zealand and all Australian states and territories. Approximately two-thirds of the participants were based at a public hospital.
Hospital site characteristics.

For survey questions not responded to by all 305 respondents, ‘n’ is provided in brackets ( ) for that question.
Table 2 shows survey responses relating to SAD use and Table 3 outlines intraoperative cuff pressure monitoring practices.
Supraglottic airway device survey responses

For survey questions not responded to by all 305 respondents, ‘n’ is provided in brackets ( ) for that question.SADs: supraglottic airway devices; LMA: laryngeal mask airway; LTS: laryngeal tube suction; CPV: cuff pressure valve; LTSD: laryngeal tube suction device.
Cuff pressure monitoring practices

ETTs: endotracheal tubes; SADs: supraglottic airway devices.For survey questions not responded to by all 305 respondents, ‘n’ is provided in brackets ( ) for that question.
For 24.3% of respondents, cuff manometers were available in each operating theatre and for 45.1%, they stated availability in another area of the operating theatre complex.
Of the 304 anaesthetists who responded to the question regarding access to a cuff pressure manometer, 237 (78.0%) confirmed they had access to one in their institution, but only 122 (40.0%) used them routinely in their daily practice. An additional 17 respondents measured cuff pressure at induction and again if any concerns arose with ventilation or the seal, during long cases, or if nitrous oxide was being used. Four respondents reported monitoring cuff pressures at induction and at regular intervals only for paediatric cases, whereas four respondents stated reserving their practice of the use of ETT cuff pressure monitoring for anterior cervical spine surgery only. Eight respondents would check the cuff pressure only if concerns arose with ventilation using either an SAD or ETT. Based on the 122 responses regarding the type or brand of cuff manometer used, the VBM® Cuff Pressure Gauge (VBM® Medical Inc. Noblesville, IN, US), Portex manometer (Smith Medical, Hythe, UK) and Covidien Mallinckrodt™ Cuff Pressure Gauge (Covidien®, Boulder, CO, US) brands were the most popular types (Figure 1), whereas 84 participants were unsure of the brand of the manometer used in their institution.
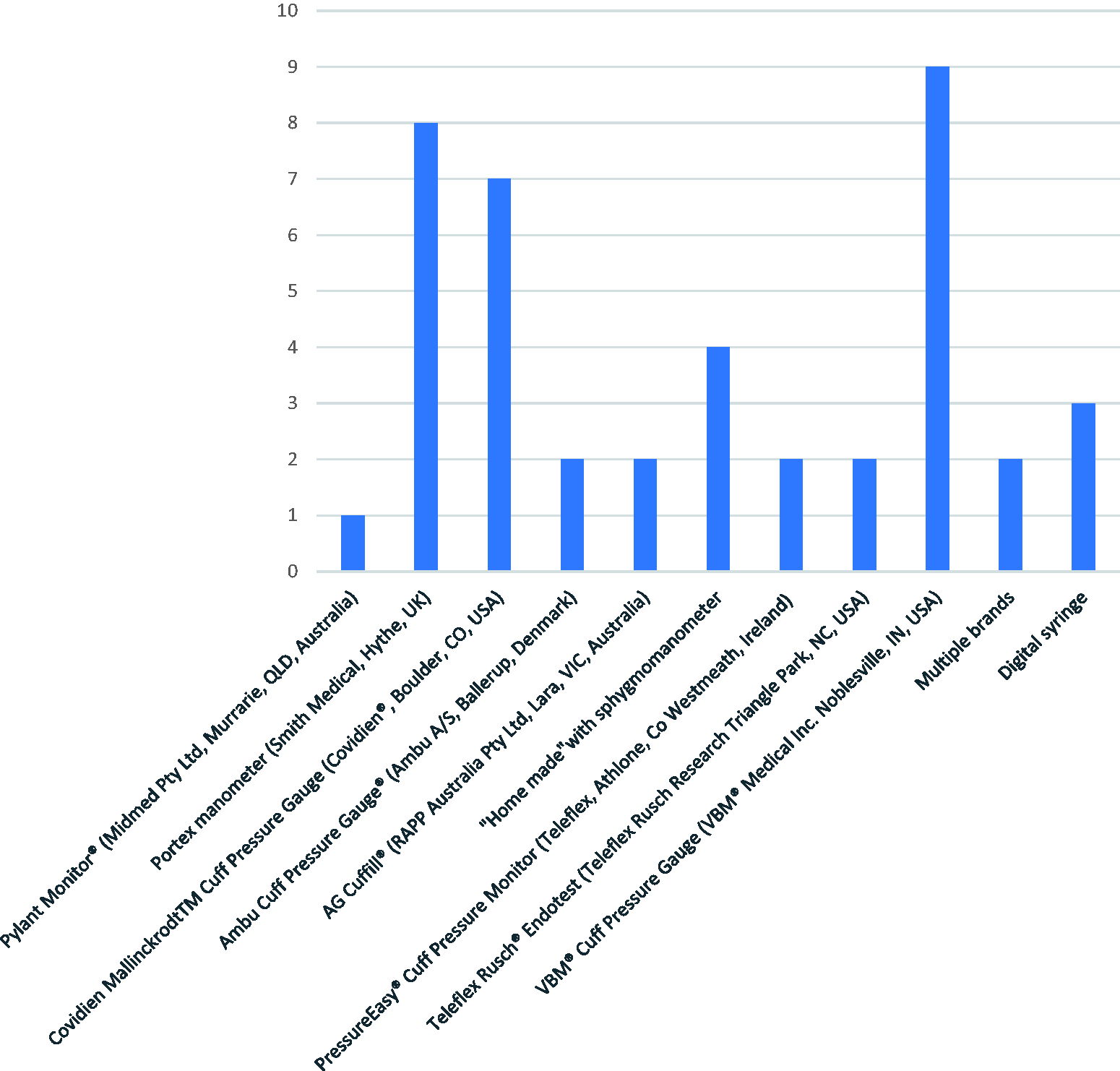
Types of cuff manometers used.
In response to this question, one respondent stated that they detest cuff pressure manometers due to their use causing leaks.
One hundred and twenty-eight (44.4%, n = 288) respondents stated they did not use intracuff pressure measurement for airway devices and of the remainder, the majority were measured at induction only (95, 33.0%, n = 288). Only 27 (9.4%, n = 288) reported measuring at regular intervals following induction in their daily practice during maintenance of anaesthesia. Optimal intracuff pressure for ETTs (20–30 cmH2O) was aimed for by 101 (39.6%, n = 255), whereas only 44 (17.6%, n = 250) of respondents aimed for optimal intracuff pressure (40–60 cmH2O) when they used SADs. Overall, 154 (60.4%, n = 255) and 206 (82.4%, n = 250) of respondents aimed for an intracuff pressure outside the optimal range for ETTs and SADs respectively.
Discussion
Our survey results provide insight into the current practice of ETT and SAD cuff pressure monitoring in general anaesthesia in Australia and New Zealand. Sixty-seven (22.0%, n = 304) respondents had no access to cuff manometers in their main hospital site. Although 30 (9.9%, n = 304) respondents indicated that they would like to have access to cuff manometers, 37 (12.2%, n = 304) did not see the need for access to one. Furthermore, there was an expression of concern for the cause of leaks by one respondent, which has been demonstrated in the literature.27,28 For example, cuff pressure drops can be experienced from the initial connection or disconnection of the manometer to the pilot balloon and less frequently by the pressure gauge of the manometer. 27 However, correct use of cuff manometers with three-way valves or the use of continuous monitoring techniques have been described to avoid this complication.29,30
Access to and the routine use of cuff manometers in Australian and New Zealand institutions is not mandatory.
Of the 305 respondents, 176 (57.7%) stated that for general anaesthesia, greater than half of their airways comprised of SADs, which was similar to the NAP4 study. 31 The LMA-Classic™ (Teleflex Medical Europe Ltd, Athlone, Co Westmeath, Ireland) was by far the most used SAD. The NAP4 study revealed the most common complication with the use of SADs was aspiration. 31 In our study, intracuff pressure measurement of airway devices is not applied by 44% of respondents and the majority measure at induction only (33.0%), whereas 9.4% will measure at regular intervals following induction in their daily practice during maintenance of anaesthesia.
For 137 (45.1%, n = 304) respondents, cuff manometers were reported to be available in the operating theatre complex if needed, and for 74 (24.3%, n = 304) respondents, cuff manometers were a routine monitoring device available in all operating theatres.
Of the 122 (42.4%, n = 288) respondents who routinely measure cuff pressures of their airway devices, 95 (33.3%, n = 288) only do so at induction. It could be argued that the pressure at induction is not representative of the whole period of anaesthesia due to changes in position or the use of nitrous oxide.
Historically, the use of cuffed ETTs was reserved for children aged eight years or older to minimise the risks of post-extubation laryngeal oedema. 32 Researchers have consistently presented evidence that appropriate use of cuffed ETTs is as safe as uncuffed ETTs. 32 Risks related to using cuffed ETTs in young children increase when guidelines for safe and appropriate use are not followed. 32 Four respondents reported use of cuff pressure monitoring for paediatric or neonatal patients, but other paediatric anaesthetists might use uncuffed ETTs rather than cuffed ETTs in their practice. The proportion of respondents who practised paediatric anaesthesia was unknown.
As with any survey-based research, a limitation of our survey was the potential for response bias, given the overall low response rate of 30.5%. Respondents may possibly be avid users of cuff manometers, or may be uninterested in their use. It is possible that recipients of the survey invitation who did not perform cuff pressure monitoring as part of their routine practice may have been less inclined to participate. Therefore, the proportion of respondents without access to cuff manometers could be higher or lower in reality. Considering that the response rate is important for the reliability of the results, it must be considered that our low response rate will limit the generalisation of our findings. However, ANZCA member surveys typically have a response rate of only 5%–40%, 33 and the degree of representativeness is important. We consider that the participants of our survey would be likely to be representative of the target population of anaesthetists in Australia and New Zealand.
Another limitation is the absence of information on the duration of the anaesthetic cases in question as a variable influencing practice. Some respondents may use cuff monitoring for long cases but not for short cases, or they may not require repeated pressure monitoring due to the short duration of cases. Furthermore, our survey did not differentiate between the use of cuff manometers for SADs and ETTs. There was one survey question that required the respondent to specify which SAD they use routinely, including an i-gel® (Intersurgical, Wokingham, Berkshire, UK), which has no cuff; thus monitoring of the SAD was not possible for the 108 respondents who used this device on a regular basis. There was also the option to list as ‘other’ any SADs that were not included in the list provided. These responses were analysed individually.
A systematic review and meta-analysis by Hockey et al. concluded that subjective measurement methods alone may lead to patient-related adverse effects and inaccuracies. 24 It is recommended that an objective form of measurement is used. 24 However, the auditory and palpation methods are commonly used by respondents (167 (54.8%) and 154 (50.5%) respectively). Cuff pressure monitoring is accurate (if appropriately calibrated), inexpensive and non-invasive and therefore there should be little contraindication to its use in most cases.
High cuff pressures were present in a large proportion of patients with in situ ETTs and SADs in multiple audits.13,34–39 Although adverse effects due to high intracuff pressures may not commonly cause serious adverse effects, it would be prudent to minimise this risk by ensuring the availability of cuff pressure manometers ideally in every operating theatre, otherwise in every institution’s operating theatre complex for routine use during general anaesthesia.
In summary, regular use of cuff pressure monitoring devices appears to occur infrequently, with 22.0% (n = 304) of respondents reporting hospital operating theatres having no availability of appropriate devices in Australia and New Zealand and 44.4% (n = 288) of anaesthetists not using cuff monitoring. Of those who monitor cuff pressure as part of their routine practice, only 101 (39.6%, n = 255) aimed for an ETT cuff pressure of 20–30 cmH2O and 44 (17.6%, n = 250) aimed for an SAD cuff pressure of 40–60 cmH2O. These findings indicate that educational programmes are required to increase the availability and use of cuff pressure monitoring devices for both ETTs and SADs across Australia and New Zealand.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
