Abstract
Background:
Shared decision making is a consultative process designed to encourage patient participation in decision making by providing accurate information about the treatment options and supporting deliberation with the clinicians about treatment options. The process can be supported by resources such as decision aids and discussion guides designed to inform and facilitate often difficult conversations. As this process increases in use, there is opportunity to raise awareness of shared decision making and the international standards used to guide the development of quality resources for use in areas of prosthetic/orthotic care.
Objectives:
To describe the process used to develop shared decision-making resources, using an illustrative example focused on decisions about the level of dysvascular partial foot amputation or transtibial amputation.
Development process:
The International Patient Decision Aid Standards were used to guide the development of the decision aid and discussion guide focused on decisions about the level of dysvascular partial foot amputation or transtibial amputation. Examples from these shared decision-making resources help illuminate the stages of development including scoping and design, research synthesis, iterative development of a prototype, and preliminary testing with patients and clinicians not involved in the development process.
Conclusion:
Lessons learnt through the process, such as using the International Patient Decision Aid Standards checklist and development guidelines, may help inform others wanting to develop similar shared decision-making resources given the applicability of shared decision making to many areas of prosthetic-/orthotic-related practice.
Clinical relevance
Shared decision making is a process designed to guide conversations that help patients make an informed decision about their healthcare. Raising awareness of shared decision making and the international standards for development of high-quality decision aids and discussion guides is important as the approach is introduced in prosthetic-/orthotic-related practice.
Keywords
Background
Shared decision making (SDM) is a consultative process designed to help clinicians and patients engage in meaningful conversations focused on making a particular healthcare decision. 1 At first glance, a consultation using an SDM approach might look like any other. However, an SDM consultation deliberately seeks to empower patients to make informed decision about their healthcare by encouraging patient participation in decision making, providing accurate information about all treatment options, and supporting subsequent deliberation. 2 In practice, this means clinicians must allow and encourage meaningful conversations that help patients participate in decision making. Patients must know that there is a decision to be made about treatment and that clinicians can only help guide the process of decision making given that the patient’s personal preferences will, to a large extent, determine the path ahead.
Encouraging participation in decision making is the first step in the SDM process given that informed decision making can only occur when patients are provided with accurate information about all the treatment options, not just those a health professional might recommend. Information includes what the different treatment options involve, as well as the risks, benefits, and likely outcomes of each. 3 Communication of detailed information about different treatment options can be supported by a decision aid. Decision aids are resources written for patients—a booklet, website, video, or flash cards—designed to communicate information about the different treatment options in a way that helps inform a specific healthcare decision. Decision aids typically include a description of the different treatment options, as well as facts about the risks of harms or likely outcomes, in a way that is easily understood by patients. Decision aids may be used during the consultation to guide patients through the different treatment options and can be taken home as a summary of the consultation for later review or to share with others.3–5
While providing accurate information about the available treatment options is important, clinicians must also support patients as they deliberate on the treatment options, both during and outside the consultation. During the consultation, clinicians can help patients reflect on the information already provided or initiate difficult conversations, such as the risk of dying. These often difficult conversations may be aided by use of a discussion guide. A discussion guide is a resource written for healthcare professionals to help initiate and guide conversations with a patient. As a companion to the decision aid, the discussion guide may include the same topics along with example questions or prompts to help initiate difficult conversations (e.g. what matters most to you at this stage?). There may be more detailed information about the research evidence (e.g. measures of relative risk) written specifically for healthcare professionals to help contextualize evidence to a specific patient (e.g. the relative risk of dying increases with older age and the number of comorbidities).
It is also important that clinicians provide opportunity for patients to deliberate on the decision outside the consultation. Patients may want to review the information provided outside of the time constraints of a clinical appointment or share the information and discuss decision making with family or friends. In this context, a decision aid can help ensure that everyone has access to the same high-quality information as well as support patients to identify what matters most to them or explore uncertainty about the decision. 3 Decision aids typically include exercises, such as multiple-choice questions about the various treatment options to check for accurate understanding or a visual analog scale anchored with the different treatment options to help patients identify a preferred decision. These exercises can promote reflection or help further discussion during a subsequent consultation as clinicians and patients share what was learned through these exercises.
Given this understanding of the process, it is perhaps not surprising that SDM can help clinicians and patients engage in meaningful conversations that, in turn, support patients to take an active part in decisions about their healthcare.2,3,6 SDM has been shown to help patients become more knowledgeable about the different treatment options, accurately perceive the risks and benefits of different treatment options, reduce feelings of conflict about the decision, and reduce frustration about feeling uninformed.3,4 SDM has also been shown to be more effective at helping inform decisions than evidence summaries, clinical practice guidelines, systematic reviews, or clinical decision algorithms, particularly in situations where the evidence is uncertain or decisions are influenced by strong personal preferences. 3
For these reasons, SDM may be particularly beneficial for people facing decisions about dysvascular partial foot amputation (PFA) given that the available evidence does not lead to a clear understanding about the most effective course of treatment, the risk of complications and reamputations are high, and treatment decisions are highly influenced by personal preferences. 7 Like other groups facing amputation surgery, people facing PFA may be uncertain about their treatment options, feel uninformed about the likely outcomes and potential risks, disempowered, and not adequately involved in the decision-making process. 8 With an SDM consult designed to address these barriers to informed decision making, we contend that many people facing the prospect of PFA may deliberately choose a more invasive transtibial amputation (TTA) to reduce the risk of complications and reamputation, particularly given an understanding of the similarity in the mobility and quality-of-life outcomes between PFA and TTA and the high rates of mortality in the years after amputation surgery. 7
As understanding of SDM has grown and resources such as decision aids have become more widely available, we have seen examples of SDM being used as a basis for informed consent and healthcare reimbursement.9,10 In the United States, several states have implemented SDM as a part of routine care, most notably in the state of Washington where the legislation describes that SDM supported by appropriately certified patient decision aids constitutes informed consent.11,12 The Centers for Medicare and Medicaid Services have also adopted the use of decision aids as part of an SDM consultation as a requirement for reimbursement of low-dose computed tomography in lung cancer screening. 13 While SDM and the use of decision aids have been adopted for healthcare decisions including disease screening, medication usage, and surgery,13–18 we are not aware of their use to inform decisions about amputation surgery or other healthcare decisions in areas related to prosthetic and orthotic care.
Given that SDM in areas related to prosthetic and orthotic care is new, there is an opportunity to raise awareness of the process used to develop SDM resources and avoid common issues that have plagued development and implementation of these resources in other areas of healthcare. For example, the information in many SDM resources is not based on high-quality systematic reviews.19,20 Without critical review of the literature to identify the most trustworthy evidence, there is potential for biased information to be included in these resources. Additionally, many SDM resources have not been tested by the intended users. 19 Without such testing, there is no way to know that the topics included are important to inform the particular decision or that the information is easily understood by the intended audience. 19 Finally, despite the important influence the process has on the final product, many SDM resources have been created without publication of the development process. 19
In this article, we aim to describe the process used to develop SDM resources to inform healthcare decisions using an illustrative example focused on decisions about the level of dysvascular PFA or TTA. In describing the development process, we have included illustrative pages from our decision aid and discussion guide and highlighted key lessons that may help inform other developers. In particular, insights into the development of a discussion guide may be particularly helpful given our belief that the development process for a discussion guide has not been previously described.
Development process
International Patient Decision Aid Standards
The International Patient Decision Aid Standards (IPDAS) were written to guide the development of patient decision aids19,21 and thereby minimize many of the common issues previously described. 19 The IPDAS guidelines and checklist specify requirements for a quality decision aid.19,21 For example, the language should be appropriate for a Grade 8 (or lower) reading level, 21 as characterized by short sentences, simple grammar, and lay language (Figure 1). Medical terminology may be presented in parentheses alongside plain language (Figure 1). SDM resources need to be visually appealing in order to be engaging. 21 For example, infographics and figures (Figure 1) can be used to break up text and, in doing so, convey complex information in a pictorial manner that may be more easily understood by patients.21–23 Decision aids should also provide opportunity for guided reflection, so that patients are able to evaluate information in the context of their own personal values and beliefs. 21 For example, use of a visual analog scale anchored with different treatment options may prompt reflection about which option is preferable to the patient. The IPDAS guidelines also describe the steps involved in producing a decision aid including scoping and design, development of a prototype, and preliminary (Alpha) testing with patients and clinicians not involved in the development process (Figure 2). 19

Example page from the decision aid illustrating simple language, lay terminology (e.g. below knee) used alongside medical terminology in parentheses (e.g. transtibial) and simple graphical presentation of data in keeping with the International Patient Decision Aid Standards.
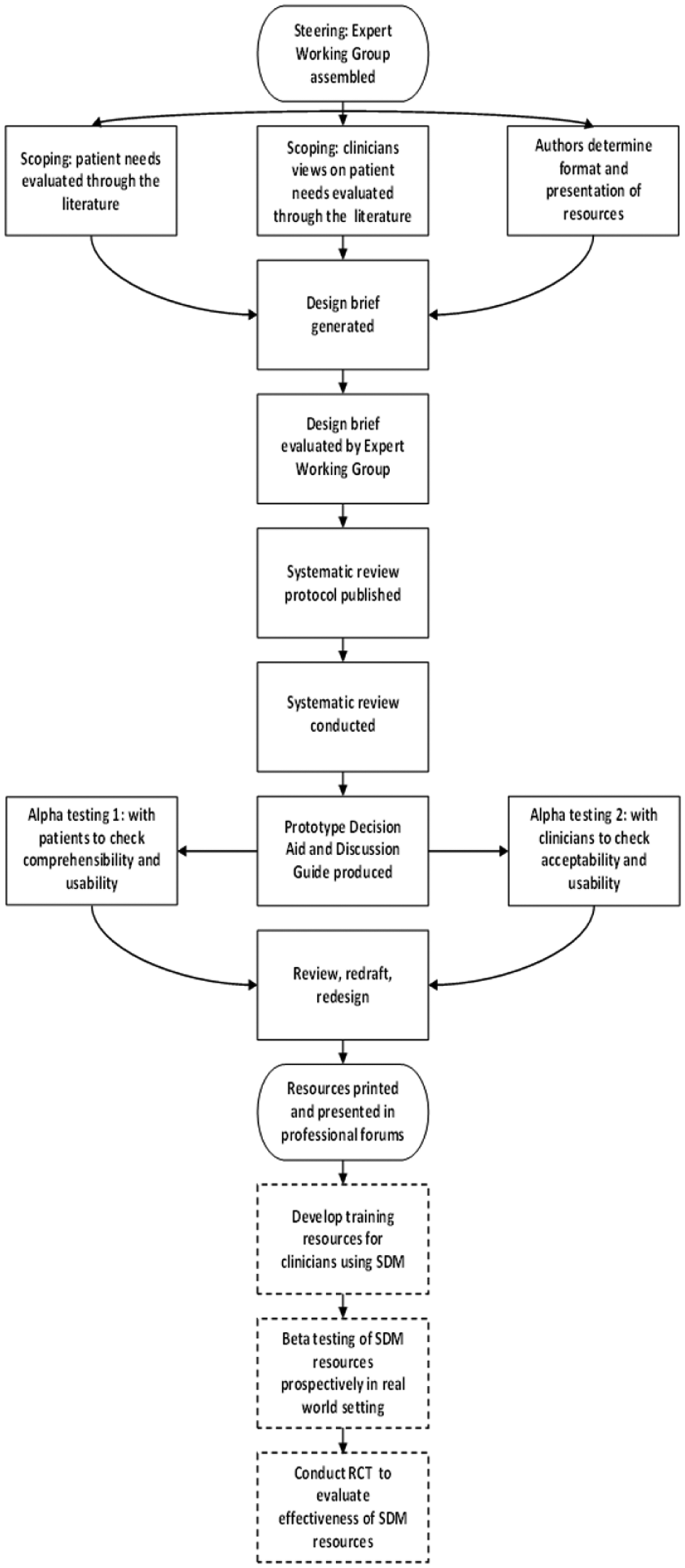
Shared decision-making resource development flowchart. The steps with dashed outlines describe testing undertaken after the decision aid and discussion guide had been developed.
While the IPDAS guidelines are specific to decision aids, they can also guide development of discussion guides. Given that we were unable to find examples of any discussion guides in the public domain nor any literature about the development process, we believe this is the first time the process for development of a discussion guide has been reported. The following subsections describe the application of the IPDAS guidelines in the production of a decision aid and discussion guide to inform decisions about dysvascular PFA, specifically decisions about the level of PFA or the choice between PFA and TTA.
Scoping, steering, and building of the design brief
In keeping with the tenet of IPDAS guidelines, we developed a design brief to define the scope of the decision aid and discussion guide. The design brief included a page-by-page outline of the proposed content (Figure 3, pane 1) as well as several “concept pages” that clearly conveyed the intended layout and design, thereby contextualizing details of the page-by-page outlines (Figure 3, pane 2).

Illustrative example showing development of one section (mobility) of our decision aid from an initial, text-based, design brief (pane 1), and concept page (pane 2) through various stages of testing and refinement based on feedback from expert and user groups (pane 3) to a final page within the decision aid (pane 4). Notable changes over time included paragraphs of text were refined to bullet points, removal of traffic light system representing confidence in the evidence, simplification of infographics showing outcomes for different proportions of the population and introduction of consistent iconography in header (i.e. person with gait aid) to aid navigation across sections of the decision aid. Full-size, high-resolution images are available as supplementary material online.
The process of developing the design brief helped clarify the decision(s) that were to be the focus of the SDM resources, the target audience, and the topics important to help inform the decision. To use the latter as an illustrative example, we turned to the peer-reviewed literature describing the experience of limb loss, outcomes after PFA and TTA surgeries, and the education needs described by patients to define the topic areas included in the SDM resources.8,24–30 Through this exploration, it was noted that those who have lived the experience of limb loss wished they were better informed about what surgery involved, potential complications, healing times and wound management, experience of pain, depression, anxiety, and what mobility would be like, as illustrative examples.8,24–30 While these needs corresponded well with data describing that the experiences of depression and anxiety were common and rates of complications and reamputation were high, it was notable that mortality was rarely discussed despite poor survival rates.31–35 While there were many more education needs identified in these studies8,24–30 (e.g. how to monitor and change the sock fit of a prosthesis), it is important to recognize that participants were sampled at some point after limb loss and as such their experience and education needs will likely differ from those who are in the process of making a decision about amputation surgery. Hence, it is important that once the SDM resources have been developed, they need to be tested with the intended users (i.e. people in the throes of making a decision about amputation surgery) to ensure that all topics necessary to inform the decision have been included.
Evaluation of design brief and concept pages
Having developed a design brief, we sought feedback from the Expert Working Group (i.e. vascular and orthopedic surgeons, rehabilitation physicians, prosthetists, academics, and a SDM expert) that we assembled to guide development. While the Expert Working Group confirmed that the included topics were appropriate (i.e. mobility; functional ability; participation; quality of life; residual limb pain and phantom pain; psychosocial outcomes, including depression, anxiety, and body image, as well as rates of mortality; wound failure; and ipsilateral reamputation), we learnt that the design brief and concept pages read like patient education material and that information was not sufficiently focused to inform the decision between the levels of PFA or the decision between PFA and TTA. In addition, members of the Expert Working Group provided feedback on a wide range of issues (e.g. paragraphs of text were too long and the language was too complex (Figure 3, pane 2)).
Once the design brief and concept pages were refined based on the above feedback, we proceeded to gather the research evidence knowing exactly what evidence was required and how the evidence would be used in the decision aid and discussion guide.
Systematic review of the evidence to be included
In order to develop SDM resources based on the best available evidence, we published a protocol 7 and then conducted a systematic review 36 that included a comprehensive range of topics: mobility, functional ability, participation, quality of life, pain, psychosocial outcomes as well as rates of mortality, wound failure, and ipsilateral reamputation.7,36 The included topics and type of data extracted were designed to provide information suitable for inclusion in the decision aid and discussion guide. For example, to inform readers about the proportion of people who died in the years following PFA, we undertook a meta-analysis to obtain a point estimate and 95% confidence interval. We presented this information graphically in the decision aid for a lay audience (Figure 1) and report it statistically in the discussion guide for health professionals (Figure 4).

Example page from the discussion guide illustrating an example patient question, conversation starter, and a synthesis of the evidence written for health professionals including point estimates of mortality rates over time.
Development of prototypes
Given the revised design brief and evidence from the systematic review, we engaged in an iterative process to produce a prototype decision aid and discussion guide in collaboration with a graphic designer. As an example, we drafted the text and mocked-up ideas for infographics and figures that the graphic designer then professionally formatted. During this process, we made a number of changes to the formatting, such as the layout of tables and graphics, to aid clarity. We included fractions instead of words in the infographics showing the proportion of people with different mobility outcomes after PFA (Figure 3, pane 3) given that the fractions were more intuitive for a lay reader. We also redrafted the text a number of times, guided by the IPDAS guidelines and checklist,19,21 to make the information simple enough to be readily understood by a lay audience.
Alpha testing, review, and finalization of prototypes
For preliminary evaluation (Alpha testing) of the decision aid and discussion guide, we used our Expert Working Group as well as recruiting a user group that included people living in the community with PFA. The purpose of Alpha testing was to fine-tune the SDM resources and check that the content was appropriate for the purpose and audience.
Much of the feedback focused on improving the clarity of the information. For example, some people found the text to be lengthy and suggested that it could be simplified using bullet points and removing numbers within the text (Figure 3, pane 4). The feedback about the conceptual or philosophical approaches underpinning the content of the resources was more challenging. For example, some people found the mortality section confronting and questioned the need for this information. Some health professionals found it unhelpful to highlight limitations of the evidence. In adapting the SDM resources in response to this feedback, we felt it was important that difficult topics, such as the risk of death in the years following amputation, be included and that gaps in the evidence be illuminated because the conversations they should prompt are important to informed decision making.
Based on feedback from Alpha testing, we revised the resources to produce final prototypes of the decision aid and discussion guide (Figure 1; Figure 3, pane 4; and Figure 4). We printed the resources to support presentations at professional meetings 37 and discussions with other interested parties regarding their further development and testing and eventual application with patients.
Discussion
We hope that by describing the development process using an example decision aid and discussion guide focused on decisions about dysvascular PFA, we can better inform others about the IPDAS guidelines, the challenges of developing these resources, and what we learnt along the way.
In keeping with the tenets of evidence-based practice and SDM, there is an obligation to be transparent in reporting both what the best evidence tells us and what it does not tell us. 38 While the systematic review that formed the basis of our SDM resources indicated that there were many studies related to mortality, there were few related to mobility and none related to pain, participation, or psychosocial outcomes. 36 To report that we do not know anything about psychosocial outcomes following PFA and offer no guidance to the patient would be disingenuous and unhelpful. To address this void, we relied on the broader body of literature describing psychosocial outcomes following limb loss that included experiences of anxiety and depression based on the assumption that these experiences may also be common in people following dysvascular PFA. 36 As such, the content of the decision aid and discussion guide highlights this gap in our knowledge and uses related research to inform our understanding about the experience of PFA. In this way, patients and clinicians can engage in discussion about the likelihood of anxiety or depression and what to do in the event they arise even if there is insufficient evidence specific to psychosocial outcomes to inform decision making about the choice of amputation level.
As previously stated, during Alpha testing, it became apparent that the section related to mortality was challenging for some patients and healthcare professionals, who questioned whether it was necessary to include such information. We accept that discussions about death are challenging, particularly given that questions about mobility, not mortality, are often foremost in people’s minds prior to amputation. However, we believe that it is important to facilitate discussion about the rate of mortality in the years after amputation given that simple statistics describing the proportion of people who die following PFA or TTA do not adequately contextualize the influence of common risk factors (e.g. older age and presence of multiple comorbidities) nor highlight that the choice of amputation level does not independently influence mortality rate. 39 As such, meaningful discussions with a knowledgeable clinician are key to helping patients use the mortality evidence to make an informed decision about amputation surgery.
As these SDM resources are a form of healthcare intervention with potential for adverse effects, their effect in the real world needs to be evaluated. 4 Therefore, next steps in the development process include the development of training resources for clinicians engaging in SDM on this topic and prospective testing of the SDM resources in a real-world setting (Beta testing), where participants are recruited at the time decisions about amputation surgery are being made. Finally, a randomized controlled trial is required to evaluate the effectiveness of the SDM resources (Figure 2). It is prudent that any decision aid and discussion guide not be used clinically until research demonstrates effectiveness (or potential harms) in a real-world setting.
By describing the process of development, as it is related to our topic regarding decisions about the level of PFA or the choice between PFA and TTA, we hope to aid future efforts to build SDM resources in other areas related to prosthetic/orthotic practice. We found the IPDAS checklist 21 and development guidelines 19 invaluable to our experience and would highly recommend their use to other developers. As academics, it was challenging to present information simply enough to be readily understood by a lay audience, while remaining true to the evidence.
While this article focuses on the process of developing SDM resources to inform decisions about dysvascular PFA, SDM approaches might be particularly useful in similar situations where the evidence is uncertain or where strong personal preferences influence decisions, such as decisions about scoliosis bracing versus surgery or decisions about plagiocephaly management using orthotic intervention, repositioning therapy, or no treatment.
Conclusion
SDM has become common in many areas of healthcare with the process improving communication and outcomes, particularly when SDM resources such as decision aids are used. We expect this may also be true for people facing decisions about amputation surgery and other areas related to prosthetic and orthotic practice. We shared our experiences of developing a decision aid and discussion guide to help inform difficult decisions about dysvascular PFA to increase awareness of the process for others seeking to develop SDM resources in prosthetics and orthotics.
Supplemental Material
752984_Figure_3_hi_res_supp_mat – Supplemental material for Development of shared decision-making resources to help inform difficult healthcare decisions: An example focused on dysvascular partial foot and transtibial amputations
Supplemental material, 752984_Figure_3_hi_res_supp_mat for Development of shared decision-making resources to help inform difficult healthcare decisions: An example focused on dysvascular partial foot and transtibial amputations by Matthew Quigley, Michael P Dillon and Stefania Fatone in Prosthetics and Orthotics International
Supplemental Material
752984_Figure_4_hi_res_supp_mat – Supplemental material for Development of shared decision-making resources to help inform difficult healthcare decisions: An example focused on dysvascular partial foot and transtibial amputations
Supplemental material, 752984_Figure_4_hi_res_supp_mat for Development of shared decision-making resources to help inform difficult healthcare decisions: An example focused on dysvascular partial foot and transtibial amputations by Matthew Quigley, Michael P Dillon and Stefania Fatone in Prosthetics and Orthotics International
Footnotes
Acknowledgements
The authors would like to extend their gratitude to the American Orthotic and Prosthetic Association Expert Working Group for their considered feedback and constructive input into the development of these resources. In particular, they extend their sincere gratitude and appreciation to Professor Tammy Hoffmann from Bond University, Australia, who provided invaluable advice consistent with her expertise in SDM. They would also like to extend their gratitude to Limbs4Life, who facilitated engagement with people living with limb loss and to the members of the User Group who provided feedback during the process of Alpha testing. The authors also acknowledge and thank their graphic designer, Jake Eadie, who worked tirelessly to improve the visual appeal of these resources through the various iterations.
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by the American Orthotic and Prosthetic Association (RFP-04012015) to develop the shared decision-making resources and was awarded to Drs Michael P Dillon and Stefania Fatone. The funding body did not have any role in the writing of this manuscript or the decision to submit for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
