Abstract
Objective
To evaluate the quantity of titanium dioxide nanoparticles released into the artificial salivary medium from orthodontic composite impregnated with 1% weight/weight (w/w) and 5% w/w titanium dioxide nanoparticles (TiO2 NPs) used for bonding metal brackets, thereby eventually comprehending the permissible levels.
Materials and Method
Eighty freshly extracted teeth for orthodontic treatment were divided into 2 groups of 40 teeth each and were bonded with brackets containing 1% w/w and 5% w/w composite containing titanium dioxide nanoparticles and placed in an artificial salivary medium. Quantification of 1% w/w and 5% w/w composite containing titanium nanoparticles was done using inductively coupled plasma mass spectroscopy for 4 timely periods 24 h, 2 months, 4 months, and 6 months.
Results
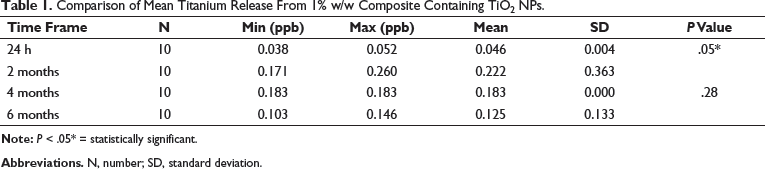
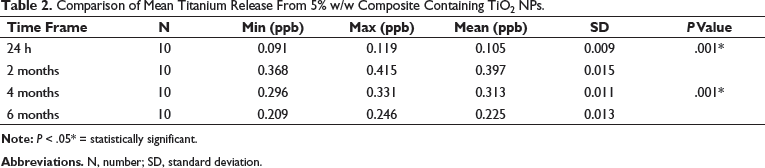
In the teeth that received 1% TiO2, the amount of titanium released was greatest in 2 months with no significant release at later intervals. In the second group that received 5%, there was a significant release of titanium at all intervals, with highest release at second month.
On comparing the 2 concentrations at 4 different time intervals, the quantities were significantly greater in the 5% group at all time frames, thus implying a significant increase in titanium released with an increase in concentration from 1% to 5%.
Conclusion
Titanium release was higher in 5% w/w composite containing nanoparticles than 1% w/w composite containing nanoparticles, and 1% and 5% concentrations can be used safely and are within the permissible limits.
Keywords
Introduction
Dental caries is the outcome of a dynamic interplay between microorganism and diet, with plaque being an indispensable precursor. It is noteworthy that plaque retention is higher in individuals receiving fixed or removable orthodontic treatments, increasing the bacterial count. 1 Additionally, orthodontic appliances, especially bands and brackets, limit the mechanical self-cleaning process of saliva and musculature and upsurge the difficulty of ensuring proper oral hygiene, eventually increasing the risk of white spot lesions. 2 Although many preventive methods such as oral prophylaxis, mouthwashes, interdental aids, and other mechanical plaque control methods were defined, all these measures demand stringent patient compliance. 3
Ever since Buonocore 4 introduced the acid etch bonding technique in 1955, many different adhesive agents such as composite, compomer, and resin-modified glass ionomer cement (GIC) are used in bonding fixed orthodontic attachments to teeth. Composites have multiple advantages: simple handling, good adhesion, reduced gingival irritation, and improved aesthetics. 5 However, the accumulation of bacteria is more on the adhesive agents due to their rough surface texture. One strategy to prevent microbial adhesion and/or enamel demineralization during the treatment is by using nanoparticles because it enhances mechanical and antibacterial properties.
Nanotechnology in dentistry has evolved over the last few decades, which works with the principle of using individual atoms and molecules to construct functional structures. 6 Nanoparticle may be defined as any intentionally produced particle with a characteristic dimension from 1 to 100 nm and has properties that are shared by nano scale particles with the same chemical composition. 7 Nanoparticles of silver, zinc, titanium, and silica have been used to improve the antibacterial properties of composites. 5 Although, composites containing silver nanoparticles provide superior antibacterial properties without compromising the shear bond strength, 8 but have shortcomings like discoloration of the composite matrix and biocompatibility. 9 Copper and zinc-based nanoparticles also produced severe toxic effects in animal studies in vitro. 10 11
Recently, there has been much attention to metallic nanoparticles composed of titanium dioxide due to its photocatalytic activity and low toxicity. Haghi et al 12 showed that the TiO2 NPs cause little pores in bacterial cell walls, leading to increased permeability and cell death, which may prevent recurrent caries and enamel demineralization. Further, bacteria are less likely to develop resistance against titanium dioxide. 13 Moreover, the addition of TiO2 NPs to dental composites also enhance mechanical properties, including elastic modulus, microhardness, flexural strength, and provide bond strength values that were equal or even higher than that of the nanoparticle free controls. 14
To measure and quantify titanium release from the composites, several methods can be used, such as inductively coupled plasma mass spectrometry (ICP-MS), inductively coupled plasma atomic emission spectroscopy, flame atomic emission, flame atomic absorption, graphite furnace atomic absorption, and cold vapor generation atomic absorption. 15 Among the above, ICP-MS has significant advantages over the others, such as multi-element capability, which allows multiple elements to be measured simultaneously in a single analysis. Furthermore, ICP-MS provides short analysis time and simple sample preparation making it a better choice. 16
Additionally, the biocompatibility of different orthodontic adhesives and their ingredients has been looked into in several studies, and most indicated the cytotoxic effect of varying intensity due to the release of unbound molecules from the structure of cured composite. Thus, the present study aimed to evaluate the quantity of titanium dioxide nanoparticles released into the artificial salivary medium from orthodontic composite impregnated with 1% w/w and 5% w/w titanium dioxide nanoparticles used for bonding metal brackets, thereby eventually comprehending the permissible levels of titanium.
Materials and Methodology
The present in vitro study was carried out after obtaining ethical clearance from the Institutional Review Board (SSDC and RI, IRB: 409/9). Anatomically and morphologically well-defined noncarious extracted premolars for orthodontic purposes were selected. Teeth with restoration, enamel caries, fractured crown, fluorosis teeth, or hypoplastic teeth were excluded. The chosen teeth were cleaned to remove blood or any tissue debris and stored in distilled water to prevent dehydration.
Eighty freshly extracted teeth for orthodontic treatment were divided into 2 groups of 40 teeth each and were bonded with brackets containing 1% w/w (group I) and 5% w/w (group II) composite containing TiO2 NPs and placed in 10 mL of artificial saliva. Quantification of 1% w/w and 5% w/w composite containing TiO2 NPs is done for 4 timely periods 24 h, 2 months, 4 months, and 6 months. Spherical-shaped anatase phase TiO2 NPs with an average size of 20 to 30 nm were considered for the study. The inherent nature of titanium dioxide nanoparticles is to form agglomerates when dispersed in aqueous solutions in most cases; hence, spherical shaped were selected due to decreased surface area and photoactivity.
Nanocomposite Preparation
To achieve orthodontic adhesive containing 1% w/w, 40 mg of TiO2 NPs, and for 5% TiO2 w/w, 200 mg of TiO2 NPs were added to the 4,000 mg of orthodontic adhesive (Enlight; Ormco Corp), respectively, and blended by using a composite mixer (high energy ball mill) at a speed of 3,500 revolutions per min in dark environment for 5 min (Figure 1). A scanning electron microscopic examination was performed on the newly mixed nanocomposite to check the nanoparticles uniform distribution within the composite paste (Figure 2).
Composite Mixer.

Scanning Electron Microscopic Image Showing Uniform Distribution of Nanoparticles in Dental Composite.

Bonding Procedure
Polishing of the enamel surface was done using pumice with a low-speed handpiece followed by etching for 15 to 30 s with 37% phosphoric acid, rinsed for 15 s and then dried with an oil-free air spray. A thin layer of primer was applied on the enamel surface and cured for 10 s using an LED unit, followed by bonding orthodontic stainless steel brackets with adhesive and cured for 5 s on each side (total = 20 s).
Placing Bonded Teeth in Artificial Saliva
Artificial saliva composition includes 0.381 g NaCl, 0.213 g CaCl2.2H2O, 1.114 g KCl, 0.738 g KH2PO4, and 2.2 g mucin in 1,000 mL distilled water and pH 7. Each sample was placed in 10 mL artificial saliva in transparent white boxes and maintained at 37°C room temperature. In order to simulate oral conditions in vitro, 37°C intro-oral temperature was maintained. As the detection of element in the sample only can be done in a minimum quantity of 10 mL, it is preferred to take 10 mL of artificial saliva. After each interval (ie, after 24 h, 2 months, 4 months, and 6 months), the samples were shifted to conical tubes color-coded with blue (group I) and green (group II) to obtain impurities free artificial saliva as well as to settle impurities at the conical base of each tube (Figure 3).
1% w/w TiO2 Nanoparticles Containing Composite (Color Coded: Blue) and 5% w/w TiO2 Nanoparticles Containing Composite (Color Coded: Green).
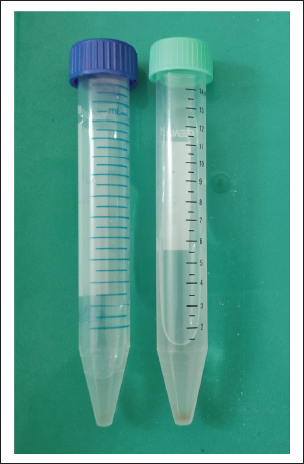
The following were the 3 step purification procedures before quantifying the amount of titanium content in the sample and removal of unwanted organic matter.
Centrifugation process: The samples are centrifuged under 3,200 rpm for 15 min at −5°C with the help of a centrifuge (REMI C23 PLUS) to obtain complete 100% pure liquid free of impurities. Dilution process: Following centrifugation, samples are diluted with distilled water from 10,000 ppm (1% w/w) and 50,000 ppm (5% w/w) to 100 ppb and 50 ppb as the quantification through ICP-MS can be done only in the range of 0 to 100 ppb. Stabilization process: The formed 100 ppb and 50 ppb through dilution process was stabilized with supra pure HCL to remove organic matter and to eradicate the interference of other elements by the detector (in ICP-MS).
Creation of Software Method
The stabilized sample containing titanium dioxide was added based on 3 specific conditions, that is, replication of a particular amount of cycles to obtain specific element ionization energy, scanning, and detecting liquid in a particular amount to get specific element mass-energy.
Quantification Process
This process was performed using ICP-MS to measure titanium at trace levels in biological fluids (Figure 4).
Inductively Coupled Plasma Mass Spectroscopy Machine (ICP-MS).

Statistical Analysis
The data was analyzed using Statistical Package for Social Sciences (SPSS) package version 20.0. An intragroup comparison was done using the Mann Whitney
Sample Distribution
The sample size was calculated using effect size of 9.2, power of the study 80% with degree of freedom 5. The Beta error was kept at 0.2 and alpha error was kept at 0.05, with confidence interval at 95%. The total sample size came to 40 (10 each group):
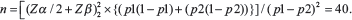
Results
A total sample of 80 extracted premolar teeth was used, wherein group I (
Comparison of Mean Titanium Release From 1% w/w Composite Containing TiO
2
NPs
Comparison of Mean Titanium Release From 1% w/w Composite Containing TiO2 NPs.
Comparison of Mean Titanium Release From 5% w/w Composite Containing TiO
2
NPs
Comparison of Mean Titanium Release From 5% w/w Composite Containing TiO2 NPs.
Comparison of Means of Titanium Release Between 1% w/w TiO
2
and 5% w/w TiO
2
Composite Containing TiO
2
NPs
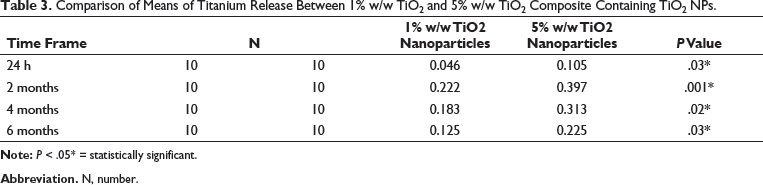
Comparison of Means of Titanium Release Between 1% w/w TiO2 and 5% w/w TiO2 Composite Containing TiO2 NPs.
Discussion
Beside orthodontic appliances, bonding materials might also retain plaque and cause a failure in the bonding of orthodontic brackets, thus delaying the treatment. Several teeth- or material-related factors can affect the bonding systems and the failure rates of orthodontic brackets. 17 Around 50% to 70% of patients undergoing fixed orthodontic appliance therapy reported enamel demineralization with an evidence of bacterial accumulation in gaps as tiny as 10 mm, at the adhesive-enamel junction. 18
GIC and composite are commonly used adhesives in orthodontics. Despite having favorable fluoride-releasing properties, GIC cannot withstand the forces generated in the posterior regions due to low fracture toughness. 19 20 21 On the other hand, composites are aesthetic filling materials, frequently used as adhesives for brackets. 22 Due to inherent property of mild roughness, they tend to accumulate more biofilms than the other restorative materials in vivo. 23 To overcome these drawbacks and improve their properties, pretreatment of inorganic fillers (nanofillers and fibers) and the addition of resin monomers 24 were introduced. Further, components like silver, tannic acid derivatives, amphiphilic lipids, and TiO2 are incorporated for antibacterial properties, fracture resistance, and surface roughness reduction. 23 Such hybrid composites, when used as adhesives, combat the invading bacteria along the tooth-bracket margins, thereby bettering the treatment outcome.
Nanotechnology that involves manipulating matter, atom by atom, is now being applied to augment the mechanical and antibacterial properties of orthodontic materials used in bonding. Compact loading of nanoparticle fillers can reduce polymerization shrinkage while increasing the mechanical properties like compressive strength and fracture toughness. 14 The reduced roughness of orthodontic adhesives significantly prevents bacterial adhesion. 23 Metal nanoparticles of 1 to 10 nm have shown the greatest biocidal activity against bacteria. 25
TiO2 NPs is the most commonly used nanoparticle with its particle size depending on the application. They constitute ultrafine particles <100 nm and fine particles of size 0.1 to 0.3 µm. It is proven to have an antibacterial effect and minimum toxicity. TiO2 NPs decompose organic compounds by the constant release of hydroxyl radicals and superoxide ions formed when exposed to non-lethal ultraviolet light. 26
Jiang et al 27 investigated that nanoparticle activity was dependent on size and phase. Nine sizes ranging from 4 to 195 nm were considered. Their results showed that the particle activity was constant above 30 nm and below 10 nm; it decreased uniformly from 30 nm to 10 nm. Likewise, the amorphous and anatase phases had the highest activity. Our study included approximately 20 to 30 nm-sized spherical anatase phase particles, thus opening a possibility for the activity and titanium release being partially dependent on both the size and phase.
A detailed review of literature12, 28, 29 by date portrayed antimicrobial properties and bond strength of titanium-based adhesives. The mean bond strength after adding 1% TiO2 is 18.17 ± 4.6564. The mean bond strength after adding 5% TiO2 is 13.9 ± 6.00256, but studies on the amount of titanium released from such adhesives over time are lacking. Hence, we measured the amount of titanium released from adhesives containing 2 different concentrations of titanium dioxide to monitor the trend in the quantities across time.
Our study being the first of its kind, we could not find any similar articles for comparing the titanium levels. However, 1% and 5% concentrations were considered for evaluation, owing to previous studies showing considerable antimicrobial and adequate shear bond strength with both the concentrations. Mechanical properties of 1% and 5% TiO2 nanoparticles composite such as shear bond strength are equal to or more than 6 to 8 MPa (as mentioned by Reynolds), and both types of TiO2 have good antibacterial property. As the percentage of TiO2 increases, agglomeration of titanium dioxide nanoparticles increases resulting in reduced shear bond strength. Hence, 1% and 5% of TiO2 have been focused.28, 29
As it is essential to consider the permissible limits also, along with the antimicrobial properties, we chose to examine the titanium released from those concentrations with established properties. Previous in vivo studies 26 revealed that after inhalation or oral exposure, TiO2 nanoparticles accumulate in the lungs, alimentary tract, liver, heart, spleen, kidneys, and cardiac muscles, and may result in liver and spleen damage, reduction in the number of microvilli, gastritis, and intestinal cancer. Cardiovascular and neurological damage also was noticed in the presence of titanium levels ranging between 10 and 50 mg/kg. In the present study, the maximum and minimum concentrations achieved were 0.397 ± 0.015 and 0.125 ± 0.133, respectively, that is, enormously reduced (around 0.0001-0.0003 mg/kg) than that required for the above adverse effects detected in the previous studies. 26 Thus, the quantities observed in our study using 1% and 5% concentrations TiO2 NPs were within permissible levels.
On comparing the 2 concentrations at 4 different time intervals, the quantities were significantly greater in the group II at all time frames, thus implying a significant increase in titanium released with an increase in concentration from 1% to 5%. The decrease in titanium release after 2 months is due to contact inhibition characteristics. 9
This being an in vitro study, the exact in vivo conditions could not be simulated. Most importantly, as dietary habits impact the release of ions, this could not be assessed. The overall number of teeth and those considered each time interval for the study was relatively less due to its novelty. In spite of considering the anatase phase, the extremely lesser values than the threshold for adverse effects derived in our study could be attributed to very few samples. However, our study could serve as a basis for future studies exploring such effects and hence adds valuable inputs to the existing literature.
Conclusions
In the present study, both the 1%(w/w) and 5%(w/w) TiO2 nanocomposite can be used safely. However, there is a difference in the release of TiO2 nanoparticles for 1% w/w and 5% w/w TiO2 nanocomposite at different intervals of time.
The mean Ti release from 5% (w/w) TiO2 nanocomposite is high compared to 1% (w/w) TiO2 nanocomposite at all the 4 intervals of time (ie, 24 h, 2 months, 4 months, and 6 months) and is statistically significant.
1% (w/w) TiO2 nanocomposite shows the highest release between 24 h and 2 months; however, there is no significant difference observed between 4 and 6 months.
5% (w/w) TiO2 nanocomposite shows highest release at 24 h to 2 months, followed by a decrease at 4 and 6 months, respectively, which is statistically significant.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent
Informed consent was not sought for the present study because no identifiable images were used.
