Abstract
Background:
The most crucial aspect of a prosthesis is the socket, as it will directly determine gait stability and quality. The current standard of care ischial ramus containment socket is reported to increase coronal stability through gait; however, socket discomfort is the primary complaint among prosthetic users.
Objectives:
The purpose of this study is to compare ischial ramus containment to alternatives in the transfemoral amputee population. All subjects were fit with three different sockets: traditional ischial ramus containment, a dynamic socket, and a sub-ischial. In this study, authors hypothesized socket skeletal motion would be equivalent across interventions.
Study Design:
Single-blind, repeated-measures, three-period randomized crossover clinical trial.
Methods:
Outcome measures were socket comfort score and skeletal motion, viewed coronally with X-ray measuring the position of the skeleton in relationship to the socket in full weight-bearing and full un-loading.
Results:
The mean age was 38.2 and mean Amputee Mobility Predictor score was 40. Mean vertical movement, horizontal movement, single limb prosthetic stance, mean femoral adduction in swing and stance, and median socket comfort score were not statistically different.
Conclusion:
The socket design did not significantly effect skeletal motion and socket comfort. All socket designs are suitable depending on the patient-centric preferences and prosthetist skill set.
Clinical relevance
The comfort of the standard of care transfemoral amputation socket has been widely reported as problematic. A comparison of alternative designs in a controlled clinical trial environment will assist the clinician in understanding the impact of design regarding skeletal motion and comfort. Users could benefit from alternatives applied in clinical practice.
Keywords
Background
There were an estimated 1.6 million persons living with limb loss in the United States, in the year 2005, with 19% to 21% of those individuals living with a transfemoral amputation (TFA).1,2 However, there should be awareness of the uncertainty in estimates of long-term amputation prevalence. 2 The prosthetic socket reportedly causes the majority of issues experienced with prosthetic use. The most highly reported problems include pain, discomfort, and dermatological conditions secondary to the socket design or improper fit.3–6 An uncomfortable socket has been deemed the primary complaint among prosthetic users.7–9 The socket interface is the most crucial aspect of the prosthesis, as it stabilizes the pelvis during stance, provides the user prosthetic control, and influences how the components will function. Despite the known problems reported, standards in socket design have largely remained stagnant. The predominant feature of the current standard of care in the United States, ischial ramus containment (IRC) socket, is incorporation of a boney lock counterforce between the femoral shaft and the ischium with a high medial and lateral socket trim line. While alternatives are clinically available and reports have demonstrated the benefits of these alternative socket design to increase patient comfort and performance, the IRC socket style has remained the standard socket design for decades.6,10–14
The advent and incorporation of newer approaches, such as vacuum, liner alternatives, flexible interfaces and advanced rigid frame designs, these sockets aim to improve outcome over the traditional IRC socket. One alternative is the dynamic IRC socket (dynamic socket (DS)), which utilizes a flexible interface to contain the soft tissue, while removing a large portion of the rigid frame by lowering trim lines and creating windows to allow muscle contraction and expansion. The remaining rigid frame is strategically placed at areas crucial to weight bearing and control. 15 This could improve comfort, range of motion and decrease skin issues. By incorporating higher trim lines, both the IRC socket and DS reportedly assist in coronal stability during the gait cycle.6,10,16 However, recent research has demonstrated that transfemoral sockets without ischial containment can still possess coronal stability via soft tissue compression and vacuum, instead of through a boney lock. Subsequently, the transferred control allows for lower socket trim lines.6,10,16 Resultantly, this eliminates the most proximal aspect of the socket which can lead to increased hip range of motion and overall comfort of the socket. Gait and balance in this sub-ischial (Sub-I) socket design have demonstrated equivalence to IRC, without any indications of coronal plane instability.6,10,16
Coronal plane stability is essential for optimal gait and socket comfort. Skeletal motion and the limb to socket relationship influence gait and socket comfort. Horizontal (lateral shifting) and vertical displacement (pistoning) of the transfemoral interface can cause discomfort, pressure, and instability in gait. An anatomically abnormal femoral adduction angle can be associated with gait deviations and problematic distal femoral pressure. Interface design strategies vary in design principles, trim lines, materials, enclosed surface area, and suspension mechanisms. There is currently inadequate data substantiating the comparative effects of the IRC, DS, and Sub-I socket designs. Socket choice for a patient has traditionally been anecdotally based on multiple factors such as cost, time, prosthetist preference or skill set, and tradition and user past experience. Therefore, the purpose of this randomized clinical trial was to compare three different transfemoral socket interface designs regarding skeletal motion and socket comfort in the TFA population. In this randomized controlled trial, authors hypothesized socket skeletal motion and comfort would be equivalent across interventions, confirming the use of alternative sockets designs may improve outcomes for the transfemoral amputee population.
Methods
The protocol was approved by the University of South Florida’s Institutional Review Board and the US Army’s Human Research Protections Office. The study was also registered on clinicaltrials.gov (clinicaltrials.gov identifier #NCT02773056). Inclusion criteria were adult unilateral transfemoral or knee disarticulation amputees with body weight between 45 and 125 kg of any race or nationality, ages 18–60 years who spoke either English or Spanish with the ability to independently ambulate at a community level and self-reported ability to walk for 20 min consecutively. Definitive prosthetic use of at least 1 year was required. Participants needed to be able to tolerate alternative interface designs from their existing design and be free of skin issues, as defined by the study protocol. Exclusion criteria included bilateral amputees, amputation level other than TFA or Knee Disarticulation (KD), those requiring an assistive device to ambulate the presence of skin issues, being out of the age range and known cognitive impairment.
The study design was a single-blind, repeated-measures, three-period randomized crossover clinical trial. In addition to rater being blinded (i.e. single blinding) by the subjects being instructed to wear loose shorts or pants which fully concealed the sockets, the study statistician was also blinded. Recruitment was from a convenience sample of multiple clinics willing to participate in the southeast United States. Following written informed consent, sociodemographic and anthropometric data were collected from 15 participants. The clinical trial start date was 6 June 2016. All subjects were separately hand casted and fit with three different sockets by the same study prosthetist for a traditional IRC brim, a DS, and a Sub-I interface. To eliminate confounding, the previously prescribed and accommodated knee and foot, tested at base line, remained consistent through all testing. The same gel liner (FSL TPE; ALPs, St. Petersburg, FL. USA.), suspension (Echo Sock; Ossur, Reykjavik, Iceland), and electronic pumps (V3; Otto Bock. Duderstadt, Germany) were utilized. All static alignments were set in 9° of adduction and bisected the proximal brim to set the outset, in accordance of Long’s 17 line. An offsite computer random number generator was used to randomly allocate the order of fitting to either (1) IRC, (2) DS, or (3) Sub-I. Participants were instructed on self-care, interface component and skin hygiene and skin inspection prior to initiating community use and accommodation of the experimental sockets. Participants determined when they felt comfortable enough to be tested using the randomly assigned interfaces (i.e. self-accommodation). The subjects guided the prosthetist as to comfort, final trim lines, and final dynamic alignment. When the subject was ready for testing, data collection for all outcome measures was scheduled and completed by a rater blinded to the interface type. Following each data collection, participants were then fit into the next randomized interface design. The process was repeated for the third time (Figure 1).

Casting and finished laminated sockets. Two separate hand casts were taken, one plaster cast for the IRC and DS sockets and one fiberglass cast for the Sub-I. The IRC/DS cast was a narrow medial lateral casting and the Sub-I was more symmetrically round. The DS sockets were designed to eliminate 50% of the rigid frame surface area. This included an anterior and posterior window as well as lowered proximal trim-lines.
Outcome measures
Skeletal and socket measures
First, the proximal trimlines of the rigid frame were lined with soldering iron. The subjects wore their prosthesis for at least 2 h and did not doff their sockets prior to X-ray. An experienced X-ray technician guided the subjects to properly position them during the process. To determine the amount of vertical interface movement (pistoning), lateral shifting, and adduction angle of the femur, two different coronal X-rays were taken of the subjects in single-limb weight bearing on (1) the prosthetic side and (2) the contralateral side, in each of the three sockets. For the purpose of this study, socket movement was defined as the numerical difference (mm) of interface to skeletal movement between simulated stance (prosthetic stance) and swing (contralateral stance) as measured by coronal X-Ray. Vertical movement was measured from the distal-most aspect of the ischial tuberosity (IT) to the proximal most aspect on the medial socket (cm). The difference between prosthetic stance and prosthetic swing was measured to determine the amount of pistoning (cm). The horizontal socket skeletal relative position was measured from the medial most aspect of the obturator foramen to the most medial aspect of the socket to determine lateral shifting (mm). All X-ray measurements were supervised by an orthopedic surgeon using a radiology software program (Osirix, Bernex, Switzerland; Figures 2 and 3).
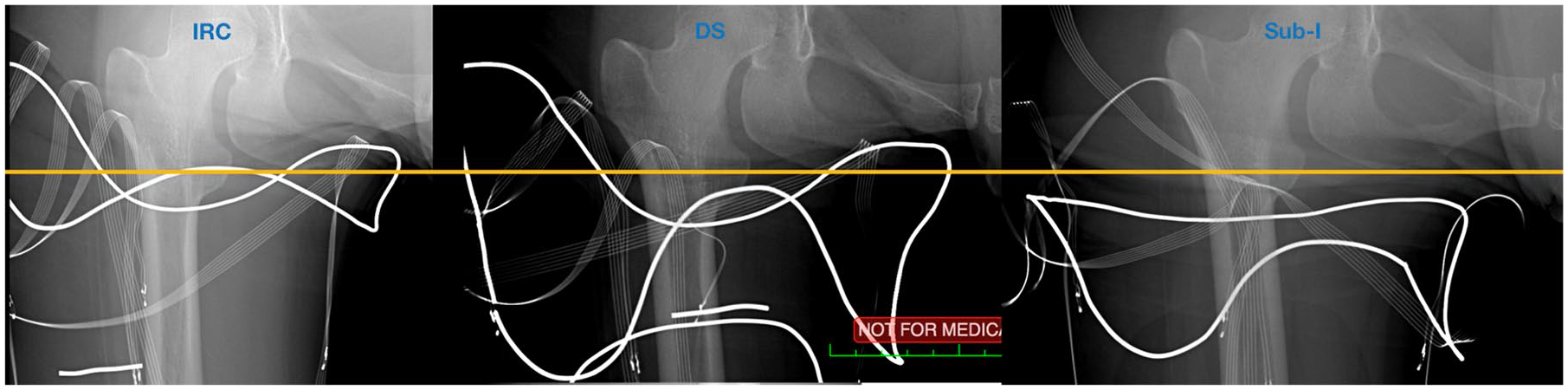
X-ray panel of socket designs on the same subject showing ischial to socket relationship. The horizontal line represents the distal aspect of the ischial tuberosity equally aligned across the X-rays of the three socket designs. The rigid frame trim-lines are lined with soldering iron.
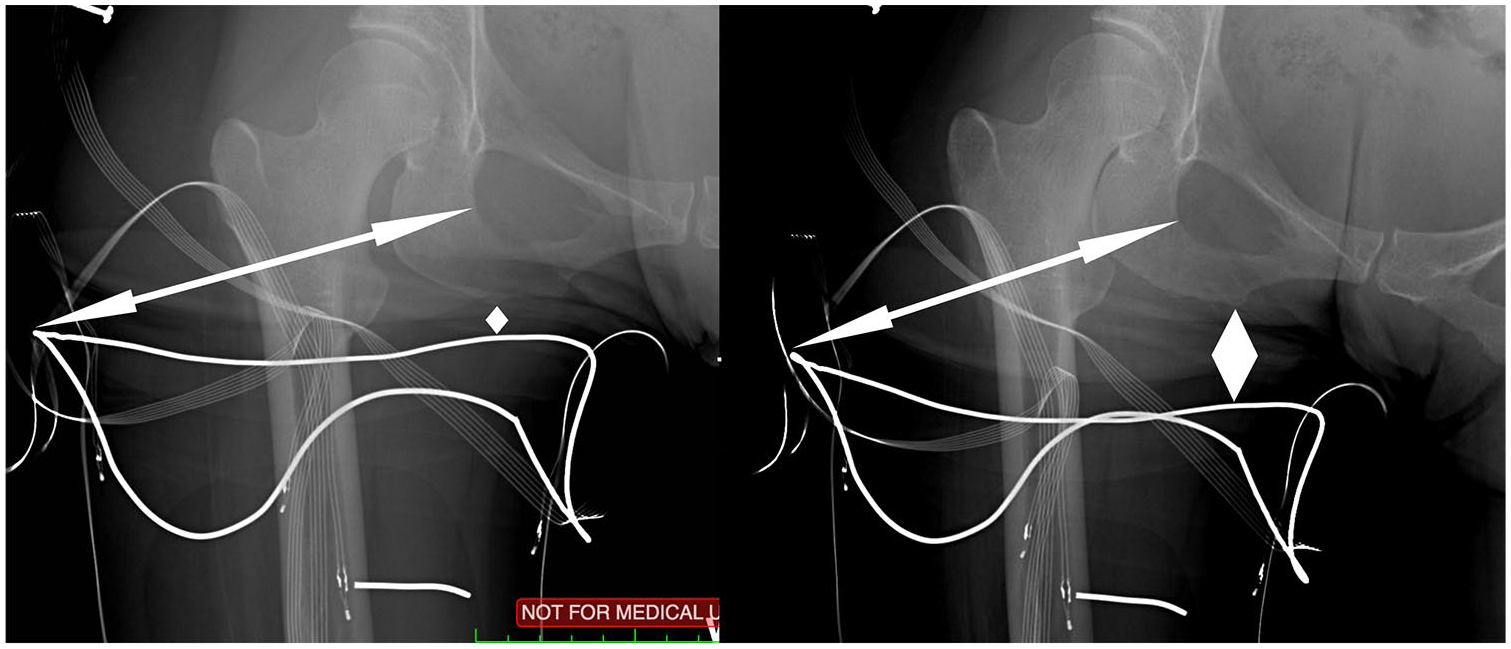
Same subject wearing Sub-I socket in weight bearing (left) and non-weightbearing (right). The distance from the lateral most aspect of the obturator foramen to the lateral most aspect of the socket was measured and compared with determine horizontal movement, represented by the arrow. The distance from distal ischial tuberosity to the most proximal aspect of the medial socket was measured and compared to determine vertical movement (pistoning), represented by the diamond shape.
Comfort
Comfort was determined following the self-reported accommodation period. The socket comfort score (SCS) was used to quantify comfort and has demonstrated validity between practitioner and patient observations as well as observation in the need for interface adjustments. 18 The SCS is sensitive to change, has a minimal detectable change of 2.73, and is reliable in terms of repeated measures observed between 20 and 60 min.18,19 Subjects’ responses on the 11-point (0–10, 10 is the socket score related to highest comfort) scale were recorded for each design and compared.
Statistical analyses
Sample size and power calculations were based on effect sizes calculated from performance outcomes (i.e. spatiotemporal walking parameters, balance, perceptive measures, and interface pressures) previously published regarding comparable interface comparisons in basic mobility tasks.6,10 These estimates indicated that 12 TFA subjects would adequately power the study with
Following all assessments, data were entered into a database and verified prior to analysis. Subjects’ performance data within a given condition (e.g. dynamic socket) were averaged (and variance calculated) to represent that condition for further analysis and comparison across conditions. Descriptive statistics were calculated (i.e. means, standard deviations) where possible. Continuous data (i.e. pistoning) were examined for normalcy and outliers using NCSS/PASS’s omnibus calculation of skewness and kurtosis (2004 edition; NCSS, Kaysville, Utah). A within subjects’ repeated measures analysis of variance model with post hoc tests (i.e. Pillai’s trace) were used to reveal statistical differences in performance between prosthetic socket conditions (dependent comparisons) when data were normally distributed. Ordinal data (i.e. socket comfort) and abnormally distributed data were compared using equivalent non-parametric statistical tests (i.e. Friedman’s Test). The protocol’s
Results
Demographics
Fifteen subjects were recruited and consented. Four subjects were female. The mean age was 38.2 ± 10.4 years (25–52 years), mean residual limb length was 29.6 ± 6.2 cm (18.4–42.0 cm), mean percent of the sound residual limb femur was 81.2% ± 16.7% (47.7%–105.6%) of the sound side femur length. Subjects mean body mass was 82.1 ± 20.0 kg and mean body mass index (BMI) was 26.2 (16.7–32.9). Subjects’ mean time with an amputation was 14.4 ± 12.1 years (2–37 years) and mean Amputee Mobility Predictor score was 40 ± 4 (33–44). Prior to the study, subjects used a variety of suspension systems; four subjects wore a traditional IRC, three subjects wore a DS, and eight subjects wore a Sub-I socket design. Following fitting and prior to data collection, two subjects withdrew citing intolerance of the liner, leaving
Demographics Table.
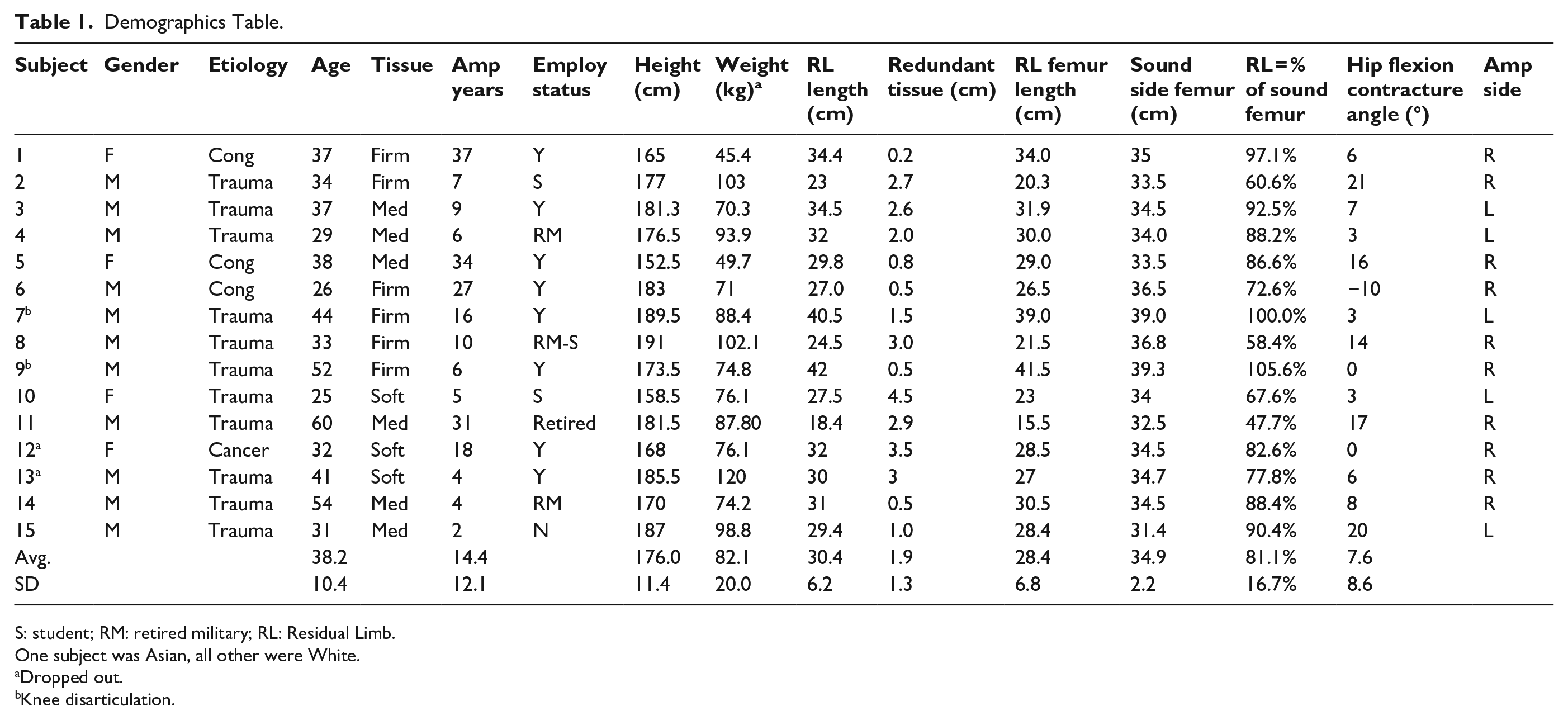
S: student; RM: retired military; RL: Residual Limb.
One subject was Asian, all other were White.
Dropped out.
Knee disarticulation.
Socket position and movement
The mean medial wall height in relationship to the IT on the IRC sockets was 0.99 ± 1.7 cm (3.3 cm distal–1.9 cm proximal) distal to the IT, on the DS sockets was 0.57 ± 1.9 cm (4.3 cm distal–1.3 cm proximal) distal, and on the Sub-I sockets was 3.93 ± 2.9 cm (1.0–9.7 cm) distal. From these measures, the medial proximal socket trim-line was significantly farther distally from the IT on the Sub-I socket compared with both IRC and DS alternatives (
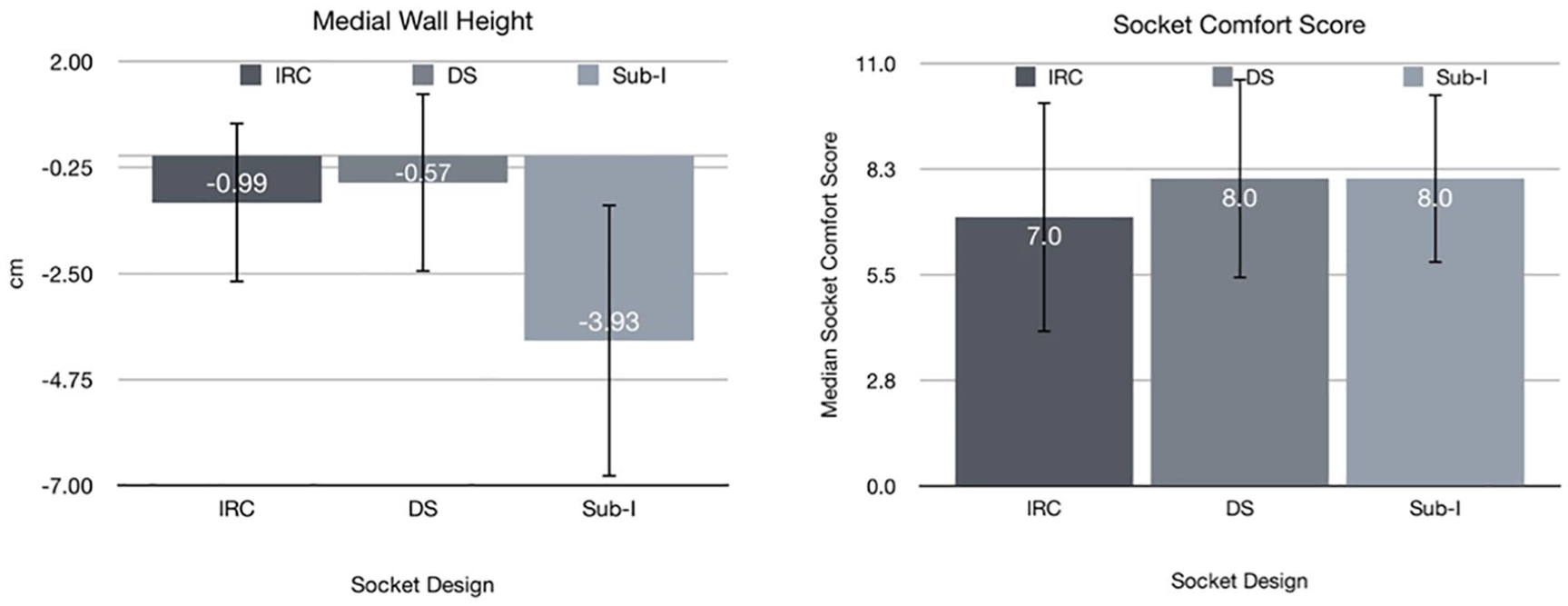
Medial wall height and socket comfort score. The mean medial wall height (ischial containment) of the rigid frame was fit to the comfort of the subject and did not extend proximal to the ischial tuberosity in this clinical trial. However, all sockets had flexible interfaces which extended at least 2 cm proximal in relation to the rigid frame.
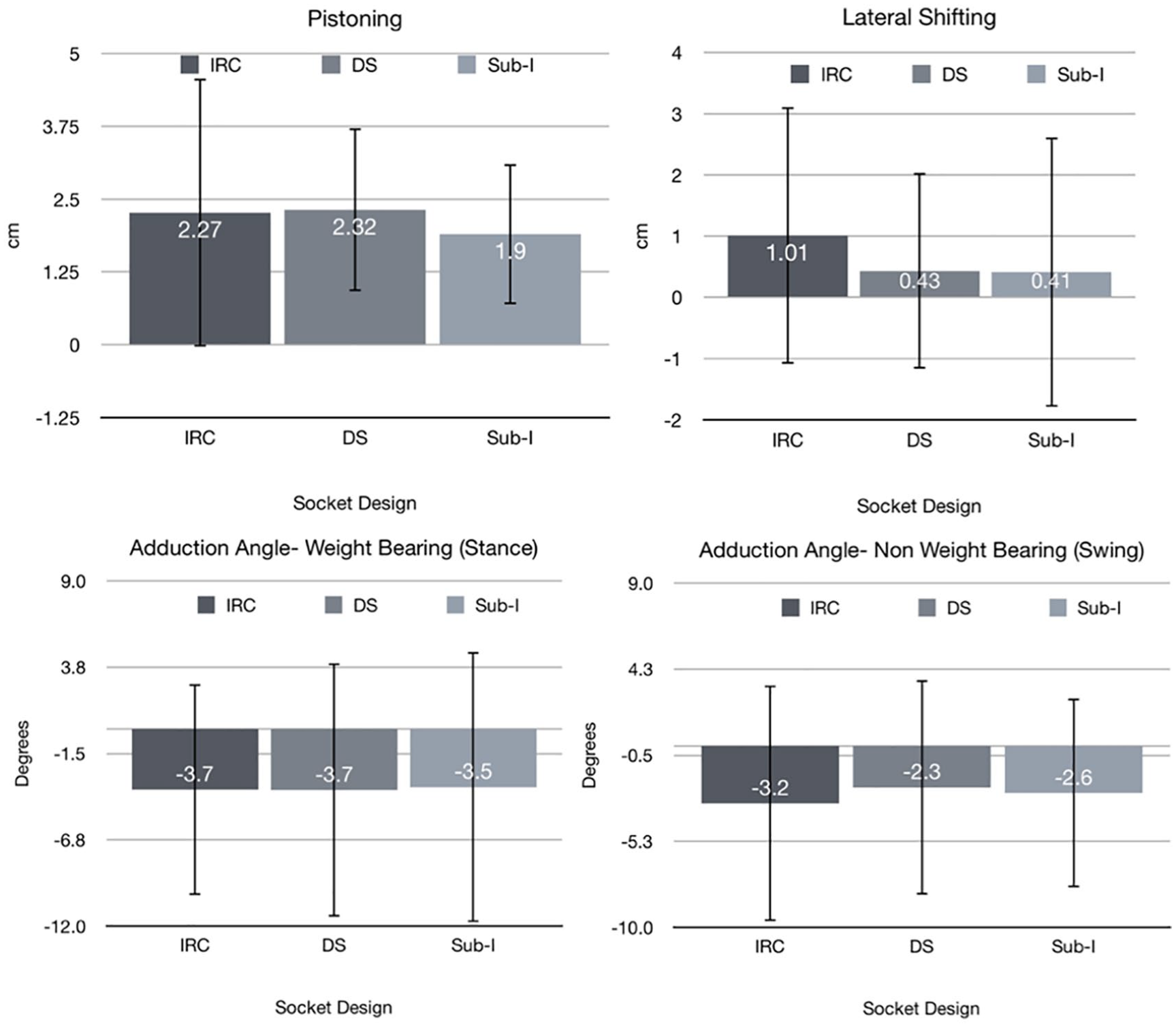
Skeletal motion; pistoning, lateral shifting and hip angle. The coronal femoral angle (adduction angle) was measured in weight bearing (stance) and non-weight-bearing (swing). The mean femoral angle did not achieve adduction, rather abduction is represented with a negative number.
Degrees of Freedom (DOF),
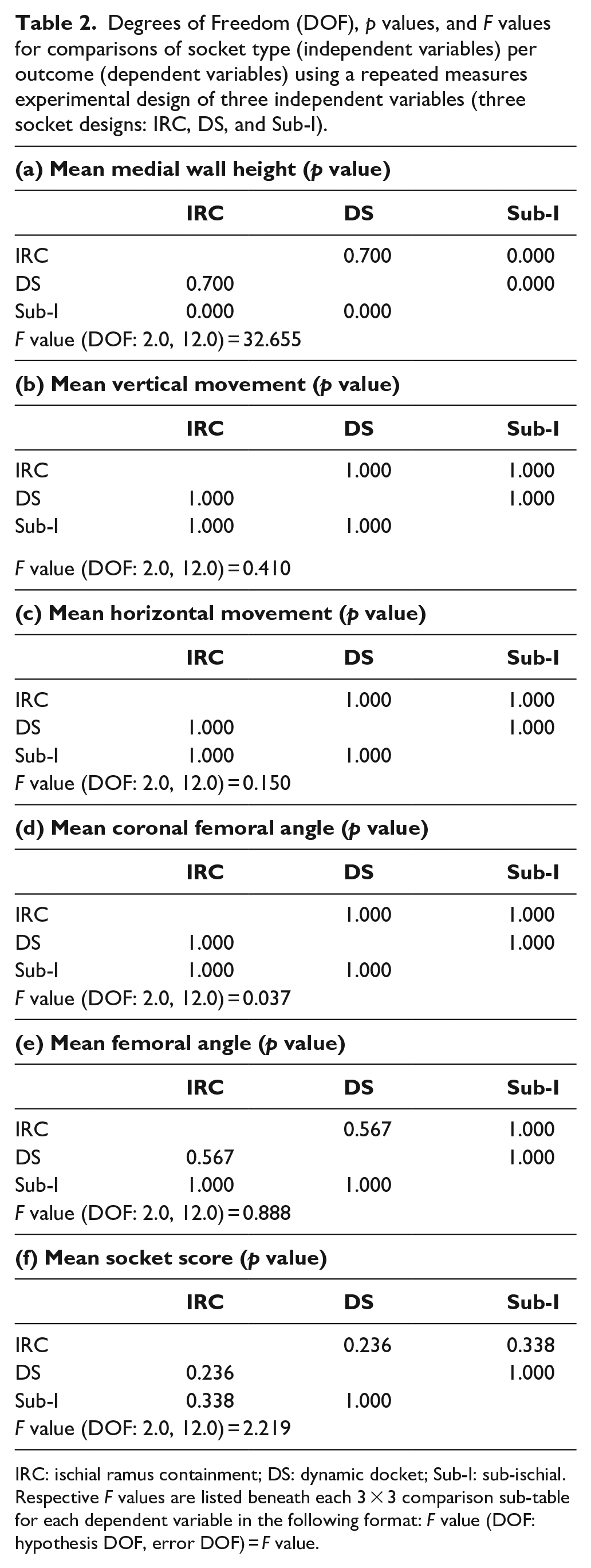
IRC: ischial ramus containment; DS: dynamic docket; Sub-I: sub-ischial.
Respective
Socket comfort
The median and range SCS for the Baseline, IRC, DS, and Sub-I sockets were 8 (3–10), 7 (1–10), 8 (1–10), and 8 (3–10), respectively. These SCS values were not statistically different. All subjects who started with a Sub-I socket scored the Sub-I and DS socket equal to or greater than their baseline socket prior to the study. Most of those scored the Sub-I socket higher than the IRC and DS designs. One third (
Discussion
In this study, authors hypothesized socket skeletal motion would be equivalent across interventions, as previously reported.6,10,24–28 This study used a defined demographic of only experienced high functioning amputees, 18–60 years of age. It was anticipated that the more active subjects might benefit from a Sub-I or DS. Understanding if an intervention is equivalent to the standard of care (IRC) is important to clinical translation of socket design options available to users and clinicians alike. In this and previous studies, there were no significant differences in pistoning, lateral shifting or femoral adduction angle in swing and stance. The subjects maintained similar skeletal motion regardless of which design was used. All designs could be a viable option, when considering the effect on the skeleton. Other clinical determinants should be used to guide the design of choice such as range of motion, appearance, urogenital considerations, past problems, and experiences with other designs and preference toward a particular feature of a design.
The Sub-I medial proximal socket trim line terminated significantly distal from the IT compared with both IRC and DS (
There were several limitations of the study. Socket studies can be difficult from a standpoint of internal and external validity. There is no possible way to blind the user to different sockets. Controlling for socket design and the components associated with it such as the liner, suspension, and materials may not be suitable for all subjects. In this study, two subjects dropped out of the study citing intolerance of the prosthetic fit, specifically the liner. These subjects had soft tissue consistency and notable redundant tissue. The liner chosen for this clinical trial was a thermoplastic elastomer, which is a highly compliant prosthetic gel, and does not have an outer cloth cover or a distal matrix. Clinically, subjects with soft tissue consistency and significant redundant tissue should be fit with less compliant liners to contain and solidify the tissue, which may translate into improved control of the prosthesis. Subjects in this clinical trial with a BMI over 28 scored the Sub-I socket comfort the highest. However, many of these subjects had Sub-I sockets at enrollment, and some of them had developed an adductor roll. This coupled with a high BMI makes fitting IRC and DS sockets more challenging for the patient and clinician alike. As a result, these subjects were not ischial contained by the rigid frame in the IRC and DS sockets when they were tested. In addition, the soldering iron designated the proximal aspect of the laminated frame socket, whereas the flexible interface extended at least 2 cm proximal to the rigid frame on the medial aspect. A LASAR Posture device would be recommended as a way to control for static alignment in future socket studies. 31 X-ray is a two-dimensional medium, where tilting or leaning could affect the perspective. The highest possible medial wall the subject would tolerate to allow accommodation was used. These limitations are evident in the large variances seen across most reported outcome measures in this clinical trial.
Conclusion
The effect a socket design may have on skeletal motion is an important factor but may not be the single best guide for socket design choice. Socket comfort should be considered individually and case specific. All socket designs are suitable if physical considerations such as residual limb length, tissue consistency, and materials are included in the decision-making process. A patient-centric approach focusing on the experience, guidance, and preference of the user is highly recommended to determine the most appropriate socket design.
Footnotes
Acknowledgements
Authors would like to thank Ann Swanson for her invaluable contributions to this manuscript’s development.
Author contributions
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Contents of the manuscript represent the opinions of the authors and not necessarily those of the Department of Defense, Department of Veterans Affairs, the Department of the Army, and any healthcare facility or academic institution.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the US Department of the Army, Department of Defense, Congressionally Directed Medical Research Program (CDMRP; Grant no. W81XWH-15-1-0410, USF Project no. 6140103200).
Trial registration
The study was registered on clinicaltrials.gov (clinicaltrials.gov identifier #NCT02773056).
