Abstract
Background and Aim:
Although upper limb myoelectric prostheses can offer improved functionality and dexterity over body-powered systems, abandonment rates remain high. User dissatisfaction in comfort and control are among the top contributors. The design of the prosthetic socket must be comfortable, while maintaining contact of control electrodes with the residual limb throughout the day. We present a myoelectric socket design that provides user-adjustable compression over electrode control sites to promote consistent control, while maintaining comfort and fit.
Technique:
A cable tensioning system was threaded through a series of paneled windows in the socket wall over electrode sites. Adjusting tension provided tuning of electrode contact.
Discussion:
A case study of a single transhumeral prosthetic user with a follow-up interview 11 months post delivery suggests that our adjustable design has the potential to address control and comfort challenges, critical factors in myoelectric prosthetic use, and abandonment.
Clinical relevance
Achieving consistent electrode contact with muscle control sites in traditional rigid sockets is a critical challenge for myoelectric prostheses. We present a unique solution via user-adjustable electrode contacts built into the socket.
Background and Aim
Advanced upper limb myoelectric prostheses have the potential for transformative improvements for users.1,2 When comparing these systems to body-powered prostheses, they offer the potential benefits of multiple grasping patterns, fewer cables and harnessing, reduced phantom pain, and improved cosmesis. 1 However, user abandonment rates still remain high with these devices. In 2007, it was estimated that approximately 23% of individuals who own a myoelectric prosthesis discontinue using their device. 3 Dissatisfaction in comfort, function, and control are often highlighted as contributors to abandonment, or identified as priorities to be addressed.3–5
The prosthetic socket is a fundamental element in addressing these challenges. The socket is the point of attachment to the residual limb and the interface that couples the prosthetic components to the user. It must appropriately compress residual soft tissue to facilitate suspension, functional range of motion, comfort, and stability.6–9 Traditional sockets are custom rigid fabrications manufactured from thermoplastics or laminates. However, residual limbs are dynamic; volume fluctuations occur early post amputation and continue in mature limbs in response to daily activity and muscle contraction.10–12 These volume changes can compromise comfort or fit and may lead to slippage or displacement of the socket, potentially encumbering control and function.
Myoelectric prosthetic control is contingent upon electrodes contacting the residual limb at specific muscle control sites with appropriate pressure. Slippage or displacement of the socket can result in electrodes losing contact with these muscle control sites. 13 Appropriate contact pressure must also be maintained, as surface electrodes are inherently separated from the muscle by intermediate layers of soft tissue. A decrease in pressure can lead to changes in impedance as the electrode is moved further from the muscle. This may decrease signal amplitude or increase signal noise. 14 In response, the user may need to increase muscle contraction to control the device, leading to fatigue. Should a gap form between the electrode and limb, motion artifacts may be introduced to the signal resulting in unintentional movement of the prosthesis, 15 or a loss of control over the corresponding motion. In contrast, if contact pressure is increased, the sensor may register higher amplitude signals and may trigger unintentional prosthetic movement. 14
Designing sockets to maintain appropriate electrode contact during daily activities and volume fluctuations is an ever-present challenge in delivering prostheses that are reliable and consistent in their control and function. This work describes the fabrication of a novel transhumeral myoelectric socket that provides adjustable tissue compression over myoelectric control sites. The fabrication strategy is described, and we present user impressions.
Technique
Client description
An adjustable compression socket was fabricated for a healthy 31-year-old male prosthetic user with a left transhumeral amputation of traumatic etiology (Table 1). A successful socket was delivered on the first fabrication iteration. The client used a myoelectric prosthesis with two muscle control sites (biceps and triceps), and suction suspension for 4.5 years. The client was clinically assessed to have good soft tissue coverage of residual limb and described minimal limb volume fluctuation throughout the day. Socket fabrication services were requested specifically to address control and comfort issues associated with the client’s previous traditional myoelectric socket. Written informed consent was provided, and ethical approval was obtained through the University of Alberta’s Human Research Ethics Review Board (Study ID: Pro00034663).
Participant demographics.
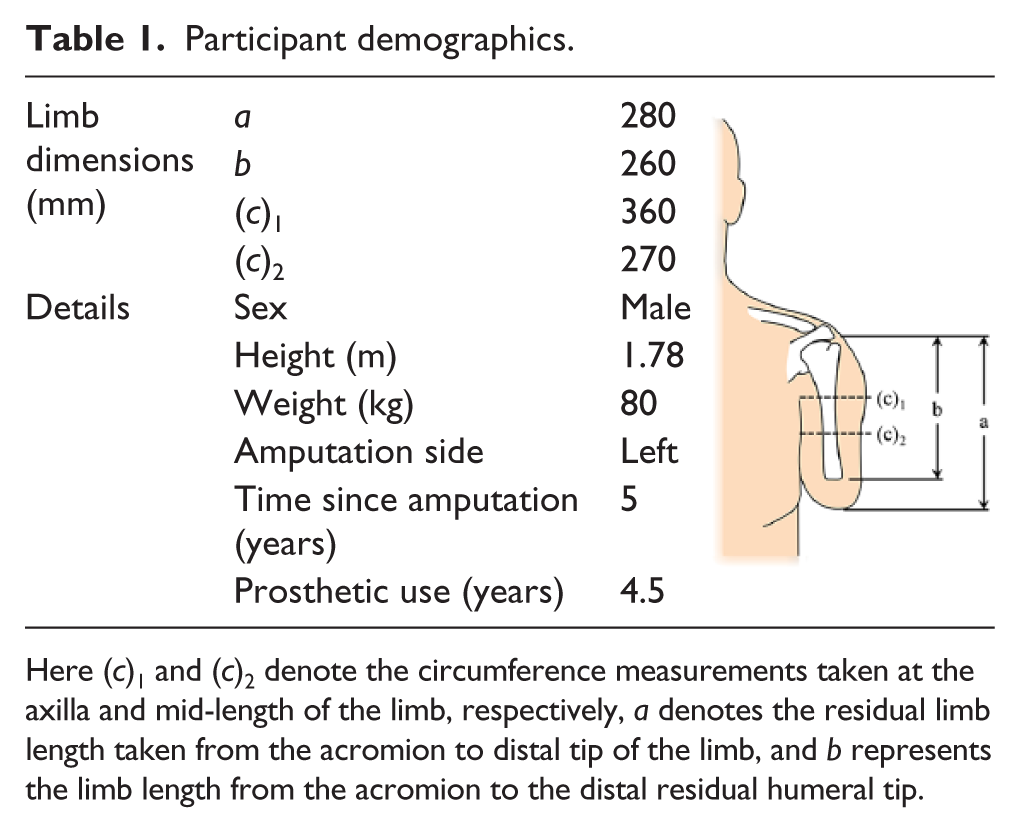
Here (c)1 and (c)2 denote the circumference measurements taken at the axilla and mid-length of the limb, respectively, a denotes the residual limb length taken from the acromion to distal tip of the limb, and b represents the limb length from the acromion to the distal residual humeral tip.
Fabrication technique
Prior to socket fabrication, a standard myoelectric assessment was performed to confirm muscle control sites. A traditional thermoplastic check socket (Orfit Trans Stiff; Orfit, Belgium) was fabricated, and the definitive prosthetic components installed. This system was trialed for 2 weeks to verify electrode placement and identify fit-based adjustments.
A definitive socket was then manufactured, which duplicated the post-trial-period check socket geometry. A flexible inner socket (ThermoLyn Soft; Ottobock, Germany) was pulled to approximately 3 mm thickness. Wiring blanks were affixed to the exterior, and a rigid outer socket was formed over this assembly creating wiring channels (carbon fiber and fiberglass impregnated with Orthocryl resin 617H19; Ottobock). Control electrodes (Triad; Motion Control, USA) and a suction one-way air valve (Mag Valve 21Y15; Ottobock) were installed in the flexible layer.
Modifications were made to the rigid layer creating adjustable panels to compress electrode control sites. This involved cutting windows over each muscle control site that extended beyond the electrodes’ footprints by approximately 10 mm (Figure 1). The removed material was set aside to be used later as a compression panel (Figure 1). On the flexible inner layer, a beveled Soleflex (SOLFLXSBK12; Vittoria Phoenix, Canada) border was added around each electrode, which extended 1 mm higher than the electrode’s top surface (Figure 1). This reduced compression forces applied directly to the electrode by the compression panel.

Schematic diagrams of the adjustable compression socket assembly; top left: medial view, top right: anterior view, bottom: view of the socket interior.
RevoFit cable guides (Click Medical, USA) were adhered to the exterior of the compression panels, a cable was threaded through the guides, and the panels were positioned over their corresponding electrode windows. The cable was then threaded into the RevoFit tensioning dial (Click Medical). The tensioning dial was positioned in a location for easy access. Finally, a lamination ring was added and prosthetic components installed (Mechanical elbow, AFB 12K44; Ottobock and myoelectric hand, Bebionic; Ottobock).
The final design allowed the user manual adjustment of cable tension, providing control over the pressure applied through the compression panels to the electrodes and underlying tissue. Unlocking the dial relieved the contact pressure to allow convenient donning, doffing, and comfort- or control-related adjustments.
Technical evaluation and case study
A follow-up interview was performed 11 months following prosthesis delivery. This included a self-reported prosthetic comfort score, 16 an Orthotics and Prosthetics User’s Survey (OPUS) device satisfaction survey 17 modified to specifically capture comfort and fit, 9 and qualitative questions on device usage, perceptions of comfort, control, and impressions of the socket (included in Supplementary Material).
The participant reported wearing his adjustable compression prosthesis 3–4 h/day and two to three times/week, approximately 1 day/week more than his previous socket. He reported a comfort score of 7/10, indicative of a comfortable and well-fit socket. 16 Modified OPUS scores are tabulated in Figure 2. Compared to his previous socket, the participant reported no change in perceived heaviness, temperature, or time to don and doff. He felt that the tensioning dial was slightly bulky while wearing dress clothes, but was otherwise unobtrusive. The myoelectric control was described as “very easy,” with the ability to “feather” control of the hand, or produce slow or small movements if desired. This contrasts with his prior socket, with which he described his control as “all or nothing.” The participant did not typically adjust the socket tightness after donning, preferring to “set and forget” the compression.
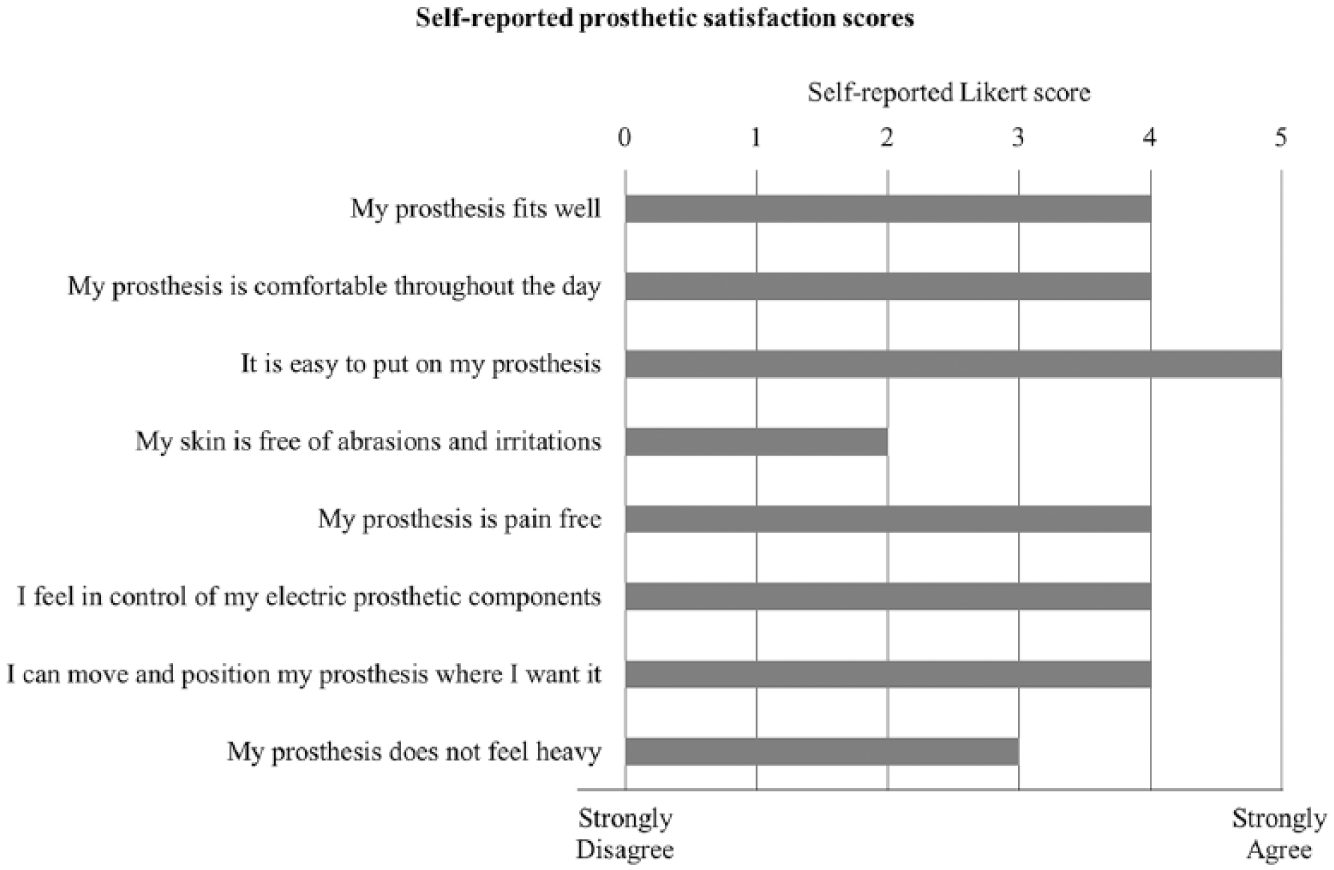
Participant satisfaction self-report scores; a 0–5 Likert-type scale was used to denote strong disagreement or strong agreement to questionnaire statements.
Discussion
Achieving consistent electrode contact is a fundamental challenge in traditional rigid upper limb myoelectric sockets as the residual limb is dynamic and may fluctuate in volume. This work presents a unique solution to provide consistent prosthetic control through adjustable tissue compression over the electrode contacts. Our participant indicated increased prosthetic usage and satisfaction as a result. This design may help augment transhumeral prosthetic suspension, improve electrode contact, and reduce unintended displacement of the socket.
Our client reported minor daily limb volume fluctuations that are anticipated in an average individual with transhumeral amputation. He suggested that he “sets and forgets” the amount of compression with each use of his prosthesis. This indicates a relatively stable daily limb volume and demonstrates a specific benefit of our design for those with between-day volume changes. However, we further suggest that those experiencing within-day volume fluctuations may also benefit from our design by enabling adjustments throughout the day as the user deems necessary.
Our techniques depart from traditional fabrication practices and involves increased technical burden with a possible 8–10 additional hours of fabrication time. However, this has the potential to address a well-known challenge in upper limb myoelectric prosthesis fitting by enabling a patient to have a more consistently functioning and comfortable prosthetic device.
Footnotes
Acknowledgements
The authors would like to acknowledge Madeline Newcomb for her assistance with figure generation.
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This work was supported by the US taxpayers through the National Institutes of Health Common Fund Transformative R01 Research Award (grant no. 1R01NS081710 – 01).
Supplemental material
Supplemental material for this article is available online.
