Abstract
Background and aim:
Patient-reported outcome measures are increasingly used to evaluate effectiveness of treatment. However, the use of ‘paper and pencil’ questionnaires is time-consuming for both patients and healthcare specialists. Therefore, the aim of this project was to develop a custom-built web-based monitoring system.
Technique:
We incorporated reliable and valid questionnaires on all domains of human functioning as described in the World Health Organization’s Classification of Functioning, Disability and Health (ICF and ICF-CY). The method of remote follow-up enables long-term evaluation of PROMs. We specified monitoring protocols for both children and adults, split for different age groups with emphasis on health-related quality of life.
Discussion:
Time-efficient evaluation of PROMs may lead to higher compliance and an increase of client-centred practice. Moreover, evaluating PROMs facilitates patient empowerment and enables patients to make informed decisions about their treatment and healthcare needs.
Clinical relevance
We developed a web-based system for evaluation of PROMs. The system has enabled better informed decision-making for our clients.
Keywords
Background and aim
Our team is specialized in treating patients with congenital arm-hand disorders or an acquired amputation of the upper extremity. The age range of our patient population is broad, from prenatal consultation, screening and providing information, to adulthood. The composition of this patient group is unique to our country and is formed by many years of collaboration between our rehabilitation centre and the university hospital, enabling lifespan care on all domains of human functioning as described in the ICF and ICF-CY. 1
Currently, our institute is one of the leading three institutes in our country regarding patient population size. Patient care for this group is preferably concentrated at a university hospital because of the low prevalence and need for highly specialized and interdisciplinary care. These patients consult our team, ExtRah (Expertise team Rotterdam arm and hand), throughout their different lifespan transition phases and are provided with expert information. Patient outcome measures are usually evaluated at a low frequency, and treatment is initiated or intensified if problems arise.
Our team’s treatment philosophy is based on the client-centred care model. In this model, patients play an active role in enabling collaboration between patients and healthcare providers (doctor, therapist, prosthetist). 2 Research findings show that client-centred practice leads to improvement of client satisfaction and compliance to health service programs. 3 Furthermore, respecting the individual client means having a high regard for his autonomy, values and cultures. 4
Regardless our patient choices whether it is to learn to use a prosthesis, a task-specific tool or to function without help or aids, it should all be based on their autonomy. Patient involvement in the care process is crucial to optimize treatment. 2 Therefore, providing feedback on the quality of their care and, above all, the regained quality of life, enhances direct influence on their care process, enabling shared decision-making and facilitating higher patient involvement. 5
Monitoring patient-reported outcome measures (PROMs) is relevant for both patients and our team. First, processing the data enables us to provide high-quality information within daily patient care facilitating well-informed choices on treatment and support in participation and quality of life. Second, by collecting the data digitally, the number of patient visits to the centre can be reduced. Third, when they fill out questionnaires prior to the visit to our centre, our practitioners have easy access to the results and are able to adjust their policy accordingly.
Nowadays, PROMs are commonly used to evaluate the effectiveness of treatment. ‘Paper and pencil’ questionnaires are time-consuming for both patients and healthcare specialists. Digital tracking and recording of PROMs has not been implemented structurally within the national working group for upper limb prosthetics care. Therefore, an interactive tool was desired.
Techniques
We developed and implemented a web-based monitoring protocol, evaluating PROMs of patients with a congenital hand difference or acquired amputation of the upper extremity. Gemstracker ExtRah offers by means of a standardized evaluation a base for research on quality and effectiveness. We aim to monitor our entire group of patients, both children and adults, on activities and participation with emphasis on and health-related quality of life (HRQoL).
Gemstracker was already operational in research projects at the departments of Rehabilitation Medicine and Plastic and Reconstructive surgery of Erasmus Medical Centre. Therefore, it was accessible and directly applicable at our institute. The disadvantages of using a system developed especially for our institute and patient group may limit generalizability for its application.
We have used LimeSurvey software, open-source software, in order to build Gemstracker ExtRah, which enabled digitalizing questionnaires and organizing them into monitoring protocols. Reliable and valid questionnaires that are widely used in rehabilitation medicine were incorporated.6,7 These questionnaires provide information on all domains of human functioning as described in the World Health Organization classification of functioning, disability and health (ICF and ICF-CY). This digital monitoring system enables patients, physicians and/or therapists to enter information in a secure online application. Patients must give their written consent to participate in this project, before they receive an email with a unique token. This token directly matches the assigned questionnaires for this particular patient at that time. Gemstracker ExtRah automatically sends an email invitation at pre-set times (e.g. the start of the rehabilitation treatment, after 1 year and subsequently on a yearly basis) to provide long-term follow-up.
Gemstracker ExtRah has been in use since February 2016, enabling comparison between patients, patient groups and treatments. Results so far show a participation of over 130 patients in the monitoring database. First analysis of the data shows the spread across the age groups (Figure 1) and an overview of the completed questionnaires so far (Figure 2).
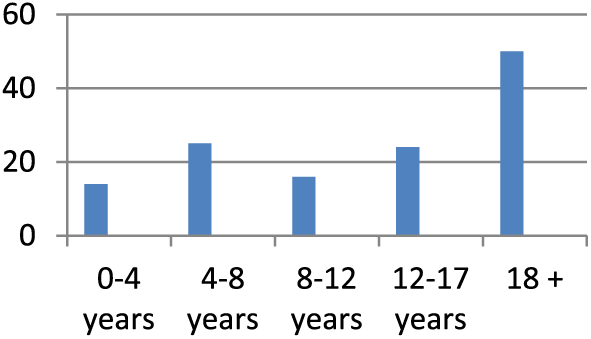
Histogram age spread.
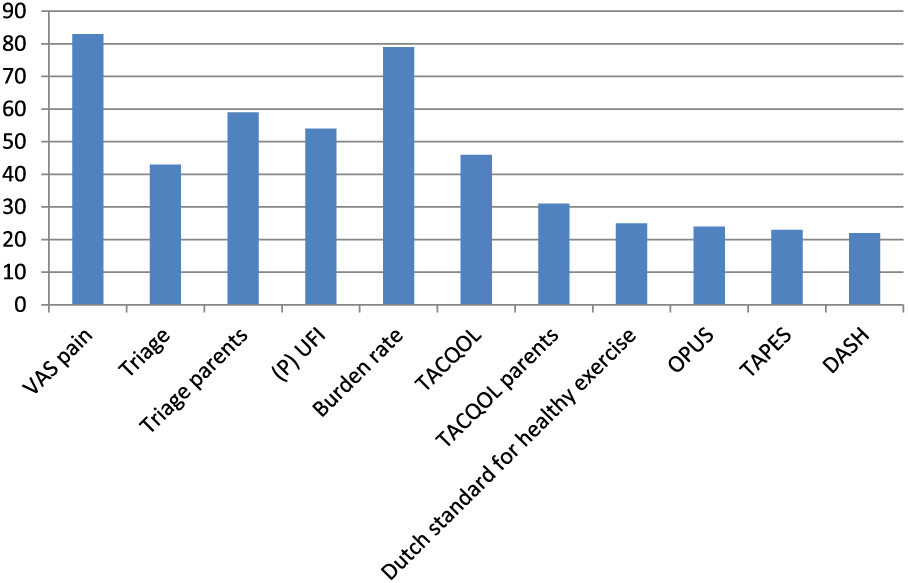
Histrogram completed questionnaires.
We use a monitoring protocol for both children and adults, specified in age groups with emphasis on HRQoL to enable age-specific questioning. Tables 1 to 6 show the questionnaires used in the protocol based on the domains of the ICF and ICF-CY.
Questionnaires used according to the ICF/ICF-CY framework.
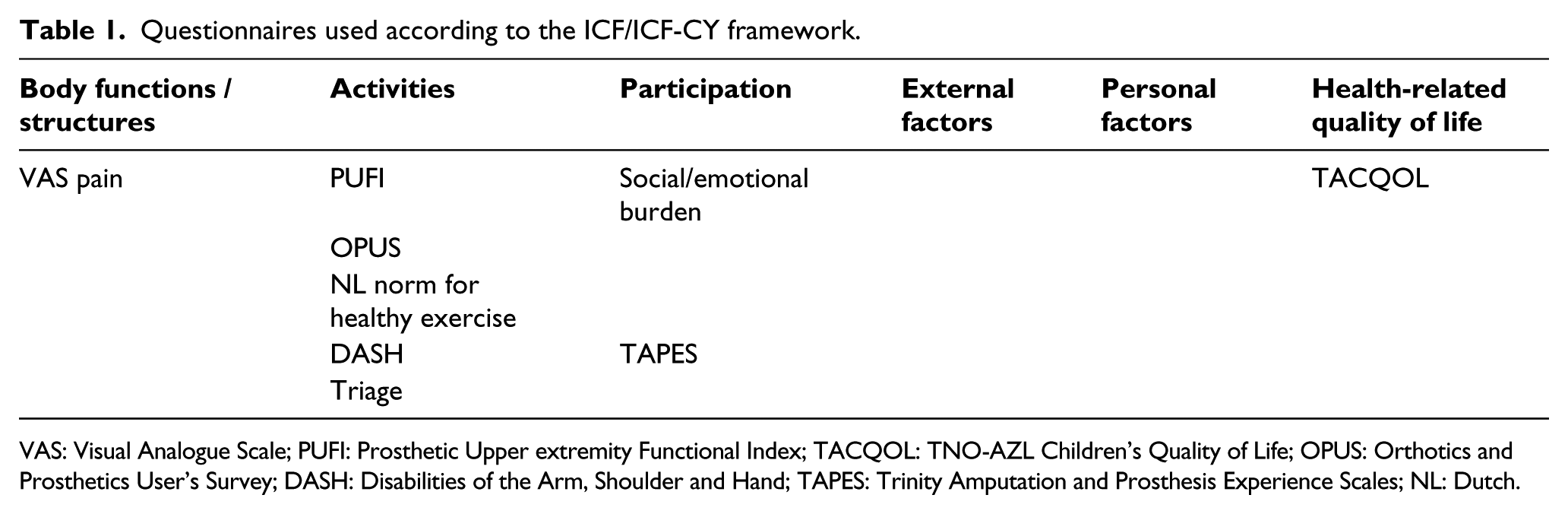
VAS: Visual Analogue Scale; PUFI: Prosthetic Upper extremity Functional Index; TACQOL: TNO-AZL Children’s Quality of Life; OPUS: Orthotics and Prosthetics User’s Survey; DASH: Disabilities of the Arm, Shoulder and Hand; TAPES: Trinity Amputation and Prosthesis Experience Scales; NL: Dutch.
Monitoring protocol child 0–4 years.
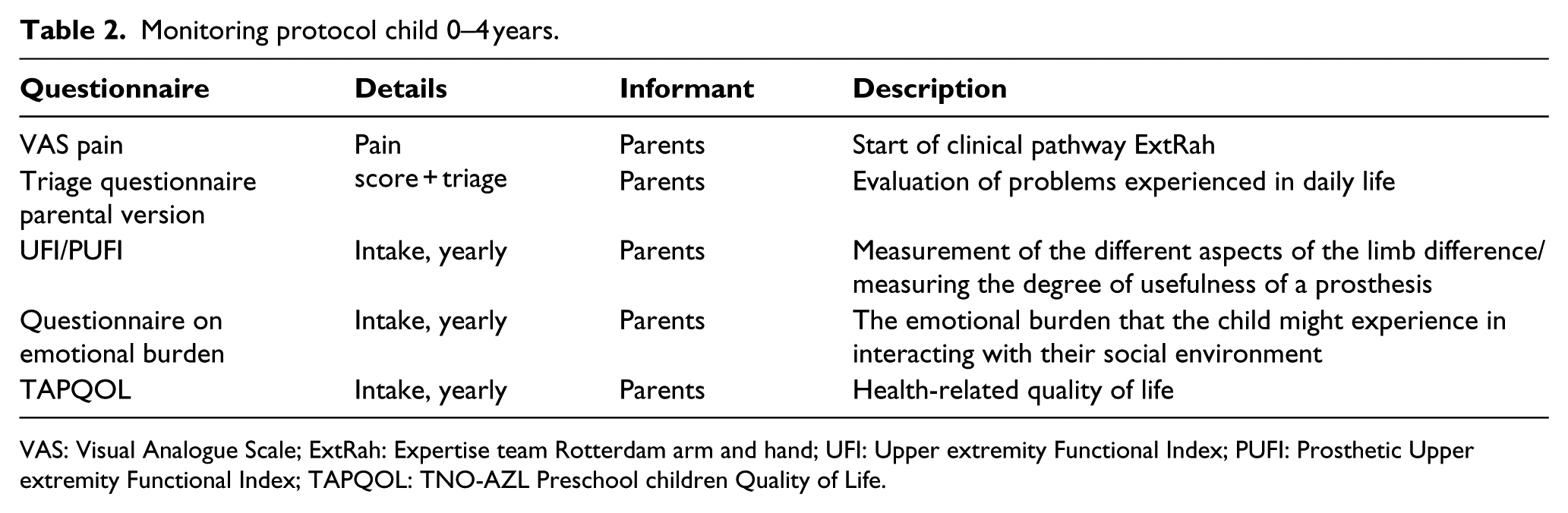
VAS: Visual Analogue Scale; ExtRah: Expertise team Rotterdam arm and hand; UFI: Upper extremity Functional Index; PUFI: Prosthetic Upper extremity Functional Index; TAPQOL: TNO-AZL Preschool children Quality of Life.
Monitoring protocol child 4–7 years.
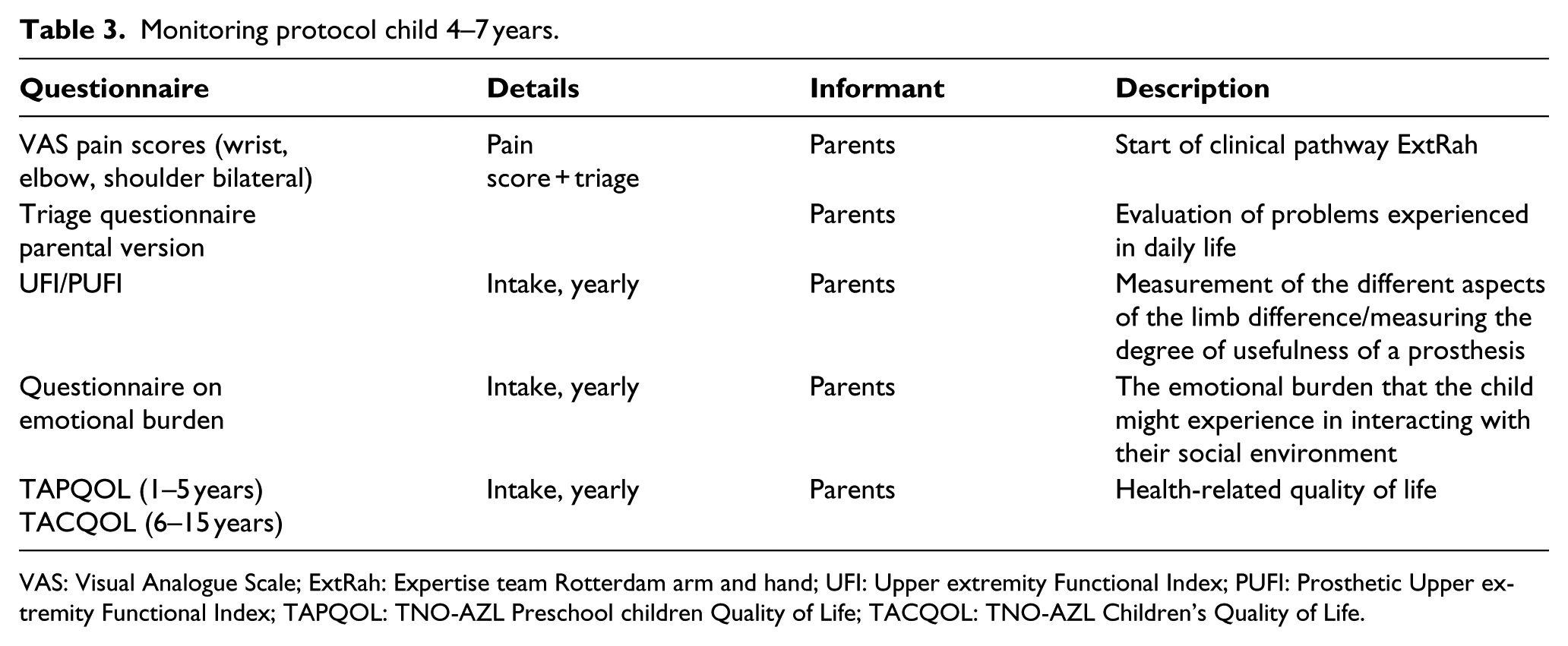
VAS: Visual Analogue Scale; ExtRah: Expertise team Rotterdam arm and hand; UFI: Upper extremity Functional Index; PUFI: Prosthetic Upper extremity Functional Index; TAPQOL: TNO-AZL Preschool children Quality of Life; TACQOL: TNO-AZL Children’s Quality of Life.
Monitoring protocol child 8–12 years.
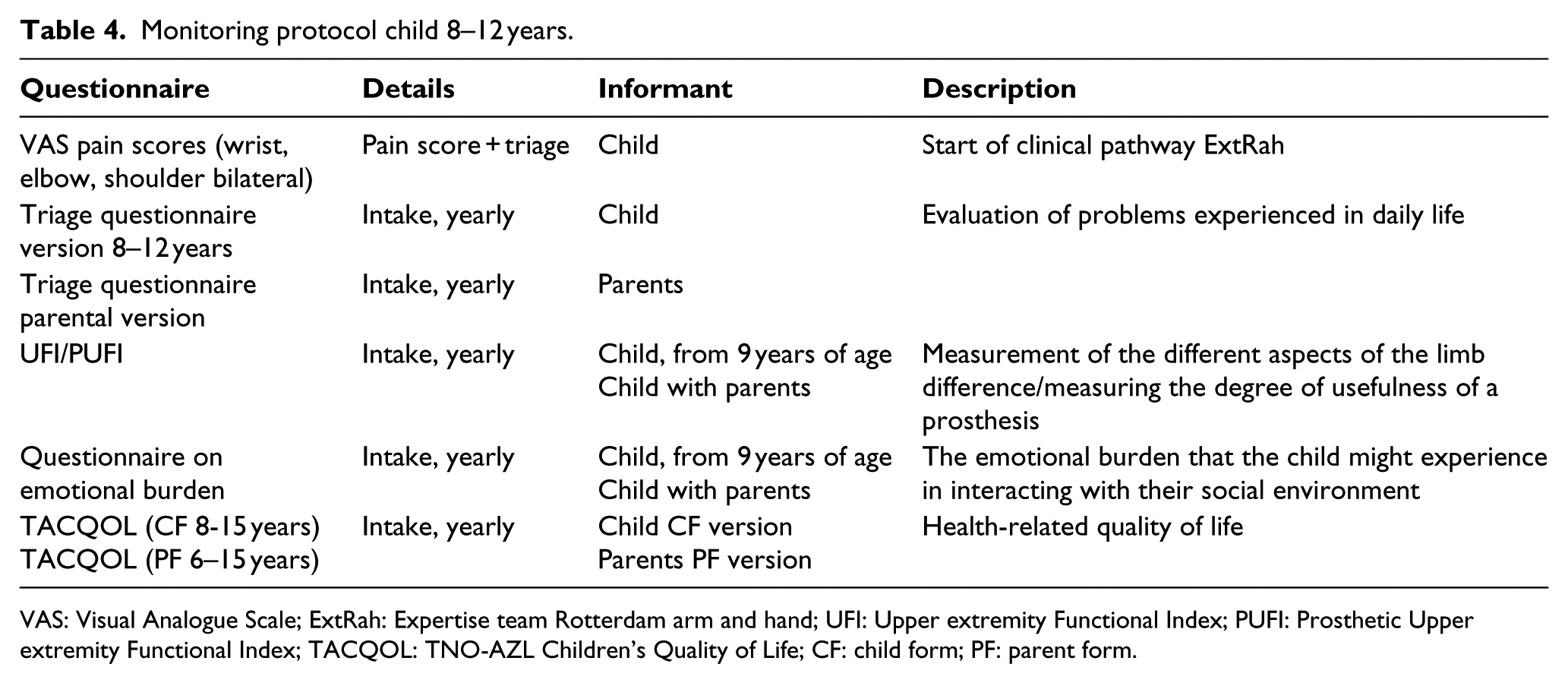
VAS: Visual Analogue Scale; ExtRah: Expertise team Rotterdam arm and hand; UFI: Upper extremity Functional Index; PUFI: Prosthetic Upper extremity Functional Index; TACQOL: TNO-AZL Children’s Quality of Life; CF: child form; PF: parent form.
Monitoring protocol child 12–18 years.
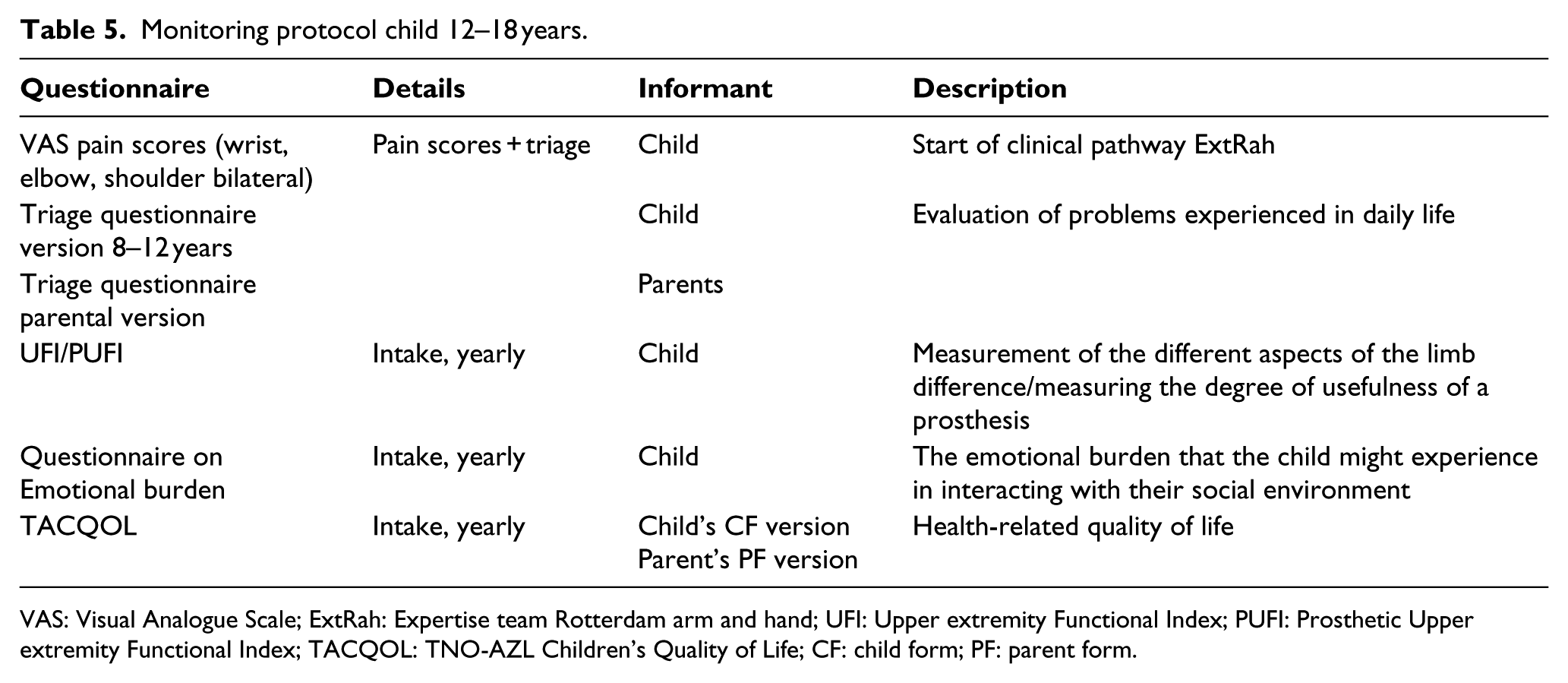
VAS: Visual Analogue Scale; ExtRah: Expertise team Rotterdam arm and hand; UFI: Upper extremity Functional Index; PUFI: Prosthetic Upper extremity Functional Index; TACQOL: TNO-AZL Children’s Quality of Life; CF: child form; PF: parent form.
Monitoring protocol 18 – adulthood.
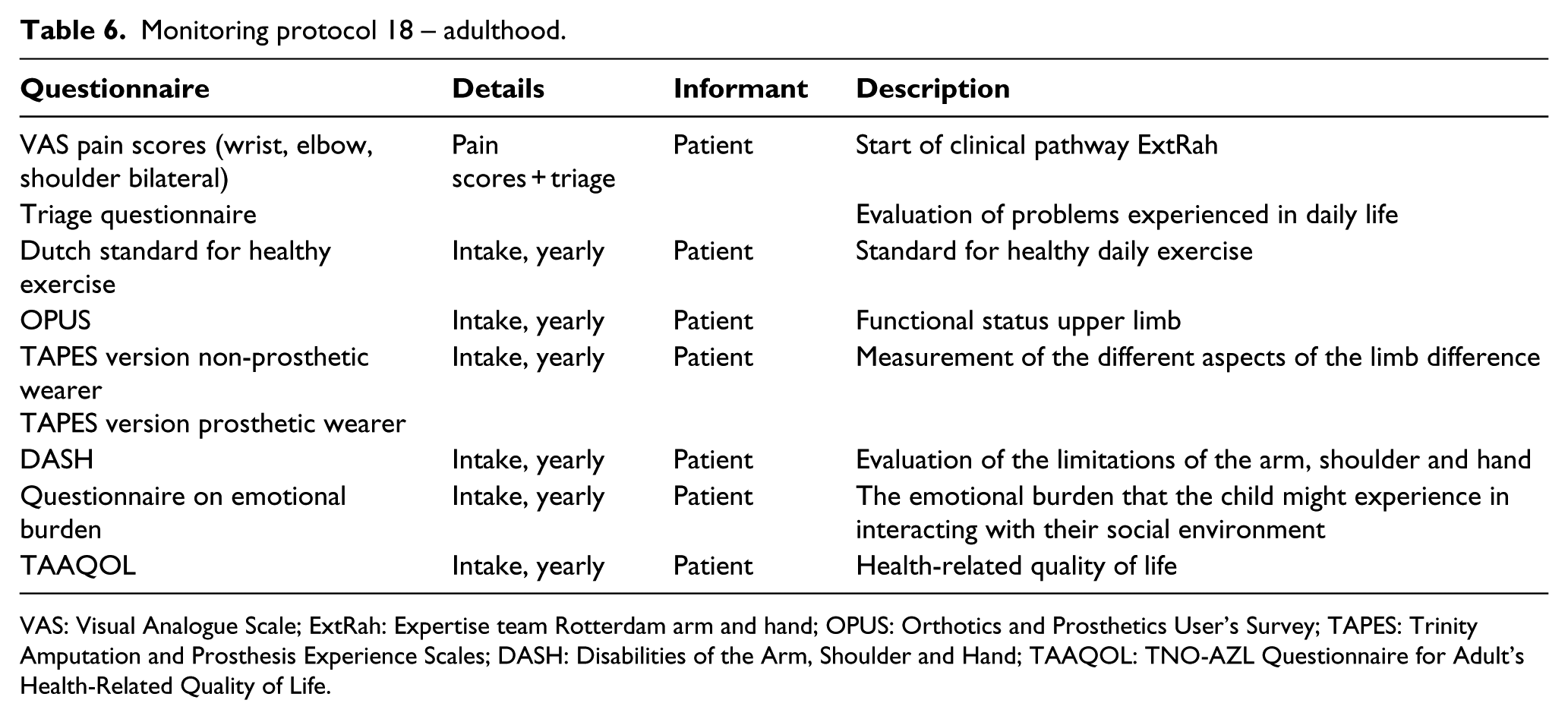
VAS: Visual Analogue Scale; ExtRah: Expertise team Rotterdam arm and hand; OPUS: Orthotics and Prosthetics User’s Survey; TAPES: Trinity Amputation and Prosthesis Experience Scales; DASH: Disabilities of the Arm, Shoulder and Hand; TAAQOL: TNO-AZL Questionnaire for Adult’s Health-Related Quality of Life.
Discussion
Gemstracker ExtRah provides, in addition to the innovative value for the direct patient care, a base for research on quality, effectiveness, prognosis and outcome on PROMs of the questionnaires used. Our web-based monitoring database provides insight in PROMs and enables continuity in the monitoring of HRQoL of our patient group. Gemstracker ExtRah can supply data and insight into quality of care towards health insurance companies.
At the start of the project, we incorporated all questionnaires in use in our daily practice and used generic questionnaires instead of diagnosis specific questionnaires. 5 We have made several choices in order to reduce patient time burden of the questionnaires.
Moreover, we changed our method of assigning questionnaires from doctor-driven to PROMs,8,9 while using a maximum of 1 or 2 questionnaires per ICF domain. These choices were made on the basis of usability requirements such as patient burden, efficiency of the system and the goals for long-term follow-up. In the very young child, we focus on parent-rated outcome, but as soon as possible, the patient’s, or in this case the child’s, opinion prevails as much as possible. 10 By making choices in which questionnaires we use, we may risk losing information.
Most questionnaires or assessments used in research papers regarding our patient group were focused on wearing or using a prosthesis. 11 However, it is our understanding that the HRQoL experienced by a person with an upper limb deficiency is not only influenced by whether or not they wear a prosthesis, or even the type of prosthesis, but by many other factors instead.12–14 They are more likely to be based on activities and social participation.15,16
However, we did incorporate the Prosthetic Upper extremity Functional Index (PUFI) because this questionnaire evaluates to what extent the child actually uses its residual arm, prosthesis or if the child is not able to perform the task.11,17 With this questionnaire we are able to distinguish performance and capacity within activities. 18 We incorporated the Trinity Amputation and Prosthesis Experience Scales (TAPES) and Orthotics and Prosthetics User’s Survey (OPUS) since these are able to describe the emotional and social status of a person in relation to whether a prosthesis is being used in daily life.19,20 OPUS assesses clinically relevant domains of patient experience that should help clinicians provide high-quality care. 20 Within the national taskforce, we have agreed to use this measurement as a part of our national Prosthesis Prescription Protocol (PPP). 21 Additionally, we incorporated Disabilities of the Arm, Shoulder and Hand – Dutch Language Version (DASH-DLV) because of its reported ability to be sensitive enough to describe the disability of the entire upper extremity without losing the ability to detect effects at each area or joint in the limb. 22 We find this information valuable because a part of our patient group consists of patients with partial hand deficiencies and thus partial hand function can be measured by DASH-DLV.
We specified monitoring protocols for both children and adults, split for different age groups so they represent school ages and the most prominent moments across transition in lifespan. Research shows that patients tend to have a higher need for specific transitional care at specific moments such as changing school, puberty, or, for example, finding a job.23,24
In addition, we composed a triage questionnaire. This questionnaire contains general questions on the domains of daily functioning such as activities of daily living, sports and work. Based on the outcome of this questionnaire, we aim to be able to select a supplementary questionnaire and add this to the monitoring protocol of this specific patient.
Our healthcare practitioners are able to gain insight in the level of functioning of the patient and the patient system with a low patient burden. The Gemstracker ExtRah database can provide data to create a reference framework (benchmarking) in which this specific healthcare can be evaluated, appropriate to an academic expertise care team.
Key points
Web-based evaluation of PROMs;
Monitoring protocols with emphasis on HRQoL;
Enabling informed decision-making.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
