Abstract
After major burn injury, once survival is achieved by the immediate excision of all deep burn eschar, we are faced with a patient who is often physiologically well but with very extensive wounds. While very early grafting yields excellent results after the excision of small burns, it is not possible to achieve the same results once the wound size exceeds the available donor site. In patients where 50%–100% of the total body surface area is wound, we rely on serial skin graft harvest, from finite donor site resources, and the massive expansion of those harvested grafts to effect healing. The result is frequently disabling and dysaesthetic. Temporisation of the wounds both passively, with cadaver allograft, and actively, with dermal scaffolds, has been successfully employed to ameliorate some of the problems caused by our inability to definitively close wounds early. Recent advances in technology have demonstrated that superior functional and cosmetic outcomes can be achieved in actively temporised areas even when compared with definitive early closure with skin graft. This has several beneficial implications for both patient and surgeon, affecting the timing of definitive wound closure and creating a paradigm shift in the care of the burned patient.
Until very recently, the early definitive closure of wounds proved problematic when full thickness burns exceeded 50% of the total body surface area (TBSA). The mainstay of burn wound repair has been the split skin autograft and, at >50% TBSA, the burn area exceeds the donor site area. A number of manoeuvres have been established to facilitate coverage of these wounds by grafting, all of which are utilised in patients with the most extensive burn wounds—serial episodes of grafting surgery, harvesting very thin autografts (to allow more rapid re-epithelialisation of the donor sites, facilitating earlier re-harvest and allowing a greater number of harvests from the same donor site) and widely meshing the grafts, or using a Meek-Wall technique (Meek, Humeca, Enschede, Netherlands). This latter technique involves using small pieces of graft, placed in a specialised holder on a cork board and run through a series of blades perpendicular to each other, to create small squares of graft each 3 mm × 3 mm. Once cut, the holding platform can be pulled apart (to differing distances—the ‘mesh ratio’), separating the tiny grafts. Although ‘fiddly’ and laborious, this technique minimises graft wastage, since even small pieces of graft can be meshed in this way. Despite their utility and necessity, these techniques guarantee that the patient with the biggest burn survives with the poorest functional, aesthetic and symptomatic outcome possible. 1
The use of cadaver skin to cover the non-grafted wounds pending donor site re-epithelialisation and ‘re-harvestability’ gained popularity in the late 20th century as issues of consent and techniques for harvest and storage (banking) were refined.2–4 The use of cadaver skin has a number of limitations. Skin banks are frequently short, or devoid, of stock. Its presence ‘passively’ temporises the wound, ‘buying time’ but not improving the wound bed, merely allowing undirected granulation. It cannot be used unless the patient is pathologically immune-suppressed, seemingly the only benefit to leaving the eschar in situ until the cellular and humoral components become inhibited by eschar-derived (Allgöwer) factors.
The dermal matrix strategy, pioneered by Jack Burke, sought to redress some of these issues. 5 In producing a ‘scaffold’ to allow autologous tissue in-growth and establish a ‘neo-dermis’ (‘active’ temporisation), he improved the outcome of the thin, meshed skin graft. 6 However, the material he (and Ioannas Yannas) produced (Integra Dermal Regeneration Template, Integra Lifesciences, Plainsboro, NJ, USA), which is a cross-linked bovine Type I collagen scaffold supported by shark fin chondroitin-6-sulphate glycosaminoglycan, physiologically closed with a bonded pseudo-epidermis of silicone, has a number of disadvantages. The first is the length of the processing time to manufacture the material, which thus renders it very expensive. 7 The second is that placing non-vascularised biological material on the surface of a wound in an immune-compromised patient, and expecting neovascularisation to occur before infection does, is ambitious and frequently unsuccessful.8,9 Its use has therefore been limited; some surgeons have been unable to make it work consistently and thus, simply don’t use it. Due to cost, those surgeons that can make it work tend to limit its use to the biggest burns and then only use it to as a ‘patch-up’ to cover the wounds that remain following the primary grafting procedure. In this situation, the material is being used to temporise, because it physiologically closes the wound, reducing inflammation/proliferation and contraction. Since its integration improves the wound bed for subsequent closure, we can consider it to be an ‘active’ temporiser.
The principal misconception demonstrated by the production of Integra (and its subsequent analogues; Pelnac [Eurosurgical, Guildford, Surrey, UK], Terudermis [Olympus Terumo Biomaterials, Tokyo, Japan], etc.), is that, because dermis is being ‘replaced’ and the dermis structurally consists largely of Type I collagen and supporting glycosaminoglycans, the replacement
During the development of this material, several unexpected outcomes were observed. The synthetic composition means that it is not prone to attack by microorganisms (it is not a ‘foodstuff’). Thus, infection is uncommon and, if it does occur, is localised and manageable without loss of the matrix.16,22,23
Increasing burns use also demonstrated that loss of skin graft over the integrated BTM is extremely rare and that the appearance of autograft mesh pattern (even with widely meshed graft) is considerably better than autograft alone. 22 This finding has also been observed with collagen-based matrices and has its basis in the principles of wound healing.
Deep wounds (deep dermal and full-thickness burns, for example) are characterised by two features—the complete absence of any residual epidermal potential within the wound and a poor dermal structure, which preclude epithelialisation. It is probably the reason that edge epithelialisation only occurs approximately 5 mm from the edge of a deep wound, and why epidermal (keratinocyte) sheets and suspensions (whether cultured or non-cultured) have no value in the primary treatment of deep dermal and full-thickness burns. In untreated deep burns, once the lengthy autolysis and separation of eschar have occurred, the wound therefore heals by secondary intention, with granulation tissue formation, myofibroblast differentiation and contraction, processes designed to approximate the wound edges so that final epithelialisation can occur. This results in prolonged healing time, contracture and hypertrophic scarring with functional disability, unacceptable dysaesthesia and symptoms of pain and itch. 18 Repeated scar breakdown and infection are common. In this group of burn patients, our priority as surgeons is to abort the inevitable secondary intention healing and replace it with a primary intention solution—the sheet autograft. Once in situ, the graft heals by primary intention around its edges and its deep margins. When we mesh autograft, the struts of the graft heal similarly, but the larger the mesh ratio, the larger the interstices (the diamond shaped gaps between the graft struts). The bed of the interstices is the same as the original wound bed and thus incapable of sustaining epithelialisation from the struts without structural alteration, and granulation tissue ‘blebs’ thus develop to facilitate epithelialisation. The wider the mesh, the more extensive the ‘blebs’, the worse (or more ‘cobblestoned’) the cosmetic appearance, and the greater the interstitial contraction. The presence of a ‘neo-dermis’ provides a bed across which interstitial epithelialisation can occur without needing granulation tissue and so the cosmetic appearance is improved. In fact, the thinner the graft, the better the appearance and the less obvious the mesh pattern.
The development of the NovoSorb™ BTM began at the end of 2004, but it was not until 2014 that it was first used in (significant) burn injury. 21 Human experience before this time was limited to small, complex surgical wounds (free flap donor site reconstruction).15,16 The burn situation is very different. The patients have suffered major trauma with complex pathophysiological insults, often complicated by smoke inhalation injury or other trauma. As such, the decision was made that the first burn exposures would be restricted—BTM would not be applied to highly important functional areas (the hands or the neck), areas of great cosmetic importance (the face), or zones where previous dermal matrix use has proven impossible or highly problematic without issues caused by untenable patient positioning (the posterior trunk/buttocks). Our early burn experience has been documented, as have our experiences in the reconstruction of significant and complex wounds (e.g. after necrotising fasciitis debridement).19,20,22,23 In the initial, five-patient significant burn trial, our concerns regarding the use of BTM on the dorsum of the hands proved unfounded, and excellent hand function was demonstrated. 21 The fourth and fifth trial patients also reassured us that the BTM was an excellent material for the posterior surface of the trunk and buttocks without needing to position the patient prone to avoid shearing and pressure effects on integration. 21 In fact, BTM seems to integrate more quickly in these areas, possibly due to compression against the bed. The results of fascial neck reconstruction with BTM in burns and necrotising fasciitis patients have made this our primary recourse.20,22
One throwback to our previous use of dermal matrices remained—we were convinced that the matrix should be used to cover areas where there was insufficient autograft for coverage, using the matrix merely to ‘patch’ and actively temporise debrided but ungrafted wounds while donor sites healed for re-harvest to allow their definitive closure. This practice had developed for two reasons; primarily, because of the exorbitant cost of collagen-based dermal matrices 7 and, secondarily, because of the predilection of collagen-based dermal matrices to become infected (30%).8,9 The thinking thus was to definitively close whatever could be closed with available autograft and temporise the rest, despite the obvious downsides elucidated earlier in this text. It was not until the 13th burn patient that this practice was demonstrated to be deleterious to patient outcome, effecting a paradigm shift in our thinking, and prompting a radical change of practice.
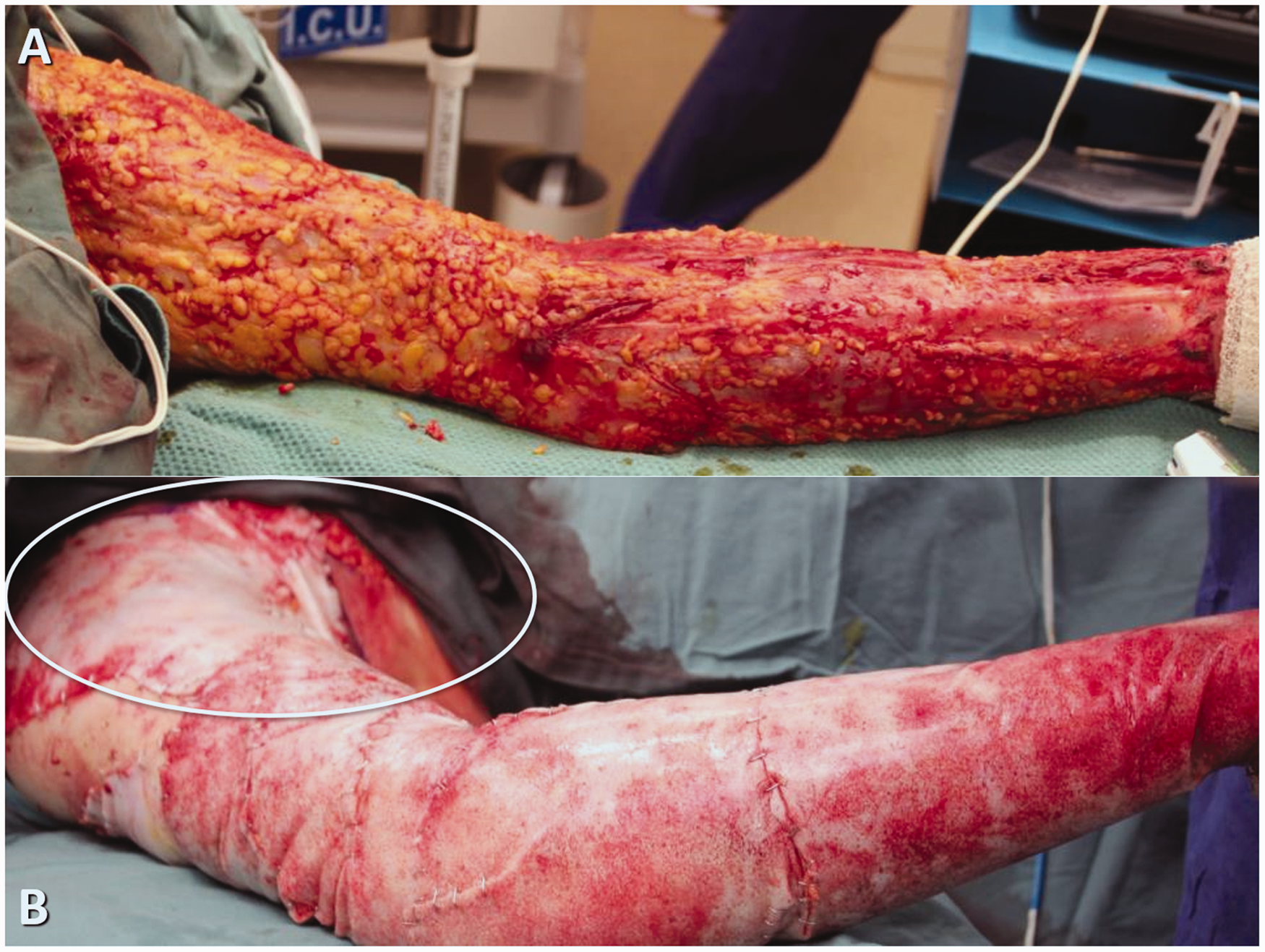
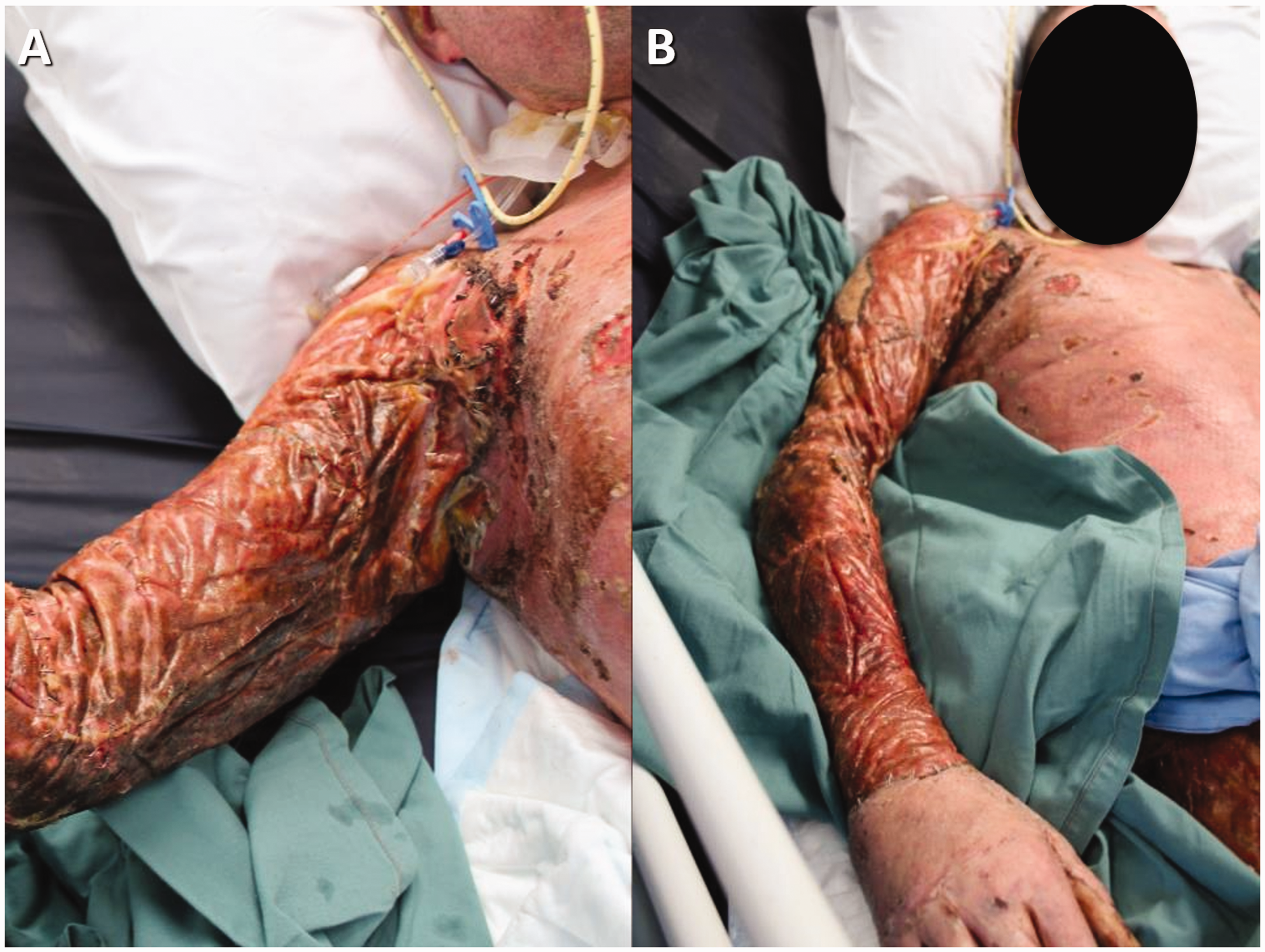
Patient 13 was admitted with 70% TBSA full-thickness burns (Figure 1(a)). The entire burn was debrided on arrival. Three days later, our confidence with BTM had grown to the point that his burns were gently refreshed and the available autograft donor sites (his back and buttocks) were harvested. Sheet autograft was applied to the dorsum of his hands and digits. The remaining graft was meshed 1:2 and applied to debrided burn on chest and abdomen (Figure 1(b)) (consent has been obtained from the patient to publish his clinical pictures). The debrided, ungrafted wounds (50% TBSA) received BTM (Figure 2(a, b)). The grafts took completely (Figure 3(a, b)). On Day 38, the BTMs of his upper limbs, shoulders and lower limbs below the knees had their seals removed, his donor sites were re-harvested and the 1:2 meshed autograft raised was applied (Figure 4(a, b)). A week later, the grafts on BTM were robustly taken (Figure 5). On Day 50, the remaining BTM (circumferential thighs) was delaminated and covered with 1:2 meshed autograft. No autograft was lost at any stage. The patient left the burns unit, completely healed, on Day 91 post-injury.
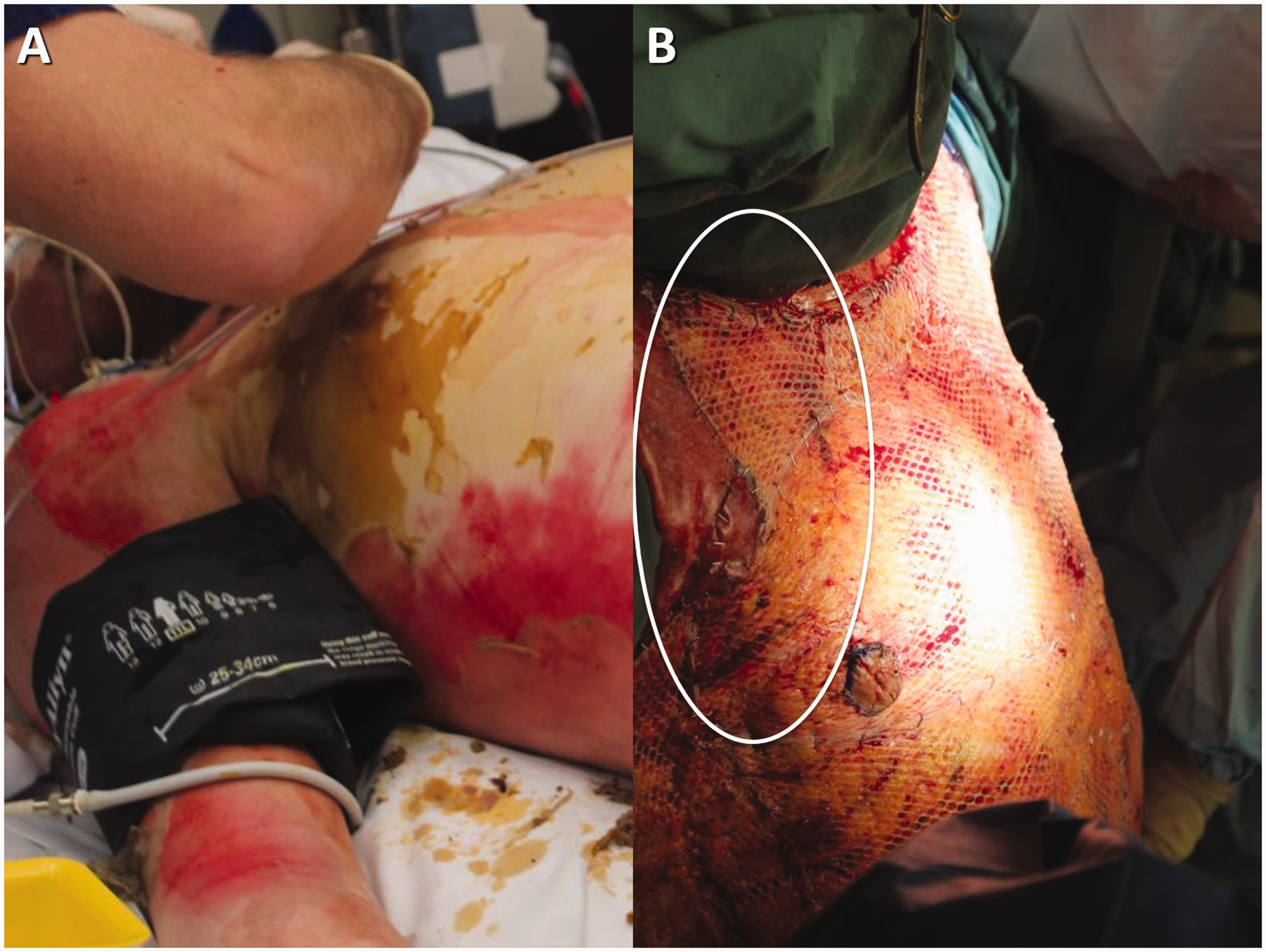
(a) On arrival, this patient’s 70% total body surface area burns included full thickness burns to the right anterior axillary fold. His burns were excised on arrival and three days later 1:2 meshed split skin graft was applied to the entire chest and abdomen. (b) Debrided areas for which there was insufficient graft received Novosorb Biodegradable Temporising Matrix (BTM) (PolyNovo Biomaterials Pty Ltd, Port Melbourne, Victoria). Circled is an area of BTM encroaching from the upper limb/shoulder onto the upper right chest.
(a) The right upper limb following burn wound refreshing on Day 3 and (b) following BTM application. Again, the right chest extension of the BTM is circled.
On Day 28 post-admission, the skin grafts on the chest and abdomen have taken well and the BTM on the upper limb, axilla, shoulder and upper right chest is in the final stages of integration (a and b).
(a) On day 38, the non-biodegradable BTM seal was removed to reveal the polyurethane-based neo-dermis. (b) Split skin graft meshed at 1:2 was applied after the neo-dermal surface was dermabraded.
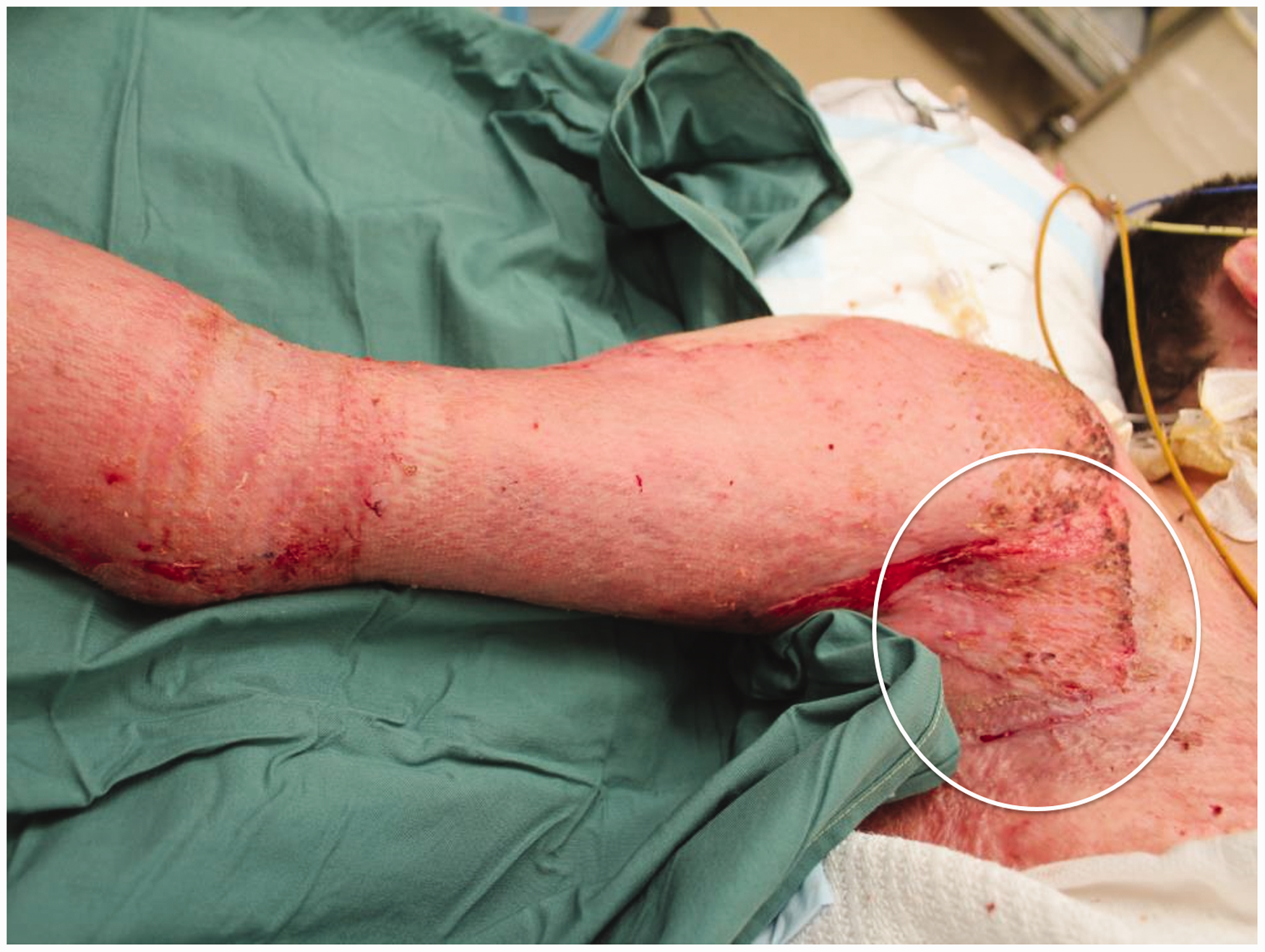
A week later (Day 45), the upper limb skin graft has taken well and is almost indistinguishable from the chest graft applied five weeks earlier. Note the ‘angled’ junction (circled).
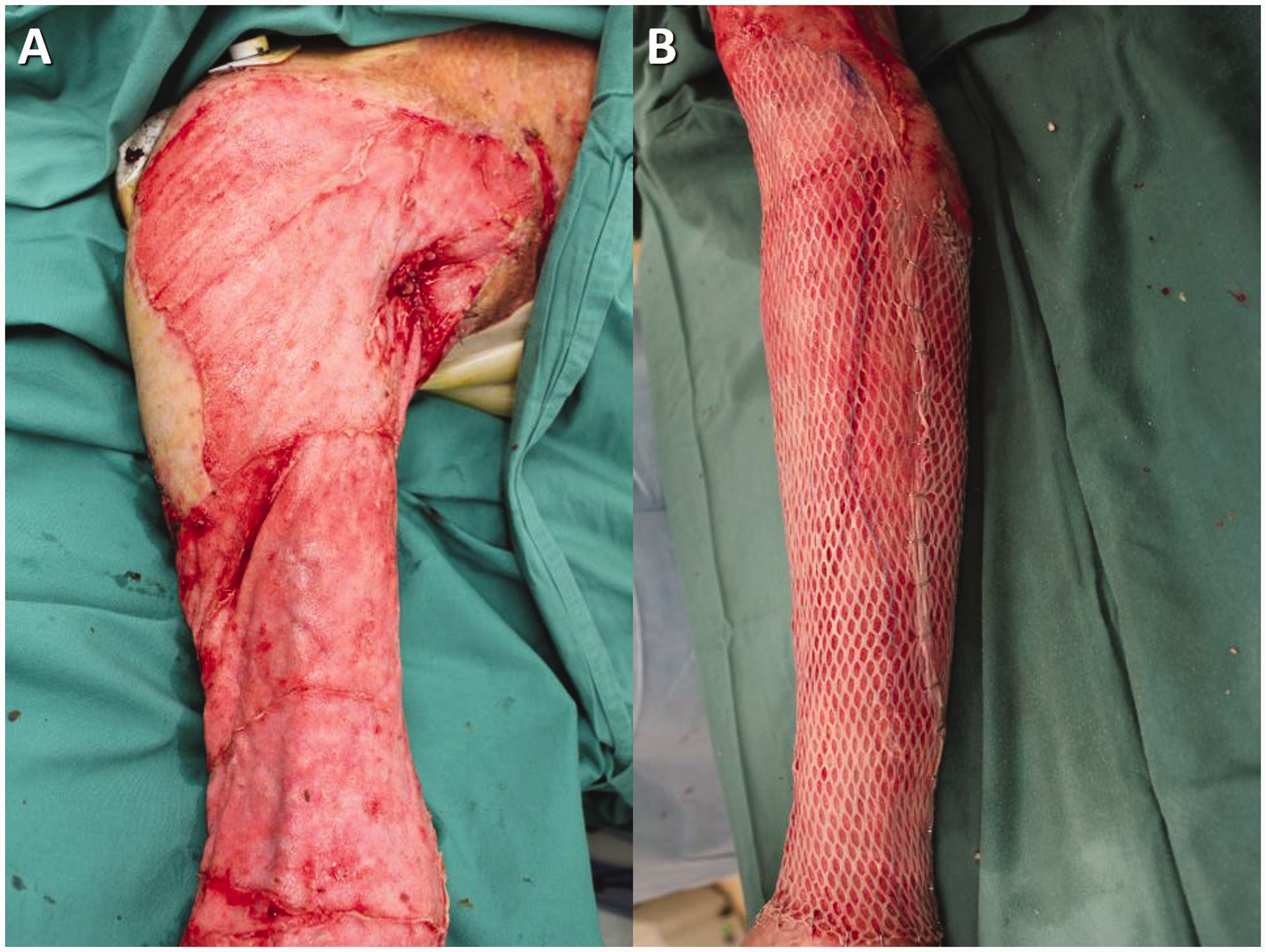
He visited frequently for outpatient review, initially from his inpatient rehabilitation facility, then from home. Over the few months after discharge, the graft over the chest and abdomen (applied at Day 3 post-injury) began to become stiff, tight and mildly hypertrophic. By nine months, this change was stark because the adjacent BTM grafts were soft, supple and elastic (Figures 6 and 7). His full range of axillary motion was marred only by tightness within his trunk grafts (Figure 8(a)). The difference in cosmetic appearance persisted (Figure 8(b)). We realised that, if graft applied over BTM five weeks after graft applied to debrided burn is softer, more elastic and cosmetically superior, and not just in the short term but also after several years, then early grafting is not only unnecessary, it is deleterious.
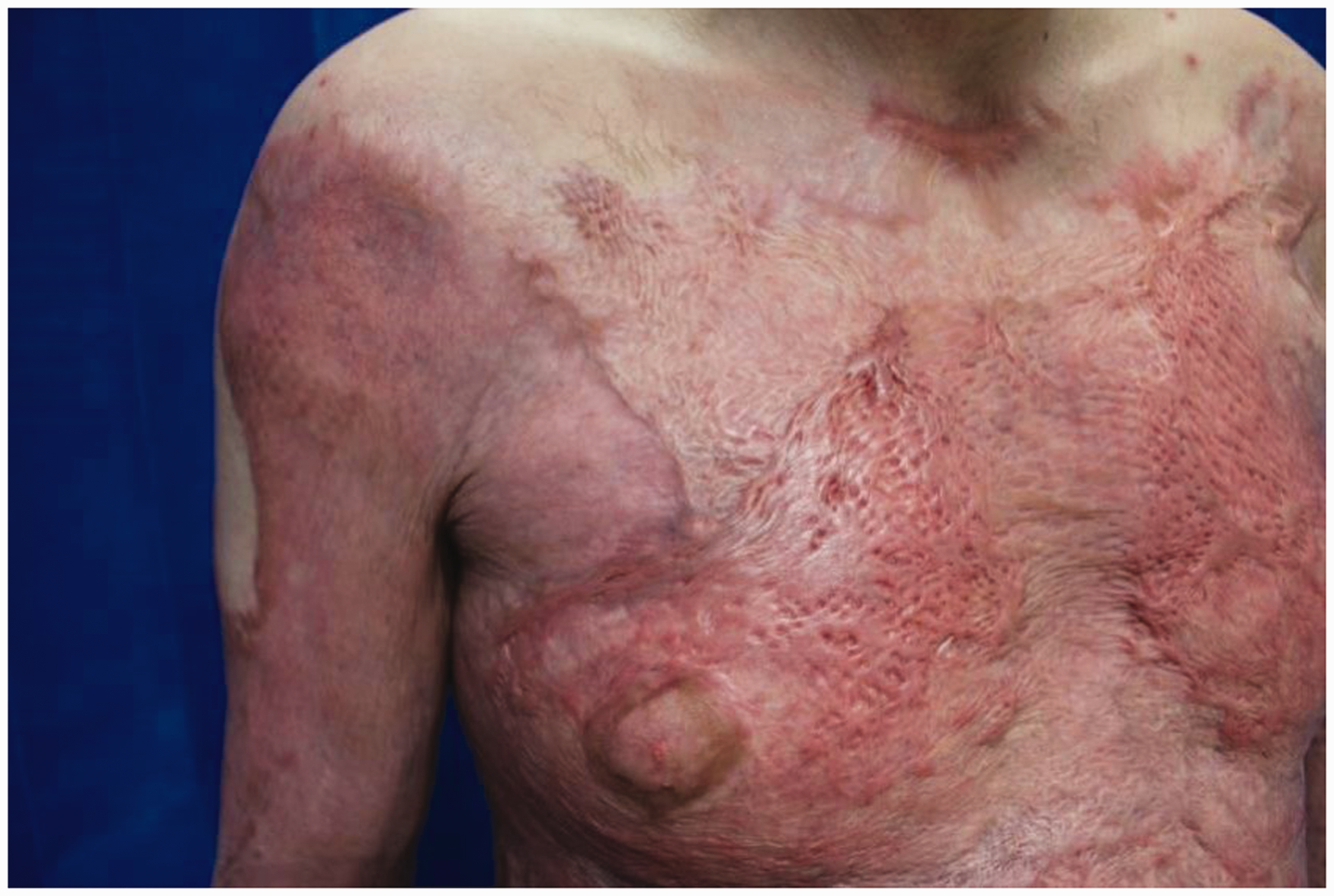
At nearly seven months (Day 200), the split skin grafts on the chest and abdomen have thickened and stiffened, but the graft on BTM is flat, soft and supple. Note the lack of graft mesh pattern and also the crease lines in the axillary fold.
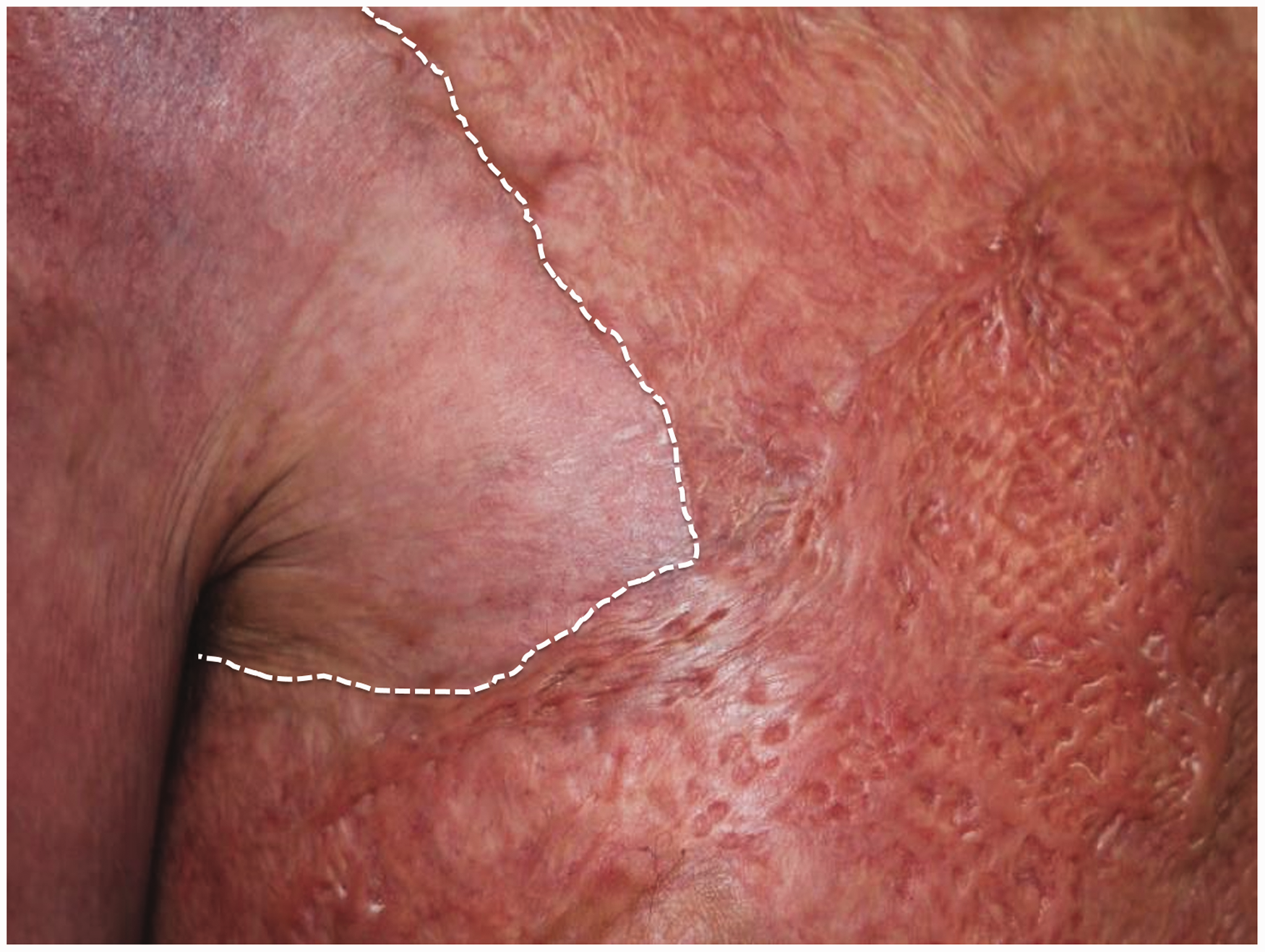
A close-up view of the junction between graft alone (to the right and below the dotted line), and graft over BTM (to the left and above the line) at Day 200. This illustrates the premise of the paradigm shift—why definitively close a burn injury very early (in this case Day 3) when a superior result can be achieved with delayed grafting over BTM five weeks later, at a time when the patient is physiologically well and stronger to cope with the insult of skin graft harvesting and donor site creation?
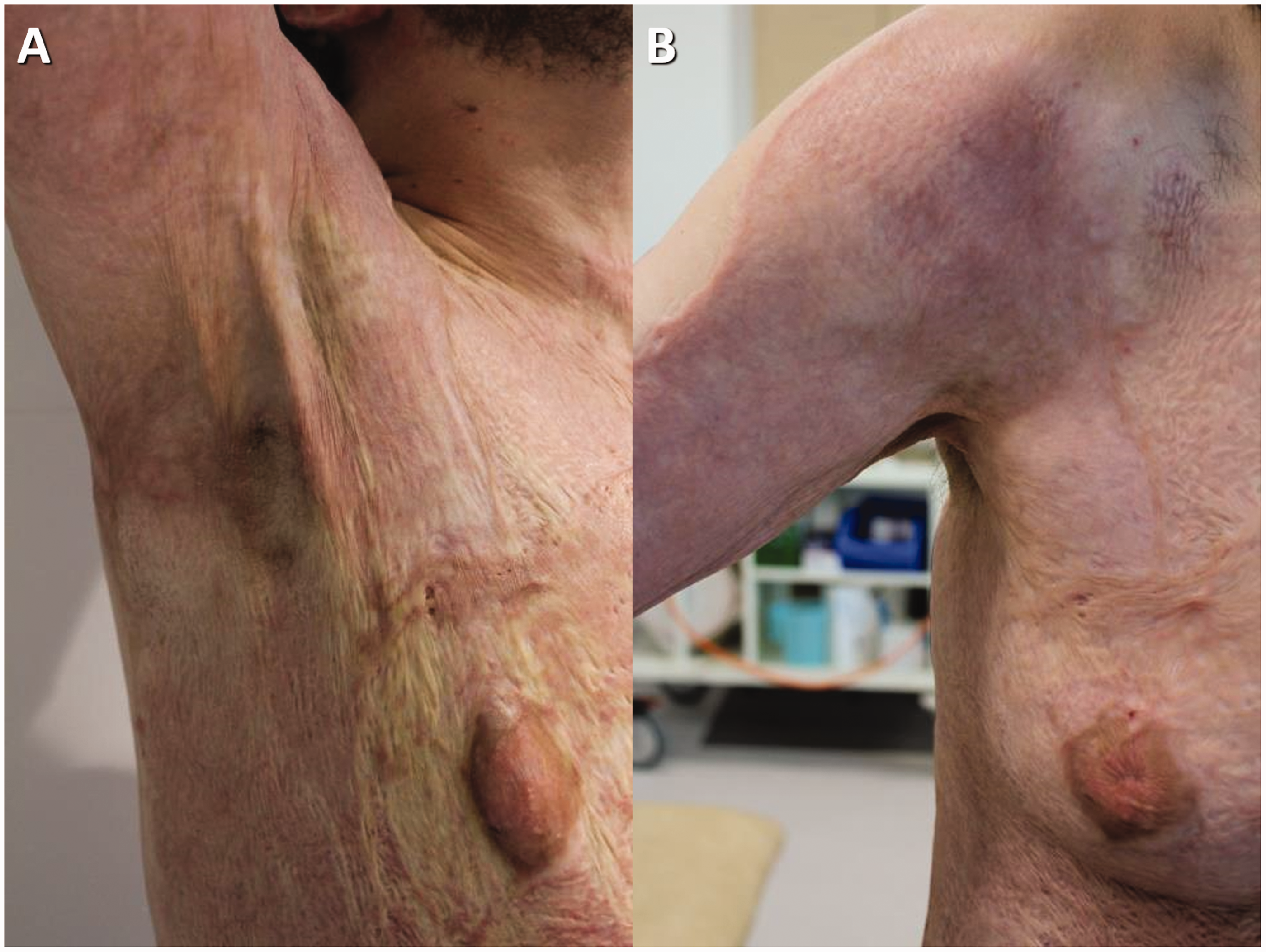
(a) At 12 months, full abduction at the shoulder joint allowed by the BTM grafted shoulder and upper right chest. Note the pallor due to tension in the graft alone scars around the nipple. These extend down the entire grafted flank. The bands in the anterior axillary folds appear as a result of this tension from the graft alone areas. (b) On Day 433, the cosmetic difference of graft alone versus BTM and graft is still obvious.
The shift in our major burn protocol was immediate. From Patient 14 onwards (Supplemental Figure 1) (consent has been obtained from the patient to publish her clinical pictures), large or complex wounds following complete deep burn excision on arrival have been covered with BTM on Day 2 or 3. Considerably superior functional and aesthetic results have been generated (Supplemental Figures 2–7). In addition, there has been an enormous reduction in the need for subsequent reconstructive surgery.
There have been other benefits. Previously, we had harvested autograft as early as possible to commence definitive wound closure. This coincided with the patient being at their sickest and physiologically most vulnerable. Early grafting resulted in longer initial surgeries and prolonged anaesthesia. The patient’s wounds were iatrogenically expanded, adding further early physiological insult. The postoperative pain and patient status created the tendency for them to remain on the intensive care unit longer. In the early phase following burn injury, cutaneous perfusion is threatened by macrocirculatory insufficiency (arising from hypovolaemia, myocardial suppression and arterial vasomotor dysregulation) coupled with local thrombotic microangiopathy and dysregulated microvascular tone. Vasopressor therapy, a common recourse in the management of hypotension in critically ill patients, while potentially protecting vital organs with greater capacity for autoregulation, can severely compromise blood flow to the skin. As a result of the superficial ischaemia, the donor sites fail to heal and become deep wounds and the skin grafts fail to inosculate and wound infection follows. In the major burn scenario, the loss of graft and donor site is devastating for the patient and the clinical condition can become irretrievable.
At the present time, the first procedure to apply split skin autograft in major burn patients at the Royal Adelaide Hospital occurs around five weeks after their admission and burn excision, at a time when they are out of intensive care and physiologically well and strong (airway injury resolved, systemic inflammatory response syndrome settled, extubated, orientated, nutrition appropriate, mobilising out of bed and already undergoing rehabilitative therapy, all areas of more superficial burn healed, etc). At this point, the patient is better physiologically equipped to tolerate large donor site creation. The BTM at this stage is well integrated, but retention of the pseudo-epidermal seal allows staged grafting (the seal is merely left intact on BTM for which there is no autograft at the first grafting operation). The outcome of staged grafting over BTM is the same as one-stage grafting over BTM. Of 29 major burn patients treated with BTM, there have been no deaths in our practice.
Supplemental Material
AIC895788 Supplemental Material - Supplemental material for A paradigm shift in practice—the benefits of early active wound temporisation rather than early skin grafting after burn eschar excision
Supplemental material, AIC895788 Supplemental Material for A paradigm shift in practice—the benefits of early active wound temporisation rather than early skin grafting after burn eschar excision by John E Greenwood in Anaesthesia and Intensive Care
Footnotes
Declaration of conflicting interests
The author(s )declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Greenwood developed Biodegradable Temporising Matrix (BTM) between 2004 and 2016 in collaboration with PolyNovo Biomaterials Pty Ltd. This manuscript represents the necessity for its development and its subsequent utility. He has no current affiliation with PolyNovo Biomaterials Pty Ltd, and does not receive any fee for consultation, presentation, travel or promotion. However, he remains a shareholder.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
