Abstract
Background:
The evolution of three-dimensional printing into prosthetics has opened conversations about the availability and cost of prostheses. This report will discuss how a prosthetic team incorporated additive manufacture techniques into the treatment of a patient with a partial hand amputation to create and test a unique assistive device which he could use to hold his French horn.
Case description and methods:
Using a process of shape capture, photogrammetry, computer-aided design and finite element analysis, a suitable assistive device was designed and tested. The design was fabricated using three-dimensional printing. Patient satisfaction was measured using a Pugh’s Matrix™, and a cost comparison was made between the process used and traditional manufacturing.
Findings and outcomes:
Patient satisfaction was high. The three-dimensional printed devices were 56% cheaper to fabricate than a similar laminated device.
Conclusion:
Computer-aided design and three-dimensional printing proved to be an effective method for designing, testing and fabricating a unique assistive device.
Clinical relevance
CAD and 3D printing techniques can enable devices to be designed, tested and fabricated cheaper than when using traditional techniques. This may lead to improvements in quality and accessibility.
Keywords
Background
Additive manufacturing (AM) is the process of joining layers of material to manufacture objects from three-dimensional (3D) model data. 1 AM, or 3D printing as it is commonly known, has been in existence since 1987 and was commercialised in the early 1990s. 2 Advantages of AM are documented as including elimination of cost and time for tooling, increased design freedom, reduced need for assembly, economic low-volume production and mass customisation.
The evolution of 3D printing into prosthetics has opened conversations about the availability, cost and design of prostheses. The benefits of AM when manufacturing prosthetic sockets was questioned by Jin et al. 3 who found that the cost involved in setting up AM may prevent the adoption of this technique. In recent years, a number of media articles have been reported where 3D printing has been used to create low-cost prosthetic devices; however, there is currently little scientific evidence about the robustness of these devices. Despite the high level of Internet and media coverage, little information can be found on prosthetic projects which involve both 3D printing and prosthetists. In addition, there is scarce evidence that 3D-printed devices are tested prior to being delivered to patients. This case report will discuss how a prosthetic team incorporated additive manufacture techniques into the treatment of a patient to create a unique prosthetic assistive device.
Case description and methods
This case report describes the process of designing, manufacturing and testing an assistive device for a patient with a partial hand amputation, which would enable him to play the French horn. This article focuses on the process undertaken rather than specific details about the device.
Playing the French horn is a bimanual task where the right hand is placed inside the bell of the horn to change the tone while the left hand is used to operate the valve keys, as well as to stabilise the instrument by placing the fifth finger under the finger hook. The patient for whom the device was made had undergone a disarticulation of his left fifth digit at the metacarpophalangeal joint following an unsuccessful fasciectomy to release a Dupuytren’s contracture. In addition, he presented with limited mobility in both hands due to other Dupuytren’s contractures. Upon assessment, the patient reported no pain and the scarring was healed. Since undergoing the amputation, the patient had been unable to stabilise his French horn and therefore could not participate in this leisure activity.
Prior to this intervention, the patient had tried a variety of commercially available assistive devices, which he had rejected as they involved making permanent or semi-permanent changes to the horn itself, were difficult to apply or were too bulky to fit into the musical instrument carry case. After a lengthy consultation, a list of design requirements was agreed which included: no changes to the subject’s playing posture/holding position; no permanent adaptations to the horn; and any solutions must fit within the instrument carrying case, be lightweight, and allow wrist movement.
Using these design requirements, a prototype assistive device was constructed from low-temperature thermoplastic (LTT). While this was too flexible to support the weight of the horn, the general design concept worked. Different materials and fabrication methods were considered. Due to the custom nature of the design, polylactic acid (PLA) and 3D printing were selected. The design, fabrication and testing process used is illustrated in Figure 1.
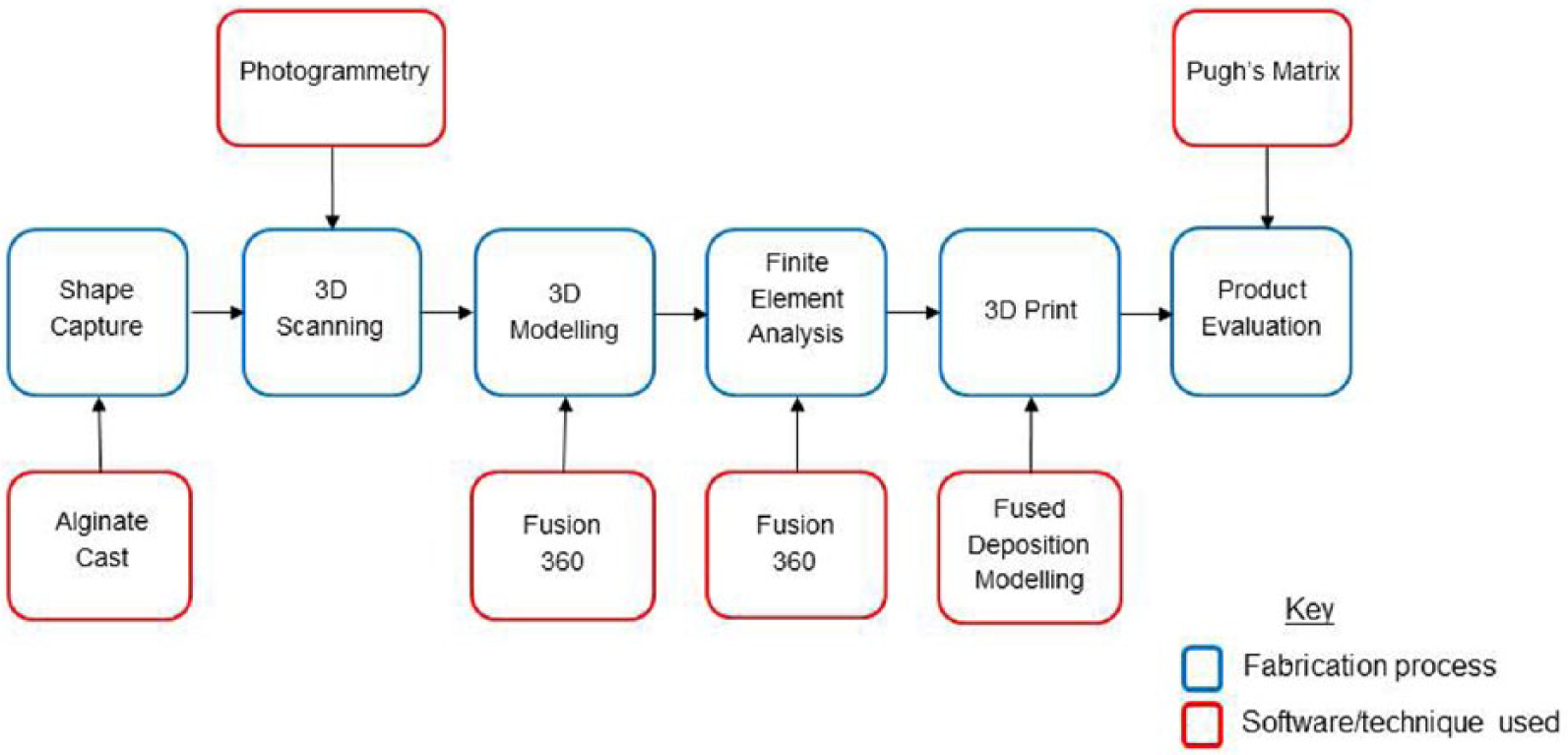
Design, fabrication and testing process.
Detailed shape capture was achieved using an alginate cast of the subject’s hand which was filled with Plaster of Paris and converted to a digital image using an Autodesk photogrammetry software called 123D Catch. This technique involved taking 18 photographs around the circumference of the cast; 12 photographs level with the base of the cast and six at an elevated height of 45°. The photographs were then uploaded to the software package and collated to give a 3D digital interpretation. The digital model was rectified using Meshmixer. The device was designed by the prosthetist using biomechanical principles to include a socket with an integral fifth digit which could hook around the finger rest of the horn. The design was then uploaded to Autodesk Fusion 360, and its structural integrity tested using finite element analysis (FEA). Considering the physical properties of the device and material specification, the displacement and static stress within the device were calculated by simulating the force which would be applied to the fifth digit by the weight of the French horn.
The device was printed on a Zmorph 3D printer using fused deposition modelling (FDM) technology. The material used was 2.85-mm diameter PLA, printed with a layer height of 0.2 mm and a 40% infill to reduce the weight of the device. The printer had a working temperature of 215°K, with the glass bed heated to 65°K.
The patient was recalled for fitting, at which point the socket fit was checked and the design was re-evaluated. The process was repeated using a further two different designs (Figure 2). The product, design and manufacturing processes were evaluated based on three criteria: structural integrity (FEA), patient satisfaction and cost.

Shows the three different devices designed: Device A, designed to have the palmar aspect left open with the main area of suspension around the pollex. With the presence of discomfort at the first web-space area, Device B was designed to wrap around the lateral aspect of the dorsal and palmar surface of the hand, with the suspension being offered by a slotted strap. To reduce bulk and improve suspension, Device C was designed to be donned like a glove with the suspension being in the form of circumferential containment.
Findings and outcomes
When the 25 N force of the French horn was applied to the model, the results showed a maximum displacement of 0.43 mm at the tip of the fifth digit which gradually decreased proximally. The result of the stress showed a maximum of 3.86 MPa on the fifth digit (Figure 3). After considering these results, the risk of failure was determined to be low. Similar results were found for the other two designs.

Results of finite element analysis.
A product evaluation form was supplied at the fitting stage, and the subject was asked to rate the devices in terms of fitting, comfort, function and cosmesis, with an option to add any of his own comments. The subject’s responses were inserted into a Pugh’s Matrix (Table 1), with the prototype LTT device being used as a baseline. A Pugh’s Matrix is a decision-making tool where important criteria are selected and alternatives are compared against those criteria. In this case, the patient scored his baseline device against the selected criteria using a scale of 0–5 and then scored the other devices as either the same, worse or better than the baseline. Responses for the three printed designs were compared against the baseline by totalling up the individual scores and weighted according to the absolute scale 4 with a higher score indicating improved satisfaction. The resulting Pugh’s Matrix showed that devices B&C were an improvement over the baseline, with the subject preferring the final design.
Pugh’s concept selection matrix showing results of patient satisfaction test.
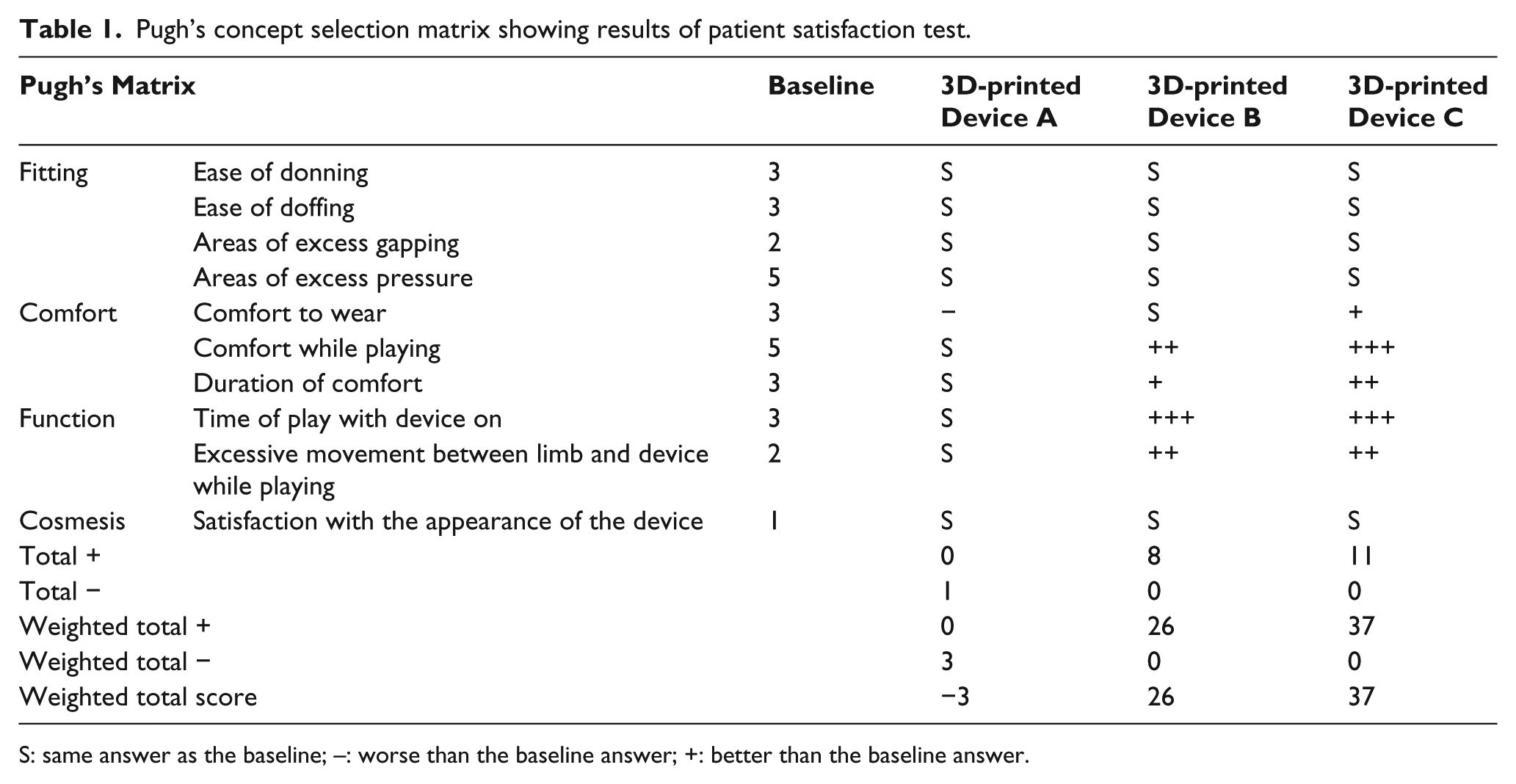
S: same answer as the baseline; –: worse than the baseline answer; +: better than the baseline answer.
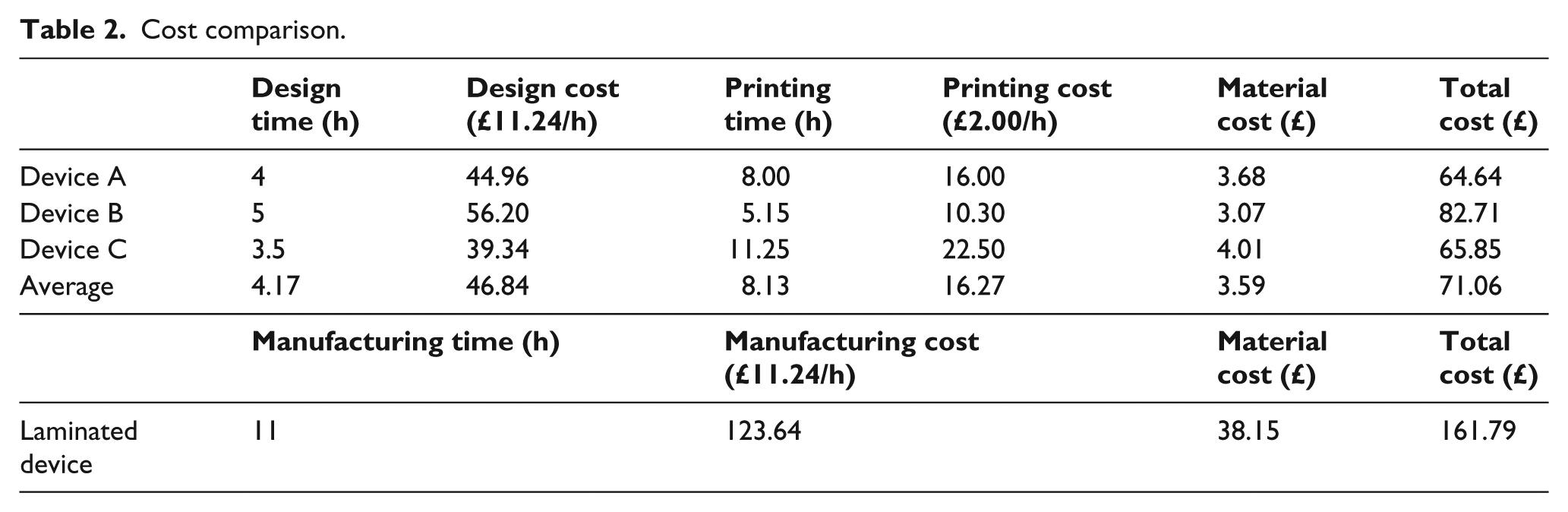
The cost of designing and printing the devices was calculated and compared to the cost of manufacturing a similar device using a lamination technique. The design time was calculated for each of the three devices as though each design had started from the unmodified Plaster of Paris model. Costs for the design and technical work were calculated using the Agenda for Change band 5 pay scale, which is commonly used for prosthetic technicians within the National Health Service.5,6 The material costs were calculated using information from regional suppliers. Printing costs were calculated by multiplying the print time by an hourly cost of £2.00 to cover the electricity and running costs of the printer. The time to print varied from 5.15 to 11.25 h according to the complexity of the design and orientation within the printer.
Table 2 illustrates that the average cost of the three printed devices was £71.06, which was 56% less than the cost for a similar device made from laminated resin.
Cost comparison.
Discussion and conclusion
Computer-aided design (CAD) has not yet been adopted as the preferred design method in prosthetics. This is partly due to the industry’s reluctance to move away from ‘hand skills’ to virtual design. Reasons given for not using CAD in prosthetics include inexperience, the cost-benefit ratio and a perception that it is difficult. 7 In this case report, we have demonstrated that with limited experience, and using online tutorials, it is possible for a willing person to master the basic skills of CAD. The process of teaching oneself basic computer-aided design and computer-aided manufacturing (CAD/CAM) skills took approximately 3 weeks and can be considered a one-time investment as the skills are transferrable to many applications and easily recalled at a later date. It should be noted, however, that for more complex designs, additional CAD expertise may be required.
Autodesk’s Fusion 360 software package was selected for use as it allowed free access and contained the features required to create the model, design and test the product. Other design software packages are available and may be better suited for creating more intricate devices.
One benefit of using CAD to create modified ‘casts’ is repeatability, as the design can be digitally stored and recalled at a later date if a remake is required. This can also reduce the issues of physical storage space often experienced by clinics. In addition, making changes to the cast or device prior to a remake can also simpler and quicker when using CAD than when using a physical cast.8,9
The shape capture process could be improved using direct scanning of the hand with the subject holding the instrument. However the scanning packages currently available to prosthetic clinics are not capable of capturing the hand in sufficient detail. Therefore, a recommendation from this case report is that scanning techniques, such as structured light or laser scanning, need to be improved so that accurate definition can be achieved in intricate areas such as the hand.
The ability to test the structural integrity of a device prior to fabrication is a useful component of the design process. While a risk assessment may be conducted, custom-made devices are rarely tested prior to being supplied to patients, and failures do occur. The regulation of custom-made medical devices is a complex issue which varies according to geographical location and governing bodies. The UK Government Medicines and Healthcare Products Regulatory Agency published guidance notes which state that it is the responsibility of the qualified person (e.g. medical professional, prosthetist or orthotist) to specify the design characteristics of a custom-made product and that the manufacturer must make documentation available which allows an understanding of the design, manufacture and performances of the product. 10 The ability to perform virtual tests on custom-made products can demonstrate conformity. In cases such as this, where the custom-made device is unique, a full risk assessment should be performed, informed by the physical limitations of the product. Using FEA, it was possible to predict the performance of the device by applying external forces to the design to expose areas of weakness. In this case, the results of the analysis were satisfactory; however, if any weaknesses had been discovered, the design could have been reviewed and retested prior to manufacture.
This study indicated that designing and fabricating a 3D-printed device was on average 56% cheaper than fabricating a similar laminated device. It is important to note when working from an already uploaded and modified scan, the time to remake a device would be significantly reduced, resulting in a further reduction of cost.
The aim of this task was to design and manufacture a device that would allow the patient to play his French horn once again, with the final indicator for success being patient satisfaction. The patient was impressed with the process and end product. This may, in part, be due to the novelty factor of receiving a 3D-printed device; however, he also commented about the lightweight of the device and improved cosmesis. Through involving the patient in the design process, clear objectives were set and the patient was delighted that he could once again play his horn comfortably and with ease.
Throughout this process, it was demonstrated that with limited prior knowledge, it was possible to use CAD software to design and test a device, and a 3D printer for manufacture. The experience has shown that 3D printing can be an effective method for supplying unique custom-made assistive devices. This project was patient-centred and prosthetist-led, which ensured that the resulting device met the patient’s expectations and that safety and quality of care were high. From this experience, it is recommended that prosthetists be involved in the development of 3D-printed prosthetic and orthotic devices and that relationships between AM groups and clinicians are further developed.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
This study was approved by the University of Strathclyde Department of Biomedical Engineering Ethics Committee (DEC/BME/2016/77).
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
