Abstract
The prevalence of transplantation is on the increase worldwide. Corneal transplantation is the most common form of human donor transplantation. Transplantation of other organs and bone marrow is established treatment for various end-organ failure and many haematological conditions, respectively. Success and survival of these patients have increased with advances in immunosuppression. Unfortunately, these patients are susceptible to cataract formation as a consequence of immunosuppressive therapy and accelerated progression of several diseases. Topical anaesthesia and regional ophthalmic blocks are ideal for cataract surgery in cooperative adults. General anaesthesia may be required in children, for extremely anxious or claustrophobic adults and for complex surgery such as simultaneous cataract and corneal transplantation. The perioperative anaesthetic management of cataract surgery in a transplant recipient is no different to a standard technique in a healthy adult, but additional challenges are posed by the underlying pathology necessitating transplantation, function of the transplanted organ, physiological and pharmacological problems of allograft denervation, side-effects of immunosuppression, risk of infection and potential for rejection. This narrative review summarises optimal anaesthetic management in transplant recipients undergoing cataract surgery.
Introduction
Over the past few decades, organ transplantation has become increasingly common worldwide as an established treatment for end-organ failure. The World Health Organization estimated that about 136,000 organ transplantations were performed in 2016; kidney was the most common (66%), followed by liver (22%), heart (6%), lung (4%) and pancreas (2%; Figure 1). 1 Multiple organ transplantation (e.g. kidney-pancreas, heart-lung, liver-intestine, multi-visceral) is performed less frequently.
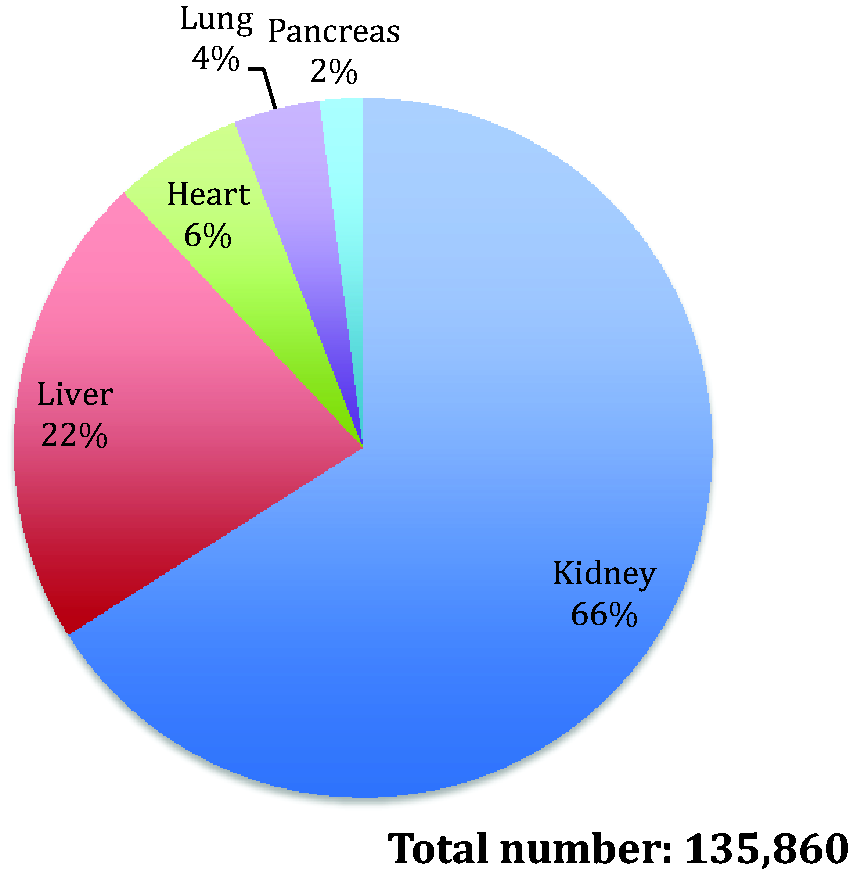
World Health Organization transplantation data 2016.
Corneal transplantation is the most common human donor transplantation. 2 There were >184,000 corneal transplants performed globally in 2012. 3 Bone marrow transplantation has also become an established treatment for a range of haematological conditions (e.g. leukaemia, bone marrow failure and myeloma), especially in children. Unfortunately, transplant recipients are susceptible to cataract formation as a consequence of immunosuppressive therapy and accelerated progression of several diseases.
Increasingly, transplant recipients present for cataract surgery as their survival has been optimised with advances in immunosuppressive therapy (the one-year survival after kidney, pancreas, liver and heart transplant is 89%–97%, while the five-year survival is 76%–90%; Table 1).4–9 Cataract surgery is usually performed as a day case, and the ophthalmic anaesthetist may have limited experience in caring for transplant recipients. There is a dearth of published material and guidance on anaesthetic provision for cataract surgery in these patents. This narrative review summarises optimal anaesthetic management techniques for cataract surgery in transplant recipients.
Outcome of organ transplant (2017 annual report USA).

Search strategy
A literature search using MEDLINE (PubMed) with the aim of identifying English language articles was conducted in May 2019. An initial search using ‘transplant recipient’ and ‘cataract’ and ‘anaesthesia’ yielded no articles. A broader search using the key words ‘transplant recipient’ and ‘anaesthesia’ identified 586 articles. The authors screened the abstracts for relevance, and hand searched their references for further relevant publications. As a result, 61 articles were included in the final review.
Cataract development in transplant recipients
The high concentration and specific arrangement of protein molecules within the human lens fibres prevent scattering of light and provide its transparency. 10 Cataract or opacity of the lens results from separation of these protein molecules by water entry or formation of large molecular aggregates. 11 Contributing factors include ageing, corticosteroid therapy and diabetes. Cataracts can be categorised into three types based on the location of lens opacity: nuclear, cortical and posterior subcapsular. A person may develop one or a combination of these three types of cataract.
Transplant recipients are susceptible to cataract formation, and bilateral posterior subcapsular cataracts (86% bilateral versus 14% unilateral) 12 are most commonly encountered after renal transplantation. 13 The reported incidence varies from 23% to 58%, with a lower incidence in the early years, and plateaus at 15 years post kidney transplantation. Contributing factors include the use of cyclosporine 13 and high cumulative corticosteroid dose (prednisolone dose >3 mg/kg/month). 14 Acute rejection episodes treated with pulsed steroids may result in a higher risk of developing cataracts. 14 After combined kidney-pancreas transplantation, new cataract formation was virtually universal at 6–10 years. 11 The incidence of cataract developing has been reported to be as high as 40% one year after endothelial keratoplasty 15 and 44%–64% within five years after penetrating keratoplasty. 16
Diabetes is prevalent among transplant recipients, either pre-existing or as a consequence of immunosuppressive therapy. The pathogenesis of diabetic cataract development is not fully understood. The current hypothesis on the initiating mechanism is the generation of sorbitol from glucose by the enzyme aldose reductase, which results in increased osmotic stress in the lens fibres, leading to their swelling and rupture. 17
Steroid-induced cataract is usually associated with glucocorticoid activity (administered locally to the eye or systemically). This is possibly related to both direct effects of glucocorticoid on lens epithelial cells and indirect effects mediated through alterations to the lens environment. 18 However, the precise mechanism remains unclear. Steroid therapy can have a more pronounced effect in children, with cataract development occurring at a faster rate and at a lower dose than that of adults. 19 Bradfield et al. found a 7% incidence of cataract developing within 9–50 months after organ or bone marrow transplantation in children. 20 Total body irradiation, an adjuvant therapy before bone marrow transplantation, is also associated with a very high incidence (95%) of cataract. 21
Despite a reduction in the total dose of steroid administered, addition of cyclosporine to the immunosuppressive regimen of azathioprine and methylprednisolone has been associated with an increase in the incidence of steroid-induced cataract after renal transplantation. 22 It has been postulated that cyclosporine might influence the transitional amount of steroid in the lens, but the precise mechanism is unclear.
Anaesthetic considerations
The perioperative anaesthetic management of cataract surgery in a transplant recipient is similar to standard practice in healthy adults, but there are additional considerations depending on the underlying pathology necessitating transplantation, function of the transplanted organ, physiological and pharmacological problems of allograft denervation, side-effects of immunosuppression, risk of infection and potential for rejection.
Preoperative evaluation
There are many important objectives of preoperative evaluation in transplant recipients, especially those who require general anaesthesia and whose health may be compromised as a result of immunosuppressive therapy and/or dysfunction of the transplanted organ. An additional careful history is required questioning potential long-term complications, transplant rejection, infection and lymphoproliferative disease.
Underlying pathology necessitating transplantation
The initial reason for transplantation is important information. Some pathology is localised (e.g. Fuchs’ endothelial dystrophy and keratoconus for corneal transplant and, likewise, cardiomyopathy for heart transplant), some may lead to other system manifestations (e.g. cor pulmonale from pulmonary fibrosis for lung transplant) and others may have multisystem involvement (e.g. diabetic nephropathy for kidney transplant, cystic fibrosis for lung transplant). A summary of common causes for transplantation is included in Table 2.
Transplantation—common causes and post-transplant complications.

Assessment of function of the transplanted organ
Thorough history and clinical examination, together with additional laboratory investigations, are required to assess function of the transplanted organ. Although routine preoperative investigations provide little benefit in otherwise healthy patients for cataract surgery,23–26 customised selective investigations may be required in transplant recipients to assess the function of the transplanted organ.
Heart transplant recipients, for instance, undergo regular follow-up investigations, including cardiac catheterisation and myocardial biopsy, to assess cardiac function and exclude rejection. Communication with the transplant team is required. A recent medical summary, including the most recent status of transplanted organ function and general health, from the general practitioner or attending physician may be available, in which case further preoperative investigations may not be necessary.
Assessment of physiological and pharmacological changes of allograft denervation and complications associated with transplantation
Heart transplant
The resting heart rate of a normal heart is 60–80/minute because the parasympathetic tone is higher than sympathetic activity. The transplanted heart, on the other hand, has a higher resting heart rate of 90–110/minute as a consequence of surgical denervation (no sensory or autonomic innervation).27–29
A normal heart increases its cardiac output, in response to hypovolaemia, by increasing the heart rate and cardiac contractility (via neurohormonal stimulation). A transplanted heart, on the other hand, can only increase its output by increasing the stroke volume (through circulating catecholamines), making it preload dependent. 27
The typical electrocardiograph (ECG) in cardiac transplant recipients has two P waves, one each from the sino-atrial (SA) node of the host and donor heart. The electrical activity from the retained host SA node cannot be conducted through the suture line. Therefore, it has no effect on the transplanted heart. 27
Bradyarrhythmia is common, and sudden cardiac death can occur in up to 35% of cardiac transplant recipients. 30 Twenty percent of heart transplant recipients have a pacemaker for bradyarrhythmia, 31 and 3% have an automated implantable cardioverter-defibrillator (AICD) for prevention of sudden cardiac death. 30 Tricuspid regurgitation (47%–98%), renal dysfunction (51%), diabetes (36%), and allograft vasculopathy (29%; atherosclerotic obstructive disease of coronary vessels) are also common after heart transplant.31–33 These patients may have significant myocardial ischaemia without ischaemic chest pain.
Summaries of physiological changes and potential complications after heart transplant are included in Tables 2 and 3, respectively.
Physiological changes of a transplanted heart.
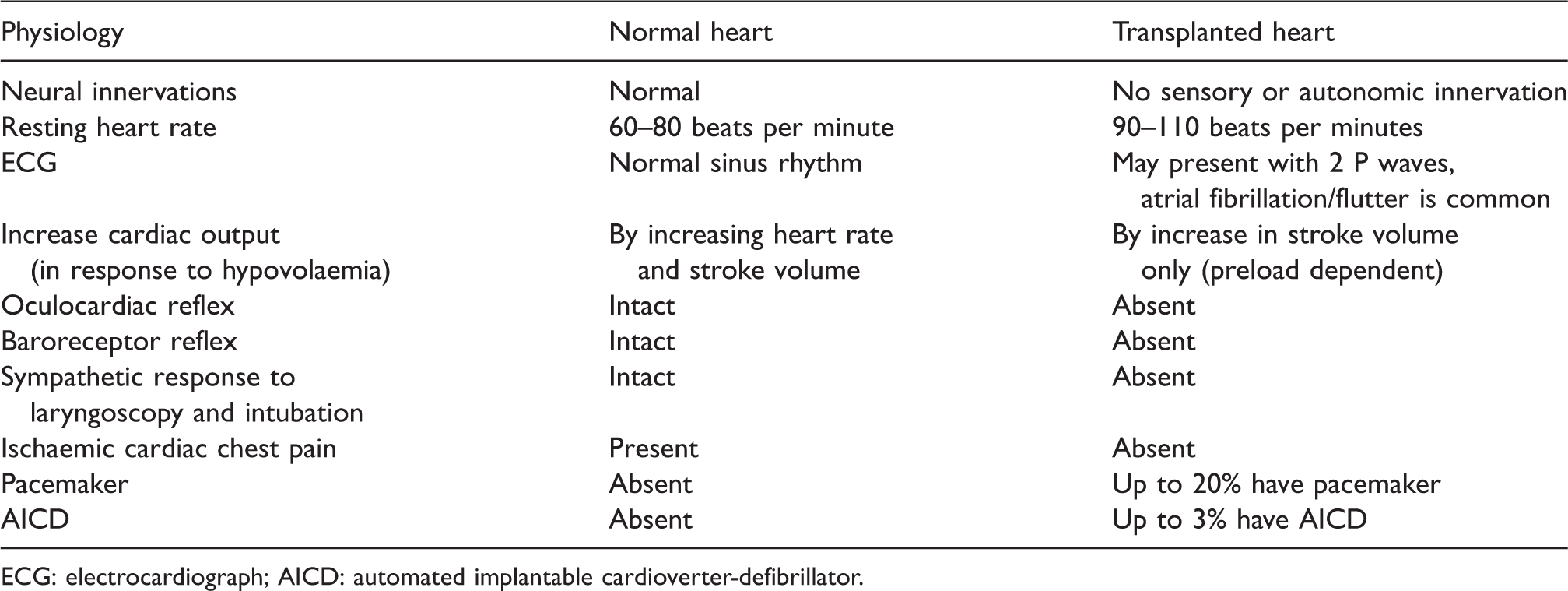
ECG: electrocardiograph; AICD: automated implantable cardioverter-defibrillator.
Anticholinergic agents do not increase the heart rate because of denervation. Direct-acting agents (adrenaline, isoprenaline) are required in the event of severe bradycardia.
Neostigmine does not produce its usual muscarinic effect of bradycardia. 31 However, although rare, cases of asystole associated with neostigmine have been reported (possibly secondary to partial re-innervation of the SA node 34 ) during concurrent administration of an anticholinergic agent. 35 The combination of rocuronium and sugammadex has been proposed to minimise cardiovascular disturbances. 36
Lung transplant
Lung denervation during transplant surgery results in diminished cough reflex and decreased mucociliary clearance, 37 therefore increasing risk of retention of secretions, silent aspiration and chest infection. Lung transplantation has minimal effect on airway tone, hypoxic pulmonary vasoconstriction 38 and ventilatory response to hypercapnia. 39 Extreme caution should be exercised on intraoperative volume replacement, as the transplanted lung is susceptible to fluid overload due to its disrupted lymphatic drainage, 38 although this is unlikely to be an issue during cataract surgery. Dissection around the trachea at the time of transplant may disrupt cardiac autonomic innervation and result in a denervated heart. 40
Each category of lung transplantation presents its own unique problems. In double-lung and combined heart-lung transplant, mechanical ventilation can be uniform to both lungs, as the transplanted lungs have the same compliance. However, the compliance in the native lung and the transplanted lung may be different in single-lung transplant, depending on the underlying lung pathology of the patient (obstructive versus restrictive).
Airway complications after lung transplantation include anastomotic dehiscence, stenosis and bronchomalacia. Therapeutic management may include balloon dilatation, stent placement, laser treatment and occasionally surgery.
Diabetes (83%), hypertension (41%) and renal dysfunction often develop within five years of transplantation as a result of immunosuppressive therapy.38,41 Approximately 10% of lung transplant recipients develop end-stage renal failure within five years. 37 Patients with pre-existing cystic fibrosis have a higher risk of renal dysfunction because of associated diabetes.
Gastrointestinal function may be significantly impaired both acutely and chronically in 40% of recipients after lung transplantation. 37 There is also evidence of a link between chronic lung allograft damage and possible aspiration. 37 Elevation of the head position (>30°) during cataract surgery is desirable, but it may not be practical for some surgeons. Airway protection with endotracheal intubation during general anaesthesia may be indicated.
Summaries of physiological changes and potential complications after lung transplant are included in Tables 2 and 4, respectively.
Physiological changes after lung transplantation and the anaesthetic implications.
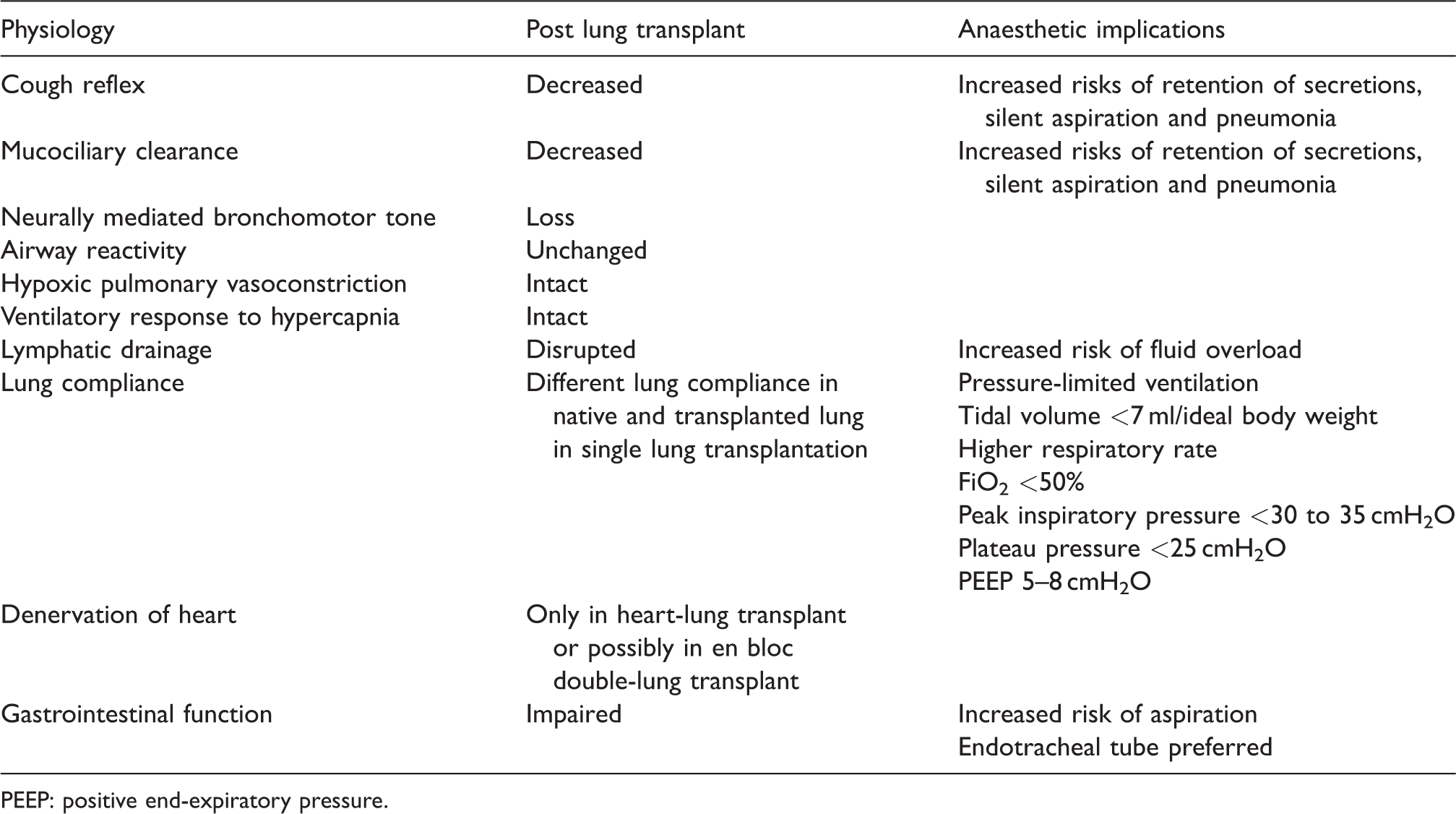
PEEP: positive end-expiratory pressure.
Liver transplant
Hepatic function rapidly returns to normal after successful liver transplantation, including coagulation and drug metabolism. Many systematic manifestations of liver disease (e.g. ascites, pulmonary shunt, hyperdynamic state) also return to normal.42,43 However, allograft liver has lost its autoregulatory ability, and is therefore susceptible to hypotensive injury. Most general and local anaesthetic agents can be safely used on liver transplant recipients.42–44
Kidney transplant
The transplanted kidney also loses its autoregulatory ability, becoming more susceptible to hypoperfusion injury. While serum creatinine level often returns to normal, the glomerular filtration rate and renal plasma flow are usually reduced on direct measurement. 45 This may have an effect on drug metabolism and excretion. It would be ideal to choose drugs (e.g. muscle relaxant: atracurium, cisatracurium) that do not rely on renal excretion during anaesthetic management unless there was an a priori plan to use rocuronium and reverse it with sugammadex.
Despite successful transplantation, pre-existing diabetes-related complications (e.g. coronary artery disease, hypertension) persist in diabetic kidney transplant patients. In fact, cardiovascular complications are the leading cause of death (32%) in renal transplant recipients. 43
Pancreas transplant
Pancreas transplantation provides an effective method for glycaemic control. It can be performed as a single-organ transplant or combined kidney-pancreas transplant (predominantly for diabetes-related end-stage renal failure). Residual requirement of insulin may still be necessary, even after a successful pancreatic transplant. Therefore, blood glucose levels should be monitored perioperatively. 42 In patients with failed pancreatic grafts, perioperative management of glucose level is the same as that of any diabetic patient.
The risk of developing diabetes-related cardiovascular disease remains high, despite successful pancreas transplant, due to the effect of previous longstanding diabetes. Furthermore, these patients are also at risk of other diabetes-related complications, such as autonomic neuropathy, delayed gastric emptying and increased risk of aspiration.42,43
Intestine transplant
Intestinal or multi-visceral transplantation is a therapeutic option for patients with intestinal failure in children and adults. It can be performed as an isolated intestinal transplant or in combination with other visceral organs (e.g. liver, pancreas, kidney). Electrolyte imbalances may be present due to chronic diarrhoea and dehydration resulting from a short native or transplanted colon. Graft rejection is the biggest problem and the leading cause of morbidity and mortality. 43
Bone marrow transplant
Bone marrow transplantation (autologous or allogeneic) has been the main therapeutic modality for patients with life-threatening haematological diseases, especially children. Total body irradiation, an adjuvant to bone marrow transplantation, may lead to subsequent pulmonary fibrosis and restrictive cardiomyopathy. 46 Graft-versus-host disease after bone marrow transplant involving any organ system, with skin, liver and gastrointestinal tract the most affected, is 5–10 times more common than following other organ transplantation. 47
Corneal transplant
There are many surgical techniques for corneal transplantation, for example penetrating keratoplasty, anterior lamellar keratoplasty and endothelial keratoplasty. Accelerated endothelial cell loss is a common problem after corneal transplantation. Surgical and anaesthetic management for cataract surgery in these patients should aim to minimise further endothelial cell damage.
In certain circumstances, corneal transplantation and cataract surgery occur during the same procedure. During penetrating keratoplasty, an ‘open sky’ operation may predispose patients to expulsive choroidal haemorrhage, a rare but catastrophic complication. 2 This risk is further increased in elderly patients with blood pressure spikes and known hypertension, as well as those with reduced coagulation ability. Coughing, gagging and Valsalva events may precipitate haemorrhage. Extra care should be taken to avoid such reflexes.
Immunosuppressive drugs: side-effects and anaesthetic drug interactions
Immunosuppressive therapy often commences prior to transplantation to reduce the risk of transplant rejection. Maintenance regimens, utilising combinations of agents, usually involve one drug from each of the following groups: calcineurin inhibitors, antimetabolites and corticosteroids. The strategy has been changing over the years, with a shift from cyclosporine to tacrolimus, azathioprine to mycophenolate mofetil, and reduced doses of corticosteroid. 43
Chronic immunosuppressive therapy can lead to diabetes, hypertension, renal impairment, increased risk of infection and malignancy, pancytopenia from bone marrow depression and poor wound healing. 43 Commonly used immunosuppressive drugs and their respective side-effects are summarised in Table 5.
Commonly used immunosuppressive agents and their side-effects.
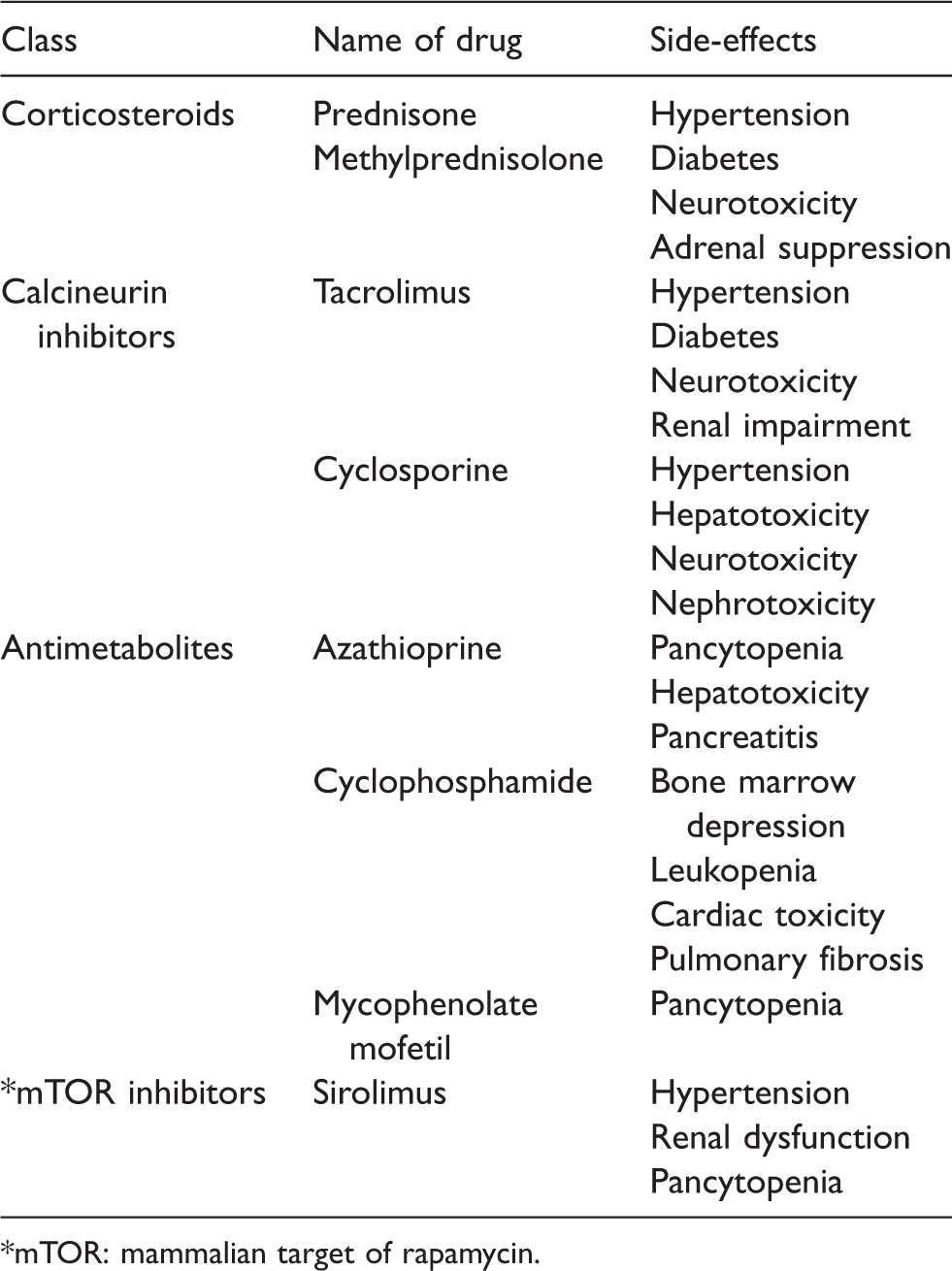
*mTOR: mammalian target of rapamycin.
Immunosuppressive drugs have a narrow therapeutic index. Subtherapeutic drug levels increase the risk of transplant rejection, while supratherapeutic levels may induce toxic side-effects and increase risk of infection. Cyclosporine and tacrolimus must be kept within the therapeutic range to achieve their desired effect. Perioperative fluctuations in plasma levels of these two drugs should be minimised.
Anaesthetic drugs that interfere with the cytochrome P450 (CYP450) enzymes may influence the plasma concentration of tacrolimus, as it is metabolised by CYP450. Barbiturates (e.g. thiopentone) induce CYP450 and therefore reduce the plasma concentration of tacrolimus. 27 Propofol inhibits CYP450. However, the clinical significant of this interaction is unclear. 27
Cyclosporine and tacrolimus increase the blood level of benzodiazepines. 43 Cyclosporine increases the analgesic effect of fentanyl, but the exact mechanism is unclear. Prolonged neuromuscular block after vecuronium and pancuronium has also been reported in patients receiving cyclosporine. 43
Risk of infection
Transplanted recipients are immunosuppressed and therefore susceptible to bacterial, viral and fungal infections, yet may not exhibit the common signs of infection (fever, elevated white cell count). Concurrent infection should be excluded before proceeding to surgery.
The incidence of chest infection, in particular bacterial pneumonia, in lung transplant recipients is higher than that of other transplant recipients. 37 The anastomosis site is subject to ischaemia because there is no revascularisation of the bronchial vessels. Infection at the site may eventually result in anastomotic stenosis and bronchomalacia.
There is no evidence to support increased use or duration of prophylactic antibiotics in transplant recipients. 42 Routine prophylactic intravenous antibiotic is not recommended for cataract surgery, as the operation is not associated with bacteraemia.
Transplant rejection
Acute rejection, treated aggressively by immunosuppressive therapy, can develop in transplant recipients. It occurs, for instance, in up to 50% and 90% of recipients in the first month and first year, respectively, after lung transplantation. 37
The incidence of chronic rejection is thought to increase progressively with time after transplantation. 48 It occurs, for instance, in 35%–50% and up to 70% of recipients one and five years, respectively, after lung transplantation. 37 Bronchiolitis obliterans syndrome, characterised by fibrous obliteration of endothelial structures and reduced forced expiratory volume in 1 second (FEV1 <80% of the peak value), is the hallmark of chronic rejection and the leading cause of mortality (40% within two years following diagnosis). 49 Chronic heart allograft rejection usually presents as accelerated coronary artery disease. 43
Chronic transplant rejection results in a progressive deterioration of the organ function and is the main cause of late mortality in transplant recipients. The presence of rejection should be excluded preoperatively, as surgery during a period of rejection is associated with higher morbidity. 39
Evaluation of other systems
Renal dysfunction, diabetes, gastrointestinal dysfunction and cardiovascular disease are commonly encountered in transplant recipients (except corneal transplant), and they may be exaggerated by side-effects of immunosuppressive therapy and diabetes. Severe renal dysfunction in post-transplant recipients may eventually require dialysis. Gastrointestinal reflux and aspiration are common after lung, pancreas and intestinal transplant.37,43,47 Diabetes-related long-term complications (e.g. autonomic neuropathy, delayed gastric emptying) should be evaluated. Special attention should also be given to cardiovascular disease, a major risk factor for morbidity and mortality among transplant recipients, especially in the presence of renal impairment or heart transplant. Many of these patients may have asymptomatic coronary artery disease.
Lymphoproliferative disorders, in particular B-cell non-Hodgkin’s lymphoma, are more common after lung transplant than other organ transplants. 37 The lung allograft, characterised by multiple pulmonary nodules, is the most common site involved.
Advice on medications
Medications, including immunosuppressants, anti-hypertensives and diuretics, should be continued as normal on the day of surgery. Additional corticosteroid administration intraoperatively is not usually required for routine cataract surgery.
Patients receiving antithrombotic agents
Transplant recipients may be on antithrombotic agents, including antiplatelet, warfarin and novel oral anticoagulant. 47 Cataract surgery is considered a low risk for bleeding. These agents may be continued and a suitable anaesthetic method employed subject to particular circumstances and surgical requirements, as explained in later paragraphs.
Glycaemic control during cataract surgery
The Joint British Diabetes Societies Guidelines and the American Diabetes Association recommend that blood glucose concentrations should be maintained between 140 and 180 mg/dl (7.8 and 10.0 mmol/l) during the perioperative period. However, currently, there is no recommendation on blood sugar concentration criteria for postponing cataract surgery due to the low-risk nature of this operation. 50
Scheduling for surgery
Cataract surgery in transplant recipients should be performed as early as possible on the operating list. Prolonged fasting and dehydration may have adverse effects in renally impaired patients and heart transplant recipients.
Choosing anaesthesia modality and intraoperative considerations
Cataract surgery can be performed under topical anaesthesia, regional ophthalmic block or general anaesthesia. In the presence of normal hepatic and renal function, there is no contraindication to the use of any general or local anaesthetic agents.39,51 Whilst topical anaesthesia and regional ophthalmic block are ideal techniques for cooperative adults, general anaesthesia may be required for children, for extremely anxious, claustrophobic or intellectually impaired adult patients and for complicated cataract surgery.
Cataract surgery is usually associated with minimal haemodynamic or stress response to surgery. Therefore, ophthalmic anaesthetists are rarely required to manage the major physiological changes, as described earlier. In addition, the surgical time is often brief. So, the issues of intraoperative glycaemic control and prolonged use of nitrous oxide may not be relevant.
Pacemakers and AICDs, if present, do not require modification during routine cataract surgery as electrocautery (bipolar) is rarely used. 52
Topical anaesthesia
Topical anaesthesia is ideal for cooperative adult transplant recipients, especially those on antithrombotic therapy. However, it may not be suitable in mature or complicated cataracts, nor the preference of some surgeons, as this technique is suited to faster surgeons and those who tolerate patients’ eye movements.
Regional ophthalmic blocks
The choice of a particular regional block largely depends on the anaesthetist’s preference and expertise. Fast injection of local anaesthetic agent around the globe may sometimes precipitate the oculocardiac reflex. 53 Patients with a denervated heart do not exhibit an oculocardiac reflex.
Modern cataract surgery is performed by a phacoemulsification technique. 54 There are few published recommendations for cataract surgical technique in corneal transplant recipients. In post-penetrating keratoplasty, Cao et al. recommended the use of femtosecond laser-assisted phacoemulsification technique. 16 Zhou et al., on the other hand, recommended the extracapsular cataract extraction technique because it was associated with significantly lower endothelial cell loss rate when compared to phacoemulsification in these patients (no difference after cataract surgery in normal cornea patients). 55 In endothelial keratoplasty recipients, a fixation suture can be applied to the endothelial graft at the beginning of cataract surgery to minimise the risk of graft displacement. 15 Regional ophthalmic block is the preferred technique in these patients in view of complicated cataract surgery.
General anaesthesia
The use of a laryngeal mask airway is acceptable. However, an endotracheal tube should be considered for transplant recipients at higher risk of gastrointestinal reflux and aspiration (lung, pancreas, intestinal and multi-visceral transplant recipients). Nasal intubation should be avoided, as it increases the risk of bacteraemia.
The majority of volatile anaesthetic agents are well tolerated unless there is significant heart failure. Prolonged use of nitrous oxide is best avoided because of the potential risk of bone marrow suppression, 43 especially in the presence of immunosuppressive drugs that have similar side-effects. However, there is no absolute contraindication to its use in short surgical procedures such as cataract surgery. Nitrous oxide should be avoided in intestinal transplant recipients, as it can increase bowel distension. 47
Although simultaneous penetrating keratoplasty and cataract surgery can be performed under regional ophthalmic block, some surgeons prefer general anaesthesia to reduce the possibility of expulsive choroidal haemorrhage exacerbated by intraoperative patient reflexes and movement. ‘Profound’ neuromuscular blockade (post-tetanic counts = 1–3) has also been advocated. 56
Provision of general anaesthesia for heart and lung transplant recipients requires further special attention.
In patients who have had a heart transplant, the aim is to maintain preload, thence cardiac output, by avoidance of hypotension and vasodilatation. Slow gentle induction is paramount. In general, invasive monitoring (arterial and central venous lines) is not required for cataract surgery. Laryngoscopy and tracheal intubation may not produce a sympathetic response because the cardiac baroreceptor reflex is not intact. 27
In patients who have had a lung transplant, preoperative administration of an anti-sialagogue is useful in reducing secretions.40,57 An endotracheal tube is preferred because of increased risk of aspiration. 40 Intraoperative capnography in single-lung transplant recipients may show a biphasic pattern: one peak from the native lung, and the other peak from the transplanted lung. 58
Pressure-limited mode is preferred during mechanical ventilation, especially in single-lung transplant recipients. The recommended ventilation parameters are: lower tidal volume (<7 ml/ideal body weight), higher respiratory rate (but avoid hypocapnia), limit peak inspiratory pressure to 30–35 cmH2O, plateau pressure <25 cmH2O, positive end-expiratory pressure 5–8 cmH2O and keep inspired oxygen concentration <50% (Table 4). 38
Postoperative considerations
Paracetamol, in its standard analgesic dose, is usually sufficient if required for analgesia after cataract surgery and is not contraindicated, even in liver transplant recipients. 42
A topical ophthalmic non-steroidal anti-inflammatory drug (NSAID; e.g. ketorolac tromethamine) is often prescribed routinely as a prophylaxis against inflammation and cystoid macular oedema. 59 Systemic NSAIDs are best avoided in post-transplant recipients, as they may worsen renal dysfunction, especially in the presence of cyclosporine or tacrolimus.39,43,47 Systemic absorption of most topical ophthalmic NSAID is minimal. Consequently, it is seldom associated with the same side-effects as oral administration. 59
Persistent nausea and vomiting, blurred vision, severe ocular pain and headache may be symptoms of raised intraocular pressure. 60 Raised intraocular pressure, potentially caused by retained viscoelastic material, 61 may warrant early surgical intervention (e.g. burping the paracentesis wound to release fluid from the anterior chamber, washout of anterior chamber to remove retained material). Medical therapy with acetazolamide or mannitol should be used with caution, as this may worsen pre-existing renal dysfunction.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
