Abstract
Background and aim:
In order to create more uniformity in the prescription of upper limb prostheses by Dutch rehabilitation teams, the development and implementation of a Prosthesis Prescription Protocol of the upper limb (PPP-Arm) was initiated. The aim was to create a national digital protocol to structure, underpin, and evaluate the prescription of upper limb prostheses for clients with acquired or congenital arm defects.
Technique:
Prosthesis Prescription Protocol of the Arm (PPP-Arm) was developed on the basis of the International Classification of Functioning and consisted of several layers. All stakeholders (rehabilitation teams, orthopedic workshops, patients, and insurance companies) were involved in development and implementation. A national project coordinator and knowledge brokers in each team were essential for the project.
Discussion:
PPP-Arm was successfully developed and implemented in nine Dutch rehabilitation teams. The protocol improved team collaboration, structure, and completeness of prosthesis prescriptions and treatment uniformity and might be interesting for other countries as well.
Clinical relevance
A national protocol to prescribe upper limb prostheses can be helpful to create uniformity in treatment of patients with upper limb defects. Such a protocol improves quality of care for all patients in the country.
Keywords
Background
In the Netherlands, annually about 40 people lose an upper limb or hand and about 50 children are born with a transverse upper limb reduction. The group of clients with an arm deficiency is thus small, but often needs long-term care because they usually have a normal life expectancy. It is important that the care of this small but intensive client group is of a high quality because all aspects of functioning may be influenced by the limb loss. Furthermore, care is associated with high costs, especially when prostheses or adaptive devices are provided. Specialized care by a multidisciplinary team is necessary.
In the Netherlands, each rehabilitation team previously followed its own prosthesis prescription process. As a result, upper limb prosthesis care differed among the diverse rehabilitation institutions in, for example, the use of educational materials, structure, and content of the prescription letter for insurance companies and advice regarding types of devices. There was a need for a standardized method of prescribing upper limb prostheses in order to improve quality of care. In 2009, the national Working Group Amputation and Prosthetics of the Arm of the Dutch Society of Rehabilitation Specialists took the initiative to develop the Prosthesis Prescription Protocol of the Arm (PPP-Arm). 1
Aim
PPP-Arm aims to structure, underpin, and evaluate the prescription of an adaptive device or a prosthesis and to create a uniform, nationally applicable, prescription policy. The intention was to use the protocol in all nine Dutch centers dealing with these patients.
Technique
The protocol has been created through the collaboration of various rehabilitation centers, patients, orthopedic workshops, and insurance companies (Figure 1). Representatives of these groups formed the Working Group PPP-Arm.
All involved parties within the project PPP-Arm.
In October 2011, the paper version of the protocol was approved by the Assembly of the Dutch Society of Rehabilitation Specialists. In order to make the protocol efficient and user-friendly, a digital version of the protocol was developed in 2012.
After obtaining funding, a national project coordinator was appointed. Furthermore, in each team, a knowledge broker was appointed, who was responsible for the implementation of the protocol within his own center.
The protocol is based on the World Health Organization’s criteria of the International Classification of Functioning (ICF) and consists of the following layers (Figure 2):
Establishing patient’s demands;
Preparation of treatment requirements;
Establishing device requirements;
Selection, try-out, and final decision;
Delivery of the device;
Use of the device (instructions and training);
Evaluation. 2
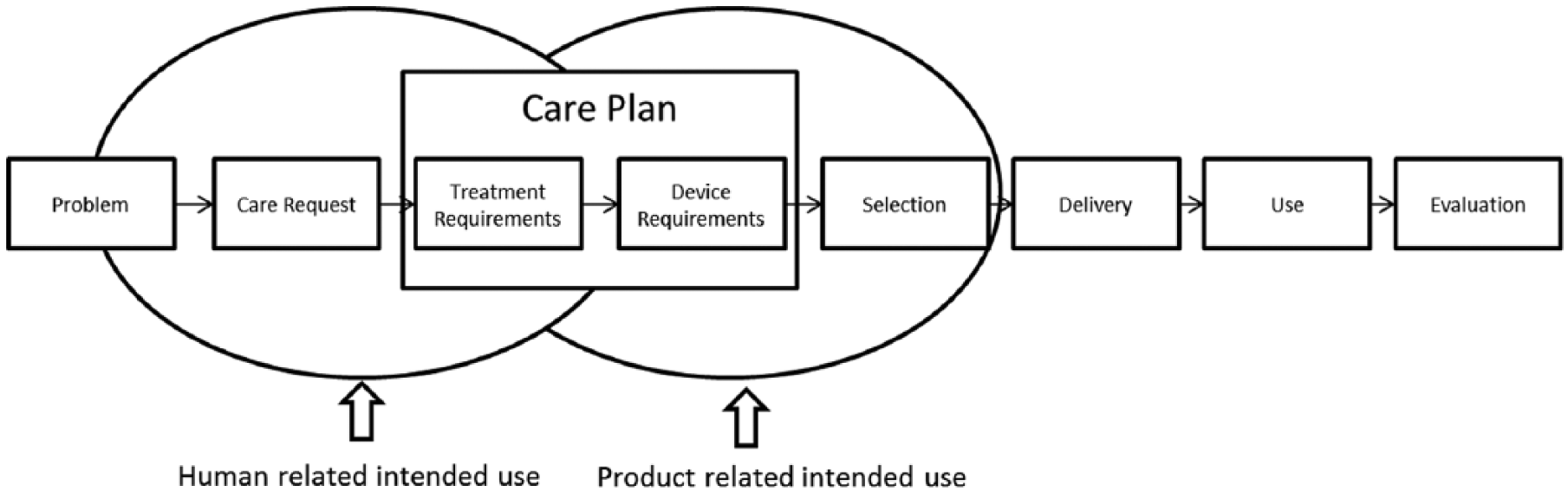
Structure of the protocol.
To fill in the digital protocol, the practitioner has to log in on a specific website (Orthofirm) with a personal username and password. The data of the client can be entered and stored in a structured manner in the various folders and tabs (Figure 3). The layers of the protocol provide the various practitioners (rehabilitation physician, therapist, and orthopedic technician) the opportunity to register relevant information quickly and easily. In the folder “Care request,” there is room to describe the client’s care request and provide information about the medical diagnosis. In the next folder, the medical analysis of the need for care is registered. In this folder, there are several tabs based on the ICF: anatomical features, body functions, activities, and participation. Most questions have multiple choice options with the possibility to add relevant information.
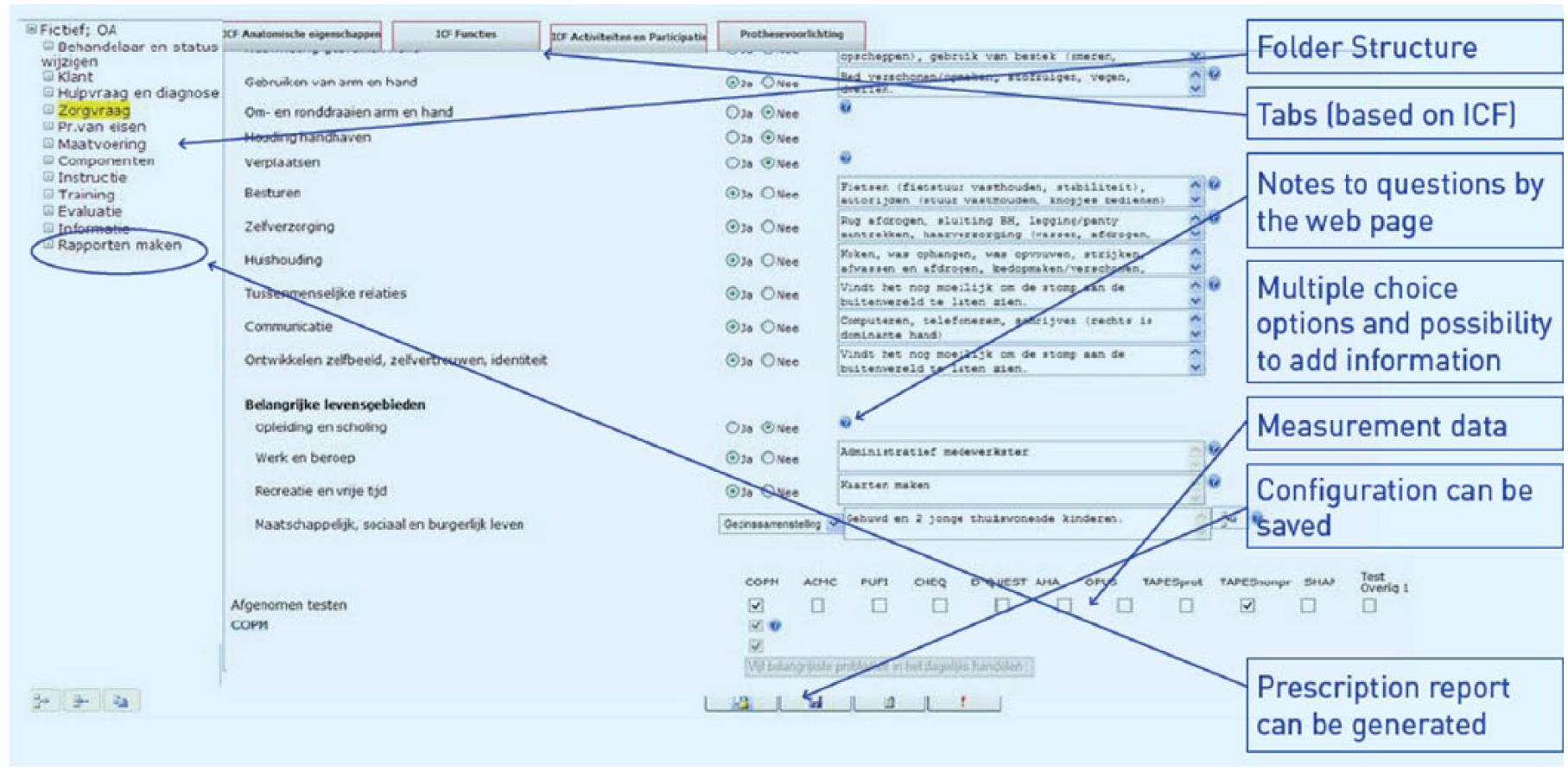
Example of a page of the digital protocol.
Results of several questionnaires and measurement instruments can be registered in the protocol, including Canadian Occupational Performance Measurement (COPM), Assessment of Capacity for Myoelectric Control (ACMC), and Dutch version of Quebec User Evaluation of Satisfaction with assistive Technology (D-QUEST). The folder “Program of demands” helps to identify which solution fits best the client’s demands and describes the requirements of the device.
On the last page of the folder “Program of demands,” the decision is made whether a prosthesis should be prescribed or not: the Go/No Go decision. The conclusion may be that a prosthesis is not a suitable solution for the client’s needs. In that case, a “No Go decision” will be chosen. If it is concluded that a prosthesis or adaptive device is the proper solution for the client’s functional problems, a “Go decision” is registered and motivated.
In some cases, it is difficult to decide which type of prosthesis should be advised, for example, when a client has bilateral upper limb amputations or when a multi-articulated hand is considered. In these situations, a trial period might be an option: the client will be able to try a temporary prosthesis for several weeks at home. After this trial period, the final decision on the prosthesis choice can be made and motivated. In the folder “Components,” information about the components of the advised prosthesis can be added by the technician. 4
After filling in all folders of the protocol, a thorough and structured analysis has been performed to achieve a suitable solution for the client’s needs. The protocol does not provide a score of some kind to sort devices. After completing all the layers of the protocol in a standardized way, it becomes clear which needs and goals the patient has and which prosthesis components may be helpful to achieve these goals and needs. A list of possible options for prosthesis components can be found in the folder “Components.”
A prescription report can be generated for the insurance company. This report consists of a main page with the client’s care request, medical diagnosis, explanation, and motivation for the prosthesis choice and the components of the prosthesis. An appendix can be added with other information concerning body functions, activities, and participation and measurement data.
After the prosthesis is delivered, the protocol can be used for instructions, training, and evaluation. In the folder “Evaluation,” the care request and need for care can be evaluated when the client has been using the prosthesis for a while. The questions are based on the ICF. Measurement data of the follow-up can be registered here and help to formulate a conclusion.
We also have developed the folder “Information.” In this folder, there are over 100 links to educational materials for clients on for example: phantom sensations, upper limb prostheses, personal care, and peer contact. This information includes photographs, movies, brochures, and PowerPoint presentations. 3
Discussion
A national prosthesis prescription protocol for upper limb prosthesis users, “PPP-Arm,” was successfully developed and implemented in nine Dutch rehabilitation teams. The protocol is used to structure, underpin, and evaluate the prescription of an adaptive device or a prosthesis. The protocol created a uniform, nationally applicable, prescription policy.
Several advantages of the protocol were experienced during the execution of the project:
Complete and structured;
User-friendly;
Using the same ICF terminology;
Applied nationally;
Digital reporting;
Workplace-independent login possibilities for all team members;
A prescription report is generated for the insurance company;
Patients gain more insight into their own treatment process;
The protocol contributes to building a national database for research.
The main disadvantage mentioned was the time investment needed to learn how to use the protocol. The use of PPP-Arm has been evaluated using a questionnaire answered by the participating rehabilitation teams and by the insurance companies. The most important conclusions were as follows:
PPP-Arm is used by all participating centers
The quality of the prosthesis applications has improved: PPP-Arm provides a more structured report with all the information that is needed for the insurance company to judge the application.
PPP-Arm has produced several positive developments: better collaboration of different team members, more structure and completeness in the prescriptions, more treatment uniformity in the country, and implementation of a temporary prosthesis trial period.
By developing PPP-Arm, we have managed to create a national uniform and structured method to advise and evaluate the prescription of upper limb prostheses. This might be interesting for other countries as well. An English version is available for usage in other countries.
Key points
Rehabilitation teams for upper limb prosthesis users work more uniformly using a national prescription protocol.
A national project coordinator and knowledge brokers in each team are prerequisites for successful protocol development and implementation.
All stakeholders should be involved in protocol development and implementation: rehabilitation teams, orthopedic workshops, patient organizations, and insurance companies.
The ICF framework can be used to structure a prescription protocol for upper limb prosthesis users.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
