Abstract
Background:
The purposes of this article are (1) to report on the overall desirability of the DEKA Arm by prototype and by level of prosthesis, (2) to report on user-perceived benefits of the DEKA Arm as compared to their current prostheses, and (3) to summarize user concerns about taking the device home.
Study design:
Qualitative content analysis of data from a multiple case study design.
Methods:
This study utilized data from 24 upper-limb amputees fit with a Gen 2 DEKA Arm and 13 fit with a Gen 3 DEKA Arm. Surveys were administered after fitting the DEKA Arm and at the end of training. Subjects recorded audiotaped comments about their experiences. All study sessions were videotaped.
Results:
In all, 79% of Gen 2 and 85% of Gen 3 users indicated that either they wanted to receive or might want to receive a DEKA Arm. In total, 95% of Gen 2 and 91% of Gen 3 prior prosthesis users reported that they were able to perform new activities that they were unable to perform with their own device.
Conclusions:
A large majority of subjects wanted a DEKA Arm, although desirability varied by amputation level.
Clinical relevance
The majority of amputees in this study expressed a desire to receive the DEKA Arm, a device which provides multiple powered degrees of freedom and is operated predominantly by foot controls. The majority reported functional advantages of the DEKA Arm over their existing prostheses.
Background
The Defense Advanced Research Projects Agency (DARPA) 1 launched its Revolutionizing Prosthetics program in 2006 and funded the development of the DEKA prosthetic arm system. After 2 years of research, DEKA Integrated Solutions Corp. (DEKA) had built and performed initial testing of the first two experimental prototypes: Generation 1 (Gen 1) and Generation 2 (Gen 2). In 2008, DEKA was charged with developing the next prototype (Gen 3), and the Department of Veterans Affairs (VA) and DARPA began collaborating on a joint study to optimize the DEKA Arm. VA Rehabilitation Research and Development funded the multisite VA Study to Optimize the DEKA Arm (Optimization Study). DARPA’s contract to DEKA provided funds to design and build the Gen 3 prototype and to support VA-funded usability studies of the Gen 2 Arm to inform the design of the Gen 3 prototype. During the study, DEKA made numerous iterative changes to Gen 2 features and software before major hardware and design changes were introduced in the Gen 3 prototype. (As of the publication date of this article, the DEKA Arm System is currently being reviewed by Food and Drug Administration (FDA) and has not received clearance to be commercially marketed.)
The primary objective of the VA optimization study was to assist DEKA in their efforts to optimize the DEKA Arm system. The purpose of this article is to report on the results of this effort. Specifically, this article will (1) report on the overall desirability of the DEKA Arm by prototype and by level of prosthesis, (2) report on user-perceived benefits of the DEKA Arm as compared to their current prostheses, and (3) summarize user concerns about taking the device home.
The DEKA Arm
A full description of the DEKA Arm and the Gen 2 and 3 prototypes are available elsewhere. 2 Briefly, the DEKA Arm is available in three configurations, or levels: radial configuration (RC) for transradial (TR) amputees, humeral configuration (HC) for transhumeral (TH) amputees, and shoulder configuration (SC) for TH amputees with very short residua as well as persons with amputations at the shoulder joint and scapulothoracic (forequarter) region. The two SC device prototypes are shown in Figure 1. The overall size of Gen 3 is smaller than Gen 2, the elbow and shoulder more contoured, and the hand and fingers are smaller looking and less blocky.

(a) Gen 2 SC on socket with harness; (b) Gen 3 SC on socket with harness.
The full SC DEKA Arm has 10 powered degrees of freedom and additional passive degrees of freedom.2–4 The SC version of the DEKA Arm employed Endpoint Control to enable simultaneous, coordinated movement of the prosthesis to bring the terminal device (the endpoint) to a desired position in space. All levels of the DEKA Arm use control inputs for the hand and wrist. At the HC and SC levels, the control scheme has dual modes enabling the user to switch between a “hand mode” of operation and an “arm mode” of operation (to control larger movements of the arm). Users control prosthetic movements with a customized combination of foot controls, myoelectrodes (electromyographies (EMGs)), pneumatic bladders, or other commonly available prosthetic input elements. There were three iterations of foot controls used in the VA Study to optimize the DEKA Arm: force sensitive resistors (FSRs), inertial measurement units (IMUs) during Gen 2 (IMU-1), and a refined version of the IMU in Gen 3 (IMU-2) which has new features including the ability to detect walking motion and automatically put the arm into standby (Walk Detect).
The DEKA Arm has six preprogrammed grip patterns (Figure 2) selected through one of two methods: direct selection or toggling. The DEKA Arm is battery powered by a rechargeable lithium-ion battery; an internal battery for HC and SC devices was introduced in Gen 3.

Six grips of the DEKA Arm (Gen 2 hand): (a) power, (b) tool, (c) chuck, (d) lateral pinch, (e) fine pinch open, and (f) fine pinch closed.
Methods
The parent study was a two-phase optimization study employing a multiple case study design and a mixed-methodology approach. 5 Phase 1 was testing the Gen 2 Arm; Phase 2 was testing the Gen 3 Arm. This article reports on a portion of the qualitative data analysis from the parent study. Data collection for this study took place between 2009 and 2012. The study coordinating site was the Providence VA Medical Center (PVAMC). Four clinical data collection sites included the NY VA Health Harbor System (NY VAHHS), the James Haley VA Medical Center, the Long Beach VA, and the Center for the Intrepid (CFI) at Brooke Army Medical Center. The protocol was approved by the institutional review boards for each of the clinical sites and the study coordinating center.
Sample
Subjects were purposefully sampled to include a range of participants who would use the three configurations of the DEKA Arm (RC, HC, and SC), males and females. Subjects were 18 years and older and single or bilateral upper-limb amputees at the TR, TH, shoulder disarticulation, or scapulothoracic level. They were recruited through clinicians, flyers and brochures, list-servs for prosthetic and rehabilitation staff, and press releases. All subjects provided their written consent.
Overview of study protocol
Study procedures were divided into two components: (1) fitting/setup and (2) training. Subjects were offered a variety of control options and the final control scheme was a collaborative decision made by the subject and prosthetist. They were initially familiarized with the arm controls by using the Virtual Reality Environment (VRE), an interactive computer software program. 3 The subjects were trained in the use of the DEKA Arm by the study therapist using a standardized protocol in 2-h training sessions. The study protocol dictated a maximum of ten 2-h training sessions for all levels of subjects, but was amended during the Gen 2 portion of the study to allow fifteen 2-h training visits for SC users when the need for additional training at that level was recognized.
Qualitative data collection
Baseline data on current prosthetic use and experience were collected at the first study visit. Subjects were asked to classify themselves as prosthetic users or nonusers and to state whether or not they considered themselves full-time users or part-time users. Subjects were also asked to report the number of years that they had used a prosthesis. Study surveys contained open-ended and closed-ended questions. Surveys were administered after fitting the DEKA Arm, after five sessions of training and at the end of training. When subjects terminated the study early, they were asked to complete the end-of-study survey. Open-ended questions asked about the overall impression and ease of use of the DEKA Arm. Both Gen 2 and Gen 3 participants were asked in the end-of-study survey whether they would like to receive a DEKA Arm in the future, what concerns they had about using a DEKA Arm at home, to list any new activities they had done in training with the DEKA Arm that they could not do with their current prosthesis, and their preference about doing activities with the DEKA Arm or their current prosthesis. Digital handheld micro-recorders were used to record comments before, during, or after study visits. All study sessions were videotaped.
Qualitative data analysis
Analysis of all qualitative data sources (audio, video, survey) was conducted by several team members. Each subject case was assigned to a primary data analyst who was responsible for tracking the subject and his or her progress through the study. Audio recordings were transcribed verbatim by a research assistant and transcripts were analyzed by the data analyst. The data analysts watched videotapes of all study sessions for their assigned subjects and documented key events and subject comments. The study principal investigator (PI) also viewed videotaped segments and read study transcripts.
At the completion of study activities, responses to the end-of-survey questions (Appendix 1) on device desirability, perceived benefits of the DEKA Arm, and concerns about home use were compiled and coded by the study analysts. Additionally, the subject comments comparing the device to their current prosthesis were identified from all data sources and comments coded and grouped into categories by the study analyst using a qualitative approach to content analysis. 6 The PI and analyst then met to discuss the coding and categorization of data and the subject’s comments, and reached consensus of categorization. In cases where two study analysts were involved in viewing videotaped data, both analysts were involved in construction of the case study.
To address the specific purposes of this article, we then constructed a cross-case analysis to compare users’ perspectives on desirability of the DEKA Arm, perceived functional benefits of the DEKA Arm as compared to their current prostheses, and concerns about home use. The cross-case analysis was facilitated by constructing a comparison matrix of subject responses by prototype and amputation level and placing the evidence within each category. Comparison matrices are an analytic tool used to visually display data in a systematic way. 7 Our analytical matrices contained key exemplars from qualitative data and responses to specific closed-ended questions. The PI verified all comparison matrices against the data to confirm the quantified numbers, coding of data, and interpretation of findings. The PI and analyst discussed any instances where their opinions differed and went back to source data to obtain additional information to support coding categories. The coding was discussed until consensus was achieved.
The accuracy and rigor of this analysis were enhanced by prolonged engagement with and persistent observation of subjects; triangulation of multiple data sources including video, audio, and written data; careful review; and debriefing of the analytical thought process.
Results
The flow of participants in each of the components of the study (Gen 2 and Gen 3) is shown in Figure 3. There were 26 fittings of the Gen 2 DEKA Arm. The first subject to participate in the Gen 2 portion of the study (an SC user) later repeated participation in Gen 2 using a different control scheme. Data from his first instance of participation were omitted from the cross-case analysis reported here. Thus, there were 25 unique participants in the Gen 2 portion of the study. One Gen 2 participant (an HC user) who dropped out of the study after two training visits did not complete the end-of-study surveys. Thus, this article reports on findings from 24 of the 26 trials of the Gen 2 Arm (8 RC, 7 HC, and 9 SC). Of the 24 Gen 2 subjects included in this analysis, 21 were current prosthetic users.
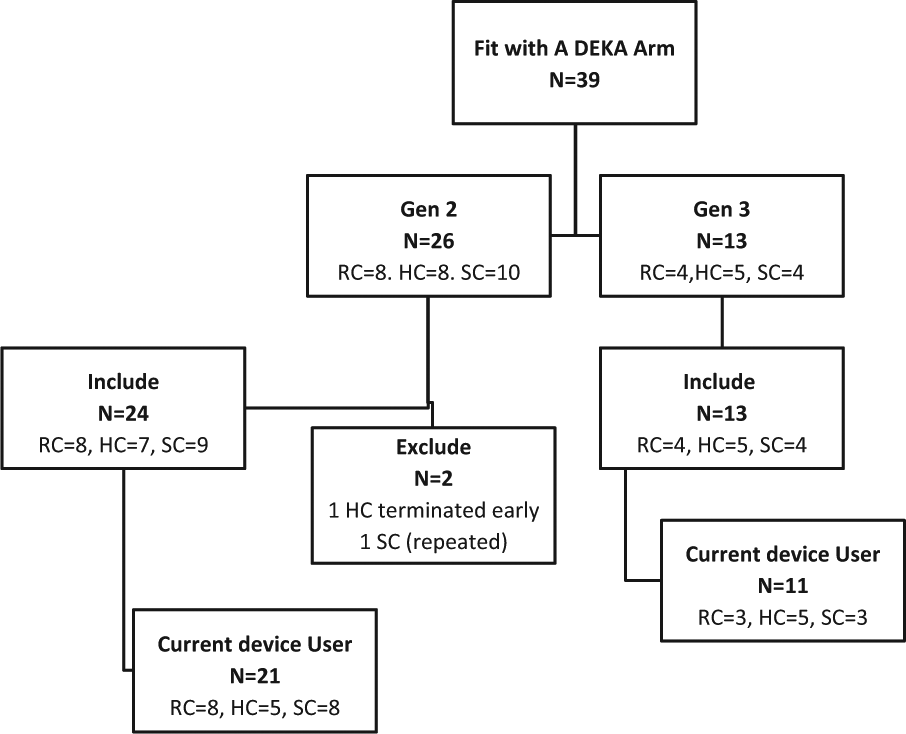
Flow of participants through study.
A total of 13 subjects were fit under the Gen 3 protocol (4 RC, 5 HC, and 4 SC). Five of the 13 Gen 3 participants (all male) also took part in the Gen 2 study (2 HC and 3 SC). Of the 13 subjects included in this analysis, 11 were current prosthetic users. All subjects included in this analysis completed the end-of-study surveys. Some subjects in the study had used a prosthesis for many years and were quite adept with it, some had basic training with their device, while others had no prior training. Table 1 shows characteristics of the subjects and training received by Arm prototype. Some subjects had less than the maximum number of training sessions because of scheduling problems or early termination.
Characteristics of all screened subjects.
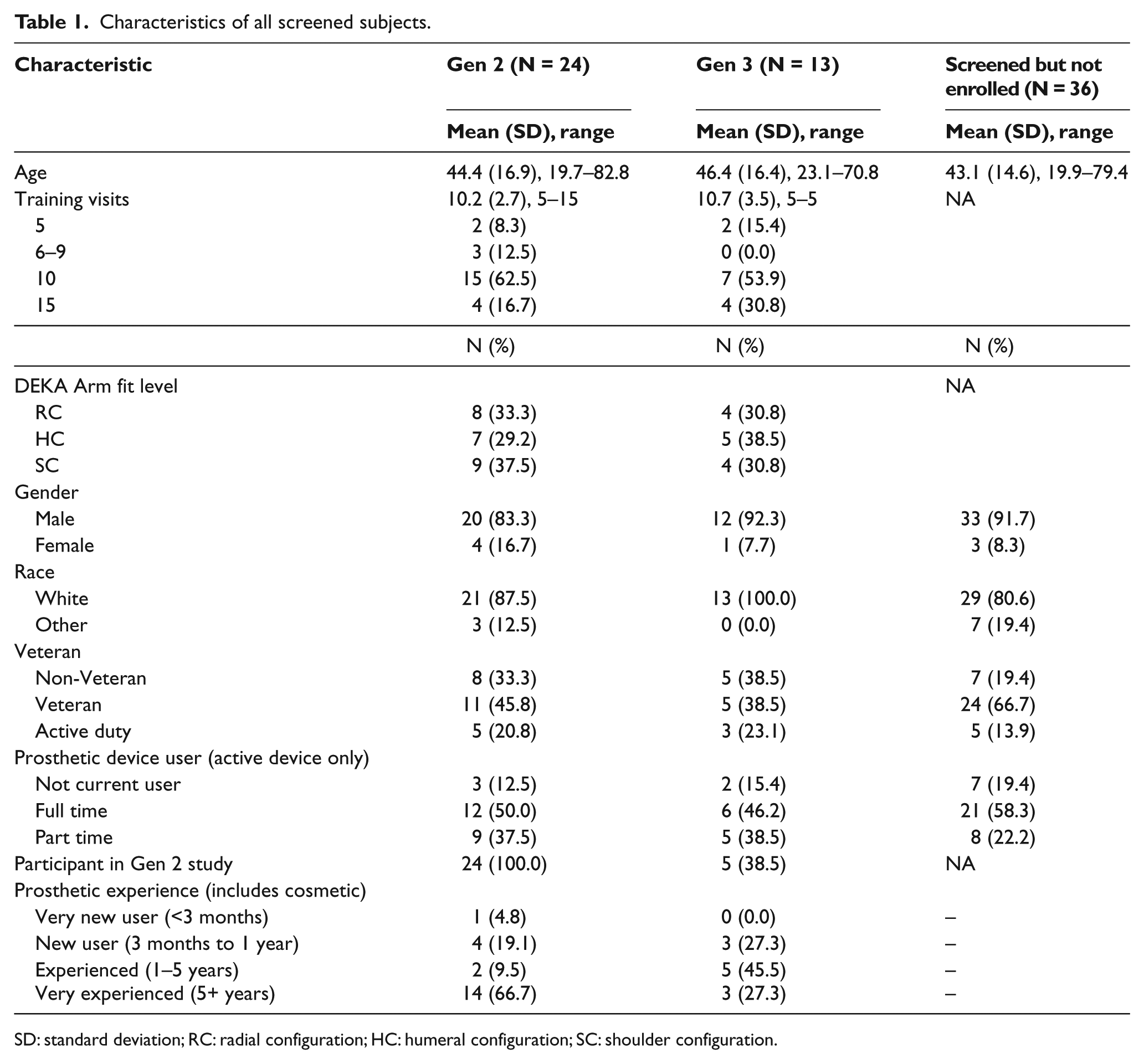
SD: standard deviation; RC: radial configuration; HC: humeral configuration; SC: shoulder configuration.
Desirability of the DEKA Arm
Nineteen (79%) of 24 Gen 2 users responded “yes” they wanted to receive a DEKA Arm in the future, 3 (13%) said “maybe,” and 2 (8%) said “no” they did not wish to receive one. This compared to 9 (69%) of 13 Gen 3 users who responded “yes,” 2 (15%) who said “maybe,” and 2 (15%) who said “no.” The response distribution by prototype and configuration level is shown in Figure 4. The desire to receive a DEKA Arm varied by level of the prosthesis fitted. Among Gen 2 users, eight (89%) of the nine subjects at the SC level said “yes” and one (11%) said “no”; six (86%) of the seven at the HC level said “yes” and one (14%) said “no”; five (63%) of the eight RC users said “yes” and three (38%) said “maybe.” Among Gen 3 users, four (100%) of the four of the SC level said “yes”; two (40%) of the four of the HC level said “yes,” two (40%) said “maybe,” and one (20%) said “no”; three (75%) of the four RC level said “yes” and one (25%) said “no.”
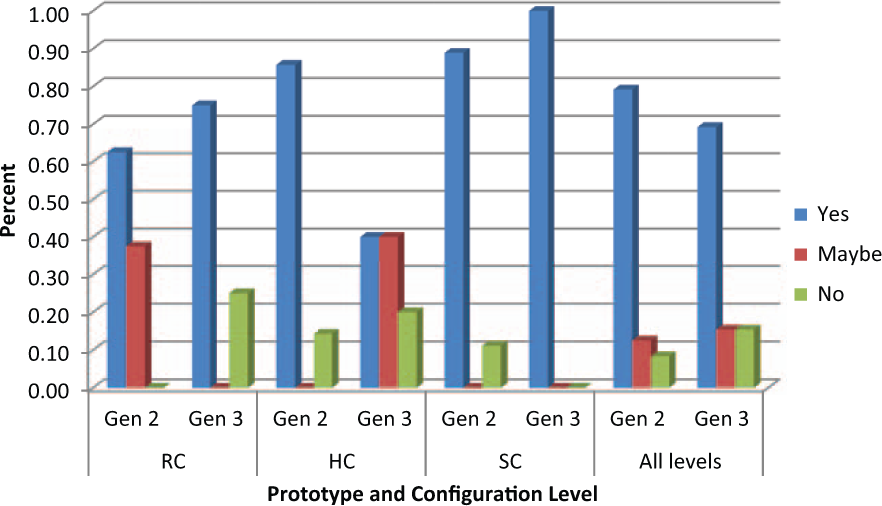
Desire to receive a DEKA Arm in future.
Among the 21 Gen 2 current prosthetic device users, 16 (76%) said “yes,” 3 (14%) said “maybe,” and 2 (10%) said “no” to wanting a DEKA Arm (Figure 5). This compared to 7 (64%) of 11 Gen 3 current device users who said “yes,” 2 (18%) who said “maybe,” and 2 (18%) who said “no.” Among the five Gen 3 subjects who also participated in Gen 2, all but one (80%) had the same response (yes) during both studies. One subject, an HC, had responded “no” during Gen 2 and “maybe” during Gen 3.
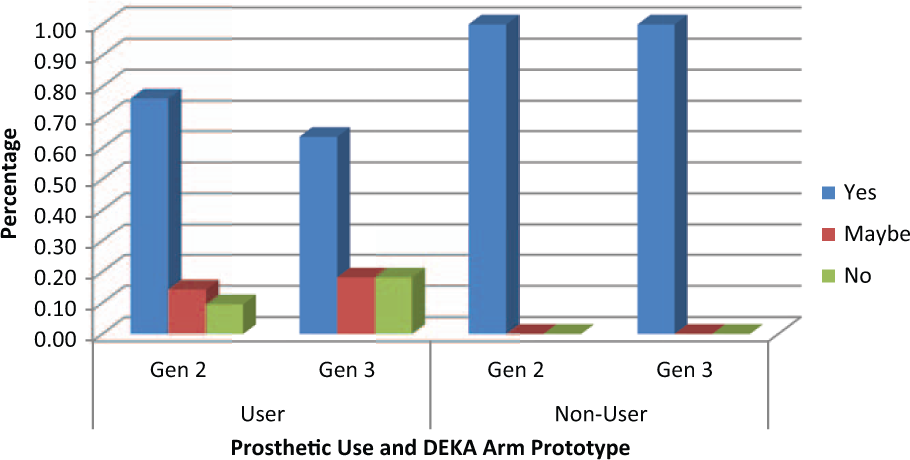
Comparison of prosthetic users’ and nonusers’ desire to receive a DEKA Arm.
All five participants who were not prosthesis users at the start of the study (three Gen 2 and two Gen 3) indicated that they wanted to receive a DEKA Arm. Among these there was a single subject (an SC) who participated in both Gen 2 and Gen 3. He was not a prosthetic user at the time of the Gen 2 study, but became a prosthetic user after the completion of Gen 2 study.
All five female amputees (four Gen 2 and one Gen 3, no duplicates) said “yes to wanting a DEKA Arm.” Fifteen (75%) of the 20 male Gen 2 subjects said “yes,” 3 (15%) said “maybe,” and 2 (10%) said “no,” while 8 (67%) of 12 male Gen 3 subjects said “yes,” 2 (17%) said “maybe,” and 2 (17%) said “no.”
Many explained that they wanted a DEKA Arm because of increased overall function, saying things like, “(it) will make everyday activities better” and “the functionality far surpasses anything that I’ve had so far.” Among the five Gen 2 users who either did not want a DEKA Arm or were not sure if they wanted one, weight was the most common reason; it was cited by three RC Gen 2 users. Other reasons included use of foot controls (one HC) and need for general further development (one SC). Among the four Gen 3 users who either did not want a DEKA Arm or were not sure if they wanted one, weight was cited as a reason by one HC user, one HC user said the device needed to be smaller with less wires, one HC user questioned functionality in comparison to their current prosthesis, and one RC user was concerned with reliability.
Comparison of the DEKA Arm to current device
Study participants who were prior prosthetic device users were asked to list any new activities that they could perform using the DEKA Arm that they were unable to do with their current prosthesis. Twenty (95%) of 21 Gen 2 current prosthetic users and 10 (91%) of 11 Gen 3 current device users reported that they were able to perform new activities. A single subject (HC level) who participated in both Gen 2 and Gen 3 reported that there were no new activities that he could do with either prototype. Illustrative responses by amputation level were grouped into six categories (Table 2). The most frequently mentioned types of new activities among Gen 2 subjects were in the categories of self-care mentioned by 14 (67%) of 21 users and everyday household/office tasks mentioned by 13 (62%). The most frequently mentioned types of new activities among Gen 3 subjects were in the categories of everyday household/office tasks, 5 (45%) of 11 users, followed by recreation, eating/drinking, and general abilities, each mentioned by 4 (36%). There were some variations in the kinds of activities by prosthetic level. For subjects who participated in the Gen 2 study, seven (88%) of eight SC users listed one or more self-care activities, compared to three (60%) of five of HC and four (50%) of eight RC. Among Gen 3 users, there were two (67%) of three SC users who listed one or more self-care activities compared to one (20%) of five HC users and zero out of three (0%) RC users. New general abilities were mentioned by 55% of all SC users (50% Gen 2; 67% Gen 3) and 50% of all HC users (60% Gen 2; 40% Gen 3) compared to 9% of all RC (13% Gen 2; 0% Gen 3). For both Gen 2 and Gen 3 RC users, the most frequently mentioned types of new activities were in the category of everyday household/office tasks, such as using a key, credit card, or combination lock.
Illustrative examples of new activities current prosthetic users reported with DEKA Arm by level.
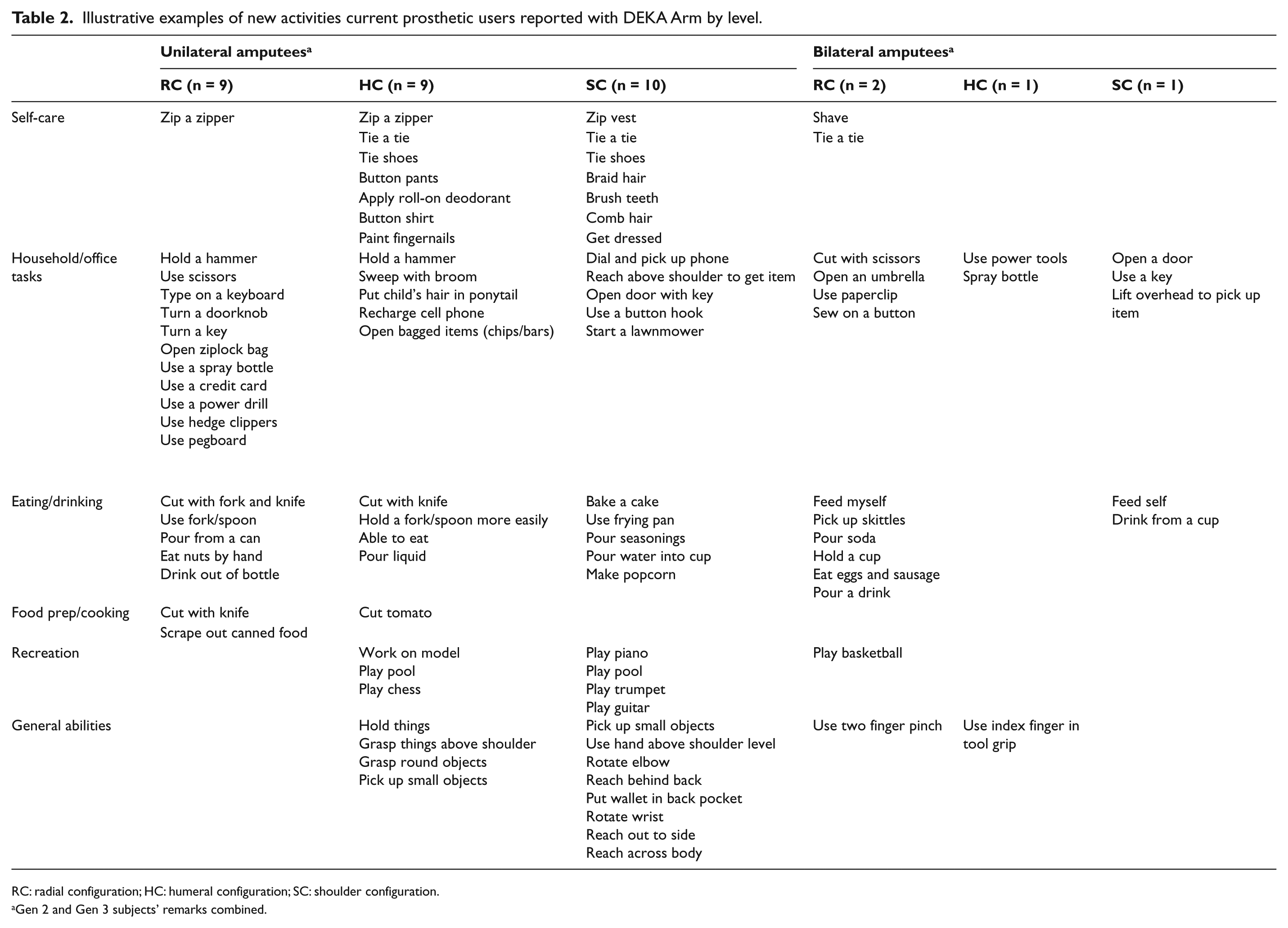
RC: radial configuration; HC: humeral configuration; SC: shoulder configuration.
Gen 2 and Gen 3 subjects’ remarks combined.
At the end of the study, participants were also asked if there were any activities that they could not do with the DEKA Arm that they were able to do with their current prosthesis. For both Gen 2 and Gen 3, the majority of users (76% Gen 2; 82% Gen 3) answered “no.” Those who did answer “yes” included one Gen 2 SC, two HC (one Gen 2 and one Gen 3), and four RC users (three Gen 2 and one Gen 3). Examples of activities from those who answered “yes” included wash myself, drive a car, ride a bike, pick up small flat objects, put on a sock, put on and take off the prosthesis, walk and use arm at the same time, and grip items with special terminal devices.
Near the end of the study, participants who were prior prosthetic device users were asked whether there were activities that they would prefer doing with the DEKA Arm instead of with their current prosthesis. They were also asked if there were activities that they preferred doing with their current prosthesis (Table 3). Among the 20 Gen 2 prosthetic device users who answered these questions (1 Gen 2 SC did not respond), 16 (80%) indicated that they preferred to do some activities with the DEKA Arm rather than their current prosthesis. A breakdown of prosthetic preferences with exemplars of preferred activities is shown in Table 3.
Prosthetic preference for specific activities.
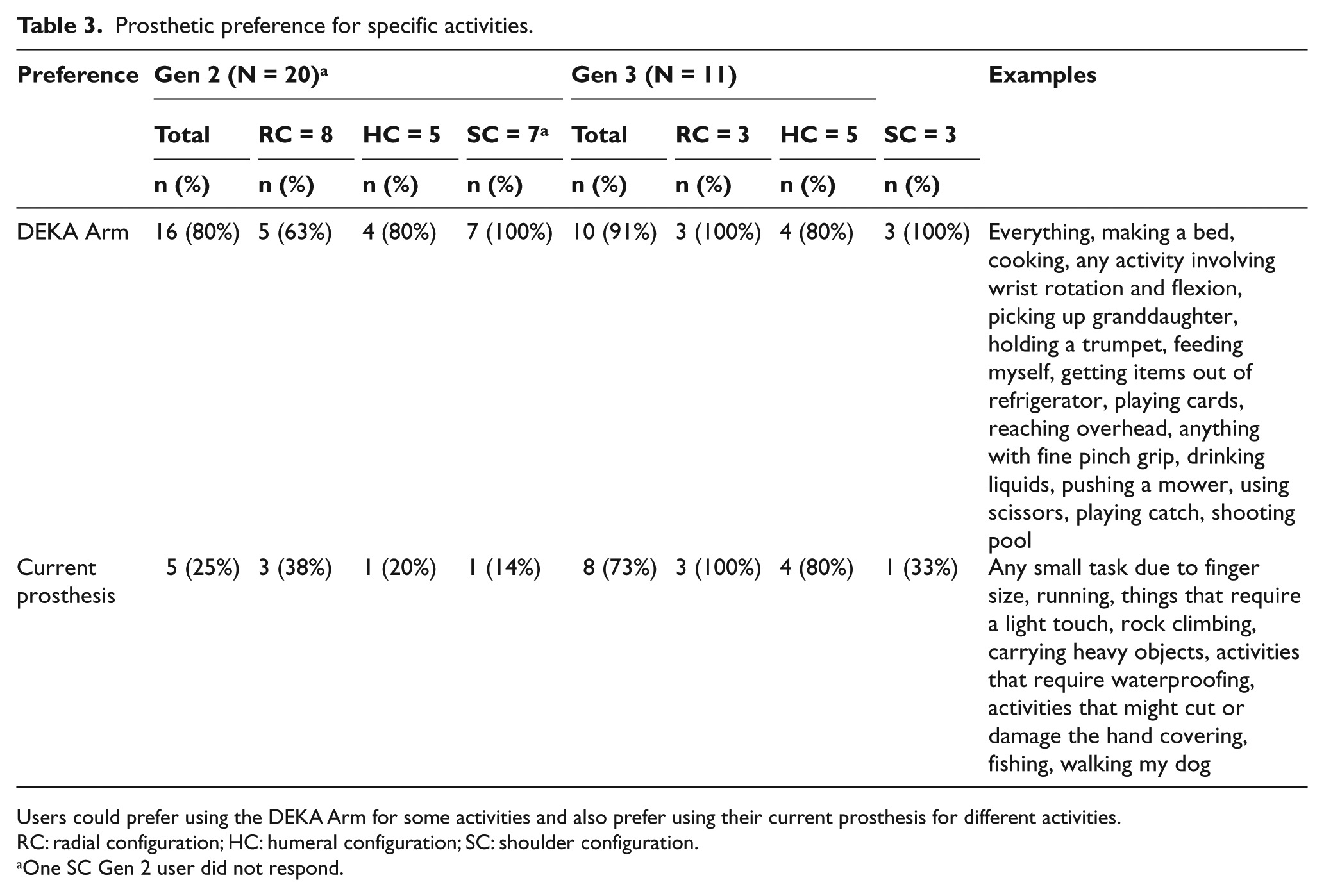
Users could prefer using the DEKA Arm for some activities and also prefer using their current prosthesis for different activities.
RC: radial configuration; HC: humeral configuration; SC: shoulder configuration.
One SC Gen 2 user did not respond.
Five (25%) Gen 2 users preferred to do some activities with their current prosthesis (one SC, one HC, and three RC). Ten of 11 (91%) Gen 3 prosthetic device users preferred to do some activities with the DEKA Arm compared to their current prosthesis. Eight of 11 (73%) Gen 3 prosthetic users preferred to do some activities with their current prosthesis (1 SC, 4 HC, and 3 RC).
All responding Gen 2 and Gen 3 SC users preferred the DEKA Arm compared to their current prosthesis to perform one or more activities. This contrasts with four (80%) of five Gen 2 and four (80%) of five Gen 3 HC users; five (63%) of eight Gen 2 and 3 (100%) of three Gen 3 RC users who expressed a preference for using the DEKA Arm instead of their current prosthesis for one or more specific activities. Only one (14%) of seven Gen 2 and one (33%) of three Gen 3 SC users indicated that they preferred to use their current prosthesis for one or more activities.
Among the four Gen 3 subjects who had also participated in the Gen 2 study as current prosthetic users, two (one HC and one SC) had a different response than in their previous trial, and two (one HC and one SC) had the same response: both who had a different response changed from not preferring some activities with their current prosthesis in Gen 2 to preferring some activities with it in Gen 3.
Comments made by each subject throughout the study that compared the DEKA Arm to their current prosthesis were compiled. Subjects were categorized as preferring the DEKA Arm, preferring their current prosthesis, or mixed preference/no opinion. Slightly more than half of prior prosthetic device users (11 (52%) of 21 Gen 2 and 6 (55%) of 11 Gen 3 users) expressed a preference for the DEKA Arm, even though most also had critical feedback and suggested improvements. Three (14%) of Gen 2 current prosthesis users and 2 (18%) of 11 Gen 3 current users clearly preferred their existing prosthesis. Finally, 7 (33%) of 21 Gen 2 and 3 (27%) of Gen 3 users had mixed preferences or no clear preference.
Advantages mentioned most often by those who preferred the DEKA Arm were related to its “superior functionality,” the utility of the six grips and joint movements, the sense that the prosthesis was “almost similar to having an actual arm,” and the fact that the device enabled them to do new meaningful activities. One joked, “The prior prosthetic I have is like having a Model T and this thing is like driving a Ferrari!” Those who preferred their current prosthesis said it was because their device was lighter, did not have foot controls, was more functional, and/or had a terminal device that they preferred to the DEKA hand. A few said they had developed proficiency over many years with their simpler device and did not want to take the time to develop equal proficiency with a more complicated prosthetic system.
Concerns about taking the DEKA Arm home
At the end of the study, participants were asked what concerns they had about using a DEKA Arm at home. The overwhelming majority, 20 (83%) of 24 Gen 2 users and 13 (100%) of 13 Gen 3 users, expressed some types of concern. Many subjects listed multiple concerns. These concerns are summarized in Figure 6. The most common concern in both Gen 2 and Gen 3 was breakage and need for repair, expressed by 10 (42%) of 24 Gen 2 users and 8 (62%) of 13 Gen 3 users. Users commented that they worried about “having it break down,” having too much “down time,” and the need for the device “to be shipped to be repaired.” The next two most frequently expressed concerns by Gen 2 users were weight and independent donning and doffing, cited by 6 (25%) of 24 users. This was followed by concerns about getting the device wet, cited by 5 (21%) of 24 users. No Gen 3 users mentioned weight as a specific concern for home use of the device; 2 (15%) of 13 Gen 3 users were concerned about independent donning and doffing; and 2 (15%) of 13 Gen 3 users expressed concern about getting the device wet.
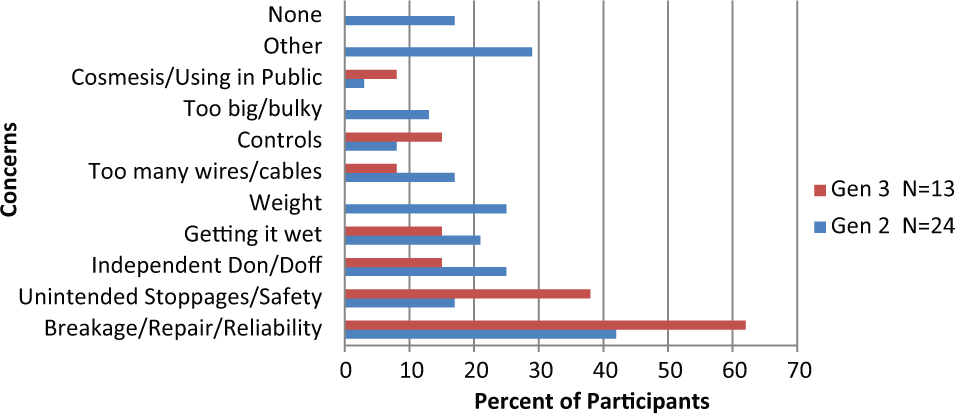
Concerns about home use.
Unintended stoppages, for example, “if the shoulder locked up or hand froze,” or faults that caused the prosthesis to not operate at all were the second most frequently expressed concern by 5 (38%) of 13 Gen 3 users and were also expressed by 4 (17%) of 24 Gen 2 users. Users were concerned that they would not be able to resolve these stoppages independently (“hard to figure out”) and were worried that they might need “either a help desk or help over the phone or computer.”
Certain aspects of the controls, the number of external wires and cables, the size and bulk of the device/accessories, the way the device looked, or how it could be heard in public were concerns mentioned by at least one Gen 2 user. At least one Gen 3 user also mentioned all of the above concerns, except the size and bulk of the device/accessories.
Discussion
This study reported on user impressions of the DEKA Arm, specifically their desire to receive one in the future, the perceived benefits of the DEKA Arm, and the concerns about home usage and compared impressions for the Gen 2 and Gen 3 prototypes. Our findings clearly suggest that the DEKA Arm is most highly desired by amputees at the shoulder disarticulation, forequarter level, and with very short TH residua—all of whom would be users of the SC DEKA Arm.
Users’ desire to receive a DEKA Arm was still strong for HC and RC users, but the proportion of users who clearly wanted to receive a DEKA Arm diminished as the amputation level became more distal. This may be explained, in part, by the complaints about the weight of the device, which was more common among users at the more distal levels. The SC socket was an X-frame which transferred the device weight (and the weight of any objects held by the arm) to the body. In contrast, weight of the HC and RC devices (and held objects) was supported by the residual limb and its musculature, and in some cases of HC devices, transferred in part, to the body through harnessing.
Clearly, the DEKA Arm offers strong advantages for users with more proximal level amputations. While there are powered elbows, and multi-degree-of-freedom prosthetic hands currently on the market, there are no powered shoulders and no devices with humeral rotators. SC users were able to utilize these new functions to perform self-care and eating activities that they were unable to do with their current prostheses. HC and RC users also reported that they were able to perform new activities with the DEKA Arm that they could not do with their existing prostheses, but these new activities most frequently fell into the areas of general (HC) or household and office tasks (RC), not basic tasks for self-care.
Our end-of-study survey asked participants to list up to five new activities that they were able to do with the DEKA Arm that they were unable to do with their current prosthesis. We do not believe that the list of new activities is exhaustive. Some subjects listed five activities, and others listed fewer than five. However, subjects were observed performing more than the activities listed. For example, no subjects at the SC level were able to cut with scissors with their existing prosthesis (a component of basis testing procedures), and all subjects at the SC level were able to cut with scissors using the DEKA Arm. However, no subject listed this activity as one of their five new activities.
These results need to be interpreted cautiously. We cannot conclude that these types of new activities could be performed with the DEKA Arm only and not with any other type of prosthesis. It is possible that some subjects might have been able to perform some of these activities using other types of prosthetic devices, or if they had additional prosthetic training with their current device.
Comparisons between the DEKA Arm and existing prostheses may have been affected by the control setup of the DEKA Arm, the amount of prosthetic and rehabilitation experience, type of prosthesis and/or terminal device used, and whether the current prosthesis was in need of repair. We were unable to control these factors in our analysis. Furthermore, some activities that users stated they would prefer to use their current prosthesis to perform were those that were restricted by study protocol and limited by the use of foot controls, such as driving a car or riding a bike. It is possible that some activities would be able to be performed with the DEKA Arm grasping an item, but turned off or put into standby to deactivate foot controls. Our study protocol did not allow subjects to attempt these types of tasks.
It is possible that subject’s attitudes toward the DEKA Arm were influenced by the intensive training provided and the attentiveness of the study personnel. However, all subjects were told that the purpose of the study was to obtain critical feedback on the device in order to refine it. Thus, research staff solicited both positive and negative feedback and suggestions for improvement on an ongoing basis.
That said, the study did not fit and train subjects with any other types of prosthesis, and thus, subjects were not able to compare the DEKA Arm to other prosthetic technology except what they already used. It is possible that if given other prosthetic options and trained in using them our subjects might have expressed a different set of preferences. Future studies could conduct head-to-head comparisons of patient preferences for a variety of technologies.
Users’ opinions were solicited throughout this study. Subjects were queried extensively about their experiences and they were asked to provide criticisms and constructive feedback to share with DEKA so that the device could be refined. Subjects were informed that they were using a preproduction device that was experimental and its final features and form had not been finalized. Subjects and staff were the “test pilots” for the rollout of new features, and they encountered numerous technical difficulties related to software and hardware glitches. It was common for the prosthesis itself or one of its components to be returned to DEKA during the study protocol for repair or parts replacement. Study prosthetists did not perform repairs. If prosthetists encountered technical problems that could not be resolved through telephone and/or video conferencing with DEKA, they needed to send the device back to DEKA for service. Many such technical support conferences took place with the subject present. Thus, it is understandable that the majority of subjects expressed concerns about device reliability and servicing.
In a clinical situation, prosthetists might manufacture dressing trees to assist higher level amputees with donning and doffing prostheses and might be able to spend more time to route the external cables and wires. However, in this study, which involved in-laboratory use only, this was not done. Therefore, it is not surprising that many subjects expressed concerns about independent donning and doffing. These concerns may have also related to the socket designs used for HC and RC level amputees, which fit more snugly, sometimes used inflatable bladders and were in some cases, difficult to don.
The results of this study may not be generalizable to a broader amputee population or to all amputees in the VA or Department of Defense (DoD). Subjects in our study were purposefully sampled to provide as much heterogeneity as possible at each of the three device levels. We tried to include bilateral and unilateral amputees as well as members of both genders. We believe that our subject pool represented the diversity of the upper-limb amputee population; however, the composition of our sample may have not been representative of broader populations.
Conclusion
Despite concerns about home usage, the majority of subjects in our study indicated that they desired to receive a DEKA Arm in the future. Desirability of the DEKA Arm varied by amputation level. A higher percentage of subjects who used the SC level clearly want a DEKA Arm as compared to users at the HC and RC levels. This desire is explained, in part, by the new self-care and eating activities that SC users could perform using the DEKA Arm as compared to their current prosthesis.
Footnotes
Appendix 1
Acknowledgements
The authors acknowledge the valuable work of study coordinator Kate Barnabe and members of the research teams at all study sites:
NY VAHHS: Christopher Fantini, Kenneth Breuer, Roxanne Disla, MaryAnne Garbarini, Dan Burgos, Edward Sliwinski.
Tampa: Melanie Harris, Samuel Phillips, Laurel Adams-Koss, Deborah Gavin-Dreschnack, Jemy Delikat, Jill Ardilla, Andrea Spehar, N. Joseph Shamp, Steve Doerr.
CFI: Ryan Blanck, Kathryn Korp, Sandra Jarzombek, John Fergason, Christopher Ebner, COL Jennifer Menetrez, Donald A. Gajewski.
Long Beach: Dana Craig, Susan Kaplan, Karen Duddy, Jack Mark, Dorene Doi, Mary Jo Van Duyn, Duane Sallade.
PVAMC: Susan Rizzo, Marcia Selinger, Crystal Davis, Debra Kelty, Shana Klinger, Katherine Etter, Matthew Borgia, Marissa Meucci.
Funding
This research was supported by VA RR&D, VA RR&D A6780 and VA RR&D A6780I DEKA’s support of the VA optimization studies was sponsored by the Defense Advanced Research Projects Agency and the U.S. Army Research Office.
Conflict of interest
The information in this article does not necessarily reflect the position or policy of the government; no official endorsement should be inferred.
