Abstract
Background:
After amputation, the brain is known to be reorganized especially in the primary motor cortex. We report a case to show changes in the corticospinal tract in a patient with serial bilateral transtibial amputations using diffusion tensor imaging.
Case Description and Methods:
A 78-year-old man had a transtibial amputation on his left side in 2008 and he underwent a right transtibial amputation in 2011. An initial brain magnetic resonance imaging with a diffusion tensor imaging was performed before starting rehabilitation on his right transtibial prosthesis, and a follow-up magnetic resonance imaging with diffusion tensor imaging was performed 2 years after this.
Findings and Outcomes:
In the initial diffusion tensor imaging, the number of fiber lines in his right corticospinal tract was larger than that in his left corticospinal tract. At follow-up diffusion tensor imaging, there was no definite difference in the number of fiber lines between both corticospinal tracts.
Conclusion:
We found that side-to-side corticospinal tract differences were equalized after using bilateral prostheses.
Clinical relevance
This case report suggests that diffusion tensor imaging tractography could be a useful method to understand corticomotor reorganization after using prosthesis in transtibial amputation.
Background
Diffusion tensor imaging (DTI), the summary of information about the degree and direction of water diffusion in multiple directions within individual voxels of magnetic resonance images, has been used to establish the integrity of the microstructure of white matter in various diseases that involve the central nervous system (CNS). A number of studies have demonstrated the usefulness of DTI for estimating the degree of neural injury, predicting motor outcomes, 1 and brain plasticity. 2
Recently, neuro-radiologic and neurophysiologic tools have developed markedly. With these advanced tools, various studies have been performed about specific conditions outside of the brain that could influence the brain’s structures and physiology. Spinal cord injury, which is one of those specific conditions, is known to induce reorganization of the cerebral cortex. 3 Amputation is also one of those conditions. Simões et al. 4 reported on the structural reorganization of callosal connections that was observed on functional magnetic resonance imaging (fMRI) and DTI analysis in nine lower limb amputees. Although the mechanism of reorganization in the CNS after amputation is not clear, it can happen because cortical reorganization can develop from the lack of afferent inputs through disuse or loss of function. 5 However, to the authors’ knowledge, only a small number of studies have been performed about the structural changes in the brain after amputation.
Here, we report the case of a bilateral transtibial amputee who showed changes in his corticospinal tract (CST) on DTI tractography after active ambulation using his prosthesis. In this study, the patient actively used his prosthesis after serial bilateral transtibial amputation. DTI tractography on bilateral CST was performed before and after providing the prosthesis to investigate the change of CST. We want to add the evidence of structural changes especially on CST after amputation using DTI tractography.
Case description and methods
The study protocol was approved by the institutional review board (IRB) of Chung-Ang University Hospital. Informed consent was confirmed by the IRB.
A 78-year-old man visited an orthopedic clinic with the complaint of gangrene on his right big toe in 2011. He had been a left transtibial amputee since 2008 because of diabetic arterial stenosis. The patient had also received a right second toe amputation in 2005. Although he was able to walk independently with his left transtibial prosthesis, he had frequent pain and small multiple wounds on his lower right extremity. Eventually, he underwent right transtibial amputation in 2011 and transferred to the rehabilitation department immediately after surgery. On physical examination, he was right-handed and had no neurologic weaknesses in his bilateral extremities, which were checked at the muscles proximal to the amputation sites. Before starting the rehabilitation on his right transtibial prosthesis, we performed the first brain magnetic resonance imaging (MRI), adding DTI to reconstruct his CST using Philips Achieva 3.0T TX (Koninklijke Philips Electronics N.V., Eindhoven, The Netherlands). The imaging parameters were field of view = 250 × 250, TR = 9213.9.0, TE = 70.0, 2.0 thk, and 1.0 NEX. The imaging data sets were transferred to a workstation (Extended MR WorkSpace 2.6.3.4, Koninklijke Philips Electronics N.V., Best, The Netherlands) to reconstruct the DTI tractography. The brain MRI showed that there were no abnormal findings such as parenchymal brain lesions or vascular abnormalities. For tracking, the upper and lower regions of interest (ROIs) were established as the upper and lower posterior limbs of the internal capsule on a fractional anisotropy (FA) color map, respectively. FA is an index of the degree of anisotropy of the biological tissue, therefore commonly used as an index of microstructural integrity. Only fiber tracking was allowed to pass through both ROIs. The FA and apparent diffusion coefficient (ADC) values of each CST and ROI were obtained. (ADC is an index of diffusivity of water.)
Findings and outcomes
There were more fiber lines in the right CST (227) than in the left (141) (Table 1 and Figure 1(a)). FA and ADC showed no differences between the right and left CSTs.
Diffusion tensor imaging variables of the patients in 2011 and 2013.
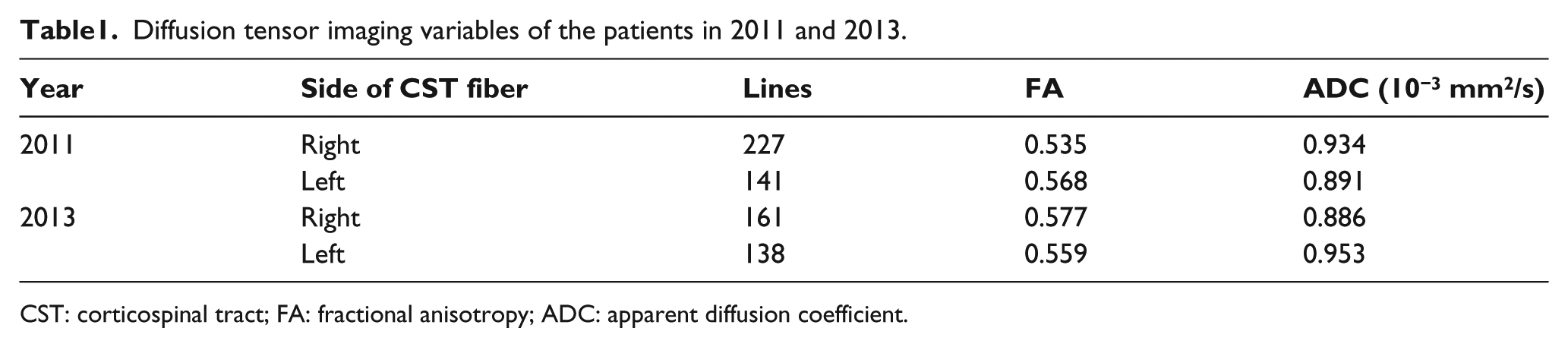
CST: corticospinal tract; FA: fractional anisotropy; ADC: apparent diffusion coefficient.
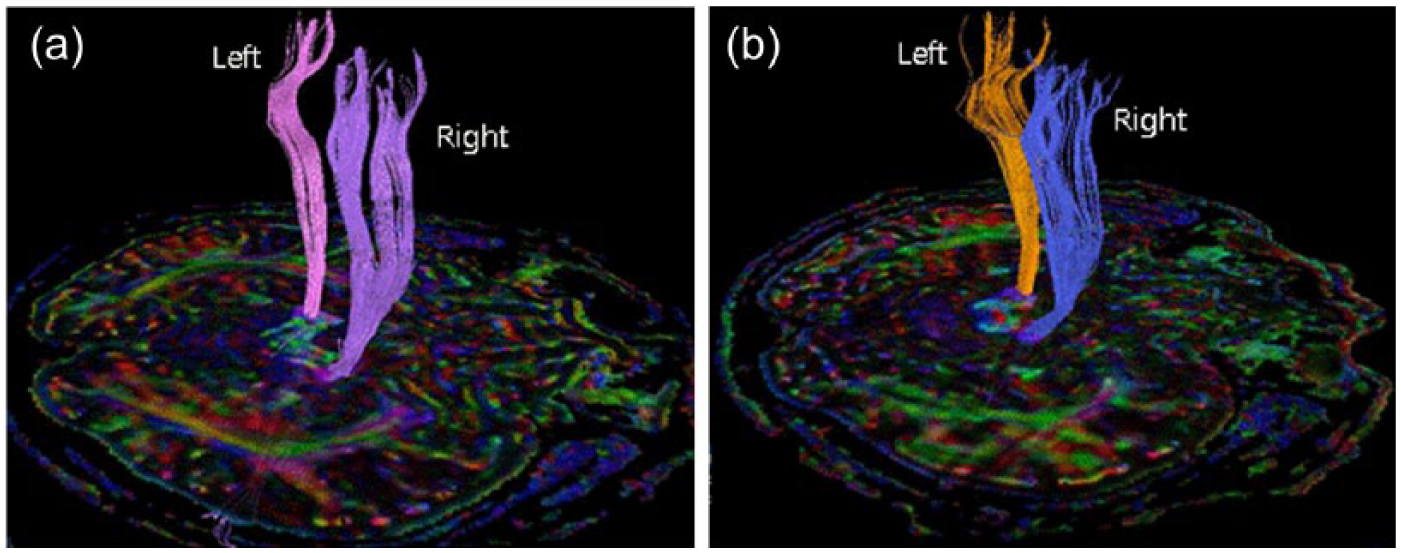
(a) Initial DTI (in 2011) showed that the number of fiber lines in right side CST was larger than that in left side. (b) Follow-up DTI (in 2013) showed decreased difference in number of fiber lines between the two sides of CST in tractography.
After an intensive rehabilitation program for 2 weeks, the patient could walk independently with a quadri-cane. He had experienced a transient phantom sensation until he grew accustomed to wearing his new prosthesis on his right leg. He was discharged from our clinic and went back to his home about 3 weeks after his second transtibial amputation.
He had regularly visited the rehabilitation clinic for 2 years, and there were no medical or surgical complications including his residual limbs (Figure 2(a)).
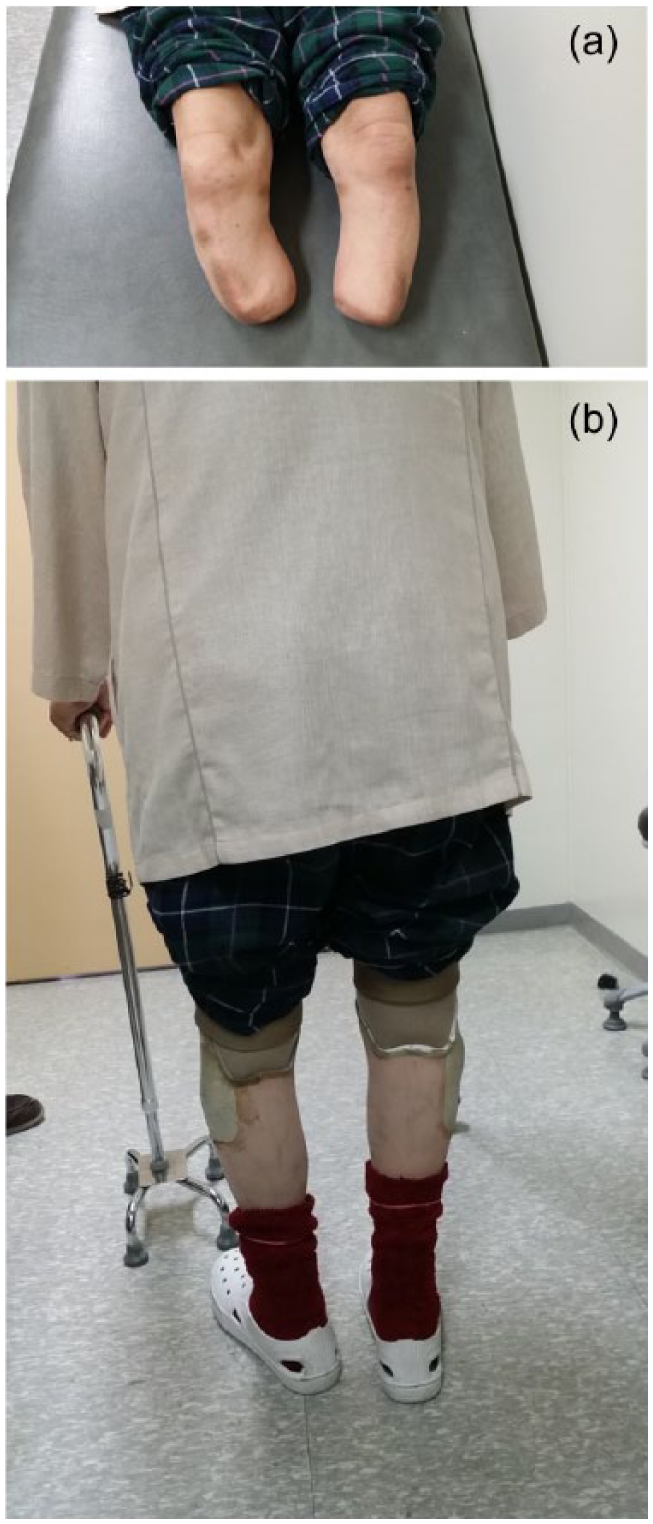
(a) Bilateral stumps of the patient were properly cared and showed no skin problem. (b) He was an active ambulator with bilateral transtibial prosthesis and single quadri-cane.
In 2013, the patient was still able to walk with a quadri-cane (Figure 2(b)), and a follow-up (second) brain MRI with DTI and tractography on bilateral CST was obtained using the same method. There were no parenchymal brain lesions on the brain MRI. In the DTI tractography, the number of fiber lines in the right CST was decreased (161) compared to the first DTI (227), whereas the number of fiber lines in the left CST was relatively unchanged (138). The difference in the numbers of fiber lines decreased (161 vs 138) compared with the result in 2011 (227 vs 141) (Table 1 and Figure 1(b)). FA and ADC showed no differences between the right and left CSTs (Table 1), and the values were not substantially different from the values in the previous study.
Discussion
Before the first DTI, we expected that there would be more fiber lines in the left (ipsilateral to first transtibial amputation) CST than on the right (contralateral to first transtibial amputation) because he had used his non-amputated right leg during ambulation more than his amputated side. However, the result showed that his right CST had more fiber lines than the left side. On the second DTI, difference in the numbers of fiber lines decreased compared with the result in previous study. With these two results, we presumed that the first result reflected that the patient had walked with his left prosthetic limb more than his non-amputated right limb to support his body weight after his left transtibial amputation in 2008. This might have been because he had repetitive problems in his right leg from the diabetic foot, which made him use his left prosthesis to support his weight during ambulation.
The second DTI, which showed a decreased difference in the fiber lines between both CSTs, reflected that the patient could walk with bilateral prosthetic limbs almost equally to support his weight after the right transtibial amputation in 2011. Although we could not confirm the exact location of the changes (myelin or axon), we can suggest that brain reorganization in the white matter was serially induced by the usage pattern of his extremities after wearing his prosthesis. There was no meaningful change or differences in either the FA or the ADC, but this result was expected because the patient had not developed any parenchymal brain lesions.
This is the first report of a serial follow-up of DTI tractography in bilateral lower limb amputation that shows the structural changes in the CST. One study showed the structural changes in the corpus callosum using DTI in amputees to determine the cause of the phantom limb sensation. 4 However, all of those subjects had either a cosmetic prosthesis or no prosthesis, which means that they were not able to use their prosthesis functionally. Other studies about motor system reorganization after amputation were usually performed using transcranial magnetic stimulation6,7 or fMRI. 8 Chen et al. 6 showed motor reorganization after lower limb amputation using transcranial magnetic stimulation and suggested that this kind of reorganization occurred at the cortical level. Hordacre et al. reported that amputees had greater excitability of corticomotor projections on the side ipsilateral to amputation. They showed increased step-time variability in amputee patients, although the participants were heterogeneous in terms of ambulatory function. 7 From this reference, we thought that gait mechanisms which influence neural plasticity would be very different from normal gait, in which using bilateral limb does not necessarily require neural control. In our study, we also expected higher functioning and increased fiber lines on the ipsilateral (left) side of the first amputation. However, our results showed higher fiber lines on the contralateral (right) side of the first amputation, maybe because the patient used his prosthetic limb to support his weight during ambulation rather than the intact limb.
One study with fMRI suggested that cortical activation of the amputees who had used their prosthesis actively was dominant in the cortex that was contralateral to the amputation side, whereas cortical activation of the non-active prosthesis user was dominant in the ipsilateral motor cortex when the patient moved the affected limb. 8 Although their approach and methods were very different from those of our study, the authors emphasized that corticomotor reorganization after amputation was influenced by prosthesis adaptation, which could support our results of the initial DTI. Our follow-up DTI, in which we observed a decreased difference in the fiber lines between both CSTs after active usage of bilateral prosthesis, would also support the importance of prosthesis adaptation.
In addition to corticomotor reorganization, somatosensory or thalamic reorganization after amputation has been reported by various studies that used neurophysiologic tools and in animal studies. 9 Because the loss of afferent input after amputation is known to influence cortical reorganization,5,9 the result of the first DTI tractography in our case might be explained by this principle. Recently, the spinothalamic tract and thalamocortical fibers were identified by DTI tractography in 23 healthy humans, 10 and additional investigations of these structures using DTI will give more information about brain reorganization after amputation.
There were two limitations in this study. First, we performed the second DTI tractography 2 years after the second amputation. The question could arise whether this 2-year period would be sufficient to confirm the change in fiber lines. There has been little study about the changes in white matter structures using DTI tractography in amputee patients. Only one study about brain plasticity in amputee patients examined the plasticity in white matter using DTI, and the time since amputation to DTI study was from 7 to 336 months. 11 In other studies of brain plasticity in amputee patients,4,6–8 the time since amputation was very diverse: 17.3 ± 9.9 years, 7 months to 53 years, 0.7–1.5 years, and 2–47 years, respectively. A study of age-related changes in CST fibers in normal populations without neurologic illness or brain lesions showed decreased fiber numbers in the 70s age group compared with the 20–50 age group. 12 Because we thought that amputation itself does not induce brain injury, this reference about the normal population is more meaningful, although there was no further reference about changes in CST in a relatively short period of time (2 years) in normal population.
The second limitation is that we only used fiber lines as the parameter of DTI tractography. Many others papers using DTI tractography have showed changes in fiber density or FA values in CST after brain injuries, and these studies performed follow-up DTI in relatively short periods of time: 9–10 months or even in 7 months.1,13 In these references about the brain injury, the FA value was effectively used to show the changes after brain injury because FA or ADC is known to effectively reflect the degree of Wallerian degeneration after neurologic injury. In our study, FA and ADC showed no difference or change, which was expected if we admit the fact that amputation itself is not related to brain injury. A study about brain plasticity in amputee patients used FA values in DTI tractography; 11 however, the age of subjects was from 18 to 60 years, and the time since amputation was very wide.
We report this case to initiate studies using DTI in amputee patients. Further studies might be needed in large number of amputee patients with similar levels of amputation and with the same level of prosthesis use.
Conclusion
We found that side-to-side CST differences were equalized after active use of bilateral prostheses in a patient with bilateral transtibial amputations. This case report suggests that DTI tractography could be a useful method to understand corticomotor reorganization after using prosthesis in transtibial amputation.
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
