Abstract
Background:
Individuals using a lower-limb prosthesis indicate that they need to concentrate on every step they take. Despite self-reports of increased cognitive demand, there is limited understanding of the link between cognitive processes and walking when using a lower-limb prosthesis.
Objective:
The objective was to assess cortical brain activity during level walking in individuals using different prosthetic knee components and compare them to healthy controls. It was hypothesized that the least activity would be observed in the healthy control group, followed by individuals using a microprocessor-controlled prosthetic knee and finally individuals using a non-microprocessor-controlled prosthetic knee.
Study design:
Cross-sectional study.
Methods:
An optical brain imaging system was used to measure relative changes in concentration of oxygenated and de-oxygenated haemoglobin in the frontal and motor cortices during level walking. The number of steps and time to walk 10 m was also recorded. The 6-min walk test was assessed as a measure of functional capacity.
Results:
Individuals with a transfemoral or knee-disarticulation amputation, using non-microprocessor-controlled prosthetic knee (n = 14) or microprocessor-controlled prosthetic knee (n = 15) joints and healthy controls (n = 16) participated in the study. A significant increase was observed in cortical brain activity of individuals walking with a non-microprocessor-controlled prosthetic knee when compared to healthy controls (p < 0.05) and individuals walking with an microprocessor-controlled prosthetic knee joint (p < 0.05).
Conclusion:
Individuals walking with a non-microprocessor-controlled prosthetic knee demonstrated an increase in cortical brain activity compared to healthy individuals. Use of a microprocessor-controlled prosthetic knee was associated with less cortical brain activity than use of a non-microprocessor-controlled prosthetic knee.
Clinical relevance
Increased understanding of cognitive processes underlying walking when using different types of prosthetic knees can help to optimize selection of prosthetic components and provide an opportunity to enhance functioning with a prosthesis.
Background
Individuals walking with a lower-limb prosthesis report that they must cognitively attend to each step they take.1,2 This is supported by a growing body of literature demonstrating a clear link between executive functioning (EF) and gait performance. Of relevance in rehabilitation settings is evidence that challenging gait tasks, for example, walking while performing a secondary task, place an increasing demand on executive functions.3,4 Research has also demonstrated that individuals with body impairments affecting their mobility have increased demands on executive functions when walking,5,6 and that this is associated with postural instability and an increased risk of falls.7–10
Executive functions are complex cognitive processes that are thought to be necessary for control of behaviour. They include cognitive processes such as impulse control, response inhibition, attention, working memory, planning and decision making.11,12 Anatomically, the frontal lobes and, in particular, the prefrontal cortex have been identified as critical for successful performance on tests of EF.13,14 EF during gait has typically been evaluated using dual-task paradigms, where gait performance is evaluated while simultaneously executing a secondary task, for example, counting backwards. Performance of two tasks simultaneously can result in a decrease in performance on one or both tasks. 15
Until recently it has been difficult to record physiological changes associated with an increase or decrease in cognitive load under dynamic conditions such as walking. Recent advances in imaging technology have, however, made it possible for researchers to measure relative activity in the brain during performance of dynamic motor tasks. One technique receiving increasing attention in the literature is functional near-infrared spectroscopy (fNIRS), a non-invasive, optical, neuroimaging technique which measures the brain’s haemodynamic response. fNIRS systems record the relative changes in concentration of oxygenated (oxyHb) and deoxygenated (de-oxyHb) haemoglobin, which result from neural activation.5,16 In response to regional brain activation, one would typically observe an increase in concentration of oxyHb and a decrease in concentration of deoxyHb. 17 The increase in oxyHb concentration results from increased regional cerebral blood flow, while the associated decrease in de-oxyHb concentration occurs when the increase in regional cerebral blood flow exceeds the metabolic uptake of oxygen. 17
Over the past 5 years, fNIRS has become increasingly popular to study the interaction between motor performance and cognition. It has been used to compare gait of individuals with pathological conditions to healthy controls, 5 to assess the relative effects of training on gait 18 and to assess the effects of dual-task performance.5,6 These studies have demonstrated an increase in activity within the prefrontal cortex associated with gait pathologies, a decrease in frontal lobe activation associated with training and an increase in prefrontal cortex activation with the addition of a secondary task.5,6,19 To date, fNIRS has not been used to study the cognitive demands of walking with a lower-limb prosthesis.
Lower-limb prosthetic rehabilitation presents an interesting opportunity for studies of EF. Miller and Deathe 20 indicate that perceived ability to ambulate without thinking is a predictor of depression and balance confidence in individuals using lower-limb prostheses. Dual-task studies have presented varied results and some research indicates that a concurrent cognitive task has a negative effect on performance when ambulating with a prosthesis on a stable, level surface,21,22 walking on an unstable surface 23 and while maintaining balance. 22 Others suggest that the relative effect of adding a secondary task is similar for both individuals using a prosthesis and healthy controls. 24
A wide variety of prosthetic interventions are available to address the functional limitations experienced by individuals with a lower-limb amputation 25 and selection of appropriate prosthetic components is considered essential in achieving a successful outcome. 26 One of the most notable technological advances in prosthetic componentry has been the introduction of microprocessor-controlled prosthetic knee (MPK) joints. A systematic review regarding use of MPKs suggests that those prosthetic knees are associated with improvements in patient-reported measures of cognitive effort. 25
In order to improve the quality of prosthetic devices, there is a need to better understand the relationship between gait performance and cognitive processes. The objective of this study was to assess patterns of haemodynamic cortical brain activity during level walking for groups of individuals with transfemoral (TF) or knee disarticulation (KD) amputations using a prosthesis incorporating either an MPK or a non-microprocessor-controlled prosthetic knee (non-MPK) and to compare this with a group of healthy controls.
It was hypothesized that the least amount of cortical activity would be observed in the prefrontal cortex in the group of healthy controls, followed by the group using an MPK and finally that those using a non-MPK would have the largest amount of prefrontal cortical brain activity.
Methods
Participants
A total of 29 (n = 29) individuals with unilateral TF or KD amputations were recruited through prosthetic and orthotic clinics in Sweden and Norway. Participants were recruited on the basis that they used either an MPK (n = 15) or a non-MPK (n = 14). One individual fitted with a Very Good Knee (VGK) was included in the MPK group. While it is recognised that this knee does not include a microprocessor, the adaptive nature of the knee is consider to be closer to an MPK then a non-MPK (Schuling et al. 2013). 27 A group of healthy controls were also recruited (n = 16). Stratified sampling was used to ensure that groups were homogeneous in terms of age and sex. Participant details are presented in Table 1. Individuals using prostheses were required to be a minimum of 1-year post-amputation and able to walk continuously for 500 m indoors with no more than a single-handed walking aid as support, that is, one crutch or one walking stick. Individuals with a bone-anchored prosthesis or those with additional physical limitations or cognitive impairments (Mini-Mental State Examination test <27) 28 were excluded from the study. Participants were required to understand written and spoken Swedish or Norwegian.
Participant details.
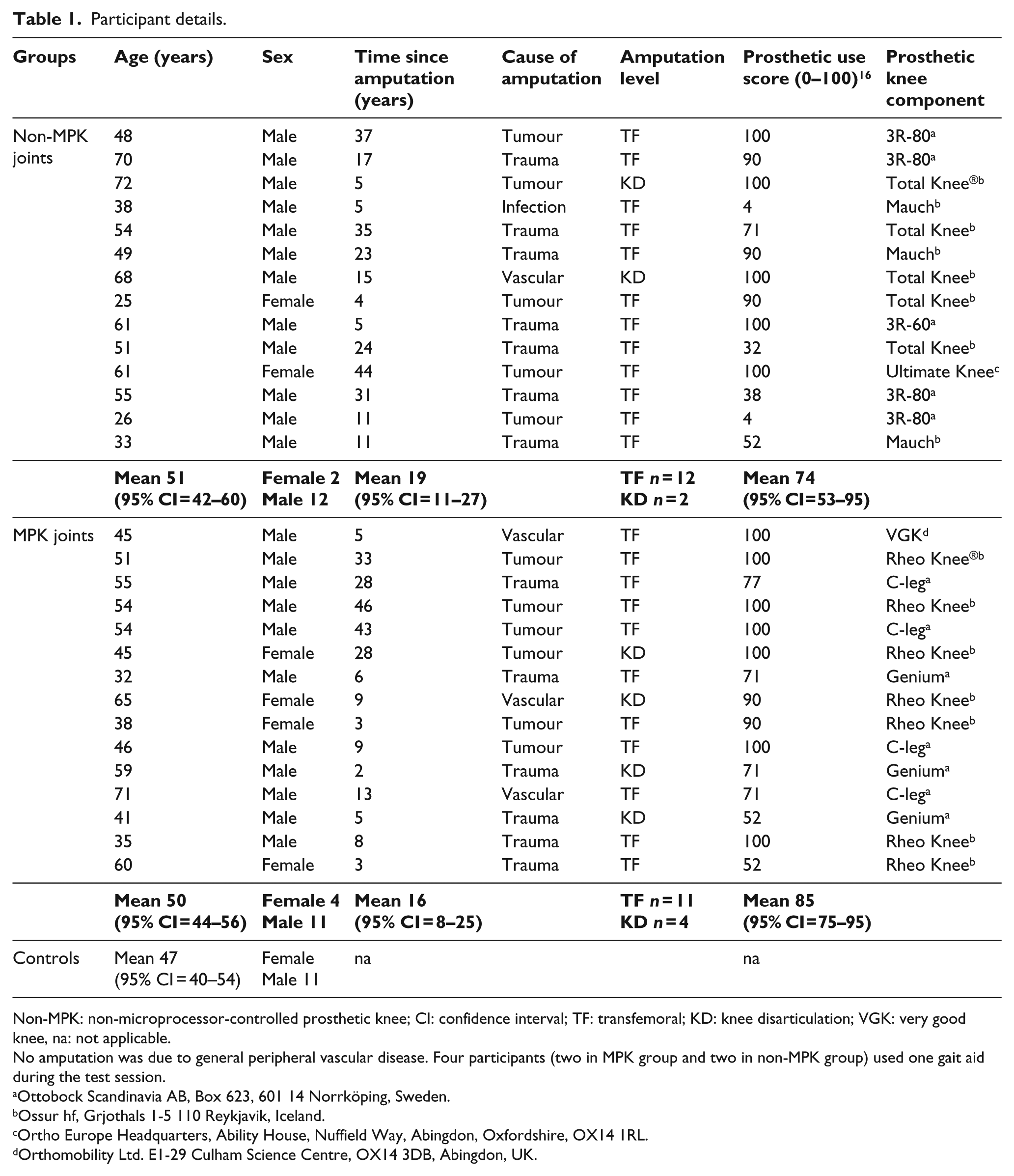
Non-MPK: non-microprocessor-controlled prosthetic knee; CI: confidence interval; TF: transfemoral; KD: knee disarticulation; VGK: very good knee, na: not applicable.
No amputation was due to general peripheral vascular disease. Four participants (two in MPK group and two in non-MPK group) used one gait aid during the test session.
Ottobock Scandinavia AB, Box 623, 601 14 Norrköping, Sweden.
Ossur hf, Grjothals 1-5 110 Reykjavik, Iceland.
Ortho Europe Headquarters, Ability House, Nuffield Way, Abingdon, Oxfordshire, OX14 1RL.
Orthomobility Ltd. E1-29 Culham Science Centre, OX14 3DB, Abingdon, UK.
Study procedures were approved by the Regional Ethics Committee in Linköping, Sweden (Dnr 2015/215-31), and the Regional Committees for Medical and Health Research Ethics, Norway (Dnr 2015/1526/REK sor-ost). Information related to the study procedures was provided and written informed consent was received prior to testing.
Procedure
Procedures in this study follow the recomendations outlined in the STROBE guidelines (see supplimentry material for STROBE checklist). Participants were requested to attend a single test session and were able to use their normal walking aid during the test session. Prior to data collection, the full testing procedure was explained, and any questions were answered. Background information related to the participant was collected, that is, time since amputation, amputation cause, prosthetic use score 29 before they were fitted with an fNIRS system (Figure 1). Details of the fNIRS system are presented below.
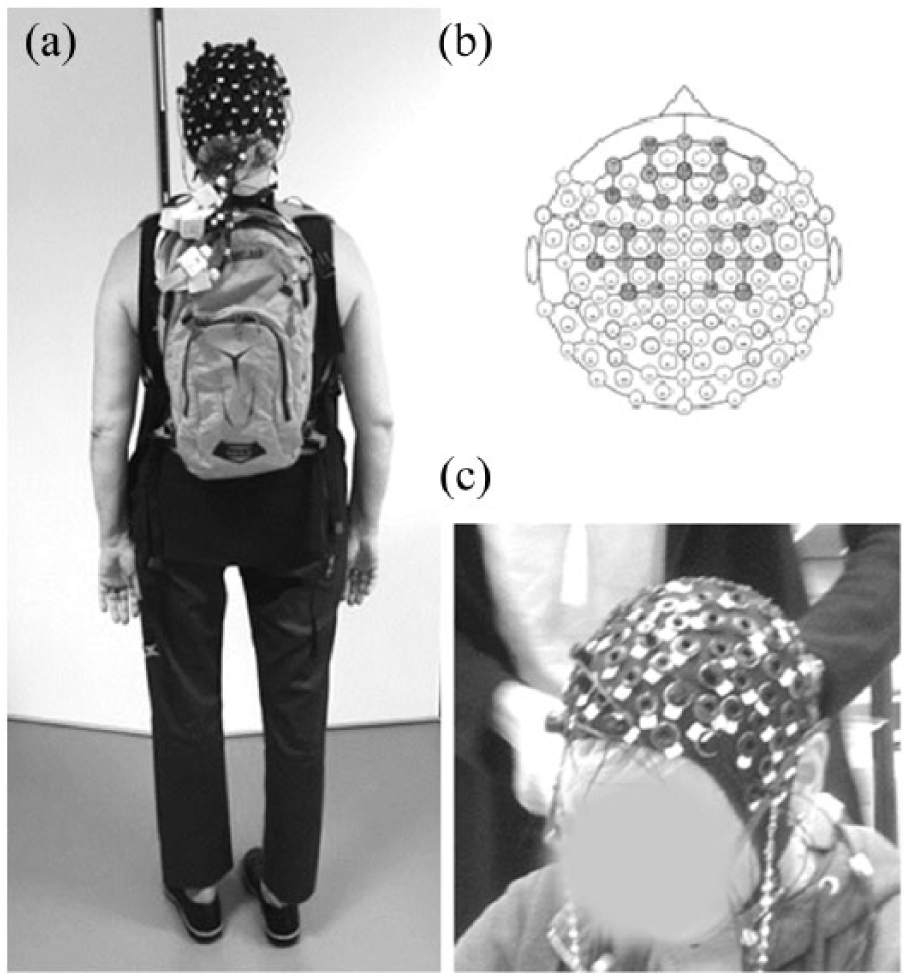
(a) Equipment worn by participants, (b) topographical-layout of the 16 source and 16 optode placement covering prefrontal and motor cortex, both left and right hemispheres resulting in 40 channels and (c) the elasticized cap with pre-determined holes.
The full testing procedure is outlined in Figure 2 and began with a 30-s baseline measurement. During this time, the participant was seated in a chair with their eyes closed. Following baseline measures, participants were requested to stand quietly for a further 30 s (refractory period) before completing the first of four walking trials. During walking trials participants were requested to walk back and forth at a self-selected velocity over a distance of 14 m. A 30-s period of quiet rest was given between each walking trial to allow signals to return to pre-test levels and to avoid non-linear effects of the haemodynamic refractory period.30,31 During walking trials, the number of steps and time taken to walk the first 10 m was recorded by a researcher. Measurement of temporospatial data was limited to the first 10 m to minimize any effects of turning. All fNIRS trials were performed in quiet rooms with no distracting noise or activity.
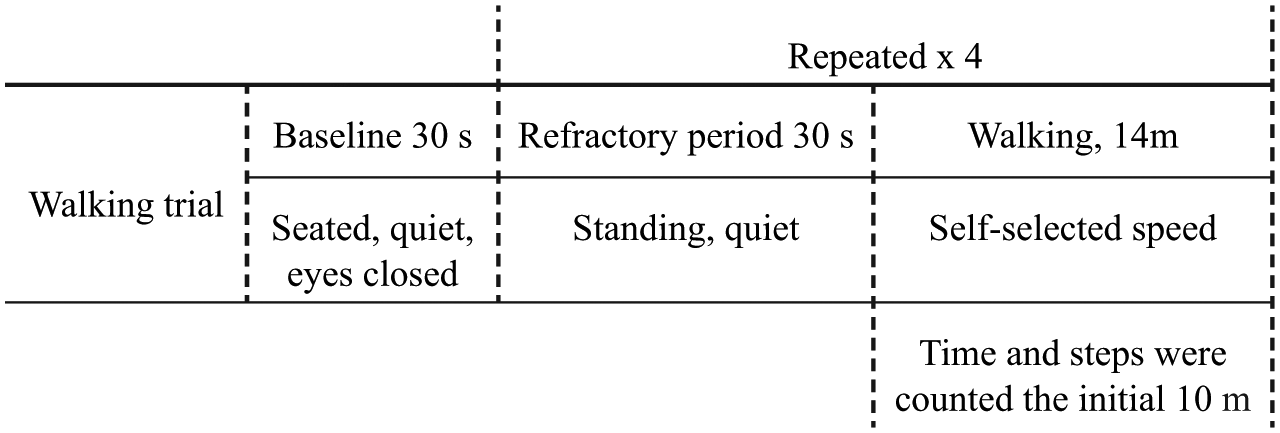
Experimental protocol.
NIRStar acquisition software (NIRx Medical; NIRx Medical Technologies LLC, 15 Cherry Ln, Glen Head, NY 11545, USA) was used to collect haemodynamic data and instructions to participants were standardized by using NIRStim software (NIRx Medical Technologies LLC), which delivered pre-recorded voice instructions to participants. The NIRStim software was also used to place time stamps in the data files when specific instructions were given. Baseline measures and walking sequences were all captured in a single data file. After the fNIRS probes were removed, participants were requested to perform a 6-min walk test. 32 Due to space constraints, the 6-min walk test was conducted on a 20-m track rather than the recommended 30-m. All participants were given the same instructions, ‘cover as much distance as possible in the 6 minutes’. The fNIRS walking trials took about 5 min to complete and the entire test session, including preparation of fNIRS equipment, took approximately 1.5 h.
fNIRS data acquisition
A wireless, portable, continuous wave fNIRS system, NIR1Sport tandom (NIRx Medical Technologies LLC), was used to record haemodynamic responses. An elasticized cap with pre-determined holes ensured secure placement of the optodes according to the international 10–20 system. 33 Optode placement is presented in Figure 1. A total of 32 optodes were used, consisting of 16 sources and 16 detectors and resulting in a total of 40 channels. Optodes were positioned using a standard optode montage setup template (prfMotor_16 × 16) provided by the manufacturer (NIRx Medical). Optodes covered the prefrontal cortex including, dorsolateral and anterior prefrontal cortices (Broadmans area (BA) 9 and 10) and the motor cortex, including primary motor cortex and premotor cortex/supplementary motor area (SMA; BA 4 and BA 6, respectively) in both hemispheres of the brain. An interoptode distance of 3.0 cm was used (Figure 1). Source optodes emitted infrared light at wavelengths of 760 and 850 nm at a frequency of 7.81 Hz. A black headcap was used to stabilize optodes during testing and reduce the likelihood of interference from ambient light. After calibration and optimization of the signal-to-noise ratio (SNR), the laptop computer used for data capture was placed in a backpack worn by the participant (Figure 1).
fNIRS data processing
NirsLAB 2016.5 (NIRx Medical Technologies LLC) software was used for post-processing and data analysis. The first of the four walking trials was considered a practice trial and was subsequently excluded from analyses. In walking trial 2, 3 and 4, 10 s of straight, level walking was selected for analysis, excluding acceleration and deceleration phases. The quality of signals recorded from each channel was visually inspected. Channels with a gain factor greater than 3 or a coefficient of variation (CV) higher than 7.5% were removed (bad channels). The CV is an indication of SNR and was calculated (using all raw data points in the measurement series) as 100 times the standard deviation, divided by the mean of all data points. A standard deviation threshold of 5 was utilized as the cut-off for eliminating discontinuities and spike artefacts. A Bandpass filter with a passband from 0.01 to 0.2 Hz was then applied to all data to eliminate fluctuations related to factors such as heartbeat and respiration as well as low-frequency signal drift.30,34
Pre-processed data were analysed to determine the relative change in concentration of oxyHb and de-oxyHb. The signals of each channel were converted to concentrations using the modified Beer-Lambert law 35 which describes the linear relationship between absorbance of light and concentrations of a molecular substance, that is, blood. Concentrations of oxyHb and de-oxyHb were calculated relative to baseline values and a canonical haemodynamic response function (hrf) with a 6-s delay used to model the waveform in the haemodynamic response.
Statistical analysis
OxyHb and de-oxyHb concentrations were analysed separately. NirsLAB software was used to perform a general linear model (GLM) analysis of the fNIRS haemodynamic-state time series and evaluate the position-dependent relationships between computed data channel responses and the temporal model (montage) used for data collection (NirsLAB 2016.5, NIRx Medical; NIRx Medical Technologies LLC). An analysis of variance (ANOVA) was performed to assess statistical significance by comparing the GLM model-fitting coefficients (β) across conditions (control, non-MPK and MPK). In instances where significant differences were found (p < 0.05), a post hoc t-test was performed with a Bonferroni multiple-comparison adjustment (p < 0.0167).
IBM SPSS Statistics 21 (SPSS Inc., Chicago, IL, USA) was used to analyse temporospatial data. As violations of normality were indicated by a Shapiro–Wilks test (p < 0.05), non-parametric tests were utilized. Median time to cover 10 m, number of steps and the 6-min walk data were analysed using a Kruskal–Wallis H test and post hoc Dunn’s test with a Bonferroni adjustment.
Results
No significant differences between groups in relation to age, time since amputation or amount of self-reported prosthetic use was observed. In total, 95% CIs of the percentage of bad channels removed per participant for each group was non-MPK (5.0%–9.3%), MPK (3.7%–9.6%) and controls (3.4%–9.2%). Figure 3 presents mean concentrations of oxyHb and de-oxyHb (mM) in the prefrontal and motor cortices for each of the experimental conditions (non-MPK, MPK and controls).
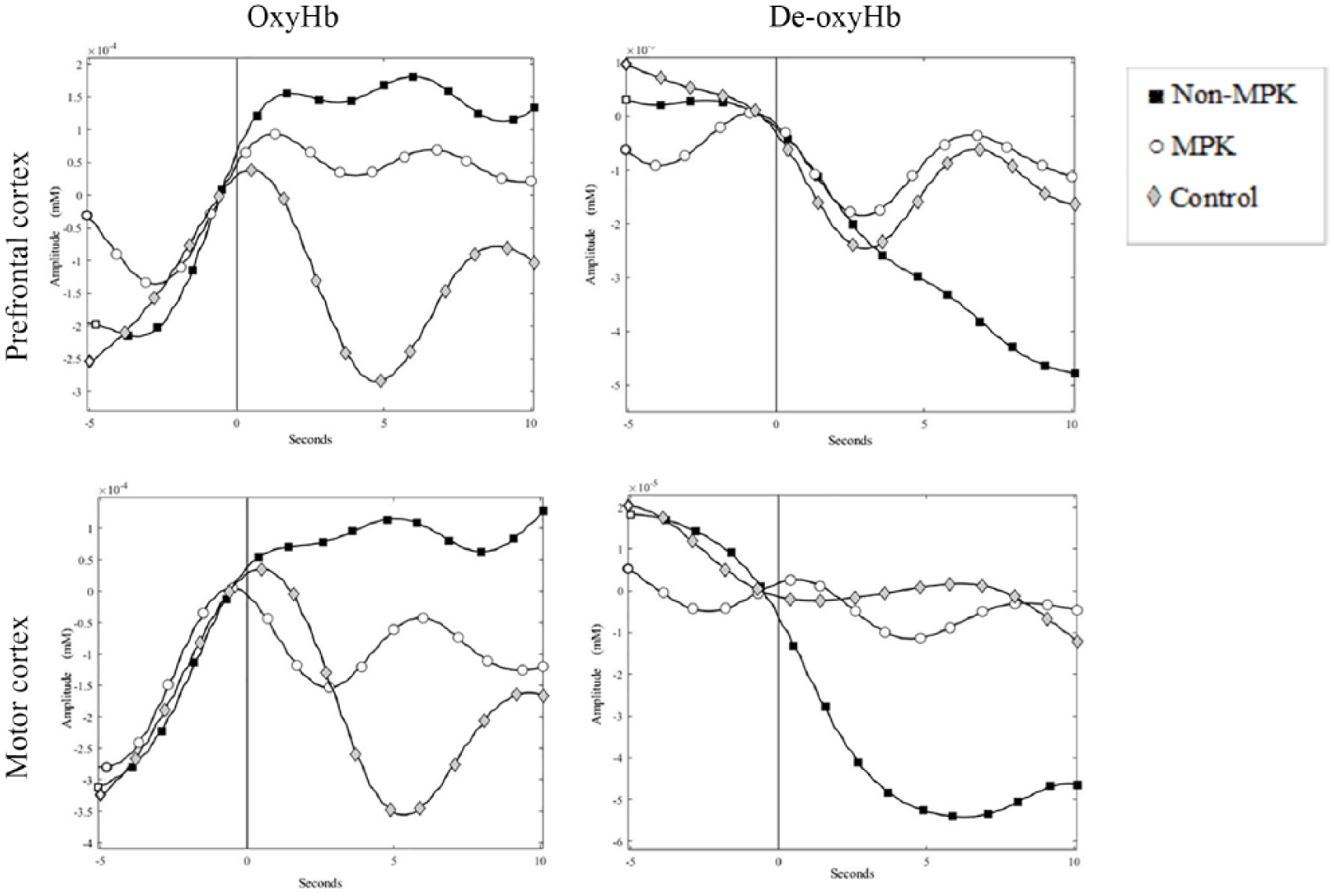
Mean oxyHb and de-oxyHb concentrations (millimolar (mM)) in prefrontal and motor cortices for each experimental condition, (non-MPK, MPK and controls). The 0 time period on the x axis represents onset of the walking trial.
Results of the ANOVA performed on GLM model-fitting coefficients revealed significant differences in both oxyHb (channels 14 and 28 in prefrontal and channel 33 in motor cortex) and de-oxyHb (channel 24 in prefrontal) between the three groups (p < 0.05). Post hoc analyses are presented in Figure 4 where each channel in which a significant difference was found (p < 0.0167) is shown. Results reveal significantly increased oxyHb and decreased de-oxyHb concentrations in both prefrontal and motor cortices in individuals walking with a non-MPK when compared to controls (Figure 4(a); p < 0.0167). Individuals walking with a non-MPK showed a significant increased oxyHb in the prefrontal and motor cortex when compared with those walking with an MPK (Figure 4(b); p < 0.0167). No significant difference was seen in de-oxyHb. No significant differences were seen in oxyHb between MPK and controls. There was a significant (p < 0.0167) decrease in de-oxyHb concentration in the motor cortex for controls when compared to the MPK group (Figure 4(c)).
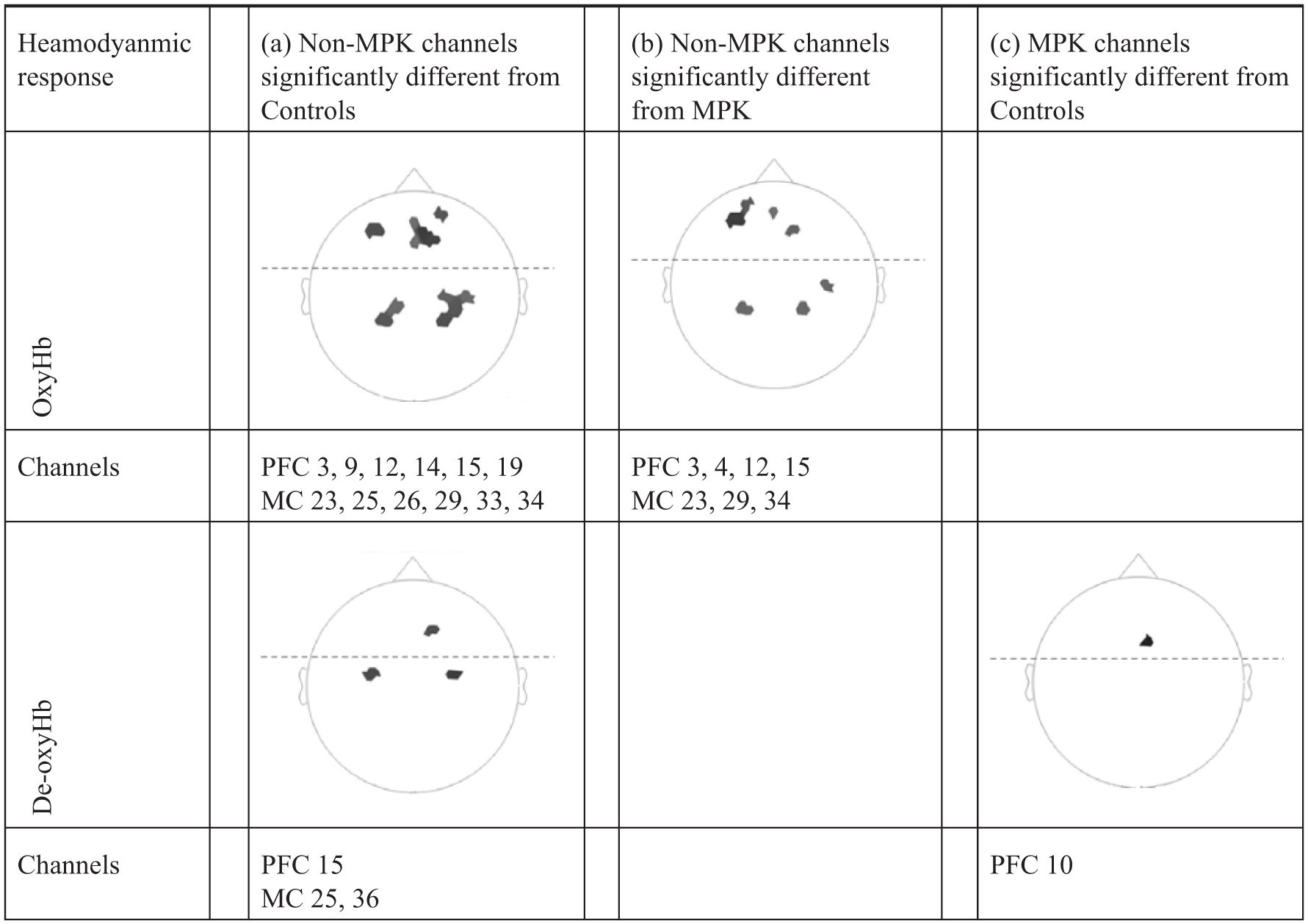
t-maps of oxyHb and de-oxyHb of all participants, only significant (p < 0.0167) channels are viewed, dark grey represents a greater degree of significance. (a) Increased oxyHb and decreased de-oxyHb for non-MPK compared with controls. (b) Increased oxyHb for non-MPK compared with MPK, no significant difference in de-oxyHb. (c) No significant difference in oxyHb, significant decreased de-oxyHb for controls compared MPK.
Analysis of temporospatial data revealed a significant difference between groups. Individuals using a prosthesis walked slower, took more steps and walked a shorter distance than the control group (Table 2). No significant difference was recorded between individuals using a non-MPK compared to those using an MPK in either walking time, number of steps taken or the 6-min walk test (Table 2).
Temporal and spatial data. Median and p value of comparison between groups.
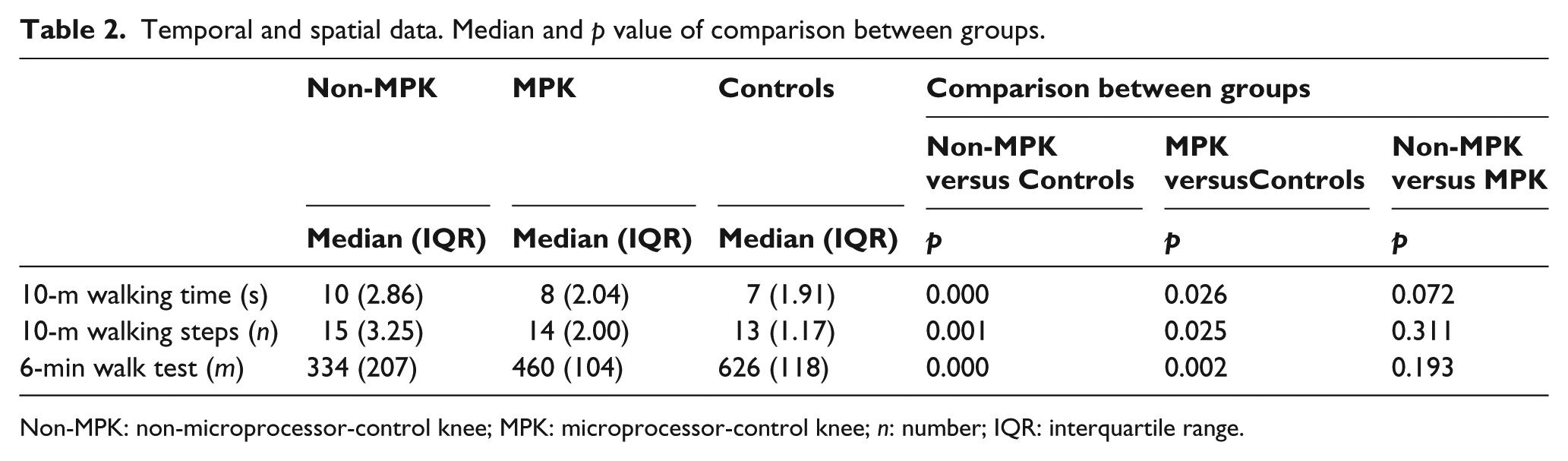
Non-MPK: non-microprocessor-control knee; MPK: microprocessor-control knee; n: number; IQR: interquartile range.
Discussion
The objective of this study was to assess patterns of haemodynamic cortical brain activity during level walking for individuals with a TF or KD amputation. Of interest was the comparison of cortical brain activity between individuals using non-MPKs with MPKs, and these groups to healthy controls. The results partially confirmed the experimental hypotheses, and an increase in cortical brain activity was observed in individuals walking with a non-MPK when compared to healthy controls. Moreover, the results demonstrated increased cortical brain activity for individuals walking with a non-MPK when compared to individuals walking with an MPK.
Patterns of cortical brain activity identified in this study are consistent with earlier work indicating that individuals with physical disabilities have a higher degree of prefrontal activation in the brain compared to healthy controls when walking at self-selected speeds.5,36–38 In this study, the group who used a non-MPK to ambulate registered significantly more activity in the prefrontal areas of the brain than healthy controls. As the prefrontal regions of the brain are associated with cognitively demanding processes,5,16 results suggest that these individuals must allocate more cognitive resources to ambulate than healthy controls. Under the assumption that there is a limit to the attentional capacity of the brain, as proposed by Kahneman, 39 this places individuals, using a prosthesis to ambulate, at risk of attentional overload which means that they may have less capacity for simultaneously using other information processes while walking.
Of particular interest in this study was the significant increase in cortical brain activation that was observed in the non-MPK group compared to the MPK group. This result suggests that persons walking with an MPK require less cognitive resources when ambulating than those using a non-MPK. Results are consistent with self-report data from patients using MPK prostheses.40,41 As increased cognitive demand when walking is associated with an increase in frequency of falls,8,9 our results support earlier research indicating that persons using an MPK would fall less than those using non-MPKs and would perform better on gait-related dual-task activities. Research on falls and dual-task walking with different prosthetic knees have indicated a decrease in number of falls, increased balance confidence 42 and decrease in self-reported cognitive load 41 when using an MPK.
In this study, no differences were recorded between the MPK and non-MPK groups in age, time since amputation, prosthetic use or temporospatial variables. This suggests that observed differences were more likely to be due to the prosthetic componentry than any influence from these factors. While no difference in temporospatial data was observed between the non-MPK and MPK users, a significant difference was observed between individuals walking with a prosthesis and healthy controls. The group who used a prosthesis walked at a slower speed, took more steps and walked a shorter distance on the 6-min walk test. It is interesting that the observed increased cortical brain activity in the non-MPK group was not reflected in the temporospatial data. This suggests that cognitive changes may not always be reflected in biomechanical variables and is likely linked to complexity of the task being performed. This premise is supported by Morgan et al. 24 who indicated that gait of individuals using a prosthesis and healthy controls was equally affected with the addition of a dual-task when walking on a stable surface. This same group further demonstrated, however, that step time asymmetry on an unstable surface was affected to a greater extent in individuals using a prosthesis for ambulation. 23 Future research should aim to increase understanding of the relationship between task complexity, motor performance and cognitive processes.
Study limitations
This study includes one of the largest sample of MPK users included in published research. 25 Nevertheless, the sample size is small which may limit the generalizability of the results. A further limitation with potential to affect generalizability of results is that individuals included in this study were relatively young and healthy as compared to the general Swedish population of individuals who have undergone TF and KD amputations. These individuals are typically older and have been amputated due to peripheral vascular diseases. 43 Given that haemodynamic activity in the prefrontal cortex during walking and conducting a secondary task decreases with age 16 it would be of interest to repeat this study with an older population.
fNIRS is a fairly new technology and there is currently no standard procedure regarding measurement protocols or data analysis procedures. While every attempt has been made to standardize procedures and minimize interference during measurement, the fNIRS signals may have been affected by artefacts such as physiological noise from superficial tissue 44 and/or motions artefacts. 45 During analysis of the fNIRS data, bad channels were identified and removed as per manufacturers recommendations (NIRx Medical; NIRx Medical Technologies LLC), but there is no consensus on post-processing and some researchers argue that all bad channels should be included and filtered instead of excluded. 45
Inspection of the distribution of bad channels did not indicate any systematic errors in the data collected and we are confident in the validity of our results based on sound methods, although, with the ongoing research into post-processing methods, one should be conscious of this when interpreting the results.
This study also utilized a cross-sectional design which limits the extent to which we can infer a causal relationship between cognitive load and prosthetic knee components. Longitudinal research is needed to confirm the results, detect causal relationships and potentially investigate the relative effects of training.
Despite the above limitations this study has established, for the first time using physiological measures of cognitive load, that individuals who utilize a TF prosthesis have increased cognitive load when walking compared to able-bodied controls as established. Of potentially even greater interest to both patients and clinicians alike is that this study suggests that individuals who use a non-MPK prosthesis have increased cognitive demand for ambulation than those who use an MPK.
Conclusion
Persons walking with a non-MPK have an increase in cortical brain activity during ambulation when compared to healthy controls. Furthermore, individuals using an MPK joint require less prefrontal cortical brain activity to ambulate when compared to those using a non-MPK joint. Findings suggest that prescription of microprocessor-controlled prosthetic knees in relatively young and healthy users of TF and KD prostheses may help to reduce reliance on executive functions during gait.
Supplemental Material
STROBE-Checklist-v4-MS-Word – Supplemental material for Reduced cortical brain activity with the use of microprocessor-controlled prosthetic knees during walking
Supplemental material, STROBE-Checklist-v4-MS-Word for Reduced cortical brain activity with the use of microprocessor-controlled prosthetic knees during walking by Saffran MMöllerller, David Rusaw, Kerstin Hagberg and Nerrolyn Ramstrand in Prosthetics and Orthotics International
Footnotes
Author contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded in part by Promobilia foundation, Össur and Team Olmed.
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
