Abstract
Background: Using the new modular socket system (MSS) to produce a prosthetic socket directly on the patient has the potential of being easier and quicker to manufacture but also incurring higher costs.
Objectives: The purpose of the study was to compare the costs of manufacturing a transtibial prosthetic socket using either a MSS or a standard laminated socket (PC).
Study design: Concurrent controlled trial.
Methods: A total of 20 patients at two orthopaedic facilities were followed with regards to the cost of manufacturing a prosthetic socket using either MSS or PC. Time aspects and material costs were considered in the cost analysis. Other factors studied include delivery time and number of visits. For the cost analysis, only direct costs pertaining to the prosthetic socket were considered.
Results: The total cost of MSS was found to be significantly higher (p < 0.01) compared to PC. However, the production and time cost was significantly lower. Delivery time to the patient was 1 day for MSS compared to 17 days for PC.
Conclusions: Our study shows that the direct prosthetic cost of treating a patient using MSS is significantly higher than treating a patient using PC. However, the MSS prosthesis can be delivered significantly faster and with fewer visits. Further studies taking the full societal costs of MSS into account should therefore be performed.
This study shows that the direct prosthetic cost of treating a patient with Modular Socket System is significantly higher than treating a patient with plastercasting with standard laminated socket. However, the Modular Socket System prosthesis can be delivered significantly faster and with fewer visits.
Introduction
Prosthetic sockets have for a long time been manufactured based on a plaster cast of the patient’s residual limb followed by rectification of the plaster cast and production of the final socket on the plaster positive. This is a rather time-consuming process and requires one or more revisits to the prosthetic facility by patients before the socket can be delivered for use. When the method of producing the socket directly on the patient was introduced with the ICEX system several years ago, one could therefore assume that it would lead to a change in production method. However, at least in the Nordic countries, the predominant method of producing prosthetic sockets is still based on a plaster cast. There are several possible reasons for this, but one that often is pointed out is the higher cost of ICEX compared to the plaster cast method. This increased cost has been confirmed in the few studies that have investigated the costs of ICEX. 1,2 The increased cost coupled with no significant difference in function 1,2 implies that the use of ICEX based on current evidence could, as Datta 1 concluded, be questioned.
Also, as the demand for evidence is increasing, 3 and cost-effectiveness has become a key criterion for decision-makers when deciding which health-care interventions should be made available in collectively funded health-care system, 4 the need for a cost comparison when changing to a new technology is of present interest. Should the cost of producing the socket directly on the patient be made more equal to that of a prosthesis produced from a plaster cast, and the immediate delivery of the prosthesis maintained, an increase in use could perhaps be expected.
The new modular socket system (MSS) is based on the same principal as ICEX of producing the socket directly on the patient. The MSS production procedure has a slightly longer curing time and uses injection moulding instead of the prepreg used in ICEX. MSS operates under the same basic principle of direct manufacture under pressure. However, the resin carbon is replaced by glass fibre, and rather than material pre-impregnated with resin, lamination is achieved by injecting the resin into the material. 5 For MSS a glass fibre braid with distal attachment is applied directly to the patient’s stump between two layers of silicon. The resin is then injected between the two silicon layers and the socket set under pressure with an Icecast.
The function and fit could be expected to equal that of the ICEX method. One major change with the introduction of MSS compared to ICEX is the cost of the material used. As the material cost was found to be the biggest disadvantage in earlier studies, it is therefore interesting to investigate the cost of MSS compared to sockets produced after a plaster cast (PC). Thus the aim of this study was to perform a cost analysis comparing the costs of MSS and PC.
Methods
Study design
Two prosthetic facilities in Sweden, one using MSS and one using PC, participated in the study. The two facilities were selected based on two criteria: long experience with their prosthetic system, and having a large number of prosthetic patients. At the PC facility the prostheses were fitted by three prosthetists, each with more than 20 years experience. As the MSS is a rather new method, the same degree of experience with this manufacturing method could not be achieved, but at the MSS facility the two prosthetists fitting the prostheses had two years experience of fitting approximately 200 patients with MSS and 10 years experience with ICEX. Ten transtibial amputees about to receive a new prosthesis were recruited at each clinic to participate in this study. The inclusion criteria for patients were that they were above the age of 18 years and had a transtibial amputation. No changes were made to the patient’s treatment due to the study. All prosthetists and patients gave written informed consent.
Patients at both the PC and MSS facilities were fitted with a prosthesis that incorporated a silicone liner. Therefore the only variable thought to differ between the two groups was the method of producing the socket. The MSS socket was fitted directly on the patient, whereas the PC was produced by plaster casting, rectification and production of a socket that was fitted at the next visit.
Cost analysis
Costs
Cost data was collected by recording the time and material used to manufacture and deliver either an MSS or PC prosthesis.
Questions for cost estimation.
The collection of time and cost data continued until the patient received a prosthesis considered to have an appropriate fit. If manufacturing a new socket was needed due to poor fit or other reasons before the patient could start gait training or use the prosthesis at home, this cost was included in the analysis.
With MSS the suspension mechanism can be chosen upon delivery, and could thus be seen as a variable cost among different patients. With PC, however, the suspension mechanism must be incorporated into the prosthesis during manufacture. To keep the cost of the suspension mechanism constant between MSS and PC, it was therefore assumed that a pin locking mechanism would be used for both systems. Other costs not directly pertaining to the socket, such as for a foot and pylon, were not included in the analysis, as this was assumed to be equal for both systems.
To see if the potentially faster delivery of MSS would be coupled with a higher number of revisits, as found earlier with ICEX, 2 we also did a retrospective review of the patients’ records three months after delivery to investigate the number of visits required after delivery.
Sensitivity scenarios
As costs often vary with quantity and decisions specific to local settings, some parameters were tested in sensitivity scenarios to elucidate their impact on the results. These factors were: the purchase of MSS material in bulk (as this gives a discount); the influence of always making a test socket for a PC; and a combination of both factors. In addition, as MSS is a rather new method, the effect of a potential future cost reduction was also tested.
Statistical analysis
All time and cost data were analysed using the independent samples t-test to identify differences between the groups with regard to total cost, material cost and time. Two-sided significance tests were used at an α-level of 0.05. The difference in number of visits after delivery of the prostheses was analysed using the chi-square test. All analyses were performed in SPSS 16.
Results
Patient characteristics.
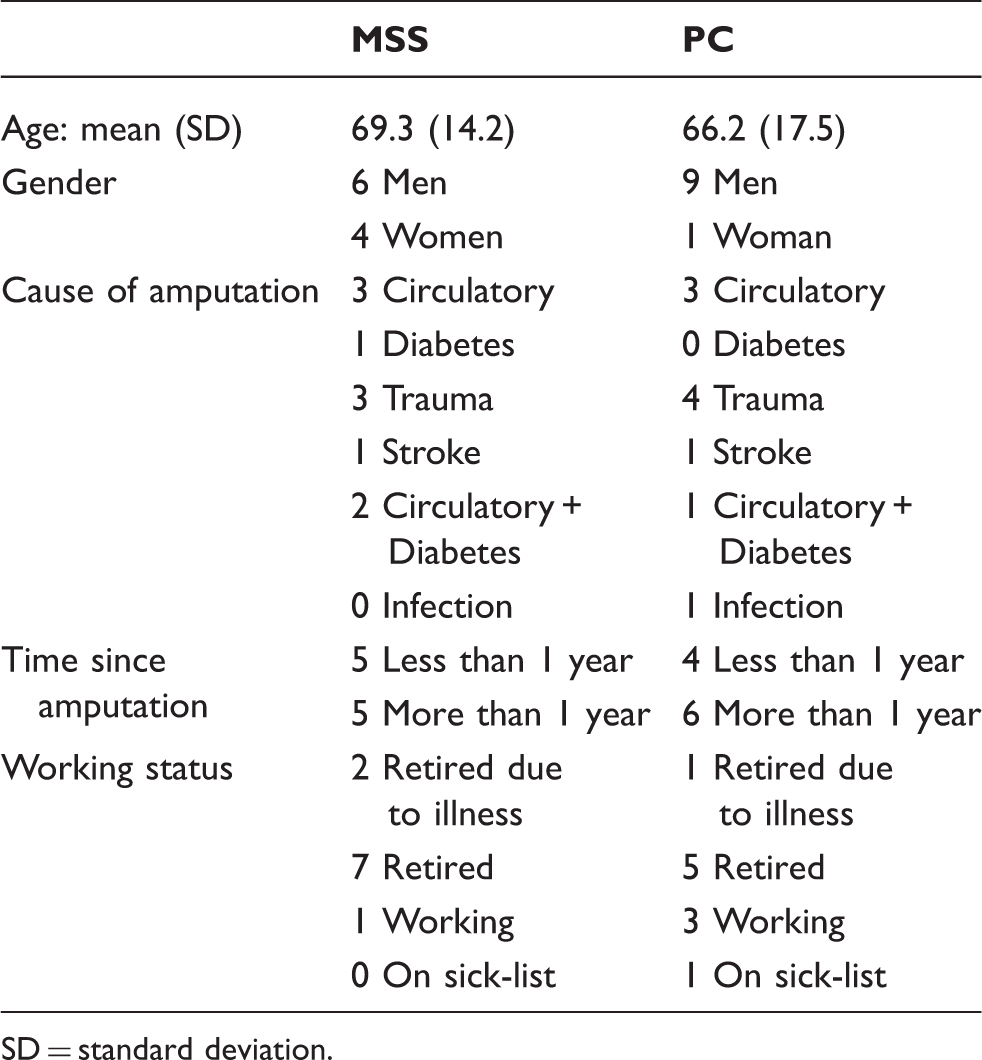
SD = standard deviation.
Total, material and work time cost of the two socket alternatives.
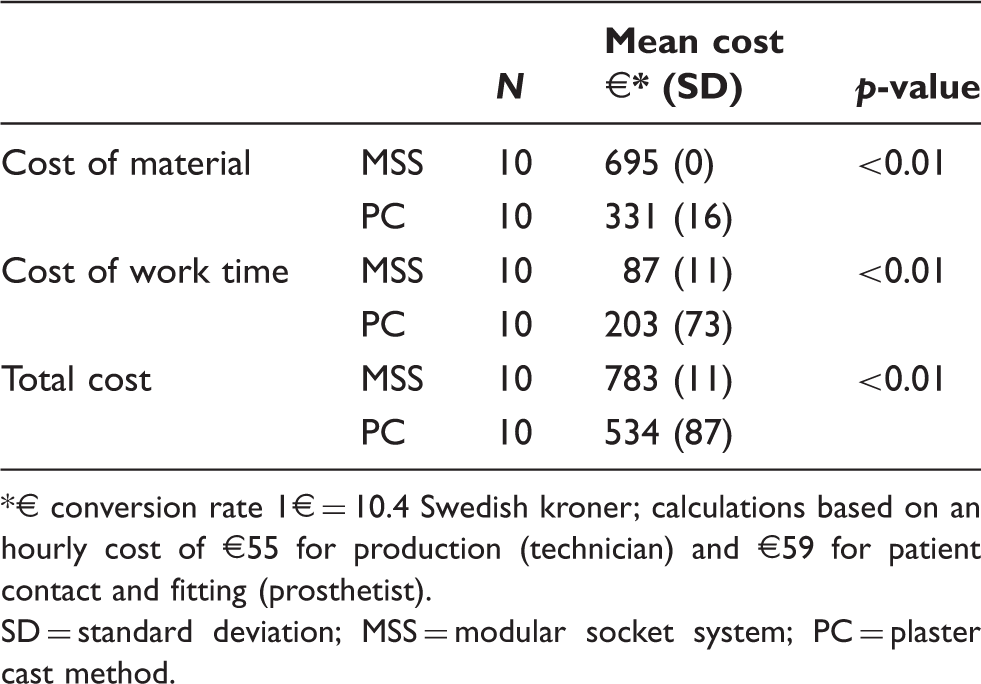
€ conversion rate 1€ = 10.4 Swedish kroner; calculations based on an hourly cost of €55 for production (technician) and €59 for patient contact and fitting (prosthetist).
SD = standard deviation; MSS = modular socket system; PC = plaster cast method.
Production time of the prostheses in minutes (SD).
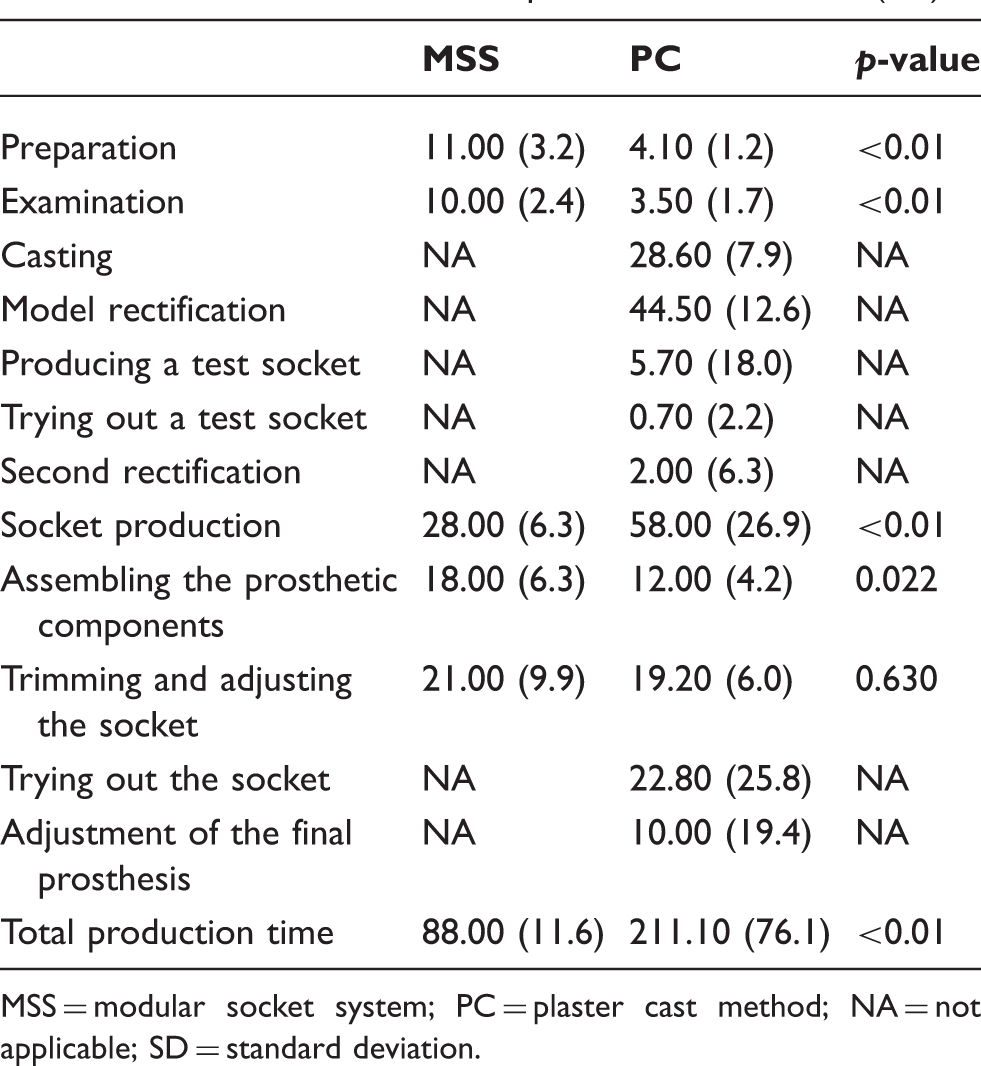
MSS = modular socket system; PC = plaster cast method; NA = not applicable; SD = standard deviation.
In addition, all MSS prostheses were delivered on the same day the initial measurements were made, whereas the mean delivery time for PCs was 17 days (SD 13.5). The MSS patients also needed only one visit to the prosthetic facility as opposed to 2.5 visits (SD 1) for the PC patients. A test socket was manufactured for only one patient in the PC group, but the sockets for two patients had to be remade before sufficient fit could be achieved.
When reviewing the number of visits for adjustment of the prosthesis three months after delivery, two patients in the PC group were lost to follow-up. Of the eight left for analysis, five had had adjustments made to their sockets. In the MSS group, eight out of ten had had adjustments made to their sockets (p = 0.41). None of the patients in either group had more than one visit for adjustment of their prosthesis.
Sensitivity scenarios
The sensitivity scenarios showed that buying the MSS weave in quantities of 50 would reduce the cost of MSS by €96. Also, if a test socket were made for all patients in the PC group this would increase the cost of PC by €115. Combining these two variables, buying 50 MSS weaves and producing a test socket for all PCs would result in a mean difference of only €29.
For the total cost of the two systems to be equal, given the current findings, the cost of MSS would have to decrease by 31%.
Discussion
This study has shown that the direct cost of providing a patient with the MSS prosthesis is significantly higher than providing the PC prosthesis. This is due to the fact that the significantly higher material cost for MSS was not outweighed by the time saved during production. It should be noted, however, that the results were highly sensitive to changes in the number of MSS weaves bought simultaneously and to the production of a test socket for all PCs. It may be impractical for smaller prosthetic facilities to buy 50 MSS weaves at once, as these should be used within one year. However, it is likely that larger facilities or several collaborating facilities could buy even larger quantities than 50, thereby reducing the cost of MSS even further.
Our results regarding production time are in line with those found by Datta et al. 1 Their study found that an ICEX took 100 minutes to produce and a PC 288 minutes, compared to our result of 88 and 211 minutes, respectively. When comparing our results to those of Selles et al., 2 the proportional difference in production time between the two socket alternatives was similar: Selles et al. found that it took 2.4 times longer to provide PC than ICEX; we found the same ratio between PC and MSS. The difference in total cost was considerably lower in our study compared to the results of Datta et al., due to the reduced cost of MSS compared to ICEX. Datta et al. found the total cost of ICEX 2.5 times higher than PC, whereas our results showed the cost of MSS just 1.4 times higher than PC. Thus our results show that the time saving of ICEX is still present for MSS, but at a reduced cost, although still higher than PC.
It should also be noted that our analysis took only the direct costs of the prostheses into account. Costs such as patient travel and secondary costs because patients could not make use of their prostheses were not included. Our finding that MSS required only one visit before delivery of a functional prosthesis compared to 2.5 visits for PC could, for example, have a significant impact on both travelling costs and loss of patient productivity. Taking the societal cost of lost production into account could quickly diminish the cost difference between the two alternatives. The average daily value of production, as calculated by the mean daily salary including social costs in Sweden, was €166 6 per day. Taking this into account, the cost to society of the two sockets would be equal if the MSS could gain 1.5 working days. Similarly, reducing the time lapse from 17 to 0 days before the patient receives the prosthesis could play a critical role in the cost of rehabilitation. If quicker provision could shorten the hospital stay due to earlier onset of rehabilitation, the use of MSS would probably save costs. However, these societal costs were not included in our analysis and therefore limited our results. To our knowledge no investigation has been done as to whether a reduction in delivery time of the prosthesis can translate directly into faster rehabilitation or faster return to work.
A further limitation of our study is that the patients were not randomly selected for MSS or PC. This does, of course, increase the risk of selection bias and factors other than the socket affecting the cost. The patient samples were not significantly different, except for gender, and were therefore not seen as likely to affect cost. Rather, the choice of producing the sockets at two different locations could be seen as a factor influencing the comparability of the two costs. However, as both methods are seldom fully incorporated in one prosthetic facility simultaneously, this approach seemed appropriate. In this way both methods can be thought to be performing optimally as found in normal clinical practice. Introducing a method to a facility that did not normally treat patients with this method could have just as large a disadvantage. For example, when investigating ICEX, Selles et al. found that due to the prosthetists’ lack of familiarity with the ICEX system, patients needed extra visits to adjust their prostheses. 2 In our study, no such differences in visits for adjustments were found within three months after delivery of the prosthesis. However, over a longer time perspective than used in this study, the number of visits and the survival time of the prosthesis could differ. To compare the full life-cycle cost of both alternatives, a trial with a longer follow-up period is required.
Conclusion
Our study shows that the direct prosthetic cost of treating a patient with MSS is significantly higher than treating a patient with PC. However, the MSS prosthesis can be delivered significantly faster and with fewer visits. Further studies taking the full societal costs of PC and MSS into account should therefore be performed.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
