Abstract
Background:
Amputation of a limb impacts on patients’ self-perception and quality of life. Prostheses directly anchored to the skeleton are being investigated, aiming to avoid soft tissue complications.
Objectives:
We report outcome data for the UK trial of the Osseointegrated Prosthesis for the Rehabilitation of Amputees Implant System with a minimum of 9-year follow-up.
Methods:
Eighteen transfemoral amputees received unilateral implants between 1997 and 2008. Five were implanted before a formalised protocol, called Osseointegrated Prosthesis for the Rehabilitation of Amputees, was developed. Mean follow-up of the Pre-Osseointegrated Prosthesis for the Rehabilitation of Amputees group is 11.4 years (1.8–18.6 years), while for the Post-Osseointegrated Prosthesis for the Rehabilitation of Amputees group it is 12.3 years (2.9–15.9).
Results:
The Kaplan–Meier cumulative survivorship is 40% for the Pre-Osseointegrated Prosthesis for the Rehabilitation of Amputees group and 80.21% for the Post-Osseointegrated Prosthesis for the Rehabilitation of Amputees group. Five implants (28%) have been removed, three (17%) for deep infection, one (5.6%) for chronic pain, later proven to be infected and one (5.6%) due to implant fracture secondary to loosening due to infection. Two patients (11%) have peri-implant infections suppressed with oral antibiotics. Eleven cases (61%) of superficial infection were successfully treated with antibiotics. 36-Item short-form health survey and Questionnaire for persons with a Transfemoral Amputation showed significant improvements in quality of life up to 5 years after implantation.
Conclusion:
This small cohort of patients demonstrates osseointegrated prosthesis allows prolonged usage and improves patients’ quality of life compared to conventional prostheses.
Clinical relevance
These prostheses may provide a future gold standard for amputees and this study provides the first outcome data over such a time period to be reported from outside of the developers group.
Keywords
Background
Amputation of a limb has a substantial impact on lifestyle and self-perception.1,2 Quality of life is closely related to the ability to use a prosthetic limb. 3 Loading the residual limb soft tissues using conventional prostheses commonly causes problems including pain or discomfort, reducing wearing tolerance over time and residual limb skin infection and ulceration which negatively affect quality of life and mobility.4–6 Solutions to avoid such complications include attaching the prosthesis directly to the residual skeleton.7–11 Such osseointegrated prostheses have been implanted into transfemoral amputees since 1990 in Gothenburg, Sweden. 12 In 1999, a protocol was developed, as part of a trial, detailing the surgical technique and subsequent rehabilitation called the Osseointegrated Prosthesis for the Rehabilitation of Amputees (OPRA). 12 The implant has been named the OPRA implant manufactured by Integrum AB, Mölndal, Sweden. Prior to this, osseointegrated prostheses have been used since 1965 in dentistry, craniofacial prosthetics and bone-anchored hearing aids. 11
The Douglas Bader Rehabilitation Centre at Queen Mary’s Hospital was the first unit to offer implantation of the OPRA implant outside of Sweden. The authors present the mid- to long-term outcome data and complications for these devices in the first such report from outside of Sweden.
Methods
Eighteen carefully selected patients received unilateral transfemoral OPRA implants between 1997 and 2008. All amputations were due to trauma and all patients underwent the transfemoral amputation in a separate procedure to the implantation of the OPRA device, often many years prior.
Ethical approval was obtained from the Department of Health and the local South West Thames ethics committee prior to the recruitment of patients. Amputees underwent a very thorough assessment at the Douglas Bader Unit at Queen Mary’s Hospital before being accepted and proceeding with surgery. The 3-day assessment process was undertaken by a Consultant Orthopaedic Surgeon, a Consultant Rehabilitation Physician, a Specialist Physiotherapist, a Clinical Psychologist, a Specialist Occupational Therapist, a Specialist Prosthetist, as well as gait analysis using the patient’s conventional socket prosthesis by a Specialist Bioengineer. After full disclosure with the patient regarding expected benefits, potential complications and the expected length of rehabilitation, informed consent was taken for the process to begin.
The procedure was not available under National Health Service funding and therefore, after patients had successfully undergone the aforementioned assessment process, separate individual funding requests (IFRs) were made to each separate patient’s primary care trust (PCT) to fund the surgery. Due to this process, patient enrolment was slow and therefore, only 18 implants were inserted at this unit. Inclusion criteria are shown in Table 1.
Inclusion and exclusion criteria for enrolment into the trial.
Surgical technique
The first five implants were inserted between 1997 and 1999 before the OPRA protocol was formalised. Despite this fact, devices in both pre-OPRA and post-OPRA groups were implanted in a similar manner and patients underwent similar rehabilitation in both groups, according to the OPRA system protocols, which were formalised in 1999 by Hagberg and Brånemark. 12
The implants were custom made for each patient from commercially pure titanium and were supplied along with the necessary surgical instruments by Integrum AB, Mölndal, Sweden. The implants were inserted using a two-stage technique. Prior to the initial surgical procedure, plain radiographs were taken along with a 1:1 CT scan according to a standardised protocol. These images were used for the fabrication of the customised implants.
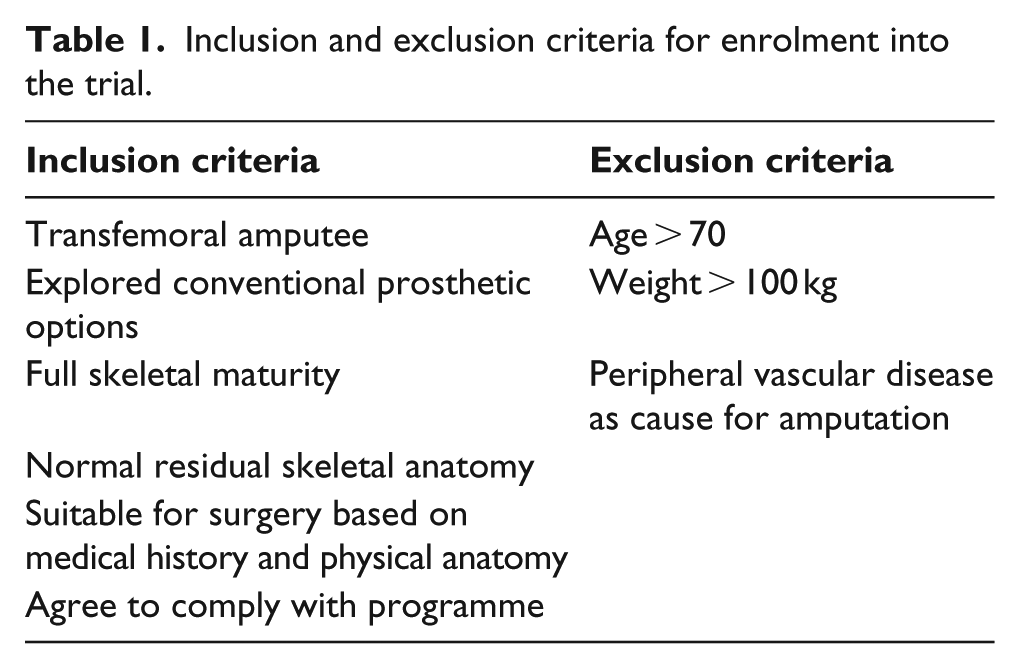
The first-stage procedure involved inserting a titanium tube, measuring 110–150 mm in length and 9–20 mm in outside diameter, with a self-tapping screw around the outer circumference, as well as an inner screw thread, into the intramedullary canal following sequential reaming (Figures 1 and 2). This portion of the implant is termed the fixture. Access was achieved through the existing terminal scar or through newly fashioned anterior and posterior flaps, with the posterior flap left slightly longer to produce an eventual terminal scar anterior to the residual bone end and second-stage penetration site. Residual muscles were defined and shaped to form a cuff around the bone end, being sutured to the periosteum, in contrast to normal amputation techniques. The soft tissues were closed with sutures and a drain was left in situ for 24 h with appropriate dressings. Antibiotic prophylaxis was administered in two post-operative doses and plain radiographs taken for a final position record.
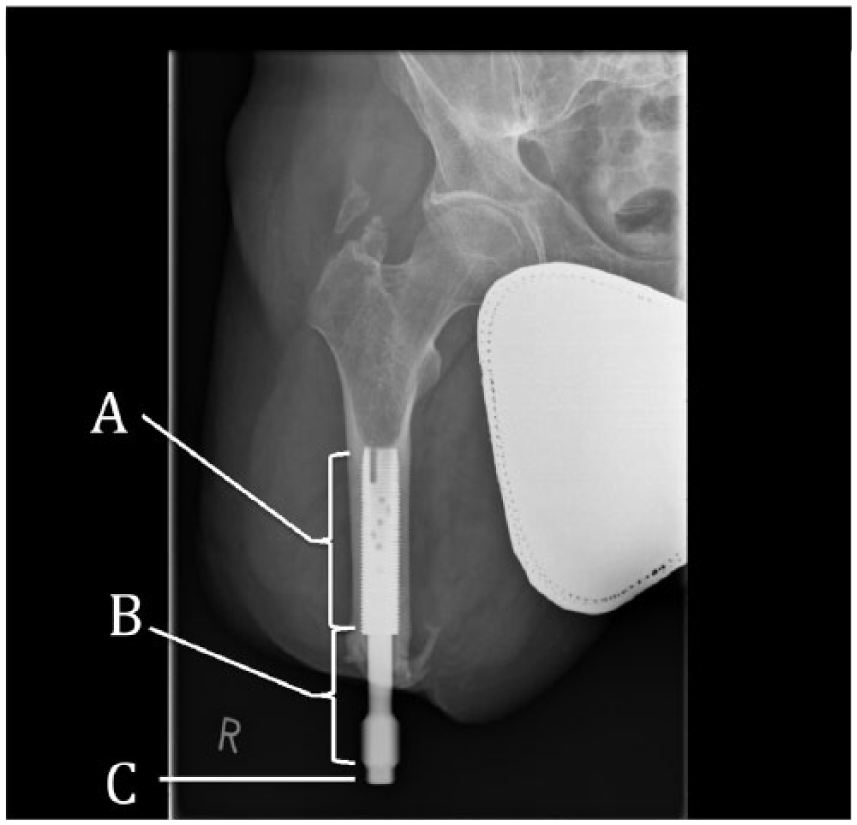
Antero-posterior radiograph showing the eleven year post-operative appearance. The fixture (A), abutment (B) and abutment screw (C) can be seen in situ. Note the short residual bone length which caused the patient difficulty in using conventional prosthesis suspension methods.
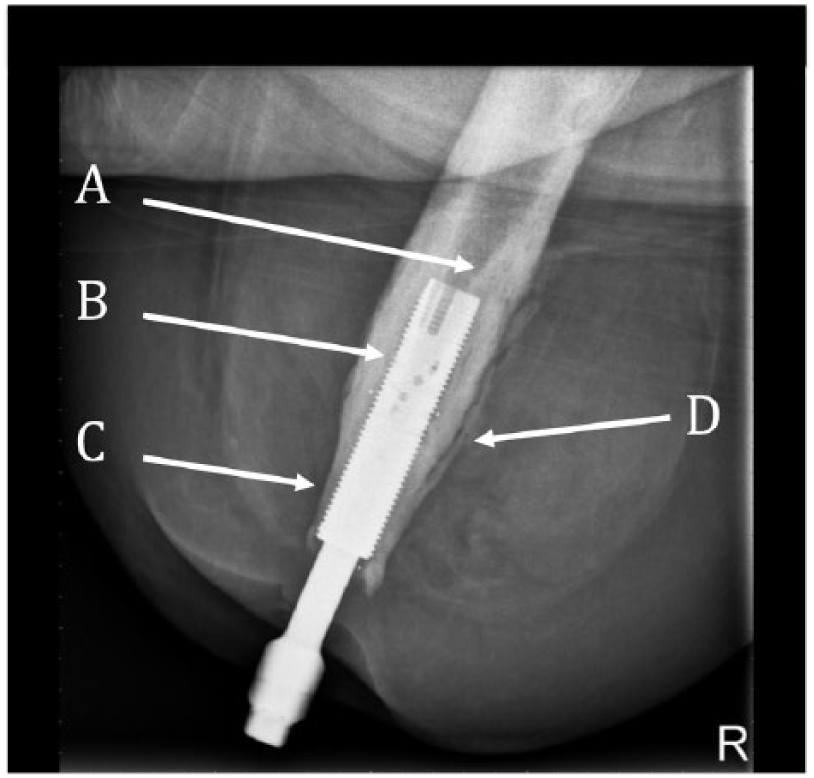
A lateral radiograph from a patient with a low grade, peri-implant infection being suppressed with oral antibiotics. Note the bone remodelling with pedestal formation (A), endosteal lucency (B), distal periosteal resorption (C) and periosteal reaction (D).
A period of 6 months was left between the first- and second-stage procedures. Patients were allowed to use conventional socket type prostheses during this period, if appropriate, depending on wound healing and comfort. The second-stage procedure was performed through the terminal scar; the soft tissue flap was mobilised and thinned at the point where it overlies the bone end. At the point chosen for the prosthesis to penetrate the flap, the dermis was defatted using the edge of a glass microscope slide. A cylindrical skin biopsy cutter was used to fashion a circular hole in the skin flap. A second titanium component, called the abutment, was inserted into the inner diameter of the fixture. An abutment-retaining bolt, manufactured from titanium alloy, was passed through the abutment to secure into the inner screw thread of the fixture, thereby securing the two components together (Figures 1 and 2). The abutment passes through the hole created in the skin flap to connect with the prosthetic limb. The abutment-retaining bolt was tightened to a torque setting of 12 N m. The skin was sutured, a drain inserted for 24 h and a soft dressing applied.
Patient rehabilitation
Rehabilitation followed the OPRA protocol. 13 During the first 6 weeks after the second-stage procedure, exercise was limited to gentle range of movement of the residual joint. General mobility was maintained with the use of crutches or wheelchair. After 6 weeks, when wound healing could be assured, muscle strengthening and core stability exercises were commenced. A short training prosthesis was used, attaching to the externally protruding abutment portion of the implant. Patients were allowed to load the training prosthesis on weighing scales. Initially, a maximum of 20 kg was applied for 15–30 min twice a day. This was continued for 1 week. Each week thereafter a further 10 kg of load was allowed through the training prosthesis until full body mass was achieved. If any difficulty was encountered, a half speed protocol was followed, with the weekly addition of only 5 kg of load. 13 Certain patients were selected to undergo a half speed protocol from the beginning depending on bone quality. 12
Once full body weight loading through the training prosthesis was consistently achieved, the patient was fitted with their full-length prosthesis. This usually occurred 2–3 months following the second-stage procedure. The final prosthesis was attached to the abutment via a failsafe device called the ROTASAFE (Integrum AB, Mölndal, Sweden), which protects the implant from rotational movements. Later a new failsafe device (Axor (Integrum AB, Mölndal, Sweden)) was introduced, designed to protect the abutment from bending moments as well as excessive torque in the event of a fall. Gait training commenced with partial weight bearing between parallel bars, progressing to crutches, walking sticks and finally walking unaided depending on individual progress. Length of time wearing the prosthesis was also increased in a gradual manner. All patients underwent regular gait analysis throughout the process, with corrective therapy as required.
Once full mobility had been achieved, patients were assessed on an annual basis with clinical examination (particularly inspecting for signs of infection or discharge as well as assessing the torque limit on the abutment screw), physiotherapy assessment, radiographic assessment and assessing the level of activity using the Specialist Interest Group of Amputee Medicine (SIGAM) 14 scoring system. As per the OPRA protocol, both the 36-item short-form health survey (SF-36) and the Questionnaire for persons with a Transfemoral Amputation (Q-TFA) 15 were recorded: SF-36 as a global assessment of patient function and Q-TFA as a condition-specific assessment. The SF-36 was used under the OPRA licence from QualityMetric.
Patient recruitment
Data for all patients who underwent implantation of a Brånemark OPRA device into a transfemoral amputated residual limb at the Douglas Bader Rehabilitation Centre at Queen Mary’s Hospital, Roehampton were analysed. Data were taken from a review of the clinical notes and internal registry of patients held at the Douglas Bader Centre from the date of the first-stage surgical procedure until the last recorded entry in the notes or registry.
Study outcomes analysed were as follows:
Time to full mobilisation using a full-length external prosthesis measured with the arithmetic mean.
Time to failure of the implant, namely removal or revision of the device, and reason for failure.
A review of residual limb or implant infections, diagnosed clinically or on microbiological swabs.
Describe the radiographic changes associated with these implants.
The change in quality of life from baseline prior to implantation of the devices using Q-TFA as a condition-specific tool and SF-36 as a generic health and quality of life assessment.
Statistical analysis
Time to failure data were assessed using the Kaplan–Meier survivorship analysis SPSS (Version 22.1 for Mac, SPSS Inc, IBM, Chicago, IL). Quality of life data was analysed using the Mann–Whitney U test (the Wilcoxon rank-sum test) SPSS (Version 22.1 for Mac, SPSS Inc, IBM, Armonk, NY).
Results
As the first five devices implanted were prior to the formalisation of the OPRA protocol data for pre-OPRA and post-OPRA, patients are reported separately. In the pre-OPRA group, all five patients were male. The mean age at the time of implantation was 26.8 years (24–30). In the post-OPRA group, 3 of the 13 patients were female (23%). The mean age at implantation was 37.8 years (21–49). Ten implants were for left transfemoral amputations and eight were on the right side.
Pre-OPRA results
The median length of follow-up for the pre-OPRA group is 11.4 years (1.8–18.6 years). The median length of time taken to achieve full mobility following the second-stage procedure with the final prosthesis was 7 months (6–9). Two out of five patients underwent the half speed rehabilitation, although it was not known as this at the time and data were not recorded as to when these patients achieved full mobility. Three of the five pre-OPRA patients had the implant removed at 1.8, 2.6 and 11.4 years post-implantation. Implants were removed due to infection in two cases and due to a fracture in one case, which was found to be due to loosening secondary to infection. There was no shortening of the residual femur at removal in any case. One patient who had their implant removed due to infection requested to have a new OPRA implant and underwent this in Sweden in 2014. One patient has returned to using their original socket prosthesis. The other patient has a short residual limb and was therefore unable to use traditional prostheses, this being the reason they underwent implantation of an OPRA device. Following removal of the implant, they were still unable to use a traditional prosthesis and therefore currently utilise crutches and a wheelchair. Of the remaining two patients, one was lost to follow up at 18.6 years and the other patient is currently 18 years post-implantation. This patient has a chronic osteomyelitis being suppressed with oral antibiotics. The Kaplan–Meier cumulative survivorship analysis shows a 40% survival at 18.6 years post-implantation and 60% survival up to 11.4 years post-implantation. However, due to the low number of patients, caution needs to be taken when interpreting these results. The Kaplan–Meier cumulative survivorship graph is shown in Figure 3.
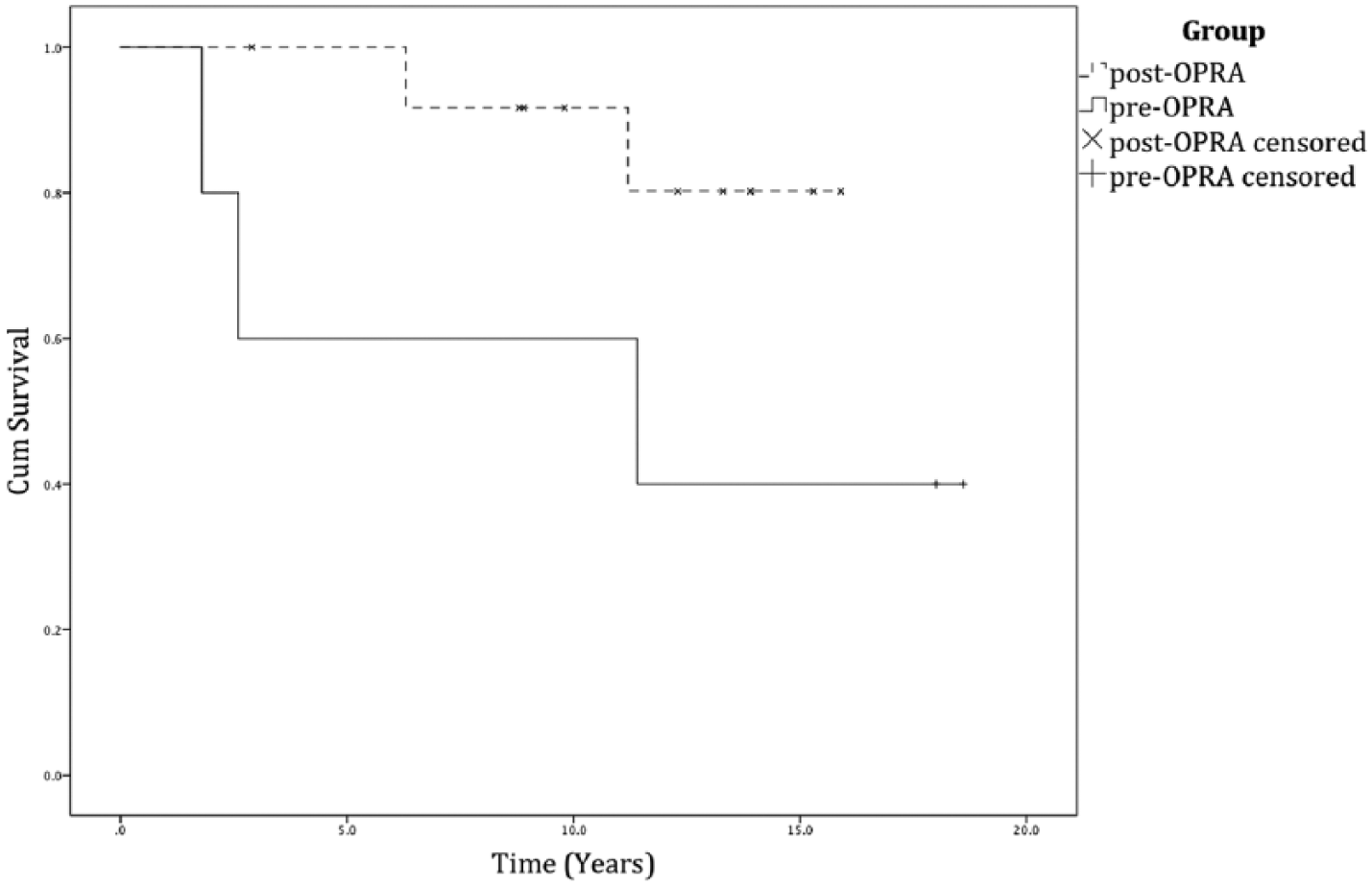
Kaplan–Meier cumulative survival graph.
Post-OPRA results
The median length of follow-up for the post-OPRA group is 12.3 years (2.9–15.9). The median length of time taken to achieve full mobility after the second-stage procedure with the final prosthesis was 9 months (4–15). Four out of thirteen patients underwent the half speed rehabilitation protocol, taking a median time of 11.5 months (10–15) to achieve full mobility. One patient died 2.9 years after surgery due to an unrelated event with the implant functioning well. Two implants were removed at 6.3 and 11.2 years, respectively. Implants were removed due to infection in one case (6.3 years) and due to persistent pain (11.2 years), although infection was discovered on tissue biopsy samples following implant removal. There was no shortening of the residual femur in either case. The remaining implants have been in situ from 8.8 to 15.9 years. One of these patients has a chronic osteomyelitis being suppressed with oral antibiotics. Both patients who had their implants removed requested to have new OPRA implants and underwent surgery in Sweden for this in 2014 and 2015. The Kaplan–Meier cumulative survivorship analysis shows 80.21% survival at 15.9 years and 92% survival at 11.2 years, although again due to the relatively low numbers of patients involved, caution needs to be taken when interpreting the results (Figure 3).
Review of infections
Across both pre-OPRA and post-OPRA groups, 17 patients (94%) have had infection-related problems. Tissue samples taken from the five patients (28%) who had the implant removed due to infection grew MRSA (Methicillin-resistant
Other noted complications
Two patients have sustained a fractured neck of femur on the ipsilateral side to the OPRA implant following a fall. The OPRA implant was not damaged by the fall in either case and both patients underwent surgical fixation, one within the authors unit and one performed at another centre. There were no known complications following these procedures; however, one patient later died due to an unrelated event as previously mentioned.
Twelve patients have required surgery for abutment changes a variable number of times (Table 2). Of these patients, 2 (11%) have fallen and fractured the abutment, 5 (27%) have fractured the abutment-retaining bolt and 11 (61%) have bent the abutment. Three patients have experienced spontaneous fractures of the abutment and of the retaining bolt without any obvious trauma. All patients have had the fractured component replaced with minimal disruption to prosthetic usage. The component is replaced by measuring the inner diameter of the remaining fixture and a new, wider abutment is inserted to ensure a correct fit. Patient details including length of implant survival, current SIGAM activity level and number of abutment changes are shown in Table 2.
Table of patients with age at time of implantation, length of survival, no of procedures to change to abutment due to either bending or fracture and SIGAM activity level whilst using their prosthesis (A = abandoned use of prosthesis, B = wears prosthesis only for transfers or therapy, C = walks up to 50m on even ground [a = with a frame, b = 2crutches or sticks, c = 1 crutch or stick, d = no aid], E = walks 50m or more without aids, F = normal or near normal walking with prosthesis).
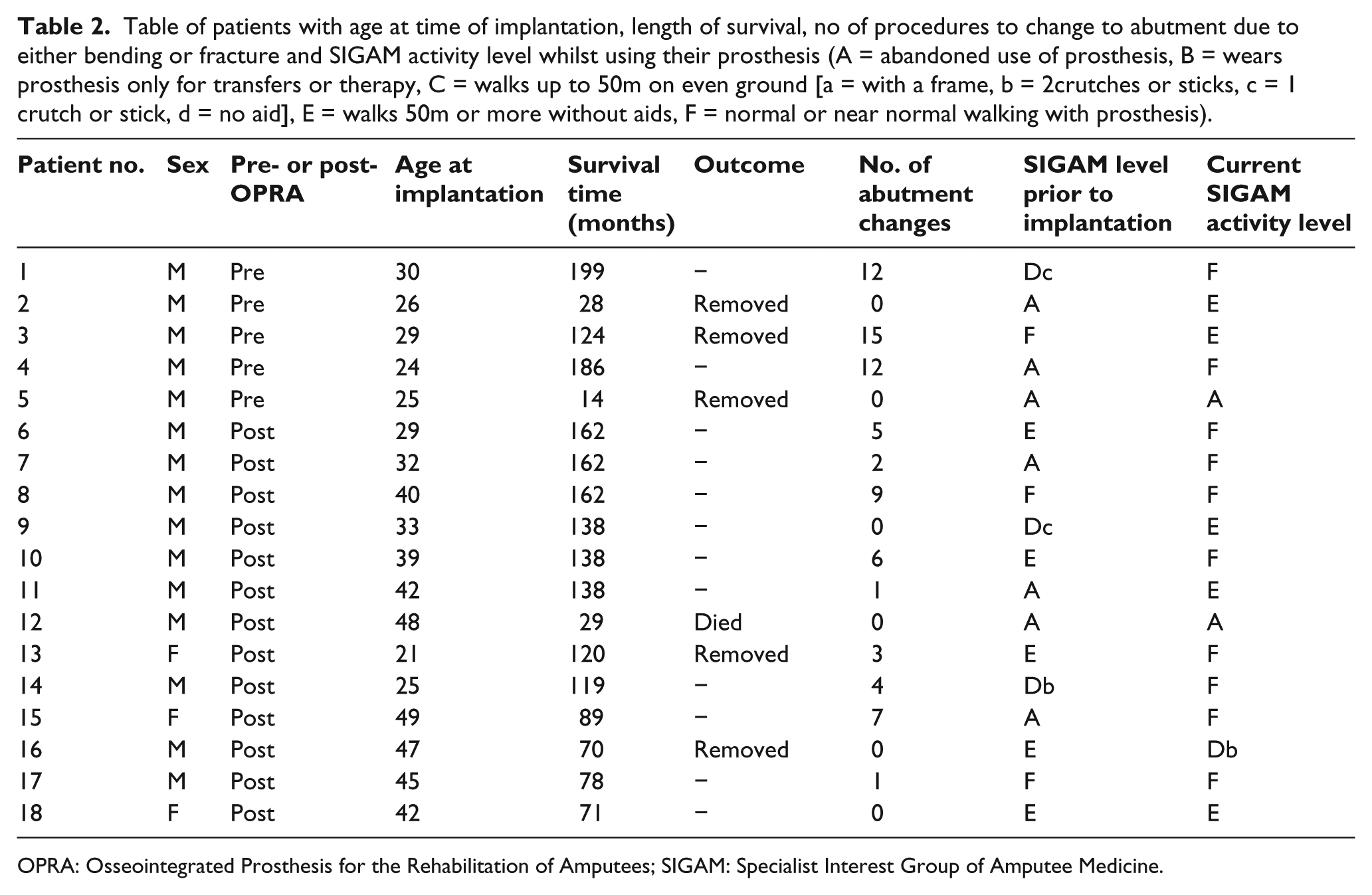
OPRA: Osseointegrated Prosthesis for the Rehabilitation of Amputees; SIGAM: Specialist Interest Group of Amputee Medicine.
Radiographic analysis
Analysis of the post-operative plain radiographs showed a general trend towards distal bone resorption in 15 patients (83% of patients). Of the three patients who did not show any distal bone resorption, one had the implant removed for infection after 14 months, one died of an unrelated cause 2.9 years after implantation and one patient still has the implant in situ 8.5 years after implantation. Six patients (33%) show signs of proximal cortical bone thickening. Twelve patients (66%) have developed radiographic signs of proximal pedestal formation with excess intramedullary cortical bone deposition. Six patients (33%) have near-implant lucency in different areas around the implant. Fourteen patients (78%) have developed a bony spur from the distal medial aspect of the residual femur. Figure 1 shows typical post-operative radiographic appearances, while Figure 2 shows radiographic appearances in keeping with a low-grade infection.
Quality of life data
As per OPRA protocol, SF-36 and Q-TFA scores were analysed pre-operatively (before the first-stage procedure) and at 2 and 5 years post-operatively. These time points were chosen, as patients were not fully independent until 18 months post-surgery for the first-stage procedure due to the graduated recovery programme. Patients who still retain their implant are now at a minimum of 8 years post-surgery; however, a complete data set was only available up to 5 years and therefore, 5-year post-operative figures were used for comparison.
The physical scores, physical functioning and physical component score in the SF-36 improved significantly between pre-operative status and 2 and 5 years post-operative time points. However, scores did not change significantly between 2 and 5 years post-operatively. There was also a significant improvement in reported pain between the pre-operative time point and 2 years post-implantation. However, this significance was not seen at 5 years post-operation. No other improvements reached statistical significance.
Table of p values for the SF-36 scores analysed using Mann Whitney U test normalized to the UK population (* = Significant values). PF = physical functioning, RP = role limitations due to physical health, RE = role limitations due to emotional problems, VT = energy/fatigue, MH = emotional well-being, SF = social functioning, BP = pain, GH = general health, PCS = physical component score, MCS = mental component score.
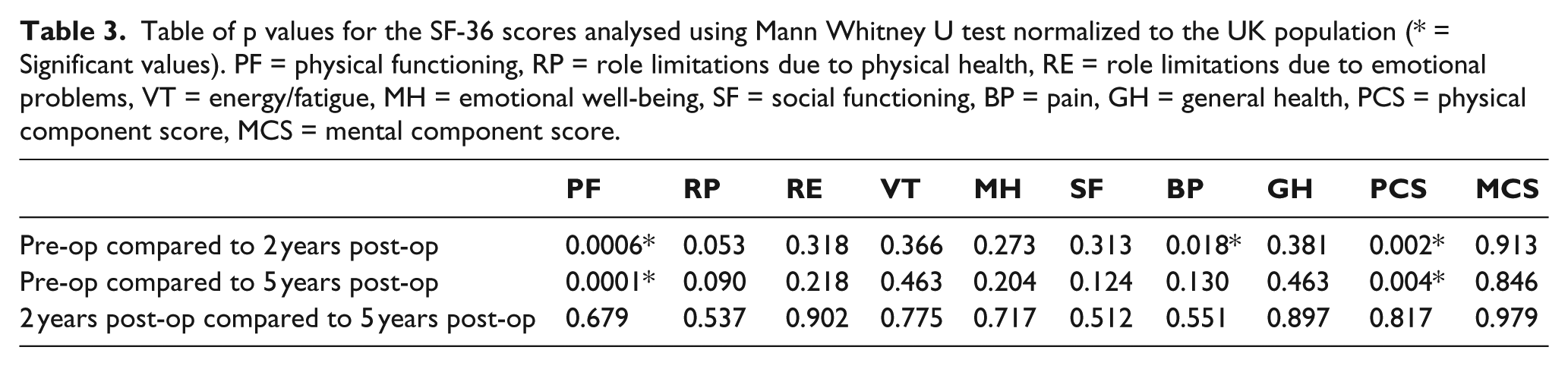
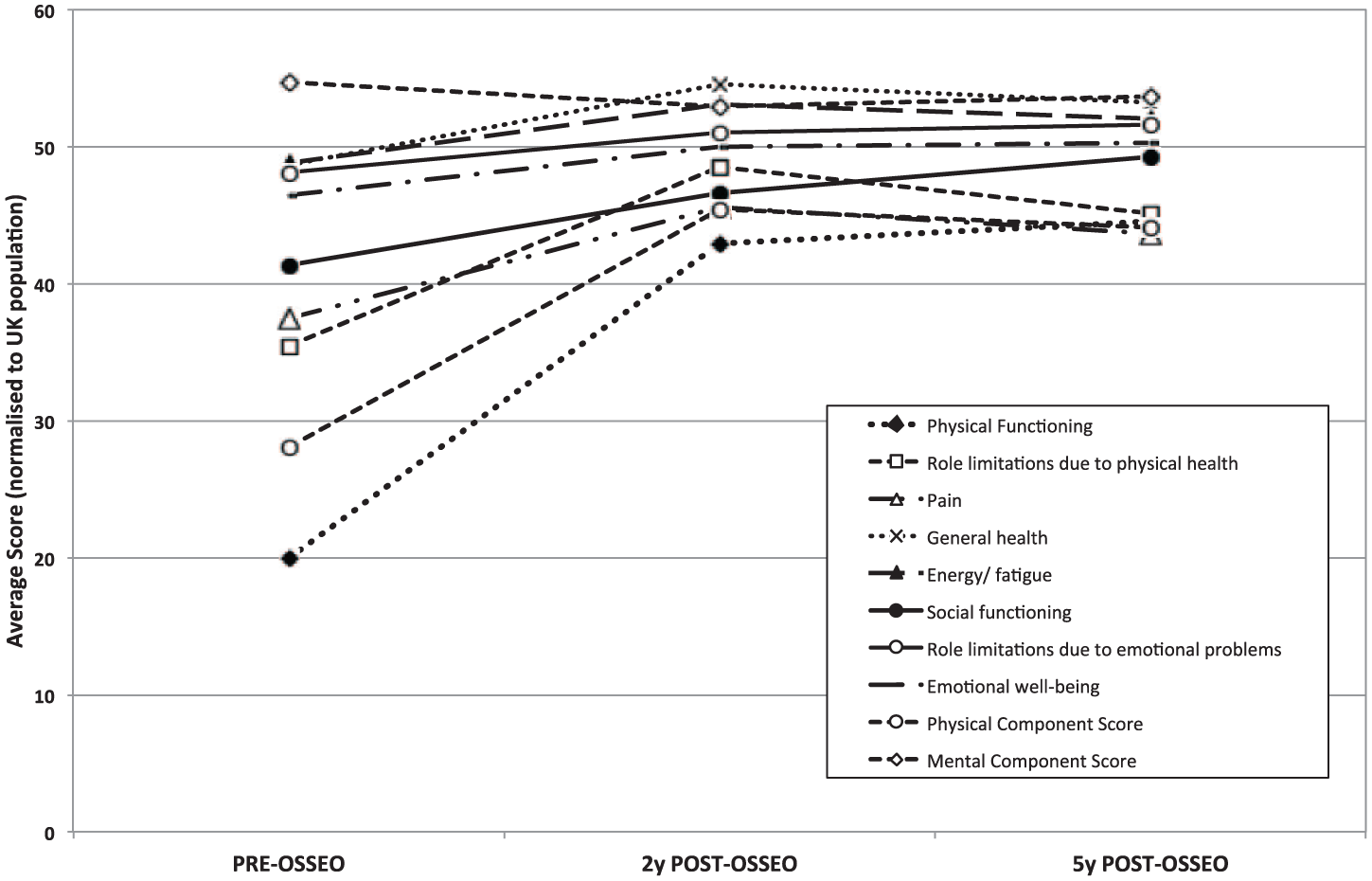
Graph showing changes in mean SF-36 scores pre-operatively, 2 years post-operatively and 5 years post-operatively.
In the Q-TFA, there were significant improvements in all of the main scores, and in two out of the three sub-scores for prosthetic mobility, between the pre-operative period and 2 and 5 years post-implantation. There were no significant differences between 2 years post-surgery and 5 years post-implantation.
Table of p values for the Q-TFA scores analysed using Mann Whitney U test normalized to the UK population (* = Significant values).
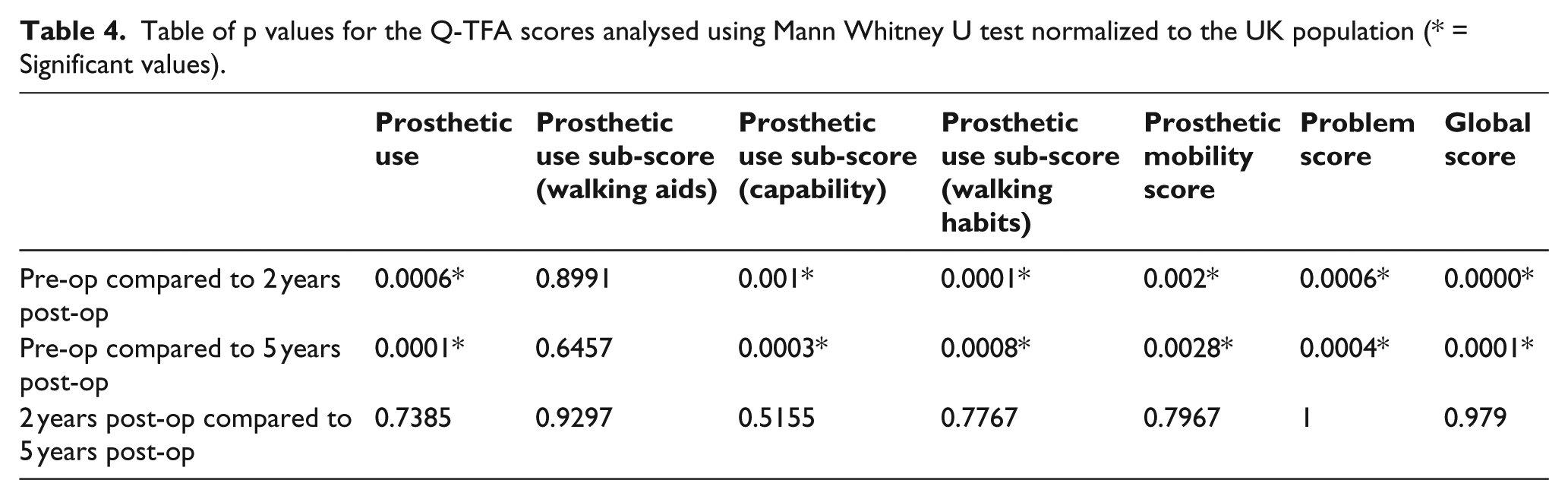
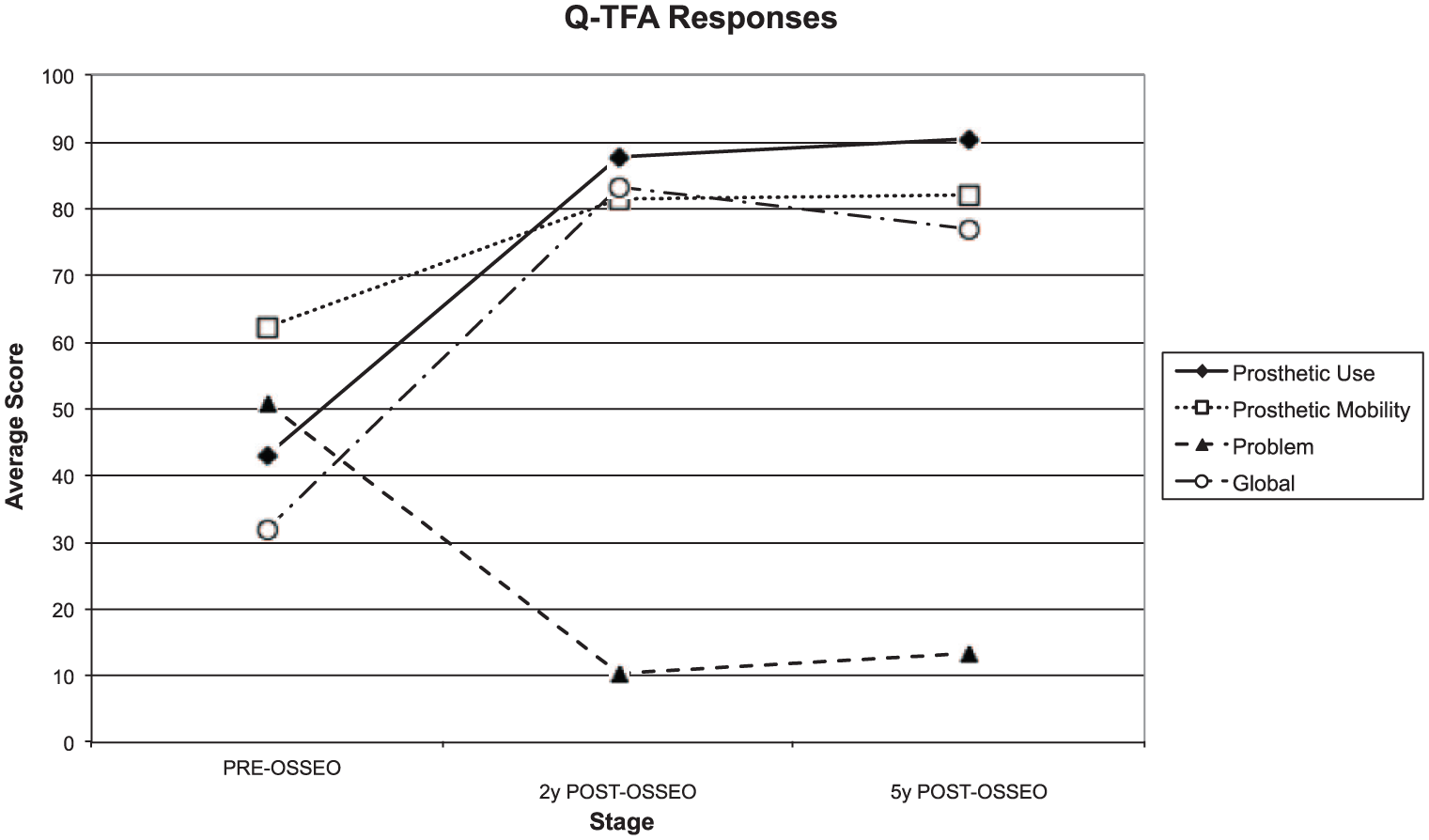
Graph showing changes in mean Q-TFA scores pre-operatively, 2 years post-operatively and 5 years post-operatively.
Discussion
This study shows that the time to full mobility with an external prosthesis attached to the implant was similar in both groups, with a median time of 7 and 9 months in the pre-OPRA and post-OPRA groups, respectively. A review of the literature did not find any specific studies examining the mean time to mobility using traditional prostheses; however, one paper commented that the average time to first attendance at a prosthetic clinic was 3–4 weeks post-amputation. 16 An amputee advice sheet advises that prosthetic fitting usually starts 2–6 months after surgery, it may take ‘several months’ to return to a normal level of function, and that an amputee may need to spend 6 months to a year working with a rehabilitation team. 17 Therefore, the use of the OPRA implant appears similar to traditional prostheses in terms of time taken to become fully mobile post-surgery.
Patients in the pre-OPRA group performed less well than patients in the post-OPRA group, with three out of five implants removed. Overall five implants (27.8%) have been removed in total across both groups, all due to infection, however, only three were known to be infected at the time of removal. Survival was 80.21% at 15.9 years in the post-OPRA group compared to 40% at 18.6 years in the pre-OPRA group; however, due to the low number of patients in both groups, these figures need to be analysed with caution. A review of the literature reporting upon the outcome of similar devices reveals explantation rates between 7.8% (4 out of 51 patients) 2 years after implantation18,19 and 10.8% (4 out of 37 patients) at 1–11 years after implantation, 20 13% (4 out of 30 patients) over 7–16 years following implantation 21 and 20% (20 out of 100 patients) within 3 months to 17.5 years after implantation. 12 Several systematic reviews of the literature have also been published. These report explantation rates of 3%–20%. 22
Across both groups all patients except one have suffered from infection-related problems. While most of these problems relate to superficial wound infections at the skin penetration site, two patients, one in each group, have known peri-implant infection with osteomyelitis being suppressed with antibiotics. However, none of these patients have developed septicaemia or become unwell due to these infections. Normal human skin, mouth and gastrointestinal commensal microbiota have been identified as the causative organisms. With conventional prostheses, soft tissue infections around the residual limb often require the patient to discontinue use of their prosthesis until the infection has been cleared; however, patients with this implant could continue to use their prosthetic limb while waiting for the antibiotics to take effect. Such soft tissue infections may occur at the penetration site due to constant movement of the soft tissue around the abutment, preventing or disrupting soft tissue integration onto the implant thereby allowing a route for bacteria to enter. Methods attempting to prevent this include good hygiene and the use of a silicone soft tissue supports disc placed around the residual limb implant penetration site in an attempt to stabilise the soft tissues. In this series of 18 patients, different cleaning methods for the abutment including the use of HiBiScrub® (Mölnlycke Health Care Limited, Bedfordshire, UK), soap and water, local antibiotic cream or water in isolation were applied daily but this did not appear to alter infection rates. The published literature reveals variable infection rates of 31.9%
23
and 5%–18% at 3 years after implantation.
24
One paper reported a superficial infection rate of 54.9% which were treated with oral antibiotics, but a deep infection rate of 7.8% with 2% removal for infection.
18
Another paper reported further surgery was required for infection in 77% of patients but did not comment on infections treated with antibiotics alone.
21
The authors of this paper did comment that patients who received implants later on in the study period with a modified technique required no further surgery for infection. Brånemark et al. published data looking specifically at the rate of osteomyelitis, which was reported as 20% 10-year cumulative risk, with a 9% 10-year cumulative risk of explantation.
25
This article identified causative organisms as being
Three of the patients in this series who had their implant removed have subsequently travelled to Sweden to have new implants inserted. Access to patients’ records from Sweden was more difficult to obtain and therefore data following the revision surgery has not been included here. It is encouraging that patients with this implant would desire to have new devices re-implanted rather than return to traditional prosthetic use.
Two patients have fallen, fracturing the abutment while five have fractured the abutment-retaining bolt. Three patients had spontaneous fractures of the abutment and abutment-retaining bolt without any trauma. This is regarded as the most serious complication as the prosthetic limb can fall off without warning, potentially leading to more serious injury from a resulting fall. However, it is reassuring that two patients fell sustaining a fractured neck of femur above the implant, without any damage occurring to the implant itself. The incidence of fractured neck of femur in the United Kingdom is 1.6 per 1000 population in women aged 65–69, rising to 32.8 per 1000 in women aged 90–94, while in men it is 0.7 per 1000 and 14 per 1000 for the same age groups. 32 Therefore, the incidence of fractured neck of femur of 11% within this cohort of middle-aged patients is high. The implant may be a risk factor for sustaining such a fracture; however, this is outside of the scope of this article. Other papers have identified fracture rates as 2%, 19 3%, 23 5.4% 20 and 7.8%. 18
There was a high level of patient maintenance required during the observed time period, with a number of patients requiring a minor surgical procedure to replace the abutment under sterile conditions using local anaesthetic. A bent abutment can lead to abutment fracture and thus requires replacement of the damaged portion with relative urgency. Since the last implantation was performed in this study, changes have been made by the manufacturers in an attempt to reduce the chance of fracture or bending of the individual components. These changes include producing the abutment from a stronger titanium alloy instead of pure titanium and producing the abutment-retaining bolt with a carbon coating, in order to reduce friction, thereby increasing the proportion of the 12 N m of torque, used to tighten the screw, which is transmitted into pre-load holding the abutment and fixture together. Other developments include an update to the failsafe device connecting the prosthetic limb to the implant. The new failsafe (Axor) is designed to protect the implant from bending moments as well as minimise torque transmission to the implant (which the previous Rotasafe provided). Similar papers have reported breakage of fail-safe devices in 27.5% of patients 23 and abutment-related complications in 7.8% of patients. 18
Radiographic analysis is in keeping with previous reports27,28 showing cortical remodelling due to stress shielding and periosteal bony resorption around the mid to distal implant as well as proximal periosteal bone thickening. None of these changes appear to have had an impact on the patients’ mobility or progress and there was no correlation with patients’ symptoms or activity levels and the radiographic appearance of the implant. Previous papers suggest this remodelling occurs rapidly after implantation until an equilibrium is achieved wherein the stress–strain environment is adequate to maintain the residual bone. 28
SF-36 and Q-TFA data show a significant improvement in patients’ quality of life, especially related to physical function and use of prosthesis, as well as an improvement in reported pain. Papers by the Brånemark group3,12,18 show similar outcomes. Previous studies utilising the SF-36 to assess lower limb amputees have shown that physical function scores are affected more than mental health scores.29–31
A further perceived benefit of using an osseointegrated implant for amputees is the ability to use it with a short residual limb, which would be unable to support a conventional prosthesis. It has been shown that an osseointegrated device used in the setting of short residual femoral bone allows a normal range of movement in the hip joint when compared to standard socket prostheses. 13
The complications in this study are higher than the reported complications in current literature. A potential reason for this higher complication rate is the relative age of the patients enrolled into this study. Most of the patients in this study were young, 21–49 years of age, when compared to the demographics of patients in other reported studies. In addition, 15 of the enrolled 18 patients were male, employed in manual jobs and returned or attempted to return to these employments following implantation. The majority of the patients in this study were also active smokers. The implantation of devices in this study was performed between 1997 and 2008, during the early development of this technology. As noted in systematic reviews, 26 earlier studies had higher complications rates, an inevitable result of the learning curve associated with new techniques. Given that this study was begun during this early period, it also has one the longest follow-up periods. The cumulative complications incurred over this follow-up period may attribute to the higher complication rates when compared to existing literature.
Limitations of study
This biggest limitation to this study is the low number of patients. The study is further limited by the fact that the first five implants were inserted prior to the formalisation of the OPRA protocol in 1999. This limits the number of patients from 18 to 13 post-OPRA. As well as being a small group of patients, this was also a highly selective group of patients and therefore further study with a wider demographic of patients would need to be performed for external validity.
Conclusion
This study shows that amputee quality of life is significantly improved with the use of an osseointegrated amputation prosthesis. There are known challenges for amputees using traditional prostheses without access to these implants. Therefore, these implants appear to be a good option for some amputees. The main complications from this study were the rate of infection as well as problems associated with the abutment and abutment-retaining screw. As previously mentioned, there have already been improvements made to the construction material of the abutment and abutment-retaining screw. Therefore, if the resistance to infection around these devices could be improved, they may become a reliable option for the management of amputees in the future.
Footnotes
Author contribution
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
