Abstract
Background:
Vertebral fractures are the most common clinical manifestations of osteoporosis. Vertebral fractures and reduced back extensor strength can result in hyperkyphosis. Hyperkyphosis is associated with diminished daily functioning and an increased risk of falling. Improvements in back extensor strength can result in decreased kyphosis and thus a decreased risk of falls and fractures.
Objectives:
The aim was to examine the effects of an active spinal orthosis – Spinomed III – on back extensor strength, back pain and physical functioning in women with osteoporotic vertebral fractures.
Study design:
Experimental follow-up.
Methods:
The women used the active spinal orthosis for 3 months. Outcomes were changes in isometric back extensor strength, changes in back pain and changes in physical functioning.
Results:
A total of 13 women were included in the trial. Wearing the orthosis during a 3-month period was associated with an increase in back extensor strength of 50% (p = 0.01). The study demonstrated a 33% reduction in back pain and a 6.5-point improvement in physical functioning. The differences in pain and physical functioning were borderline significant.
Conclusion:
The women demonstrated a clinically relevant improvement in the back extensor strength. The differences in pain and physical functioning were clinically relevant and borderline significant.
Clinical relevance
The results imply that Spinomed III could be recommended for women with vertebral fractures as a supplement to traditional back strengthening exercises. It is essential that the orthosis is adjusted correctly and that there is an individual programme concerning the amount of time the orthosis has to be worn every day.
Introduction
Osteoporosis is a systemic skeletal disease characterized by low bone mineral density (BMD) and structural changes in bone tissue. 1 The most common clinical manifestation of osteoporosis is vertebral fractures. 2 In the general population, approximately one-fourth of women aged 50 years or above have one or more vertebral fractures. 3 Vertebral fractures and reduced back extensor strength can result in hyperkyphosis. 4 For both men and women, the combination of sarcopenia and reduced physical activity can lead to the development of fragile bones and an increased risk of falling. 4 Women in early adulthood have lower muscle strength 5 and BMD 6 compared with men and thus are more vulnerable to changes in muscle and bone mass. Reduced BMD and decrease in back extensor strength can cause hyperkyphosis with or without the presence of vertebral fractures. 4 Hyperkyphosis is associated with diminished daily functioning,7,8 decreased quality of life,7,9–12 reduced lung capacity 13 and increased risk of falls. 14 Medical treatment is essential in people with osteoporosis; however, it cannot stand alone when it comes to preventing fractures. The vast majority of fractures are caused by falls, and it is thus important also to reduce the risk of falls. 4 There is evidence that physical exercise increases BMD, while the combined training of the trunk muscles and balance training prevent the risk of falling and new fractures. 15 Back extensor strength has been shown to play an important role in preventing fractures. 12 The degree of thoracic kyphosis in women with osteoporotic fractures may be influenced especially by changes in back extensor strength. It has been suggested that as long as the back extensors are strong enough, spinal deformities will not appear despite decreased BMD. 16 An improvement in back extensor strength reduces the degree of kyphosis, thus reducing the risk of falls and fractures in women with osteoporosis.17,18 A supplement to the recommended back extensor strength exercises is use of the active orthosis Spinomed III. This orthosis consists of a back brace and a belt system and can be worn on top of the clothes (Figure 1). Spinomed III is claimed to increase proprioceptive feedback in back and abdominal muscles, thereby keeping the spine in an upright position. Spinomed III thereby differs from traditional, rigid spinal orthoses. The use of rigid thoracolumbar braces in osteoporosis is limited by factors such as the atrophy of trunk muscles and restricted respiration leading to low compliances. 19
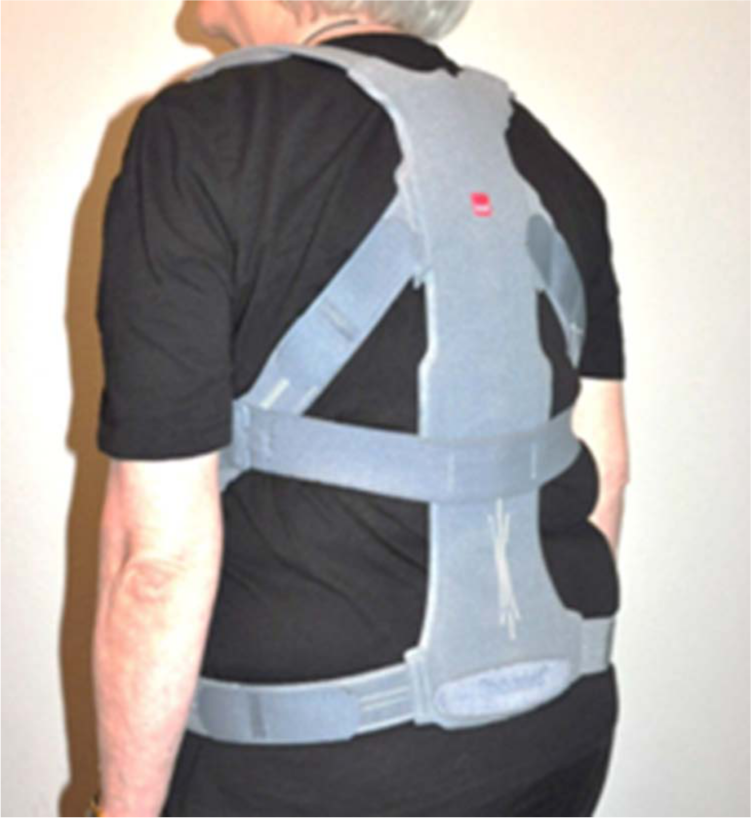
Spinomed III consists of a back brace and a belt system. The active orthosis is adjusted individually to the patient by a physiotherapist or orthopaedic technician.
The effects of Spinomed have earlier been examined in two randomized controlled trials (RCTs) conducted by the scientists who developed the orthosis. In the first trial from 2004, the authors found a 73% increase in maximal isometric back extensor strength, a 38% reduction in chronic back pain, a 27% reduction in limitation of daily activities and a 15% increase in quality of life over a period of 6 months. 3 The results from the second trial in 2011 were almost identical with the results from 2004. 19
The aim of this study was to examine the effect of Spinomed III on back extensor strength, back pain and physical functioning in a population of women with osteoporotic vertebral fractures.
Materials and methods
Design
The project was an experimental follow-up study.
Study population
Participants were women aged above 50 years receiving standard medical treatment for osteoporosis. The inclusion criteria were as follows: minimum one X-ray verified thoracic low-energy fracture with height loss of at least 20% of the front of the corpora and persistent back pain lasting at least 3 months prior to baseline. Exclusion criteria were as follows: severe exacerbation of back pain within 6 weeks prior to baseline and/or new onset of X-ray verified fractures, hyperparathyroidism, hyperthyroidism, major co-morbidities such as malignancies of the spine, neurological diseases, severe congenital scoliosis or inability to put on Spinomed III.
Data collection
The project was approved by The Danish Data Protection Agency. The Regional Scientific Ethical Committee stated that the project was not notifiable. All investigations were in accordance with the protocol and followed the ethical and humane principles of research. Women were recruited through a list of past participants in an osteoporosis programme. Data were collected from medical records and radiographs. The women who met inclusion criteria were provided with written information about the project including a questionnaire. All participants gave written informed consent prior to participation.
At baseline, women were given verbal information about the project. The women completed questionnaires about clinical characteristics, pain intensity and health-related quality of life (Short Form Health Survey–36 (SF-36)). Finally, back extensor strength of each participant was measured. At the second appointment, Spinomed III was adjusted and participants were instructed on the use of the orthosis. Participants recorded daily use of the orthosis and pain intensity once a week in a diary. The women were instructed to wear Spinomed III for 15 min a day in the first 14 days to avoid straining the muscles. In the next 14 days, the use of the orthosis progressed with up to 2 h daily, depending on individual physical abilities. In the following 8 weeks, participants wore the orthosis for 2–4 h daily. Participants underwent inspection and adjustment of the orthosis after 2 and 6 weeks, respectively. Participants were contacted by phone every 14 days to ensure compliance. In several cases, additional adjustments were made to ensure optimal fitting of the orthosis. The project was completed after 3 months with follow-up tests. The data collection took place from June to October 2011.
Test procedures
Tests were conducted in accordance with standardized procedure. The same equipment and sequences were used at baseline and at follow-up.
Back extensor strength
The maximal isometric muscle strength of the back extensors was measured with a handheld dynamometer (PowerTrack II Commander).
Testing procedures were standardized and the dynamometer was calibrated before each test. Each testing day was initiated by a short warm up of the back muscles.
The test position recommended by Limburg et al. 20 was used. The handheld dynamometer was placed on the midline between the two angulus superior scapulae. There was one warm-up trial followed by three test trials lasting 5 s, with a 60-s pause between each test trial. If the last measurement was more than 5%, higher than the second to last, another attempt was made. The highest value of the three tests was presented. An improvement of back extensor strength by 30% was considered clinically relevant. This was based on findings from other trials indicating that an increase in back extensor strength by 20%−50% was associated with an improvement in quality of life by 10%−12% in the study population.3,12,19
Back pain
An 11-point numerical ranking scale (0–10) was used to measure pain intensity. Information on the use of the scale was given at baseline. The women reported pain intensity at baseline and at follow-up, and weekly in the diary.
Pain intensity was assessed as an average of three questions about pain intensity to increase validity. 21 Based on literature, a reduction in pain by 30% is clinically relevant in patients with chronic back pain. 22
Physical functioning
The SF-36 health survey was used to examine physical functioning. SF-36 is a generic questionnaire on overall health status covering the physical, mental and social functioning. 23
Statistical analysis
Double data entry was performed to validate data. All statistical analyses were done in STATA 12. Baseline data and differences in muscle strength tests between baseline and follow-up were calculated. Since data could not be described as a normal distribution, the median and range were presented, and Wilcoxon signed-rank test was used to assess statistical significance in differences. The differences between muscle strength tests were presented as absolute and relative differences. Changes in physical functioning were evaluated by the physical component score (PCS) from SF-36. Data were analysed using the intention-to-treat principle.
Results
Participants
Figure 2 shows a flow chart of the participants in the trial. A total of 13 women were enrolled at baseline. One participant stopped using the orthosis after 10 weeks due to persistent pain in the thoracic spine, which was exacerbated by the use of Spinomed III. Control X-rays showed no sign of new fractures, but showed further collapse of existing fracture in the thoracic spine.

Flow chart of the participants in the trial.
Table 1 presents the study population characteristics. Figure 3 illustrates back extensor strength for all included women at baseline and at follow-up.
Baseline characteristics.
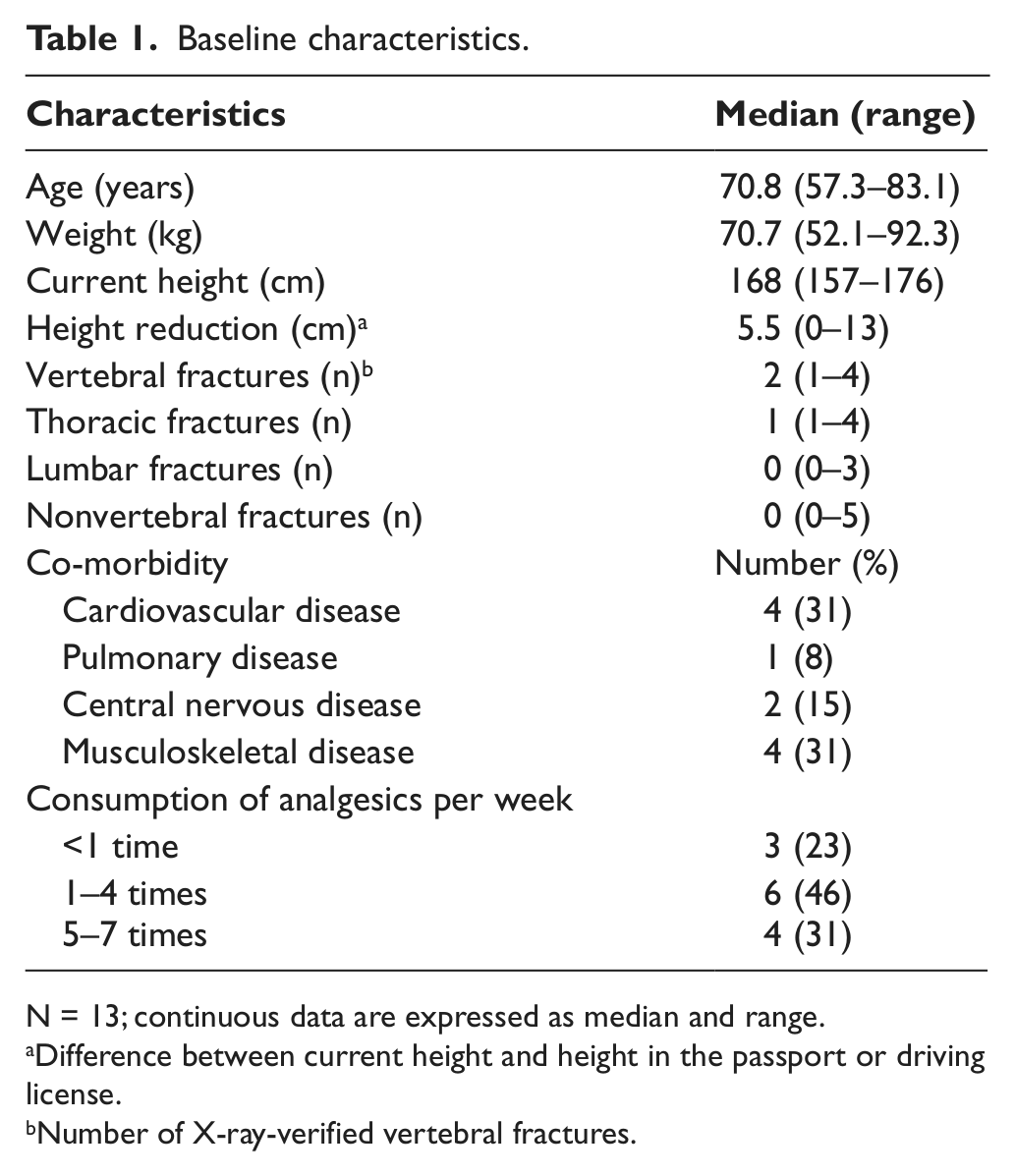
N = 13; continuous data are expressed as median and range.
Difference between current height and height in the passport or driving license.
Number of X-ray-verified vertebral fractures.
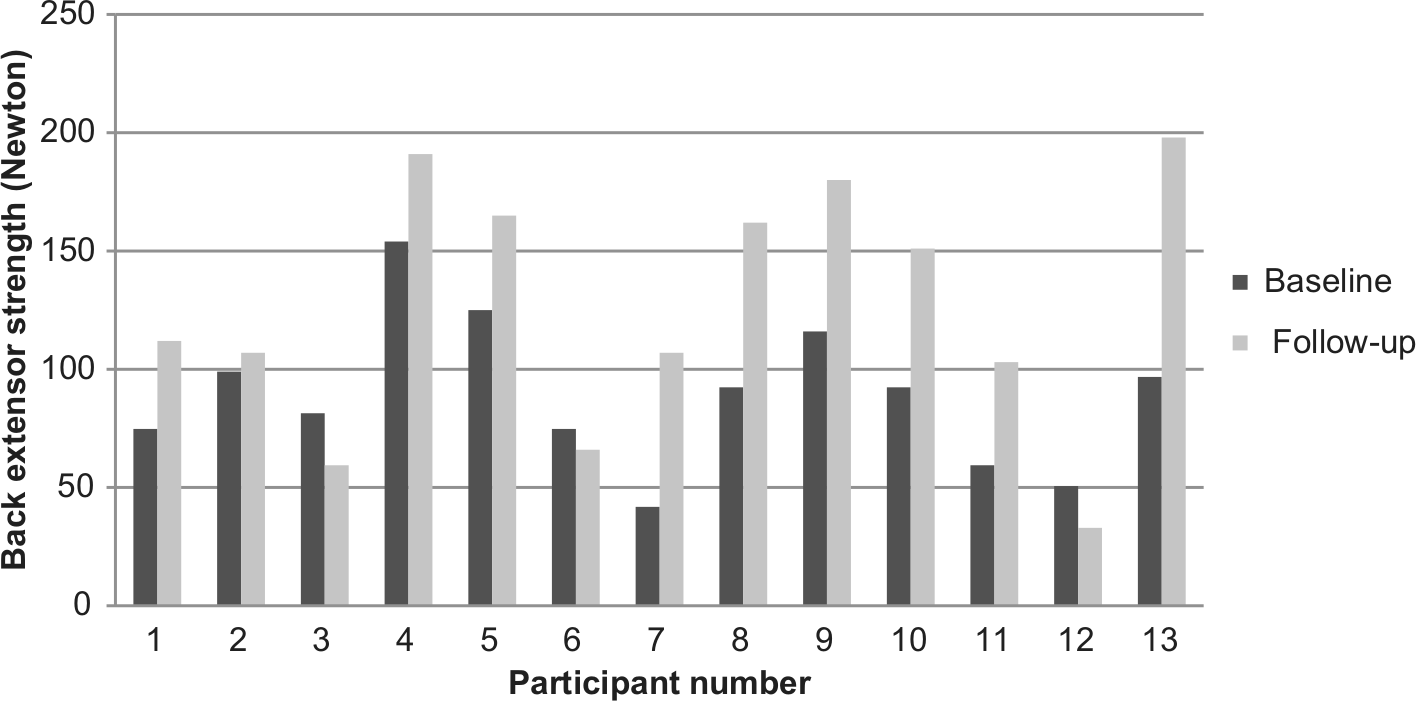
Back extensor strength at baseline and at follow-up.
Back extensor strength at baseline and at follow-up is presented in Table 2. The median difference between follow-up and baseline was 40 N. The relative median difference in back extensor strength was 50%. Eight of the 13 participants had a clinically relevant improvement in back extensor strength. The difference in back extensor strength between baseline and follow-up was statistically significant (p = 0.01).
Back extensor strength at baseline and at follow-up.

N = 13; data are expressed as median (range).
Wilcoxon signed-rank test.
Figure 4 illustrates back pain for all included women at baseline and at follow-up. There was considerable variation in the development of pain. At the end of the trial, a reduction in pain intensity was seen in nine of the women.
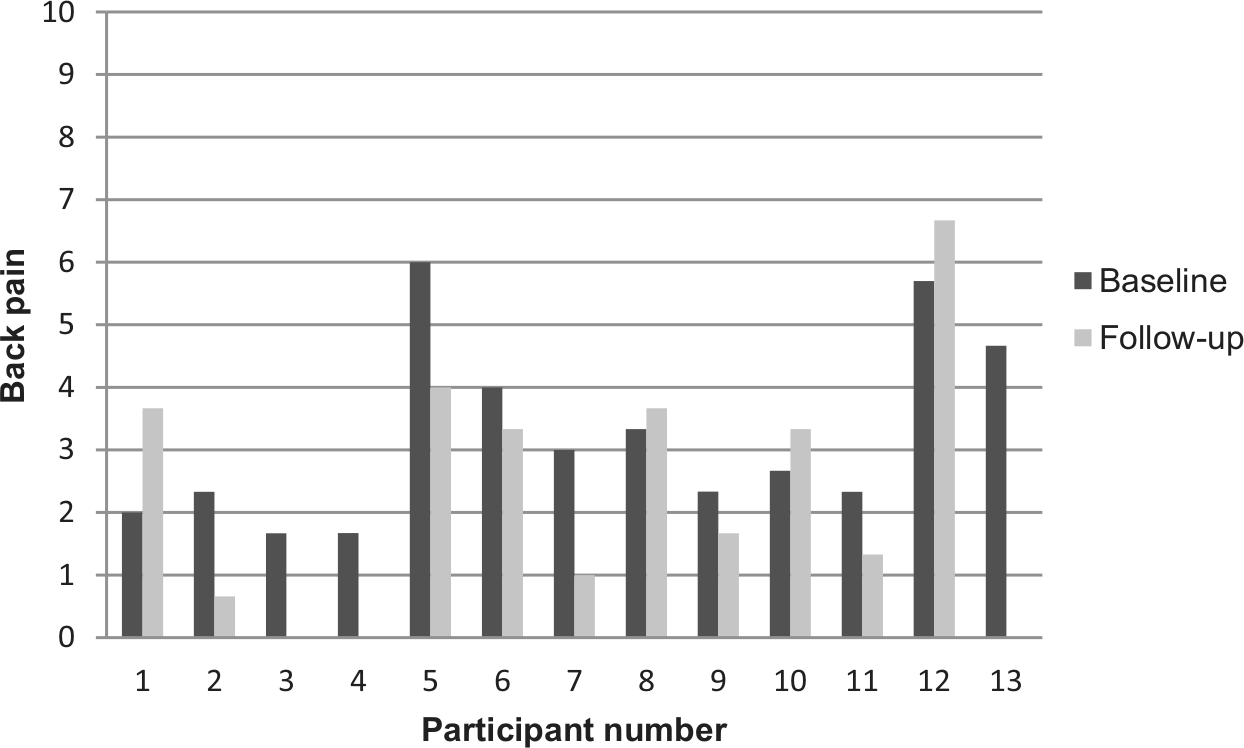
Back pain at baseline and at follow-up.
Table 3 illustrates that the median difference in pain from follow-up to baseline was 1 point, which corresponds to a relative reduction of 33%. The reduction of back pain was considered clinically relevant. Statistically, the difference was borderline significant.
Pain at baseline and at follow-up.

N = 13; data are expressed as median (range).
Wilcoxon signed-rank test.
Table 4 illustrates the differences between follow-up and baseline for the eight health domains of the SF-36 and the two component summary scores : physical component score and mental component score. Table 4 shows an improvement in physical component score of 6.5 points at follow-up. The difference was not statistically significant (p = 0.07).
SF-36 physical and mental functioning at baseline and at follow-up.
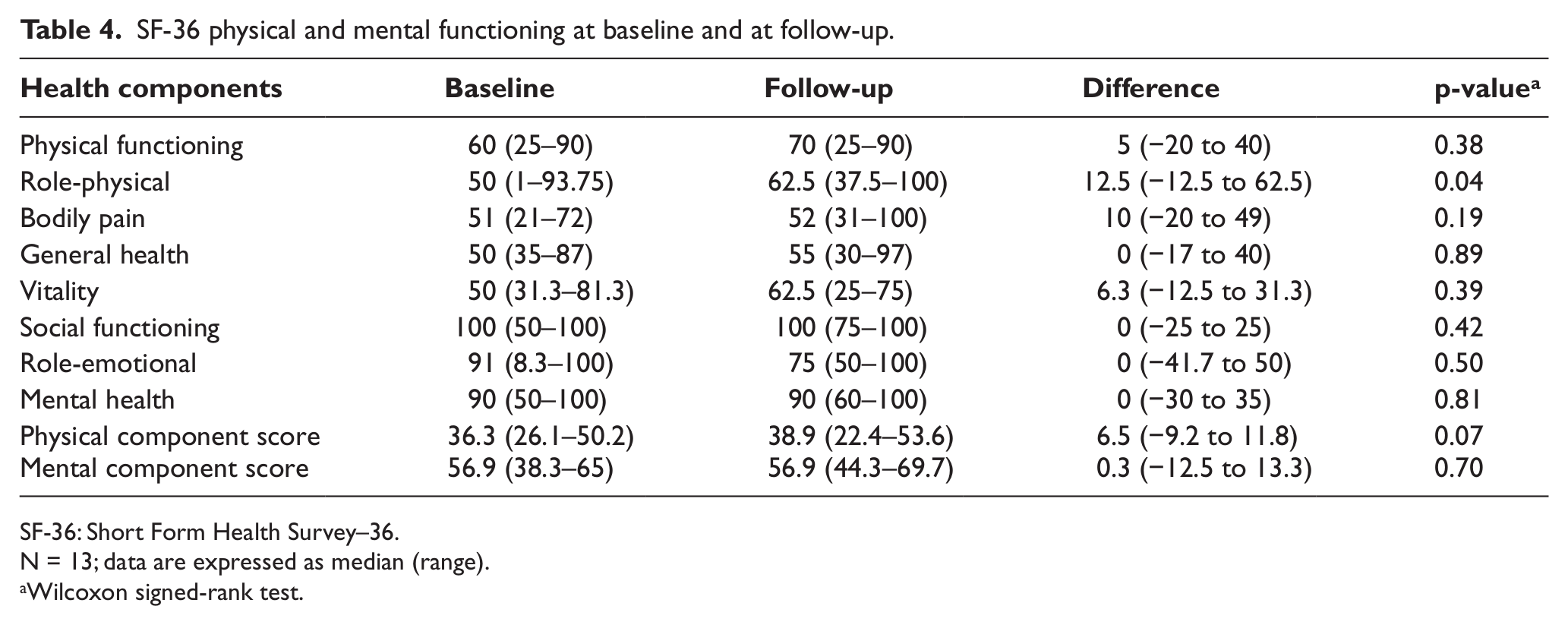
SF-36: Short Form Health Survey–36.
N = 13; data are expressed as median (range).
Wilcoxon signed-rank test.
Discussion
The main findings in this trial were a clinically relevant and statistically significant improvement in back extensor strength after a 3-month use of Spinomed III. The observed differences in pain and physical functioning were clinically relevant and borderline significant. With a sample of 13 people, the risk of type II errors was high. There was a large biological variation among the women, with a particularly large variation in age, number of fractures, back extensor strength, pain and physical and mental abilities. There were also substantial differences in the effect of the orthosis. Due to the low number of participants and the large biological variability in this study, the results should be interpreted with caution. In this study, two-thirds of the women had a clinically relevant improvement in back extensor strength after a 3-month use of the orthosis. The considerable changes in muscle strength in this group suggest that use of the orthosis has a positive effect on back extensor strength in women with osteoporotic vertebral fractures. The reduction in pain and the improvement in physical functioning were close to significant. A larger study population could perhaps prompt statistically significant differences. There were five women in the study with no clinically relevant improvement in back extensor strength. Two of these women had poor walking abilities, which meant that they were not able to be physically active while wearing Spinomed III. This may explain the lack of effect on back extensor strength in these participants. One participant had Scheuermann’s disease and she was unable to position her back in an upright position. Therefore, she did not activate her back extensors while using the orthosis, which could explain the lack of efficacy. One participant scored 8% higher in muscle test at follow-up. The observed change of 8% was not considered to be a clinically relevant change in muscle strength. The reason for the lack of efficacy is unknown. The last participant of the five had an improvement of 24%. She stopped using Spinomed III after 10 weeks due to persistent pain in the thoracic spine, which could explain the relatively low improvement in the back extensor strength. The results of this study imply that Spinomed III could be recommended for women with osteoporotic vertebral fractures. The following describes our experiences from the study with a view to possible future implementation of use of Spinomed III. If Spinomed III were to be recommended as part of the rehabilitation of women with osteoporotic vertebral fractures, it is important to identify those who can benefit. This study found no effect in women with poor walking abilities. It seems important that the patient is able to stand or walk for a longer period of time. Furthermore, it is important that women are able to actively and passively adjust for the increased thoracic kyphosis of the spine. A large proportion of women in the project mentioned that the orthosis was not comfortable to wear during activities like cleaning and gardening. Experiences from this trial indicate that Spinomed III is suitable for use in activities such as walks, cooking or ironing. It is essential that Spinomed III is adjusted correctly and that there is an individual programme for each patient concerning the amount of time the orthosis has to be worn every day. The patient must be prepared for muscle soreness in the beginning of the process. Two earlier studies have examined the effect of Spinomed.3,19 These studies showed a 73% improvement in back extensor strength after 6 months. Most of the improvement occurred during the first 3 months. The observed improvement in back extensor strength of 50% in this study is consistent with these results. Pain reduction in the studies was approximately 30% after 3 months.3,19 This study found a median pain reduction by 33%, which is also consistent with the two former studies. The RCTs found a reduction in limitations in daily activities of approximately 20% over 3 months.3,19 In our study, we looked at physical functioning, and thus it was not possible to compare these two results. It may be noted that the health component role-physical of the SF-36 in this study showed a significant difference. Overall, there was high agreement with the results of this pilot study and the RCTs.
Conclusion
The primary hypothesis was that use of Spinomed III would result in a clinically relevant improvement in back extensor strength in women with osteoporotic vertebral fractures. At follow-up, the median difference in improvement in muscle strength was 50% (p = 0.01); two-thirds of the women in the project had obtained an improvement in back extensor strength of more than 30%. The observed improvement in back extensor strength in the women was clinically relevant. The secondary hypothesis was that women would have a clinically relevant reduction in pain and a clinically relevant change in their physical functioning using Spinomed III. The study showed a reduction in pain of 33% and an improvement in physical functioning of 6.5 points. The observed difference in pain and physical functioning were clinically relevant and borderline significant. The results are based on a small study population, and the effect of Spinomed III has to be tested in a larger study sample.
Footnotes
Conflict of interest
The author declares that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
