Abstract
Early excision of deep burn eschar and the expeditious closure of the resultant wounds have become established as gold standard burn care. However, early burn excision has been accepted as up to four days post injury based on a series of misconceptions, not least that the patient is too unwell to undergo surgery and tolerate anaesthesia too soon after injury. There are several reasons why immediate burn excision yields superior survival outcomes, and these are expounded in this article. The systemic pathophysiology following major burn injury, especially when complicated by the respiratory pathophysiology accompanying smoke inhalation, evolves. The hours immediately after burn injury offer several windows of surgical opportunity, windows closed by the pathophysiological events that peak 24 hours later and make surgery and anaesthesia at that time both dangerous and ill-advised.
The two edicts held sacrosanct in the burn surgeon’s mantra are (a) excise the burn early and (b) definitively close the wound as quickly as possible. The basis of the first edict has been demonstrated clinically by (amongst others) the superior survival outcomes reported following early tangential excision by Jackson in the UK in the 1950s 1 and the work of Janzekovic in the 1970s. 2 The observed clinical benefits were somewhat explained experimentally by the mouse studies of Allgöwer over several decades in his published descriptions of the toxic effects of retained eschar (probably best summarised in his 1995 paper 3 ).
The second edict also has a pathophysiological basis derived from studies of wound healing, and in particular the tendency to healing by secondary intention of large deep wounds. 4 This process is protracted and characterised by granulation tissue formation and wound contraction, complicated frequently by recurrent episodes of infection and resulting in unacceptable hypertrophic scarring which is disabling, dysaesthetic and symptomatic. 5
The first edict raises challenges, demonstrated by several groups in the 1940s and 1950s who failed to achieve an improvement in mortality with early fascial burn excision (removal of the burn by excising all tissue superficial to the muscular fascia, including potentially viable fat). The problems were predominantly death from infection, facilitated by the lack of means to close the wounds. 6 When tangential excision (removal of the burn by serial ‘shaving’ of burn eschar parallel to the skin surface until only viable tissue remains in the wound bed) restored interest in early excision, there developed the anecdotal (and mistaken) belief that excision cannot be performed during the resuscitation phase, and that burn excision has to be performed piecemeal to reduce blood loss and anaesthetic time, as well as maintaining core temperature. 7 The avoidance of excision during the fluid resuscitation phase has evolved into practice where early burn eschar excision is considered to be within the first four days of burn injury. In truth, the immediate post-injury phase is characterised by a number of ‘windows of opportunity’, and immediate eschar excision (within hours of the injury) actually confers a number of distinct advantages.
The ‘anaesthetic’ window
The significant pathology that characterises the response to smoke inhalation (lower airway) injury is chemically mediated, when the products of combustion are transported into the lower airway on particles of soot (Figure 1). These then dissolve in the secretions of the respiratory mucosa, establishing a chemical pneumonitis, bronchitis and alveolitis, the response to which is a complex cascade of inflammatory processes, and protein exudation and fibrin cast production that results in airway occlusion and severe ventilation–perfusion mismatch. Secondary bacterial infection and pneumonia are common. Very recently, we have administered a nebulised solution of 10,000 IU of heparin with 3 ml of 20% N-acetylcysteine (HEPNAC), via the endotracheal tube, every four hours to patients with smoke inhalation intubated on the intensive care unit (ICU) with great success—a valuable technique but lacking literature support. 8 Although water-soluble compounds in smoke readily dissolve in respiratory secretions and can cause early damage in the proximal airways, poorly water-soluble components (such as phosgene) can take up to 48 hours to cause severe distal airway damage by later interaction with respiratory cell phospholipid membranes. 9 In the first 24 hours, there is a ‘window’ where anaesthesia can be delivered much more safely and more easily than it can once the chemical pneumonitis is established.
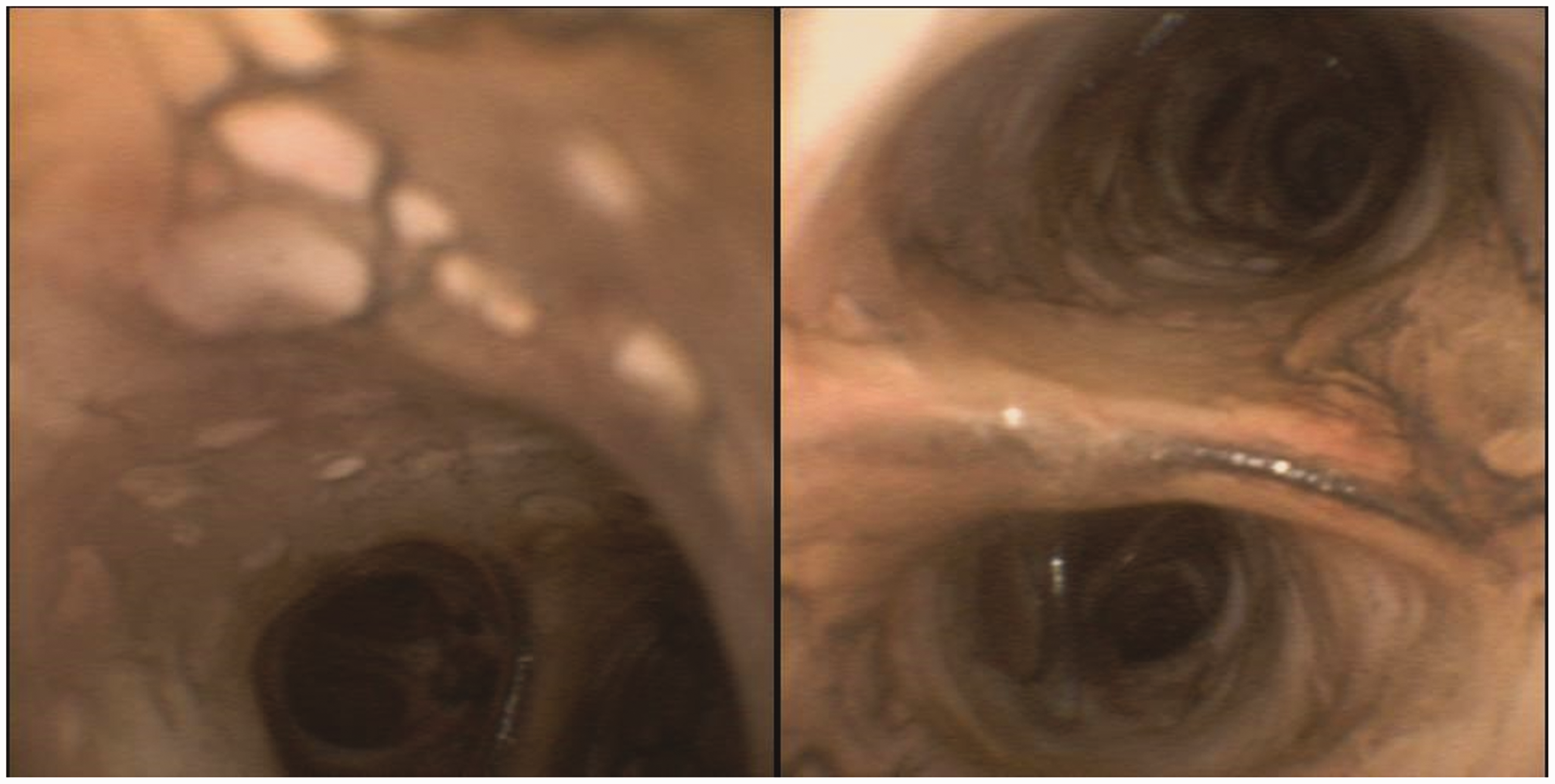
Smoke inhalation imaged at bronchoscopy on a patient with 93% full-thickness burn injury. He was the first at our institution to receive nebulised heparin/N-acetylcysteine for smoke inhalation amelioration.
The ‘haemodynamic’ window
Additionally, there is less blood loss in the very early stages of injury, partly since there are several vasoactive metabolites, such as the arachidonic acid metabolite thromboxane, as well as vasopressin, adrenaline and noradrenaline circulating in high concentrations during the first 24 hours after injury so that the post-burn hyperaemic phase takes a couple of days to establish fully.10–12 Any blood loss during immediate surgery can be further reduced by sub-eschar tumescence with adrenaline-containing solutions13,14 (Figures 2–5; consent has been obtained from the patient to publish his/her clinical pictures). Once in the hyperaemic phase, from 24 hours onwards, there appears in my experience to be markedly less response to tumescence in the burn areas, although it remains effective in reducing bleeding from freshly created skin graft donor sites.
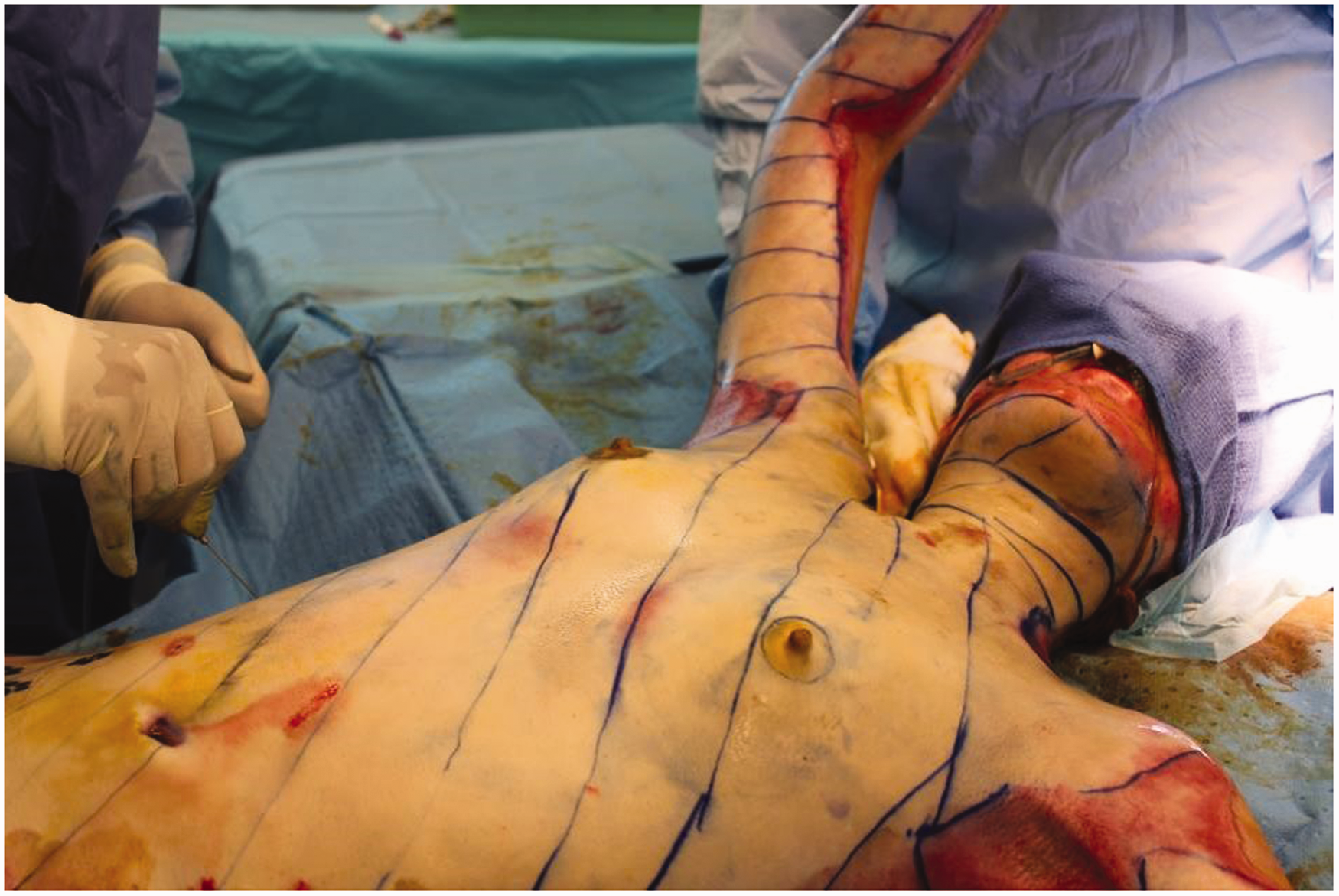
A full-thickness burn marked and undergoing tumescence with an adrenaline-containing solution prior to tangential burn excision.
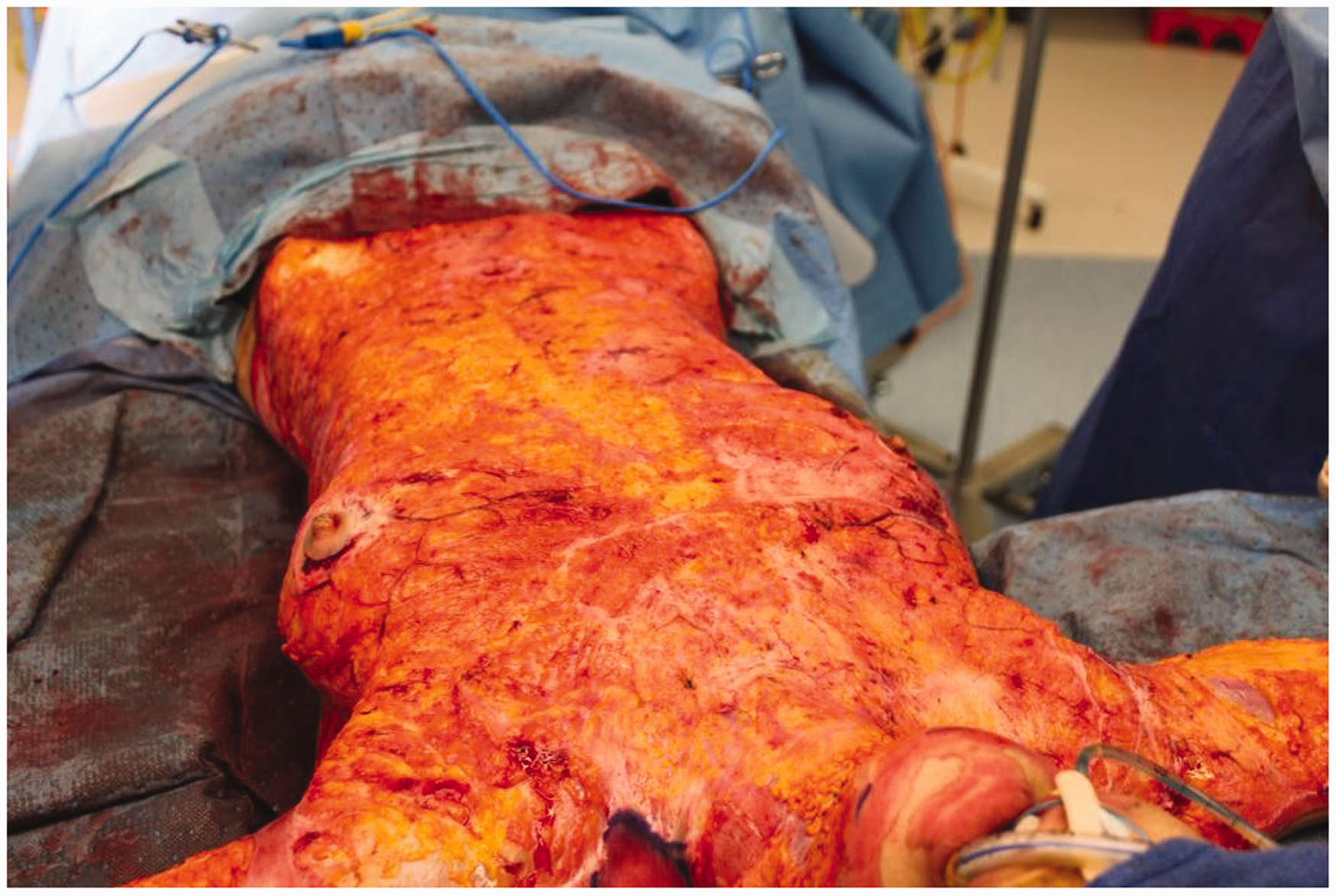
The patient from Figure 2, immediately after tangential excision of her entire burn. Note the lack of blood both on the wounds and the surrounding drapes.
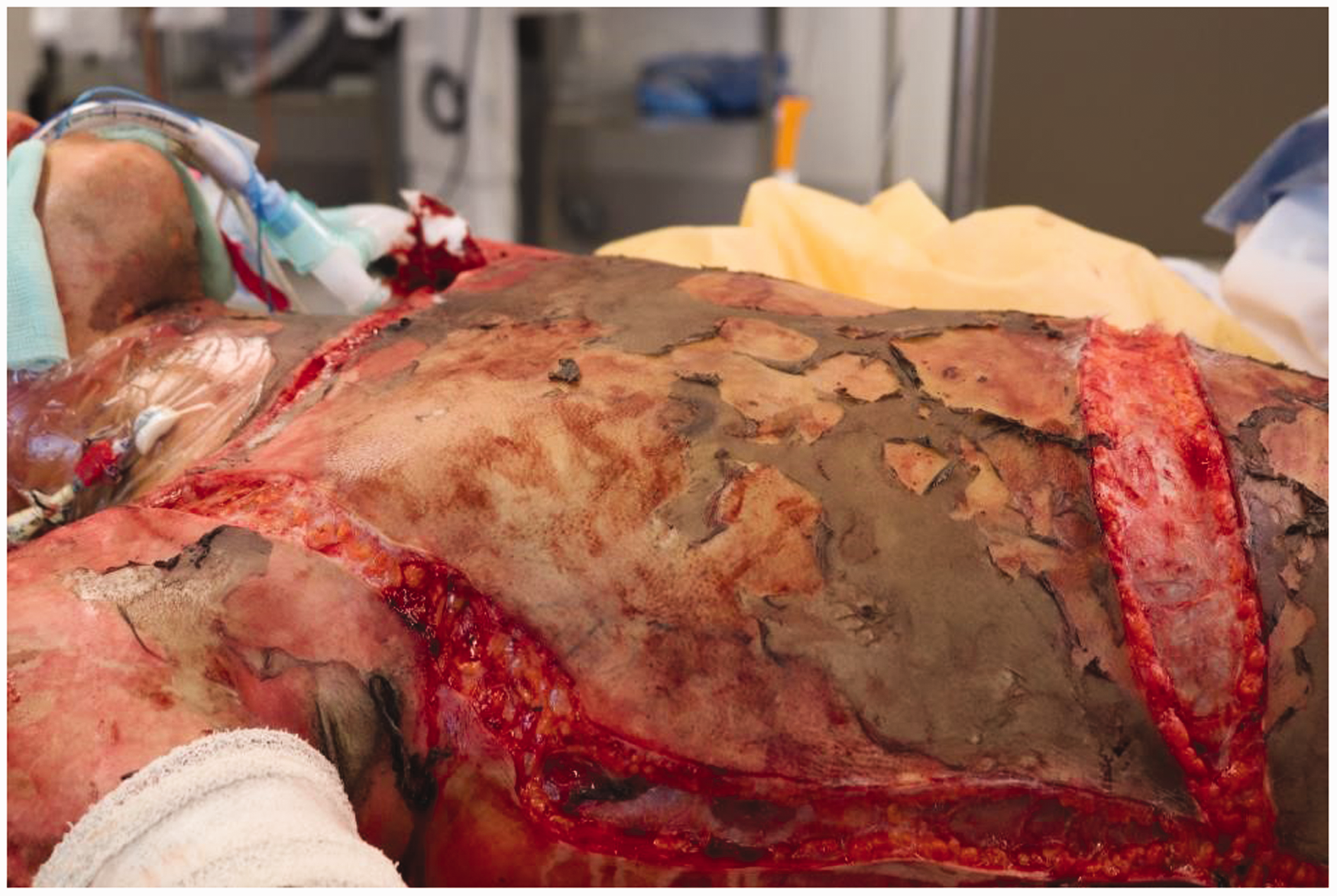
The 93% full-thickness burn patient from earlier figures. Escharotomies visible prior to tumescence and complete fascial burn excision.

After fascial excision of the anterior trunk (note the muscular delineation). In the ‘old days’, following burn excision, there was often a solid ‘meniscus’ of clotted blood extending from the flank to the operating table, but there is very little blood loss following appropriate burn tumescence with adrenaline-containing solutions.
The ‘cellular/serum health’ window
For those practitioners who harvest autologous tissue for in vitro culture, the sooner that tissue is harvested after injury, the more likely culture will be successful, since both cells and serum become more unhealthy and unsuitable for culture with time.
Additional benefits
A slightly more theoretical reason for immediate excision revolves around a potential reduction in the volume of fluid resuscitation required by these significantly burn-injured patients. The volumes of fluid required in the first 24 hours post injury to resuscitate burn patients, maintain core volume and peripheral perfusion are enormous and, by some calculations, are 100-fold greater than can be explained by alterations to the Starling Forces alone. 15 Part of the explanation for this lies in interstitial compliance, which doesn’t appear as a factor in Starling’s original equation. The molecular structure of the dermis (predominantly collagen types I and III and several glycosaminoglycans, including hyaluronic acid) exert hydraulic forces which stabilise the interstitial space whilst in their normal ‘coiled’ configurations. The destruction of the dermal macromolecules after deep burn injury alters the osmotic force in the dermal interstitium (because fragmentation increases particle number and ‘uncoiling’ allows expansion and reduces density), creating a highly negative interstitial tissue hydrostatic pressure (in the order of 19 mmHg for each milliosmole of solution). This could have the effect of creating an interstitial hydrostatic pressure of –40 mmHg according to some authors,16,17 but as low as –135 mmHg by other experimental models. 18 Since positive interstitial hydrostatic pressure is one of the effectors of fluid movement into the vasculature, a markedly negative value creates a ‘sucking’ force, moving potentially large volumes of fluid from the vasculature and into the interstitial space. Removal of the eschar immediately, or at least as soon as the patient arrives and is trauma cleared, should negate the development of this force and reduce the volume of resuscitation fluid required, potentially many fold.
Leaving dead tissue (eschar) in situ for any length of time has no basis in physiology and no role in good surgical practice. Eschar is porous, allowing fluid loss (signalling the presence of a wound and propagating inflammation) and bacterial ingress, colonisation and infection with a range of Gram-positive and endotoxin-producing Gram-negative organisms.
Finally, and perhaps most compellingly, the experimental work of Martin Allgöwer demonstrated the deleterious effect of retained eschar in terms of its effect on the immune system (disruption of both cellular and humoral components) and on general organ function (progressive renal dysfunction, failure and a multi-organ failure cascade), enhanced in the presence of Gram-negative endotoxin. 3
For these reasons, in my practice, total burn eschar excision occurs as soon as trauma clearance is complete, facilitated by adrenaline tumescence. Frequently, extensive escharotomies are performed first (either in the emergency department, or immediately upon arrival in the operating theatre), with cutting monopolar diathermy via a Colorado needle (a fine needle point on a combined cutting/coagulating handpiece) to ‘buy time’ for the subsequent eschar excision. Since major eschar excisions are often lengthy, the operating theatre is warmed to 32–34°C, the patient lies on an induction heating device and burn excision progresses serially, with each area covered with a heated air blanket (BairHugger, 3M Australia, North Ryde, New South Wales) initially, exposed only for its eschar to be removed and then re-covered. At the end of the procedure, the resultant open wounds are dressed and the patient returned to the ward, or ICU, to regain what temperature has been lost and to recover physiologically from the insult of their injury and the eschar removal. Within 48 hours, these patients are physiologically well by all measurable parameters.
The immediate and total excision of eschar leaves extensive fresh wounds and brings us on to problems inherent in the second edict: that those wounds should be definitively closed as quickly as possible. These issues, and modern solutions, are addressed in the second part of this two-paper series.
Footnotes
Declaration of conflicting interests
The author(s )declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Greenwood developed Biodegradable Temporising Matrix (BTM) between 2004 and 2016 in collaboration with PolyNovo Biomaterials Pty Ltd. This manuscript represents the background concepts that led to BTM development. He has no current affiliation with PolyNovo Biomaterials Pty Ltd, and does not receive any fee for consultation, presentation, travel or promotion. However, he remains a shareholder.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
