Abstract
Acute kidney injury (AKI) is common in intensive care patients. While creatinine definitions for AKI have been validated, oliguria criteria are less well evaluated in children. Our study compared the validity and agreement of creatinine and oliguria criteria for diagnosing AKI in a large mixed medical, surgical and cardiac paediatric intensive care unit (PICU), and assessed the significance of their independent and combined effects on predicted mortality relative to paediatric index of mortality (PIM risk of death) on admission. Creatinine measurements during PICU admissions in 2005 and 2015 were obtained from the electronic medical record. Urine output was reviewed to identify periods of oliguria of more than eight hours. We used the PIM3 model for predicted risk of death. AKI based on creatinine rise occurred in 23.6% of the total 2203 admissions (10.0%, 8.2% and 5.6% for mild, moderate and severe categories, respectively). Oliguria occurred in 11.4% (8.4%, 1.8% and 1.2% for mild, moderate and severe categories, respectively) and overlapped only partially with creatinine criteria. Mortality relative to predicted mortality increased with increasing creatinine and oliguria severity, but was lower than predicted where oliguria occurred without creatinine rise. AKI by creatinine criteria and/or oliguria are common in the PICU, but criteria overlap only partially. Increasing severity of creatinine rise and oliguria confers increasing risk-adjusted mortality, especially for admissions with low PIM3 risk of death. The mortality of patients with AKI defined by oliguria alone is low. Defining AKI by oliguria alone has less clinical utility and may not represent true AKI.
Introduction
Acute kidney injury (AKI) is a commonly encountered complication in critically ill children. 1 , 2 Previous data demonstrate AKI to be an independent predictor of mortality, increased duration of mechanical ventilation and paediatric intensive care unit (PICU) length of stay.2–5
Multiple definitions for AKI exist, with three current:
The paediatric adaptation of the RIFLE (risk, injury, failure, loss, end-stage renal disease) criteria (pRIFLE) classifies AKI based on change in estimated glomerular filtration rate (eGFR; severity groups 25%, 50% and 75% reduction) or oliguria duration <0.5 ml/kg/h (severity groups 8, 16 and 24 hours).6 The AKIN (Acute Kidney Injury Network) Classification criteria rely on rise in serum creatinine (not eGFR) within 48 hours (rise of 1.5, 2 and 3 times baseline) and oliguria <0.5 ml/kg/h duration (6, 12 and 24 hours).7,8 The Kidney Disease: Improving Global Outcomes (KDIGO) classification stratifies AKI by increase in serum creatinine (1.5, 2 and 3 times baseline) and urine output <0.5 ml/kg/h (6, 12 and 24 hours).9,10 These criteria have been applied in a recent large study of AKI epidemiology in critically ill children.2,11
When compared, AKI incidence varies across definitions, but all criteria demonstrate progressively higher mortality with increasing AKI severity. 12 Multiple studies validate these criteria 8 , 10 ,12–14 but are substantially weighted towards creatinine assessment, and there is a paucity of data robustly interrogating or validating oliguria criteria. In a systematic review, only five studies included urine output 14 and only three included patients with indwelling urinary catheters. Recently, the AWARE study compared creatinine and oliguria criteria, concluding that oliguria of six hours or more alone defined clinically significant AKI and identified increased mortality risk despite an absent creatinine rise. 2 , 11 In adults, the addition of oliguria criteria has increased patient numbers defined as having AKI and identified AKI earlier. 15
Expectedly, renal injury may manifest with oliguria and/or a creatinine rise. While current paediatric AKI definitions place equal weight on both, in our clinical practice, we observe bias towards creatinine for diagnosing AKI in the PICU. There are, in our view, good reasons for this. Short oliguria periods occur frequently and are often challenged with fluid or diuretics before the six to eight hours of oliguria needed to ‘diagnose’ AKI. These shorter periods, below the threshold for AKI definitions, trigger close scrutiny for AKI development. Further, in our experience, some patients demonstrate prolonged oliguria without other AKI features, for example patients with antidiuretic hormone secretion who have respiratory or cerebral pathology (either appropriate or inappropriate) or patients receiving exogenous vasopressin. Our study compared the validity and agreement of creatinine and oliguria criteria for diagnosing AKI in the PICU, and assessed the significance of their independent and combined effects on predicted mortality.
Methods
This study received ethics and governance approval through the Sydney Children’s Hospital Network Human Research Ethics Committee prior to commencement (LNR/17/SCHN/59).
Study population
We retrospectively reviewed medical records for all admissions to the PICU at the Children’s Hospital Westmead in 2005 and 2015. Our PICU cares for a mixed population of cardiac (bypass and non-bypass), general medical and surgical patients. Two cohorts a decade apart were chosen to compare validity of definitions over time and because AKI incidence and standardised mortality rate (SMR) may change a little from year to year. 16 Studies in adults have shown considerable differences in case mix and AKI incidence across 30 years. 17
Data collection
All clinical data for admissions were prospectively recorded in an electronic clinical information system for intensive care, including hourly fluid balance and laboratory data. We reviewed exported admission data for occurrence of elevated plasma creatinine and oliguria. This study, initially examining 2005 data, was commenced prior to the KDIGO definitions and before the AWARE study was published. As in the KDIGO classification, 9 we classified AKI as a creatinine rise ≥1.5 times from baseline with severity stratification categories: no AKI <1.5 times baseline, mild ≥1.5–2 times, intermediate ≥2–3 times and severe ≥3 times. Where a baseline creatinine was not available, the upper limit of normal creatinine for age was used, or the final measured creatinine after recovery if lower. Where creatinine rise ≥1.5 times baseline occurred but creatinine values remained within the normal range for age, results were scrutinised by the authors by assessing the creatinine rise pattern to assess for a true rise and fall.
We used creatinine values rather than eGFR, which is essentially a reciprocal of creatinine (eGFR = k×height/plasma creatinine, the Schwartz formula) because height is unavailable for many of our patients. Creatinine has been measured enzymatically since 2005, giving lower values than the kinetic Jaffe method (on which the Schwartz formula is based) and with wider reference ranges. Creatinine rises to ≥1.5, ≥2 and ≥3 times baseline represent ≥33%, ≥50% and ≥67% GFR decrease, whereas the pRIFLE criteria use 25%, 50% and 75% reduction.
We defined AKI by oliguria as urine output <0.5 ml/kg/h with severity categories: none, mild ≥8 hours, moderate ≥16 hours and severe ≥24 hours (as per the pRIFLE definition available when data collection and analysis commenced). 6 For patients without urinary catheters and hourly urine output measures, recorded urine output (e.g. weighed nappies) was averaged and converted to ml/kg/h. For admissions with ≥16 hours of oliguria, urine output data were manually screened to ensure data integrity.
Illness severity on admission was assessed using the paediatric index of mortality (PIM risk of death). PIM318 (2015 recalibration) was used for both years, with variables calculated retrospectively for 2005.
Statistical analysis
We used R for statistical computation (R Foundation for Statistical Computing, Vienna, Austria). Logistic regression (generalised linear modelling) was used in univariate and multivariate analysis to determine the individual and combined effects of PIM3 risk of death, creatinine category and oliguria category on the binary outcome variable ‘died in PICU’. The predictive performances of the models were compared using the receiver operating characteristic area under the curve (ROC AUC). Continuous variables including PIM3 risk of death for the two years were compared by chi-square or Wilcoxon two-sample test with normal and t approximations. AKI severity categories, both overall and by year, and by admission type (both years combined), were assessed with chi-square tests. The presence of a creatinine rise and/or oliguria were compared by McNemar’s test of agreement. PIM3 risk of death, in intervals of 0.05 from 0 to 1.0 (both years), was assessed against creatinine and oliguria categories by Kruskal–Wallis chi-square test.
Results
There were 957 admissions in 2005 and 1246 in 2015. Creatinine data were available for all admissions. Urine output data were available for 90% of admissions and were missing for only 22 admissions that had a significant creatinine rise. All other admissions without urine data had no creatinine rise and had illnesses with low risk and where urine measurement was not prescribed.
Patient age and weight distributions were broadly similar between years (see Supplemental Material). Admissions post cardiopulmonary bypass (CPB) were constant (24.5%), but non-bypass postoperative admissions rose from 24.5% to 30.5% and medical admissions fell from 51% to 45% (3 × 2 chi-square, P = 0.005). There was no significant change in mean PIM3 risk of death between years (3.57% and 2.57%, P = 0.36, not significant; median 0.46% and 0.45%; range 0.0001–0.999; Table 1).
Demographic and admission data across two year cohorts.
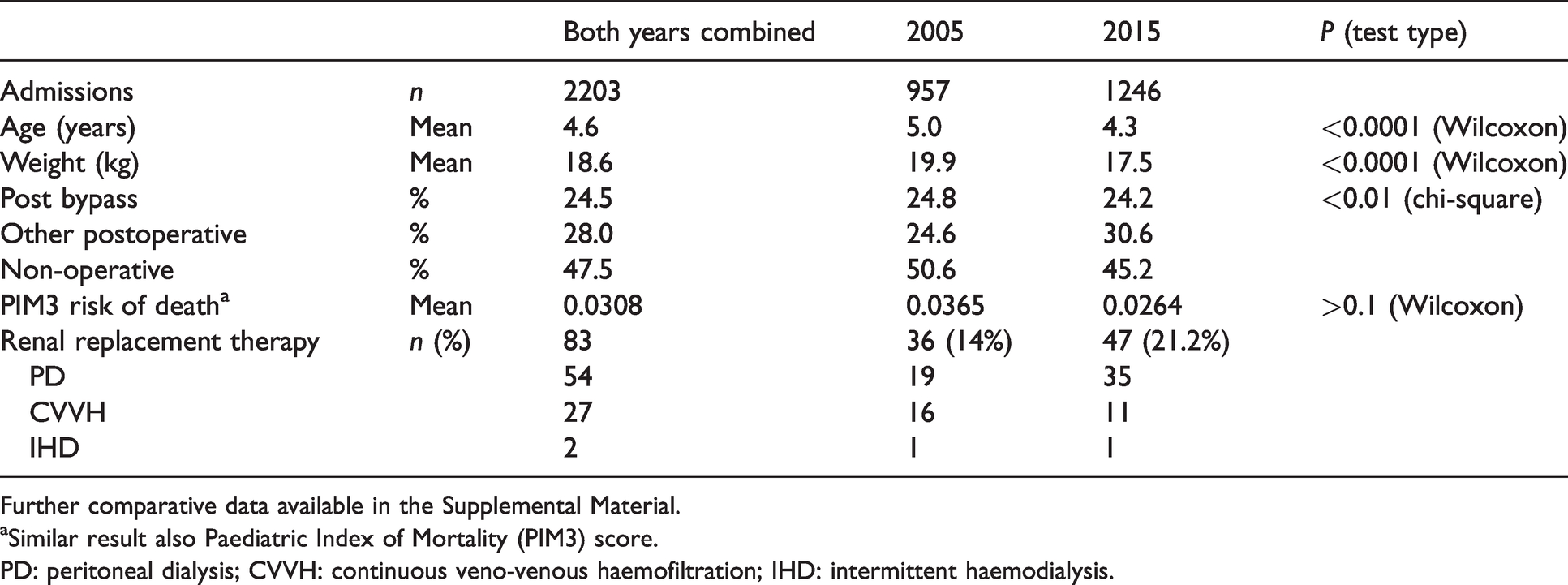
Further comparative data available in the Supplemental Material.
aSimilar result also Paediatric Index of Mortality (PIM3) score.
PD: peritoneal dialysis; CVVH: continuous veno-venous haemofiltration; IHD: intermittent haemodialysis.
In 2005, 27.3% of admissions had AKI by creatinine criteria, with severity subgroups shown in Table 1. In 2015, 21.2% of admissions had AKI by creatinine criteria. The reduction in overall AKI incidence was statistically significant (P < 0.0005; Table 2).
Incidence of acute kidney injury by creatinine and oliguria by year cohort and admission subtype.
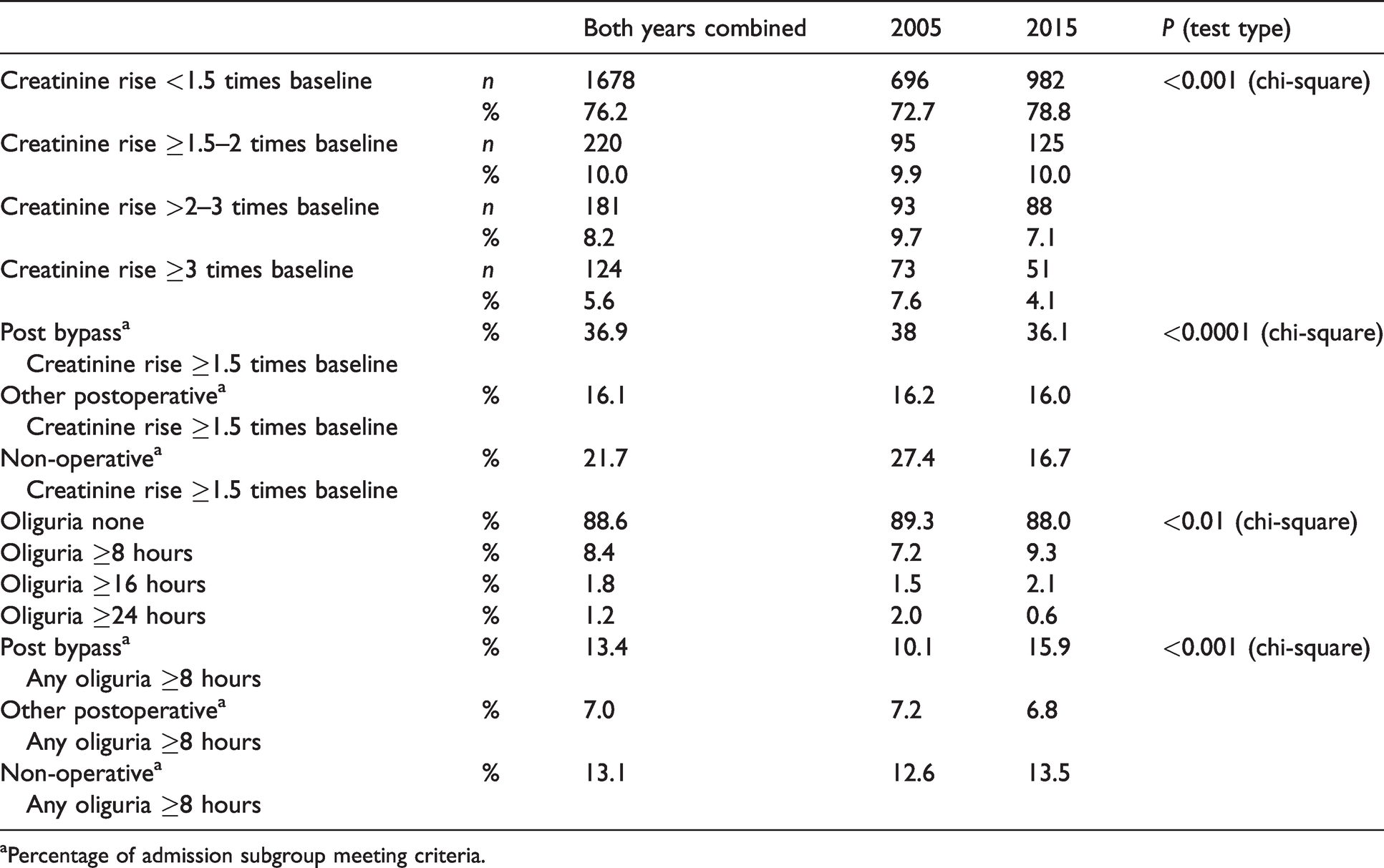
aPercentage of admission subgroup meeting criteria.
Of admissions with AKI by creatinine criteria, 74 (29%) had oliguria of eight hours or more in 2005. An additional 28 (2.9%) of admissions had oliguria of eight hours or more with no associated significant creatinine rise (Figure 1). In 2015, 64 (24.0%) admissions with AKI by creatinine criteria also had oliguria of eight hours or more. Eighty-six (7.0%) of total admissions in 2015 had oliguria of eight hours or more without an associated significant creatinine rise. For AKI by both criteria, a period of oliguria of eight hours or more had a high specificity (92%) but low sensitivity (26%) for AKI by creatinine criteria.
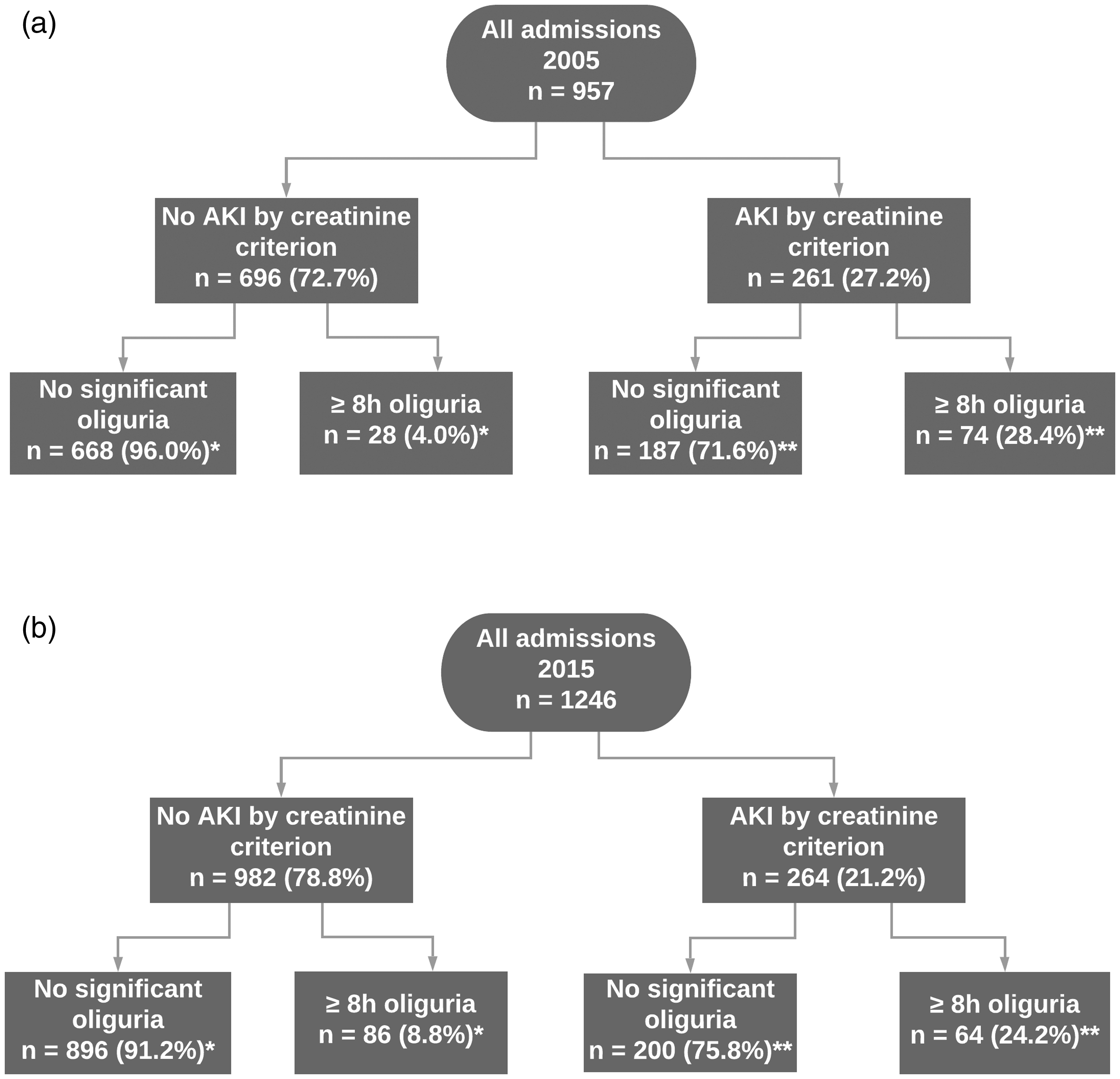
Total occurrence of acute kidney injury (AKI) by creatinine and oliguria criteria for each year cohort. *Percentage of admissions with no AKI by creatinine criteria. **Percentage of admissions with AKI by creatinine criteria.
Oliguria and creatinine rise occurred together in 138 (6.3%) admissions over the two years. Table 3 demonstrates the association between AKI diagnosed by oliguria and creatinine criteria, with only limited overlap between the two criteria. Out of 61 deaths, 50 were associated with creatinine rise and 23 with oliguria, of which only one had oliguria alone (Table 3 and Supplemental Table S9).
Acute kidney injury by creatinine and oliguria criteria in all admissions for 2005 and 2015, and mortality.
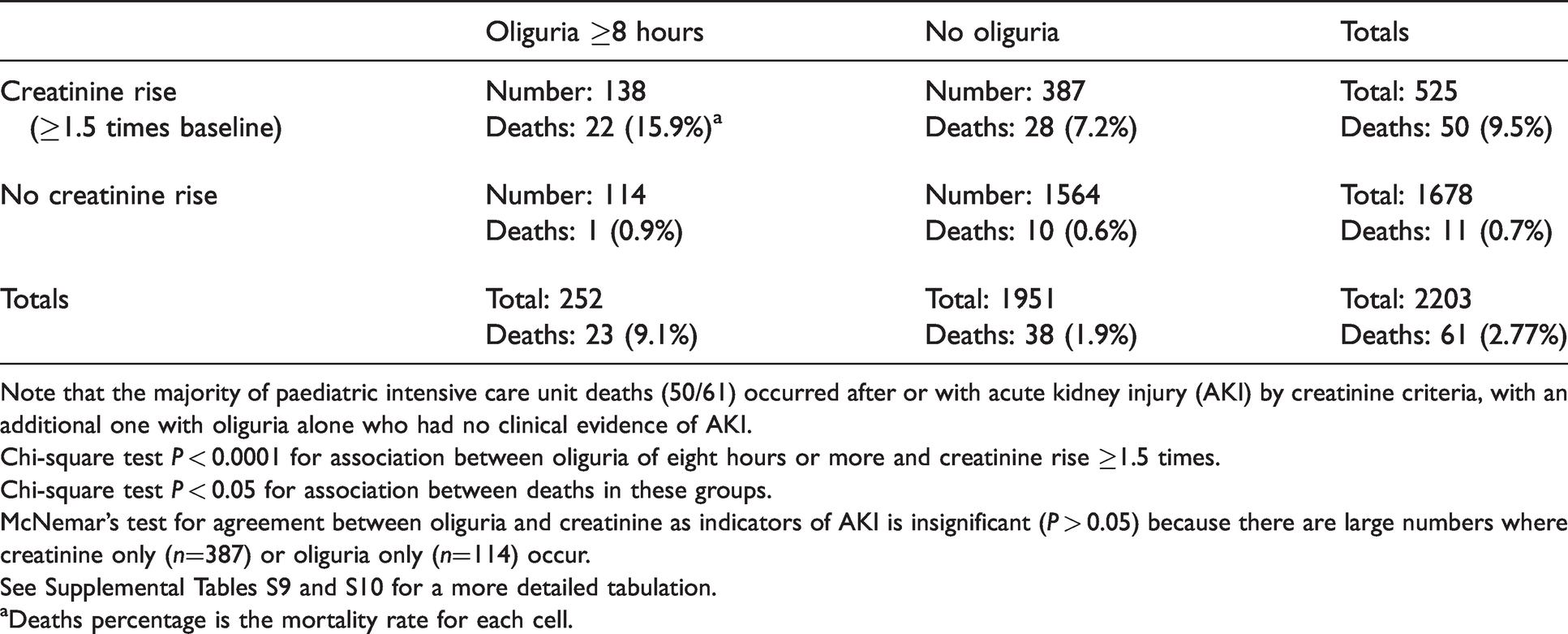
Note that the majority of paediatric intensive care unit deaths (50/61) occurred after or with acute kidney injury (AKI) by creatinine criteria, with an additional one with oliguria alone who had no clinical evidence of AKI.
Chi-square test P < 0.0001 for association between oliguria of eight hours or more and creatinine rise ≥1.5 times.
Chi-square test P < 0.05 for association between deaths in these groups.
McNemar’s test for agreement between oliguria and creatinine as indicators of AKI is insignificant (P > 0.05) because there are large numbers where creatinine only (n=387) or oliguria only (n=114) occur.
See Supplemental Tables S9 and S10 for a more detailed tabulation.
aDeaths percentage is the mortality rate for each cell.
Oliguria of eight hours or more occurred in 11.4% of admissions across the two years (Table 2, P = 0.3, not significant). Across both years, oliguria was not temporally related to AKI as detected by creatinine criteria. Oliguria of eight hours or more occurred within two days prior to a ≥1.5 times creatinine rise in 11 admissions only (six in 2005 and five in 2015). Similarly, for admissions with oliguria following a creatinine rise, this occurred within two days following the creatinine rise in only 29 admissions in 2005 and 36 in 2015. Across both years, oliguria was often temporally unrelated to the initial creatinine rise, not occurring within 48 hours before or after a creatinine rise in 53.5% (76/142).
Across both years, increasing oliguria duration was associated with increasing severity of creatinine rise. For admissions with AKI by both criteria, mean creatinine ratios (ratio between peak and baseline creatinine) were 2.7, 3.9 and 4.4 for oliguria durations of 8, 16 and 24 hours (Kruskal–Wallis chi-square P < 0.0001). Oliguria of more than 16 hours was uncommon in both year groups (Table 2). Three admissions in 2005 and 12 in 2015 had oliguria of more than 16 hours without a significant creatinine rise. No admissions demonstrated oliguria of more than 24 hours without also having a significant creatinine rise.
We compared creatinine and oliguria criteria in admission subtypes. AKI defined by creatinine occurred in 36.9% of admissions post CPB and in 21.7% of non-surgical admissions and 16.1% of non-bypass surgical admissions (Table 2; chi-square P < 0.0001). The higher incidence in the CPB and non-surgical categories was mostly of mild and moderate severity (see Supplemental Material). AKI by oliguria criteria was also most common in admissions post CPB (13.4%; Table 2). The majority of admissions with oliguria alone were in non-surgical admissions (68% across both years).
Patients who developed AKI by either criterion during their admission had higher illness severity on admission according to the PIM3 risk of death (Table 4). Severity of creatinine rise and of oliguria were strongly associated with increasing mortality after adjusting for the PIM3 risk of death on admission (Figure 2; P < 0.05).
Paediatric Index of Mortality risk of death in acute kidney injury categories.
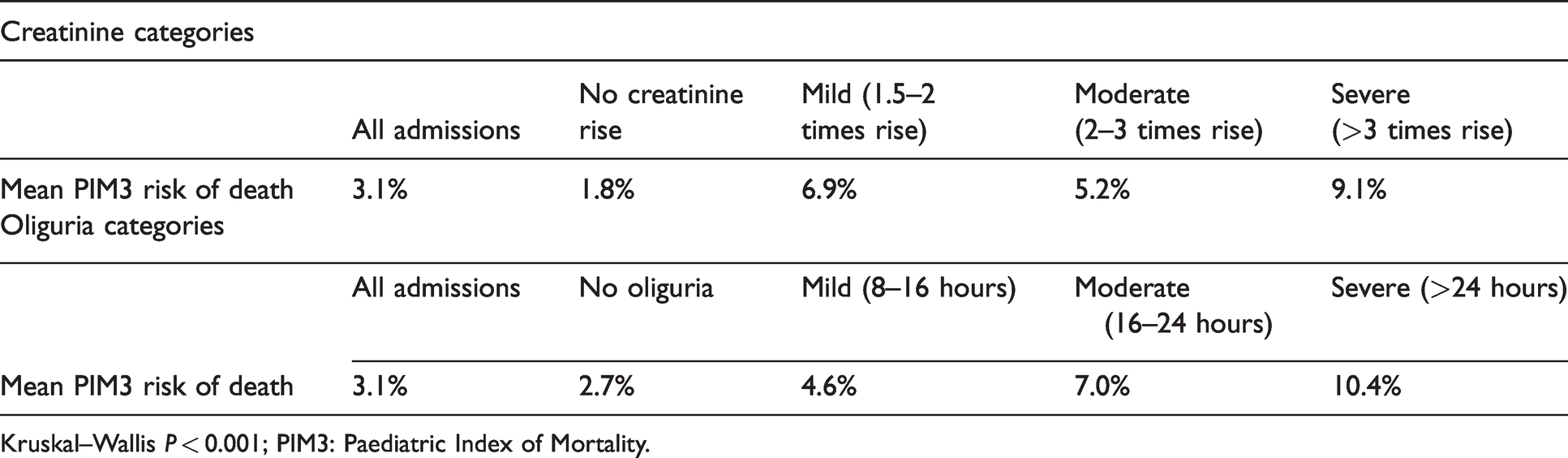
Kruskal–Wallis P < 0.001; PIM3: Paediatric Index of Mortality.
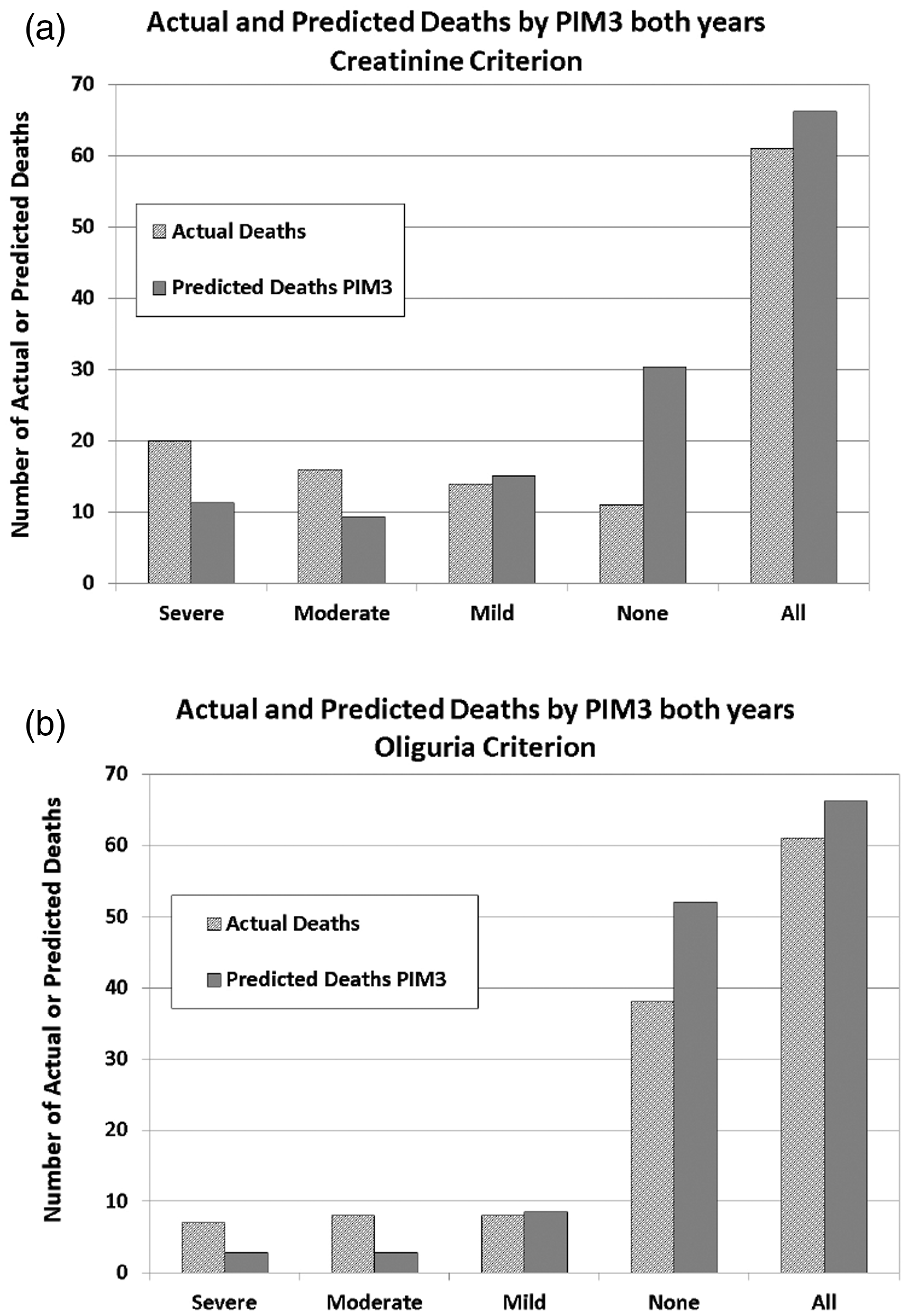
Actual and predicted deaths for the two years combined.* (a) Creatinine criteria. (b) Oliguria criteria. *Note that the columns for all admissions are not equal in height because Paediatric Index of Mortality (PIM3) is calibrated for all paediatric intensive care units in Australia and New Zealand for two-year cohorts, and our standardised mortality ratio was slightly <1.0 in this dataset.
Oliguria without any creatinine rise had a low risk-adjusted mortality (Figure 2(b)). Among 114 admissions with AKI defined by oliguria alone without creatinine rise, there was only one death in a patient with severe spinal muscular atrophy and pneumonia. The SMR for this group was 0.32, whereas the SMR for 1564 admissions with no oliguria or creatinine rise was 0.37.
Finally, logistic linear regression models were run on the dataset to explore the relationship between death in the PICU and the explanatory variables of PIM3, creatinine ratio category and oliguria category. When oliguria or creatinine ratio was added to the model with PIM3 score (PIM3 score=loge(PIM3 risk of death)/(1–PIM3 risk of death)), creatinine increased the association with mortality more than oliguria did, with this effect greater still when both criteria were added together (detail in Supplementary Table S12). Figure 3 shows the ROC AUC for baseline PIM3 risk of death model (AUC=0.902) and for a model with combined PIM3, creatinine ratio and oliguria categories and an interaction term between PIM3 and ratio (AUC=0.946). The increased mortality risk associated with creatinine rise was greatest in admissions with a low risk of death on admission (Figure 4).
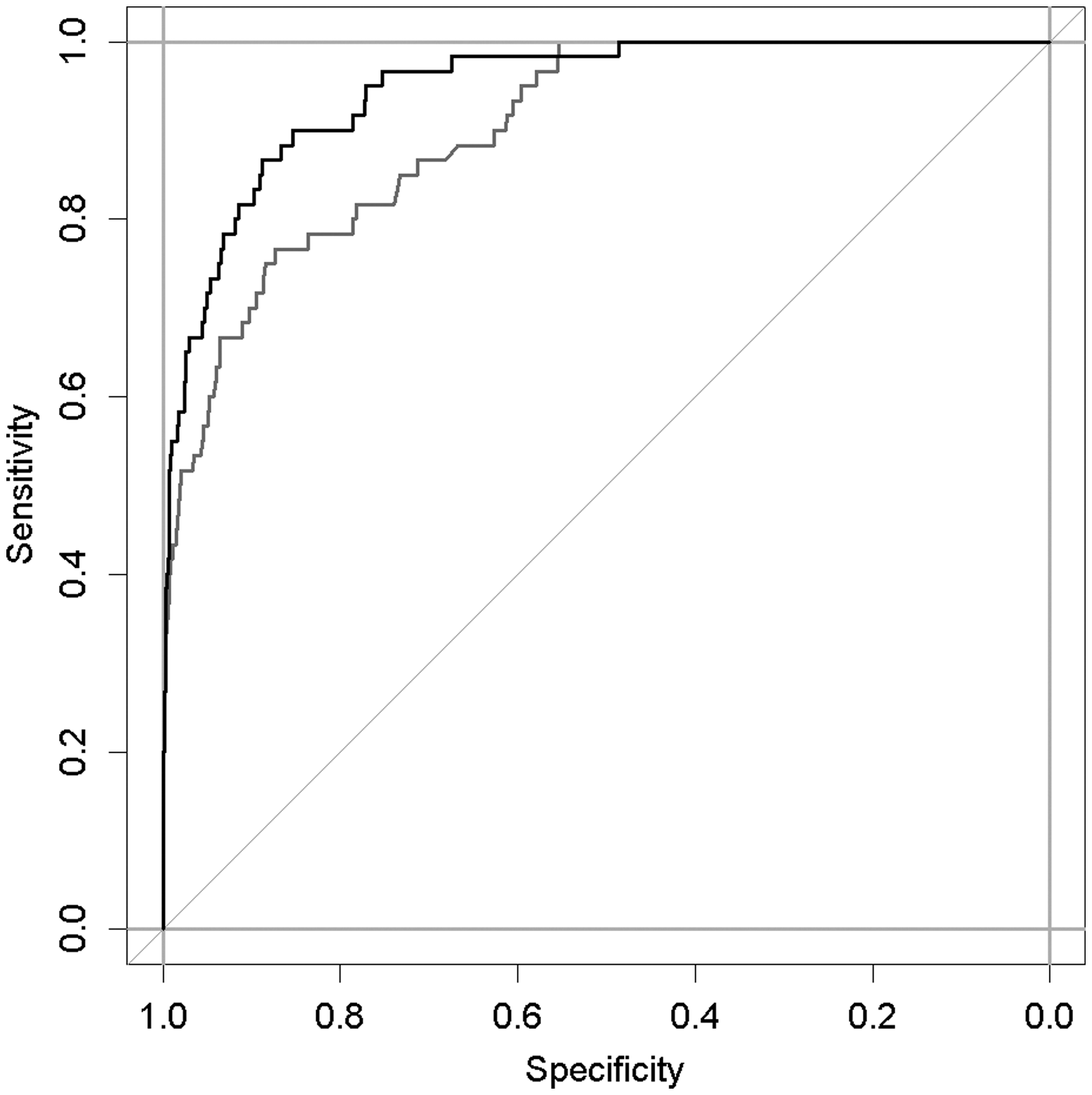
Receiver operating characteristic (ROC) curves for the baseline Paediatric Index of Mortality (PIM3) model (grey) and for a model with PIM3, creatinine ratio and oliguria categories and an interaction term between PIM3 and ratio (black).
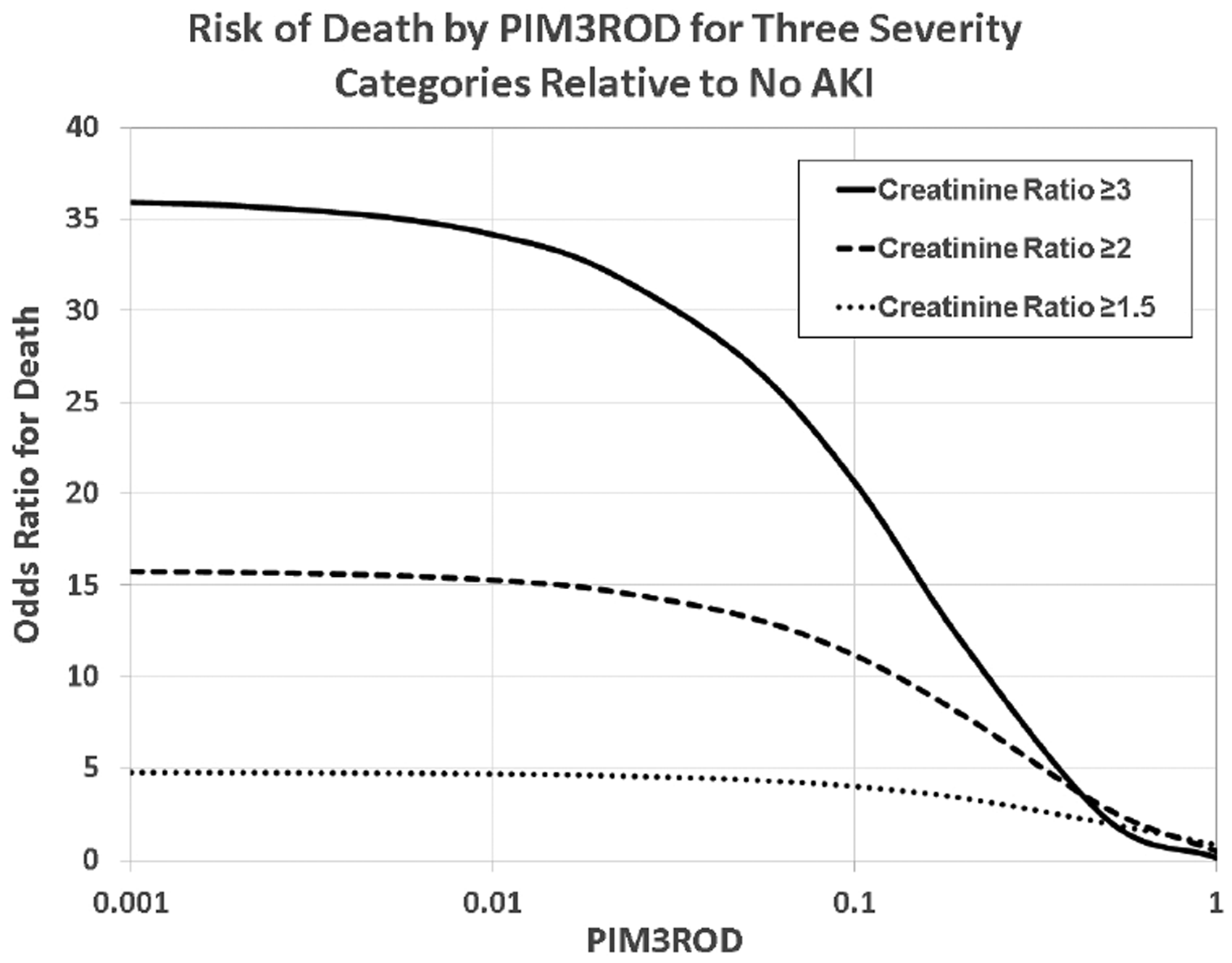
Odds ratio for death for admissions with creatinine rise compared to no creatinine category. PIM3ROD: Paediatric Index of Mortality 3 Risk of Death; AKI: acute kidney injury.
Discussion
Our study evaluates a comprehensive, prospectively collected dataset in a large mixed PICU in two cohorts ten years apart. There have been minor changes in demographics and case mix, but cohorts are comparable. Our data demonstrate that AKI, defined by creatinine criteria, remains common in the PICU, with an average incidence of 25%. This incidence is similar to previous literature, including the AWARE study, the largest AKI assessment in PICU to date. 2 , 12 , 19 Our data show a higher frequency of mild and moderate AKI in admissions admitted post CPB, which is also comparable to the literature. 20 , 21
Until recently, the utility of oliguria criteria for AKI diagnosis in children has received limited attention. The recent AWARE study found that oliguria alone identified a further 18% of severe AKI admissions. 2 , 11 In our study, 18% of AKI was also diagnosed by oliguria of more than eight hours alone, with a high proportion of these admissions having respiratory or neurological diagnoses. Where both oliguria and creatinine criteria are met, oliguria did not provide a temporal benefit in diagnosis, which is in contrast to the findings in an adult population. 15 Oliguria was rarely a sentinel indicator for development of AKI defined by creatinine criteria, providing no apparent clinical benefit over creatinine criteria for AKI diagnosis. Furthermore, patients with oliguria alone had low mortality. We therefore question whether AKI defined by oliguria alone carries equal clinical significance.
Our data support the finding that AKI by creatinine criteria increases mortality relative to predicted risk of death on admission.2–4, 14 , 22 Additionally, our data indicate that the impact of AKI defined by creatinine on mortality is greatest for those with lower PIM risk of death on admission (Figure 4). Increasing risk-adjusted mortality is associated with increasing severity of AKI by creatinine and oliguria criteria, and one compounds the other. Notably, only one patient died who had oliguria without creatinine rise (0.8% incidence).
Our study highlights an important question of how oliguria should be weighted in the clinical diagnosis of AKI. In PICU, urine output is actively managed to maintain water homeostasis via intravascular volume augmentation or diuretic use. This may limit utility of oliguria per se as a diagnostic tool. Recent studies suggest that the response to furosemide may be a useful alternative to risk stratify admissions with AKI. 21 In our experience, periods of oliguria shorter than the threshold to define AKI (i.e. <6 hours or <8 hours) are used as a trigger for fluid management and to provoke awareness of possible AKI. Our data supports this approach as AKI defined by oliguria alone is of uncertain clinical significance.
Our dataset, although slightly smaller than the AWARE study, has some specific advantages. The AWARE study assessed a shorter time frame (seven days only after admission 2 ), and 20% of their cohort did not have urine output data available. Our granular dataset provided urine output data on 90% of admissions, allowing detailed interrogation of oliguria frequency and its relationship to creatinine rise. Our mortality criterion was death in the PICU, not 30-day mortality. To our knowledge, this is the most comprehensive comparison of oliguria and creatinine criteria for the diagnosis of AKI performed to date.
The challenges and limitations of our study include, first, in diagnosing AKI, we included admissions with elevated initial creatinine and a subsequent drop back to baseline. Thus, urine output measures were unavailable before AKI onset in these admissions. Second, we included admissions with a creatinine rise of 1.5 times baseline but with absolute values within normal limits as having AKI. Although consistent with current criteria, the significance of such creatinine changes is unclear.
Concluding key messages
AKI by creatinine criteria and/or oliguria are common in the PICU. Increasing severity of creatinine rise and of oliguria confer an increasing risk-adjusted mortality. Mortality is low in patients with oliguria but no creatinine rise.
Supplemental Material
sj-pdf-1-aic-10.1177_0310057X20979718 - Supplemental material for Diagnosis and incidence of acute kidney injury in a mixed paediatric intensive care unit: Retrospective analysis, 2005 and 2015
Supplemental material, sj-pdf-1-aic-10.1177_0310057X20979718 for Diagnosis and incidence of acute kidney injury in a mixed paediatric intensive care unit: Retrospective analysis, 2005 and 2015 by Shreerupa K Basu in Anaesthesia and Intensive Care
Footnotes
Acknowledgements
We thank Elizabeth Barnes and Bruce Tabor for statistical assistance, Rae Fernandez for data downloading and the PICU clinical staff for original data collection.
Declaration of conflicting interests
The author(s) declare no conflict of interest relevant to the preparation of this manuscript.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
