Abstract
Background:
The stump–socket interface is of utmost importance for prosthetic function in transhumeral amputees. Stability of this connection may be improved using a newly designed subcutaneous implant.
Objectives:
The purpose was to determine the effect of the implant together with customized socket designs on the range of motion of the shoulder and the prosthetic function compared to conventional fitting.
Study design:
Case series.
Methods:
The range of motion was measured with scaled metrics and the prosthetic function evaluated with the Southampton Hand Assessment Procedure and the Box and Block Test. Maximal loading was measured in straight and 90° flexion of the elbow.
Results:
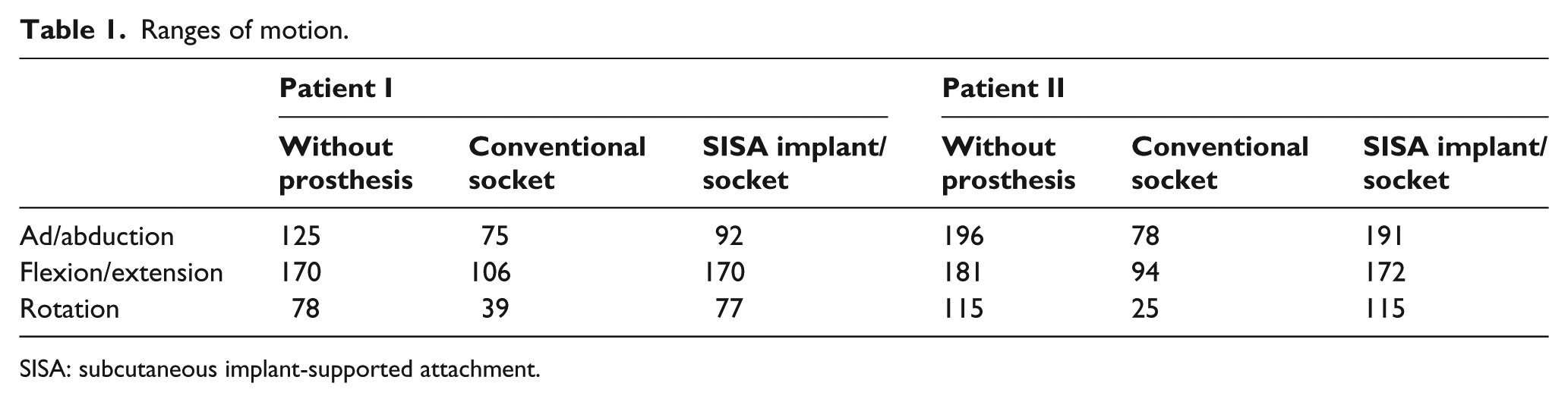
The restriction of range of motion after conventional fitting was decreased from 42.55% ± 6.56% to 9.23% ± 14.89% in Patient I and from 62.18% ± 15.19% to 2.51% ± 2.49% in Patient II using the implant with customized sockets compared to range of motion without prosthesis. Both patients showed improved prosthetic function with the new system compared to conventional fitting.
Conclusion:
The presented subcutaneous humeral implant, together with customized socket designs without straps and harnesses to the contralateral shoulder, can maintain almost complete range of motion of the shoulder. This resulted in improved prosthetic function and comfort for the patient without constant risk of infection.
Clinical relevance
Discomfort and limited prosthetic function are the main reasons for abandonment especially in transhumeral amputees. Shoulder straps and harnesses within conventional socket designs may not only lead to pain and skin irritations at the contralateral shoulder but also limit the range of motion of the shoulder joint and therefore prosthetic function.
Keywords
Background
Prosthetic fitting in transhumeral amputees is challenging. Uncomfortable and unstable socket constructions together with clumsy control mechanisms are responsible for unacceptably high rejection rates with many patients deciding to go without any prosthetic support.1,2 The advent of targeted muscle reinnervation (TMR) 3 and recent developments in prostheses have drawn needed attention to the man–machine interface.4,5 Notably, the functional outcome of prosthetic fitting strongly depends on a stable stump–socket connection.
The lack of distal insertion of the residual muscles and the cone shape of the humeral bone allows the muscle bellies to rotate around the bone in transhumeral amputees. 6 To limit these pseudoarthrotic movements between stump and socket, straps and harnesses to the contralateral shoulder are needed within conventional socket constructions. (Figure 1) These harnesses limit the range of motion of the shoulder joint, can evoke skin irritation, lead to uncomfortable wearing of the prosthesis, as well as myosignal instability resulting in prosthetic malfunction.2,7,8
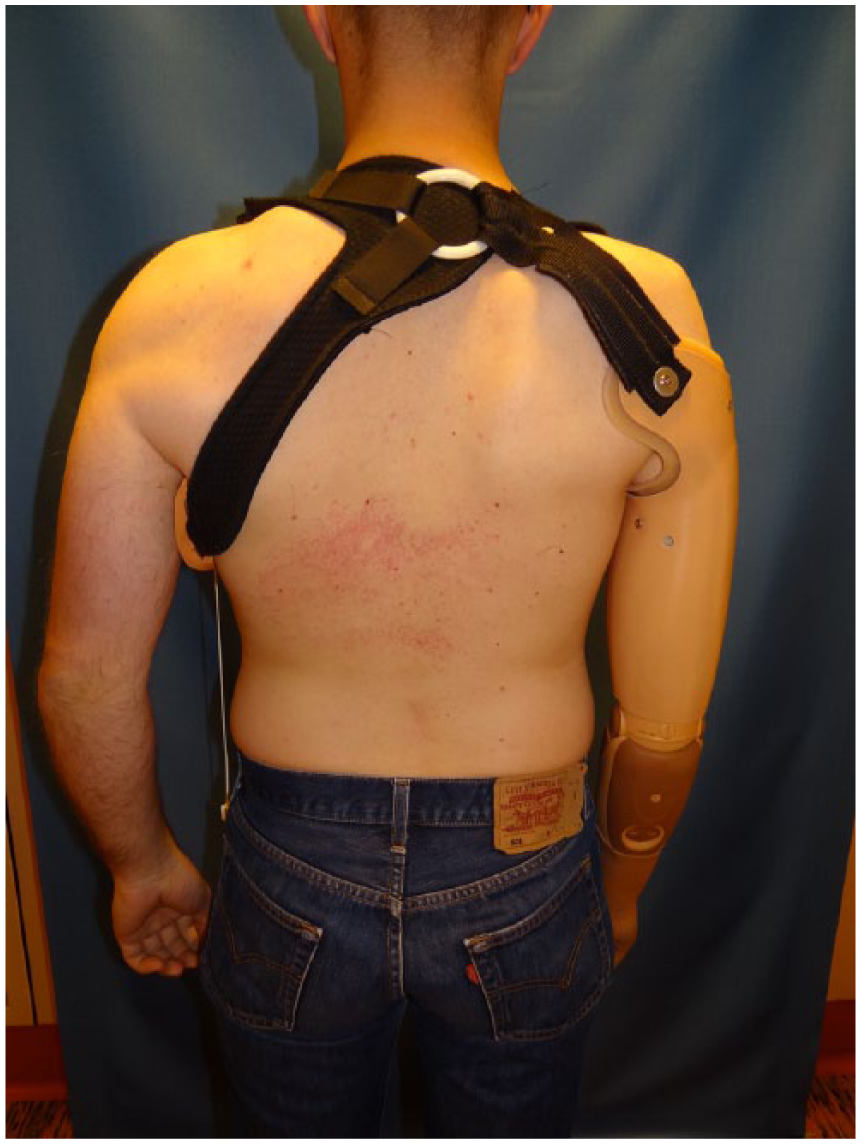
Conventional socket.
The ideal transhumeral stump should enable a reliable and rotationally stable connection to the prosthesis without restricting range of motion in the shoulder joint. To achieve this, different attempts to improve this interface have been made.
In the 1970s, Marquardt and Neff 9 introduced the angulation osteotomy to imitate a condyle-like stump end. With this contoured stump, mechanical coupling is enhanced, resulting in less relative movements and increased range of motion of the shoulder. However, this technique inevitably shortens the remaining limb and the angulated bone segment often straightens within time. 8 Thus, a sufficient length of the humeral bone would be about 50%–70% of the normal length; 10 this technique should not be applied to patients with shorter stumps.
Patients with elbow disarticulation, and therefore intact condyles, offer an amputation stump that aids prosthetic suspension. However, fitting of elbow disarticulated amputees with myoelectric prosthesis results in poor cosmetic and also functional results due to the excessive length of the final fitting.
To avoid shortening of the stump and enhance prosthetic attachment, Witsø et al. 11 invented a T-shaped subcutaneous implant to create artificial humeral condyles. The intercondylar diameter simulated the normal anatomic humeral intercondylar diameter. However, patients reported pain over the new condyles and superficial pressure wounds. 11
Direct prosthetic bone anchorage may minimize socket complications including skin irritations as well as obtain full range of motion in the shoulder joint. 12 However, the required skin penetration of the metal implant involves the constant risk of superficial and also deep infections. 12
To avoid skin penetration and the risk of infection, a modified subcutaneous implant based on the T-shaped implant demonstrated by Witsø et al. has been developed (in collaboration with ImplantCast GmbH, Buxtehude, Germany). The new subcutaneous implant was designed to achieve better force distribution and to resemble the anatomical shape of humeral condyles. Within an experimental clinical pilot study, this newly designed subcutaneous implant called the “subcutaneous implant-supported attachment” (SISA) was tested in two transhumeral amputees. The functional outcomes were studied after creating a new type of socket design, suspended and stabilized by the implant.
Methods
Design of the subcutaneous implant
The idea of the implant was, similar to Witsø, to create artificial humeral condyles without penetrating the skin. Aware of the limitations and complications from the Norwegian group, the new subcutaneous implant was designed to offer better pressure distribution and increased loading surface, while maintaining rotational stability (Figure 2).

SISA system with bony and subcutaneous implant mounted together.
The loading surface for axial tension loading is about 1000 mm2 for the T-shaped implant and could be increased by a factor of 2–3.5 with the butterfly-shaped SISA implant depending on the specific size of the subcutaneous implant. The smallest one has a load bearing surface of about 2000 mm2, the biggest implant of about 3700 mm2.
The surface of the subcutaneous titanium implant was polished to minimize soft tissue irritation and bacterial colonization. To limit relative movements between subcutaneous implant and surrounding tissue, the implant was covered with an adhesive polyester tube (Trevira GmbH, Augsburg, Germany) that was applied intraoperatively.
The intramedullary fixed bony implant is based on the Modular Universal Tumour And Revision System (MUTARS) from ImplantCast GmbH routinely used for oncological reconstructions. 13
The implantable subcutaneous system was designed in a modular fashion with five different sizes of the subcutaneous implant and matching dummies to determine the best size intraoperatively.
According to the regulations of the Austrian Ministry for Healthcare Safety, all implanted parts of the SISA system have passed ex vitro mechanical tests, for example, static loading, fatigue, assembling, and abrasion.
In vitro testing
In a first phase, different concepts of the subcutaneous implant were analyzed “in-silico” using finite element analysis (FEA). These models showed the different pressure distributions at the implant–soft tissue interface for different standardized loading forces. These results were linked with findings from an in vitro stump model that served to test different sockets, together with different implant designs. The purpose of this stump model was to evaluate the reproducibility of the socket by the orthopedic technician as well as the stiffness of the stump–socket interface.
Prior to mechanical testing on a servo-hydraulic machine (Instron, Norwood, MA, USA), an analysis had to be conducted to quantify the forces acting on the subcutaneous implant during heavy-duty activities of daily living (hdADL). For this purpose, a special measurement orthosis was built to record all forces and torques stemming from hdADL such as lifting a heavy suitcase. These data were used to perform strength analyses using FEA as well as the above-mentioned mechanical tests. As the results showed a potential risk of overloading for the subcutaneous implant as well as for the bone while performing some hdADL, there was the need to implement a protecting mechanism in case of overload. This was solved by adjusting the stiffness of the socket’s side-gates as they are able to distort and release in case of overload. As a consequence, we detected a decrease in maximum loading compared to the conventional socket fitting technique during the clinical study.
In vivo testing
Implantation of the system was performed in two human cadavers to test the different surgical tools and complete the individual steps of the procedure prior to implantation in the first patient of the clinical study.
No additional in vivo mechanical tests have been conducted, as the bony implant is frequently used in oncologic prosthesis and previous results have been published by the Norwegian group. 11
Clinical study
The study was reviewed and approved by the institutional review board at the Medical University of Vienna, Austria, and the Austrian Ministry for Healthcare Safety. Two patients gave informed consent and implantation with subsequent prosthetic fitting was performed. The inclusion criteria were minimal length of the humeral bone of 10 cm, sufficient skin and soft tissue quality especially at the distal third of the stump, and healthy bone quality in x-ray.
Patient I
Male, age 50 years, right dominant side amputated through the proximal half of the humerus epiphysis 23 years prior to the study due to a traumatic traffic accident.
Patient II
Male, age 30 years, right non-dominant side amputated at approximately half of normal humerus length 9 months prior to the study due to a working accident with a hydraulic press. At the time of inclusion, both patients used myoelectric arm prosthesis for 5 months and 21 years, respectively, fixed with straps and harnesses around the neck and the contralateral shoulder/axilla. These strap fixations led to pain in the neck region as well as discomfort and skin irritation at the contralateral shoulder.
Soft tissue at the stump region was of good quality in both patients without any split skin grafts or massive scars. Neither of the patients reported any amputation neuroma pain. Prior to implantation, prosthetic function, range of motion of the shoulder with and without the prosthesis, maximal weight loading in straight and 90° flexed position of the prosthetic elbow, as well as pain levels at the stump region (visual analog scale (VAS)) were evaluated. These measures were repeated at least 6 and 18 months after prosthetic fitting with the SISA system.
The range of motion of the shoulder was measured with standardized photographs in a 90° with the patient in front of a scaled metric. Internal and external rotations are measured with the help of a large clamp around the stump and angles were measured at maximum internal, neutral, and maximum external rotation.
Prosthetic function was evaluated with the Southampton Hand Assessment Procedure (SHAP) and the Box and Block Test (BBT). The SHAP is a clinically validated hand function test and was developed to assess the effectiveness of upper limb prostheses. 14 It is made up of eight abstract objects and 14 activities of daily living with each task timed by the participant themselves. Normal hand function is regarded as 100 points in the SHAP. 14 The BBT is made up of a wooden box divided into two compartments, one filled with 100 blocks. The BBT score is equal to the number of cubes transferred from one compartment to another in 1 min. 15
The surgery was performed under general anesthesia in a supine position with the stump circumferentially prepared on a bracer. The present scars were excised and a sharp dissection down to the bone was performed. The distal humeral bone was prepared by an osteotomy to open the intramedullary cavity. The bone marrow was removed and the appropriate intramedullary implant was selected using a dummy template. The bony implant was inserted with a press-fit technique. According to the available amount of tissue, the appropriate size of the subcutaneous implant was chosen again with the help of implant dummies. The definitive subcutaneous implant was then inserted and mounted to the bony implant. The subcutaneous implant was coated by a special adhesive polyester layer (Trevira GmbH, Augsburg, Germany) to reduce movements between the implant and soft tissue and placed under the deep fascia. The patients received intra- and post-operative antibiotic prophylaxis with amoxicillin. After 3 weeks, skin sutures were removed and the patient received a compression garment. The patients were followed up at 1, 3, 4, 6, and 12 months after implantation, and thereafter in yearly intervals. In addition to the implantation in Patient II, TMR was performed to improve prosthetic control. 4
The customized socket was constructed with computer-aided design (CAD). An exact offset of the implant, including the thickness of the subcutaneous layer, was produced and three-dimensional (3D)-printed using selective laser sintering (SLS) technology (Materialise HQ, Leuven, Belgium). This socket was superior to all other attempts regarding the fit, uniform pressure distribution, stiffness, and comfort during donning and doffing. This new socket technology consists of a distal cup with two hinged flaps to cover the subcutaneous implant, a custom-made medial support, and a hinged lateral support. The flaps were covered with soft gel pads for a better pressure distribution and could be opened and closed with sliders for donning and doffing without requiring an application aid (Figure 3). The textile liner in combination with the open framework structure led to a light socket with larger surfaces for skin perspiration resulting in increased comfort for the patient.
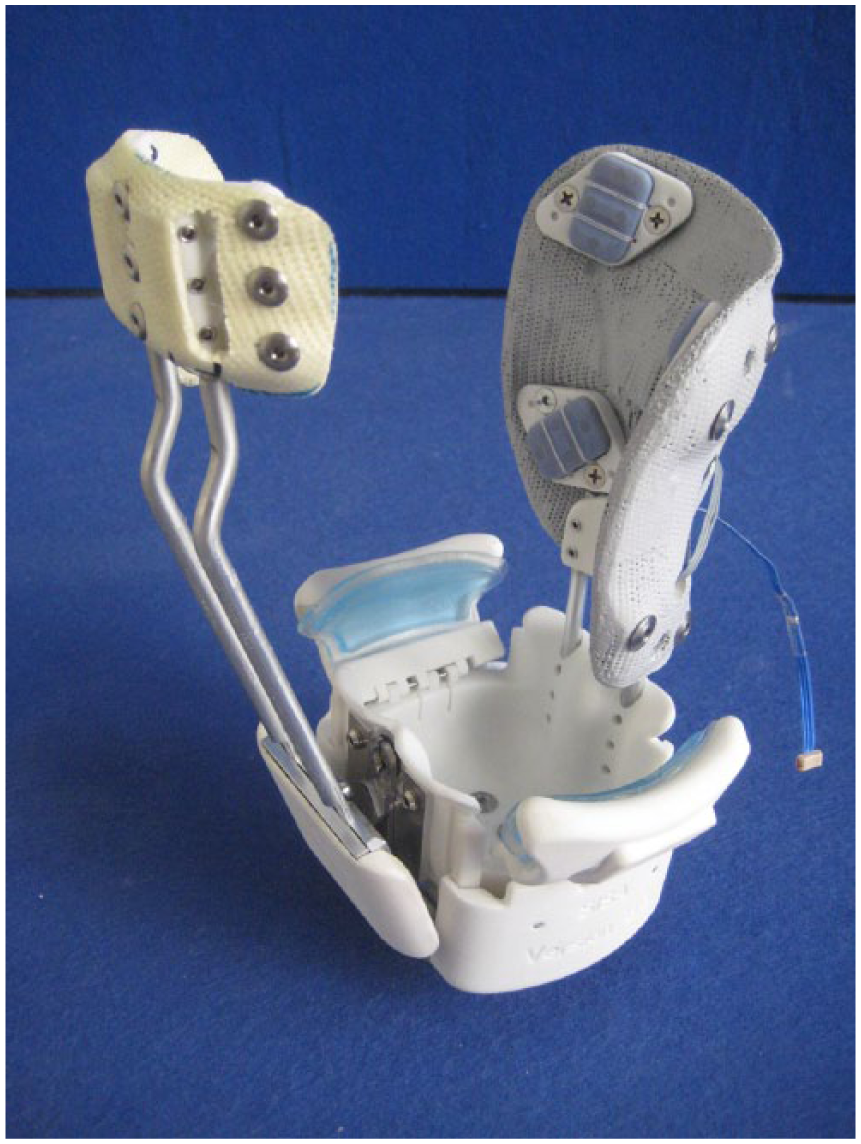
Customized socket.
Results
Clinical study
Both patients received an SISA implant of the same size. The osteotomy of the humerus to open the cavity was performed at 3.5 cm in Patient I and at 1 cm in Patient II, to the distal amputation level. This shortening of the stump is compensated by the height of the subcutaneous implant (2.5 cm). There were no pre- or post-operative complications, apart from seroma formation in Patient II, which resolved after multiple aspirations and compressive garment treatments. Post-operative x-ray of Patient I is shown in Figure 4. The patients were discharged after 5 and 6 days, respectively. Weight bearing training of the implanted system started at 3 months. The first fitting using the implant suspension started at 4 months. Both patients were fitted with myoelectric upper arm prostheses (dynamic arm, electric wrist rotator, sensor hand speed; all from Otto Bock HealthCare GmbH, Germany).
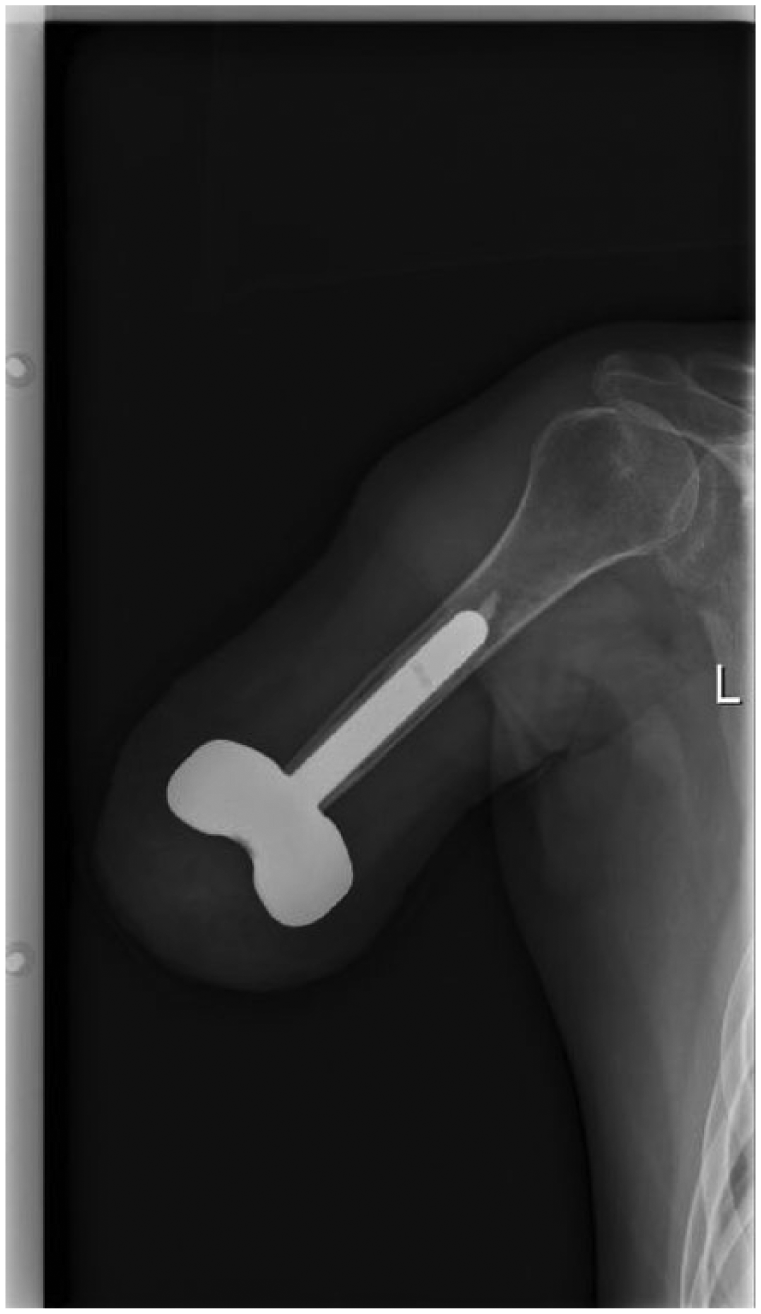
Post-operative x-ray Patient I.
There were no signs of fixture or implant loosening in both patients within 3 and 4 years after implantation. There were no pressure wounds over the artificial condyles. In Patient II, the wearing time has been measured with pressure sensors integrated into the socket for a period of 5 weeks. The average wearing time within these 5 weeks was 9.5 h, with a maximum of 14.5 h. In comparison, with conventional fitting, Patient II reported a wearing time of about 6 h/day.
The ranges of motion without any prosthesis, pre-operative with a conventional socket, and post-operative with the customized SISA socket are summarized in Table 1. Compared to the range of motion without a prosthesis, Patient I showed a mean restriction in the different axis of shoulder motions of 42.55% ± 6.56% with the conventional socket. Wearing the customized socket using the SISA implant as an anchorage, the range of motion was only restricted to a mean of 9.23% ± 14.89% compared to the pre-operative measurements without prosthesis.
Ranges of motion.
SISA: subcutaneous implant-supported attachment.
Patient II showed a mean restriction in the different shoulder motions of 62.18% ± 15.19% with the conventional socket, compared to 2.51% ± 2.49% with the SISA implant and the customized socket (Figure 5(a)–(c)).
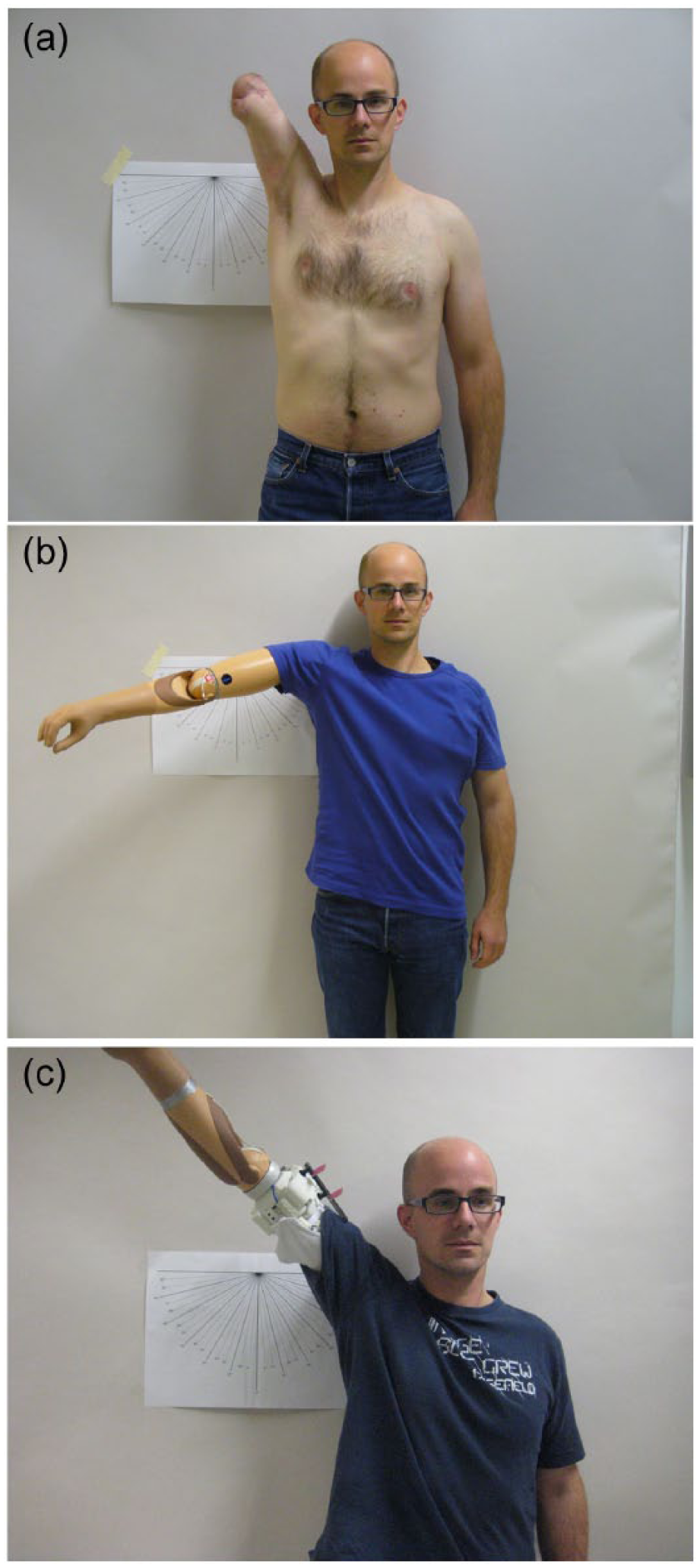
(a) ROM without a prosthesis, (b) ROM with conventional socket, and (c) ROM with SISA system.
Functional outcome scores of SHAP and BBT as well as maximal weight loading in neutral extended and 90° flexion of the prosthetic elbow are summarized in Table 2.
Functional outcomes.
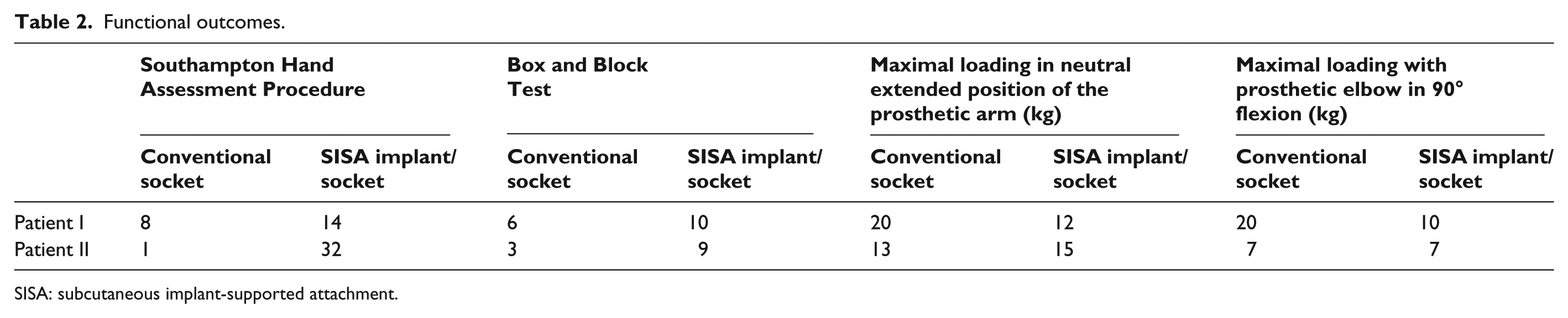
SISA: subcutaneous implant-supported attachment.
Patient I reported mild pain (VAS 3) at his stump prior to implantation. This pain level decreased to VAS 0 at 12 months post-operatively. Patient II reported no pain (VAS 0) at the stump region either before nor 12 months after implantation. During the first months after final prosthetic fitting, both patients reported moderate pain (VAS 4) while wearing the prosthesis for several hours. However, both patients and their stumps adapted to the new loading forces as well as both patients intuitively evolved habitual pressure-releasing movements such as folding hands above their head or crossing arms. Two years after implantation, both patients reported no pain at the stump while wearing their prosthesis on a daily basis.
Discussion
Function and comfort are the most important factors for successful prosthesis use, both from amputees’ and from prosthetic experts’ perspective. 16 As shown in this study, range of motion of the shoulder joint is hardly restricted with the use of the SISA implant together with individualized socket designs compared to conventional fittings. As positioning of the prosthesis in 3D space is mandatory in fulfilling activities of daily living, patients reported overall improved prosthetic function and comfort, as straps and harnesses were no longer needed. Additionally, using the novel socket design, patients do not need to undress to apply the prosthetic device, as this can be done while wearing a shirt.
Most upper limb amputations are due to trauma. 17 In such cases, skin closure is often achieved by shortening of the humeral bone resulting in a short stump and reduced soft tissue quality. As proposed by Witsø et al., 11 the use of tissue expanders within a two-stage procedure to enlarge the soft tissue envelope to make room for a subcutaneous implant is not an option from our point of view. Tissue expansion will lead to even poorer skin quality, whereas the soft tissue quality is of major importance for the success of such a subcutaneous implant to support prosthetic attachments. We propose using such subcutaneous implants only in patients with excessive soft tissue of good quality at the distal stump end, or patients where a shortening of a long residual humeral bone is acceptable.
The skin at the upper arm is not used and also not built for pressure loading, as is the case in such SISAs. Although the patients reported discomfort while wearing the prosthesis for several hours without the chance of releasing the pressure on the artificial condyles, both patients definitely did not want to revert to a conventional prosthetic attachment. Thus, to avoid pain on the artificial condyles, both patients intuitively evolved habitual pressure-releasing movements such as folding hands above their head or crossing arms. Additionally, the patients had the possibility, for example, in sitting position, to loosen a side-gate of the socket to release pressure while the prosthesis is not needed for heavy loading or precise movements. With time, the skin adapted to the new circumstances and painless wearing time of the prosthesis increased constantly within the first 2 years.
Although osseointegration with a skin-penetrating metal implant would achieve a more stable stump–prosthesis interface, as well as the best possible range of motion in the shoulder joint, the constant risk of infection limits its indication. Within a 5-year follow-up in the most experienced center for osseointegration of transhumeral amputees, 38% of the patients revealed at least one superficial infection with a cumulative implant survival rate of 80% at 5 years. 12 Moreover, this concept involves a two-stage surgical procedure. 12
The non-existing sensory feedback represents one of the major reasons for abandonment of current prosthetic devices. 18 However, this limitation may be compensated to some extent by osseoperception. 19 The principle of osseoperception necessitates at best a direct prosthesis–bone interface. Our patients reported an ability to identify various sensations through their prosthesis. As both patients had a fully sensate distal stump end and minimal soft tissue around the subcutaneous implant, mechanical stimuli can be translated onto the humeral bone. Although not as good as in directly bone anchored prosthesis, the phenomenon of osseoperception improved prosthetic function as well as bodily integration of the prosthesis in our patients.
In the modern era of TMR surgery with up to six myoelectric sites, the mechanical interface between stump and socket becomes even more important. As signals are currently recorded via transcutaneous surface electrodes, the position of these sensors must be very stable, as relative movements of the socket could not only result in vague guidance of the device but also severe malfunction due to signal loss. Improving this stump–socket interface will not only improve prosthetic function but also hopefully decrease the high rejection rates of expensive prosthetic devices in upper limb amputees.
Conclusion
The presented subcutaneous implant-supported prosthetic attachment together with customized socket designs can maintain almost the full range of motion of the shoulder joint. However, patient selection with regard to their activities of daily living, functional expectations, and stump conditions is crucial. Although the design of the implant was modified compared to previous studies by Witsø et al., the skin surrounding the implant still represents the limitation of the concept of a subcutaneous prosthetic anchorage. Future technologies such as implantable myoelectric sensors will further improve the biotechnological interface and again change the indication for certain surgical modifications of the transhumeral stump.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
