Abstract
Aim and Objectives
The study aimed to evaluate the antibacterial efficacy of titanium dioxide (TiO2) nanoparticle-coated stainless-steel orthodontic brackets and to assess cytological changes in the adjacent oral mucosa. The goal was to determine if TiO2 coatings reduce bacterial colonization and biofilm formation while ensuring biocompatibility with surrounding tissues.
Materials and Methods
This split-mouth randomized controlled trial was conducted on a sample of 30 patients who needed orthodontic treatment, aged between 16 and 30 years. TiO2-coated brackets were bonded on one maxillary quadrant, and non-coated brackets on the opposite side, with sides alternated to avoid bias. Brackets were coated using a dip-coating method followed by the process of annealing. Plaque samples were collected at 30 and 60 days post-bonding and cultured on blood agar for bacterial quantification. Microscopic analysis was performed using oil immersion field counts. Buccal mucosa samples were collected before bonding (T0) and at 30 days (T1), in order to assess cytological changes via exfoliative cytology using Giemsa staining.
Results
The TiO2-coated bracket group showed a slight but significant increase in bacterial culture grading from 1 (interquartile range [IQR]: 0-1) at T1 to 1 (IQR: 1-1) at T2, whereas the non-coated group increased from 2 (IQR: 2-3) to 3 (IQR: 2-3). TiO2-coated brackets consistently demonstrated significantly lower bacterial culture gradings than non-coated brackets at both T1 and T2 (
Conclusion
TiO2 nanoparticle coatings on stainless-steel orthodontic brackets have been shown to markedly reduce bacterial colonization and inhibit biofilm formation. Importantly, these coatings preserve the cytological integrity of the surrounding oral mucosa, indicating their biocompatibility. By limiting microbial accumulation, TiO2-coated brackets may contribute to improved oral hygiene during orthodontic therapy. This preventive effect has the potential to reduce common complications such as white spot lesions and gingivitis. However, further long-term clinical studies are necessary to validate these promising outcomes throughout the entire course of orthodontic treatment.
Keywords
Introduction
Orthodontic treatment primarily aims to correct malocclusions, enhance dental function, and improve facial aesthetics. Among the various orthodontic appliances, stainless steel brackets remain the most commonly used due to their excellent mechanical strength, durability, cost-effectiveness, and corrosion resistance. 1 However, one major drawback of stainless-steel brackets is their tendency to harbor bacterial plaque and promote biofilm formation, posing challenges to maintaining optimal oral hygiene during treatment. 2
Orthodontic brackets create retention sites that trap food debris and microorganisms, leading to plaque accumulation. This bacterial buildup increases the risk of enamel demineralization, white spot lesions, gingival inflammation, and periodontal diseases. 3 White spot lesions, which are early signs of enamel decay, are particularly common in orthodontic patients and can significantly compromise treatment aesthetics. Moreover, the prolonged presence of pathogenic bacteria can cause gingivitis and periodontal inflammation and may even increase systemic health risks in vulnerable individuals. 4
Orthodontic appliances disrupt the natural oral microbiome by favoring the growth of harmful bacteria, prompting the need for effective antimicrobial strategies. 4 In recent years, nanotechnology has offered promising solutions to enhance the antibacterial properties of dental materials without compromising their mechanical integrity.5, 6 Among various nanomaterials, titanium dioxide (TiO2) nanoparticles have gained attention due to their potent antibacterial activity, biocompatibility, and photocatalytic properties. 7
TiO2 nanoparticles exert their antibacterial effects by generating reactive oxygen species (ROS) upon exposure to ultraviolet (UV) or visible light. These ROS damage bacterial cell walls, interfere with their metabolism, and prevent biofilm formation, leading to bacterial cell death. Additionally, TiO2 coatings possess self-cleaning properties and high durability, making them suitable for long-term clinical applications in orthodontics. 8
Several in vitro studies have explored the application of TiO2 nanoparticles in dentistry, especially for coating implants, restorative materials, and orthodontic brackets. While these studies demonstrate significant antibacterial potential, further clinical research is necessary to assess their long-term effectiveness, mechanical resilience, and behavior in real-world oral environments. 9
Alongside antimicrobial benefits, the biocompatibility of TiO2-coated brackets must also be carefully examined. Particular attention should be given to their interaction with adjacent oral tissues. Continuous exposure to nanoparticles from these coatings raises concerns about their potential cytotoxicity. The oral mucosa, which is in direct contact with orthodontic appliances, may be affected if nanoparticles leach over time. 10
To assess safety, exfoliative cytology—an established, non-invasive method—can be employed to detect cellular changes in oral epithelial tissues. This technique helps identify inflammation, nuclear abnormalities, and other cytological indicators of potential adverse reactions. Studying exfoliated cells near TiO2-coated brackets can thus offer vital insights into the biological response of oral tissues to prolonged nanoparticle exposure. 11
This study aimed to evaluate the antibacterial efficacy of stainless-steel orthodontic brackets coated with TiO2 nanoparticles and to assess the cytological changes in the oral mucosa adjacent to these brackets. Specifically, it investigated whether TiO2 coatings reduce bacterial adhesion and biofilm formation compared to uncoated brackets, it also analyzed the cytological response of the oral mucosa to determine any impact on epithelial cells, and assessed the clinical viability and long-term benefits of using TiO2-coated brackets in enhancing oral hygiene, minimizing treatment-related infections, and improving overall clinical outcomes in orthodontic patients.
Material and Methods
Study Design (Chart 1)
This study was conducted as a split-mouth randomized controlled trial (RCT)
12
at the Department of Orthodontics and Dentofacial Orthopaedics. The research protocol was approved by the Institutional Ethics Committee and registered in the Clinical Trials Registry. In this design, each participant received both the experimental and control interventions in different maxillary quadrants, allowing intra-patient comparison to reduce variability. TiO2-coated brackets were bonded to one maxillary quadrant (Figure 1), and non-coated brackets on the opposite side (Figure 2), with sides alternated, and random allocation (
Titanium Dioxide (TiO2) Coated Brackets.

Non-coated Brackets.

Consolidated Standards of Reporting Trials (CONSORT) Chart.
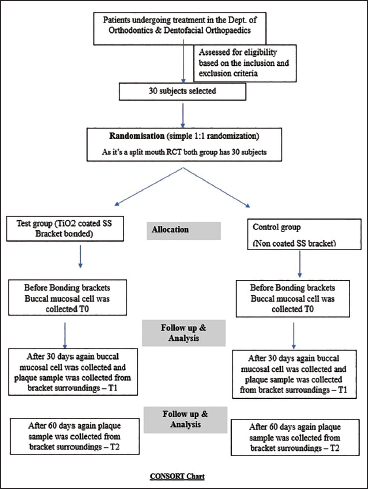
Sample Size and Sampling Technique
The sample size was determined based on data from a prior study by Avula and Padmanabhan. 14 With an effect size of ~1.0, a power of 80%, a significance level of 0.05, and a standard deviation of 1.5, the estimated sample size was calculated as 30 patients. The sampling was done randomly by selecting individuals who fulfilled the study’s inclusion and exclusion criteria relevant to orthodontic treatment.
Inclusion and Exclusion Criteria
Participants aged between 16 and 30 years, maintaining sound systemic and dental health, with no prior history of orthodontic treatment or signs of periodontitis, were included. Individuals with enamel defects, systemic illness, recent antibiotic usage, tobacco habits, prostheses, pregnancy, or poor oral hygiene were excluded. This ensured a homogeneous and compliant sample population, crucial for obtaining reliable results.
Study Protocol and Hygiene Instruction
Prior to bracket bonding, all 30 selected participants underwent prophylactic dental cleaning. They were instructed to maintain oral hygiene by brushing twice daily with a non-fluoridated dentifrice, since fluoridated formulations possess antibacterial properties that could interfere with the study outcomes. To minimize potential confounding factors in the microbial analysis, participants were also advised to refrain from using chewing gum, mouthwash, or antibiotics throughout the 60-day study period.
Bracket Coating and Preparation 15
Stainless steel brackets were sent to Technicoat (Pune, India) for TiO2 nanoparticle coating via the dip-coating method. Prior to coating, the brackets were cleaned in an ultrasonic bath using acetone or ethanol, rinsed, and optionally etched using 10% nitric acid. A suspension of TiO2 nanoparticles was prepared in an ethanol-water solution, with polyethylene glycol (PEG) or polyvinyl alcohol (PVA) as a binder, sonicated for uniform dispersion, and the pH was adjusted for stability.
Dip Coating and Annealing Procedure16, 17
The brackets were immersed in the TiO2 suspension for 30-60 s, withdrawn slowly, and air-dried for uniform coating. This process was repeated 2-3 times to achieve adequate thickness. Coated brackets were then dried at 80°C-100°C and annealed in a furnace at 400°C-600°C for up to 2 h to improve crystallinity and adhesion. Coated brackets were inspected for defects and reprocessed if necessary.
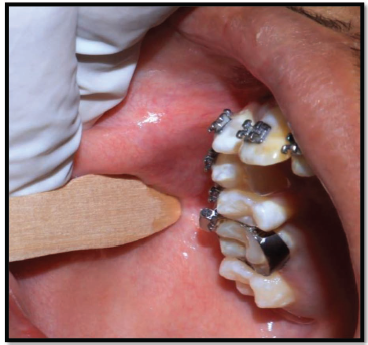
Cytological Sample Collection18, 19
Buccal mucosa cell samples were collected at two time points: T0 (before bonding) and T1 (30 days after bonding) (Figure 3). Subjects rinsed their mouth and fasted for 30 min prior to sampling. Using a sterile cytobrush or wooden spatula, epithelial cells were gently scraped from the buccal mucosa using circular or up-and-down strokes. The samples were then transferred to slides, fixed in ethanol, and stained using 5% Giemsa stain for microscopic analysis.
Plaque Sample Collection 20
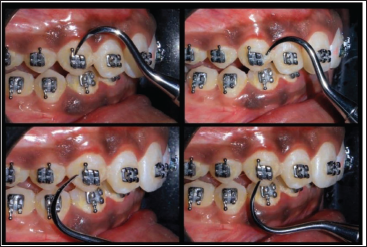
Plaque was collected from the maxillary quadrants around the brackets (Figures 4 and 5) using the four-pass technique with a sickle scaler at two time points: 30 and 60 days post-bonding (Figure 6). This method ensured comprehensive plaque collection. Samples were immediately placed in sterile containers with normal saline to preserve microbial integrity and transported to the microbiology lab for further analysis.
Buccal Mucosal Cell Collection with Wooden Spatula.
Non-coated Brackets Bonded.
Titanium Dioxide (TiO2) Coated Brackets Bonded.
Four Pass Technique of Plaque Sample Collection Adjacent to Brackets.
Microbiological Processing of Plaque Samples
In the microbiology lab at Jawaharlal Nehru Medical College (JNMC), plaque samples were homogenized by vortexing. A 10 µL aliquot was plated onto blood agar and incubated for 24-48 h. Bacterial colonies were counted post-incubation, and further staining was performed to facilitate microscopic quantification using oil immersion.
Bacterial Quantification and Grading21, 22
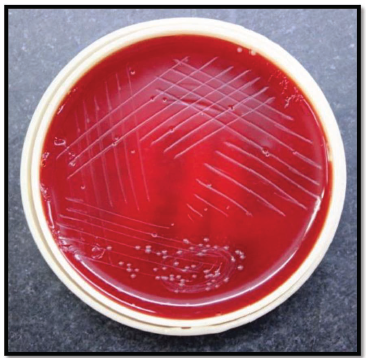
Culture plates were graded based on the number of quadrants showing growth: 3+ (all quadrants), 2+, 1+, or 0 (no growth). Microscopic grading based on oil immersion field (OIF) included: 1+ (<1 bacterium/OIF), 2+ (2-10), 3+ (11-50), and 4+ (>50). This detailed quantification enabled the assessment of microbial colonization in response to the different types of brackets over the course of the treatment (Figures 7 and 8).
Bacterial Colony Growth on Blood Agar from the Plaque Sample Collected from Non-coated Brackets.
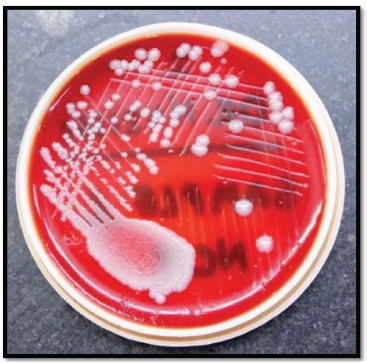
Bacterial Colony Growth on Blood Agar from the Plaque Sample Collected from Titanium Dioxide (TiO2) Coated Brackets.
Statistical Analysis
Data were tabulated in Microsoft Excel 2021 and analyzed using IBM Statistical Package for the Social Sciences (SPSS) Statistics for Windows, version 27.0 (IBM Corp., Armonk, NY). Graphs, box plots, and pie charts were created with GraphPad Prism version 10.1.2 (GraphPad Software, La Jolla, CA). Descriptive statistics included frequencies and percentages for categorical variables, and medians with interquartile ranges (IQRs) for quantitative data. Non-parametric tests were used due to ordinal observations. Wilcoxon’s test was applied for intra- and inter-group comparisons in the split-mouth design, while McNemar’s test analyzed categorical variables. A
Results
Bacterial Culture Gradings (Table 1)
Median (Interquartile Range) and Comparisons of the Bacterial Culture Gradings (Intragroup and Intergroup).

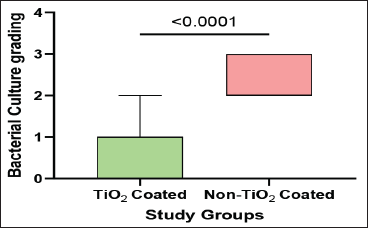
In the TiO2-coated bracket group, the median bacterial culture grading increased significantly from 1 (IQR: 0-1) at time point T1 to 1 (IQR: 1-1) at T2 (Wilcoxon signed-rank test,
Median (Interquartile Range) and Comparisons of the Bacterial Cell Counts (Intragroup and Intergroup).

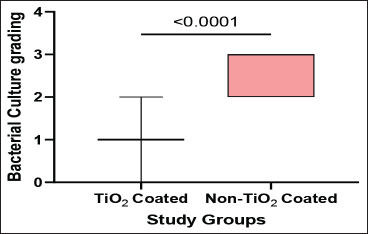
Conversely, in the non-TiO2-coated bracket group, the median grading rose from 2 (IQR: 2-3) at T1 to 3 (IQR: 2-3) at T2; however, this change was not statistically significant (
Comparisons between groups revealed significantly lower bacterial culture gradings in the TiO2-coated group compared to the non-coated group at both T1 and T2 (Box and Whisker Plots 1 and 2). At T1, the median grading was 1 (IQR: 0-1) versus 2 (IQR: 2-3) (
Bacterial Cell Counts (Table 2)
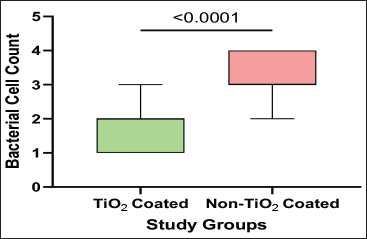
In terms of bacterial cell counts, the TiO2-coated group showed no significant change between T1 [2 (IQR: 1-2)] and T2 [2 (IQR: 1.25-2)] (
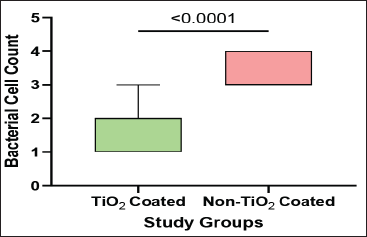
However, when comparing groups, the TiO2-coated group consistently demonstrated significantly lower bacterial cell counts than the non-coated group at both T1 and T2 (Box and Whisker Plots 3 and 4). At T1, the median count was 2 (IQR: 1-2) versus 3 (IQR: 3-4) in the non-coated group (
Cytopathology Characteristics (Table 3)
Analysis of polymorph status revealed minimal changes over time within each group. In the TiO2-coated group, samples showing occasional polymorphs increased slightly from 96.7% at baseline (T0) to 100% at T1; this increase was not statistically significant (McNemar’s test,
Cytopathology Characteristics and Comparisons (Intragroup and Intergroup).
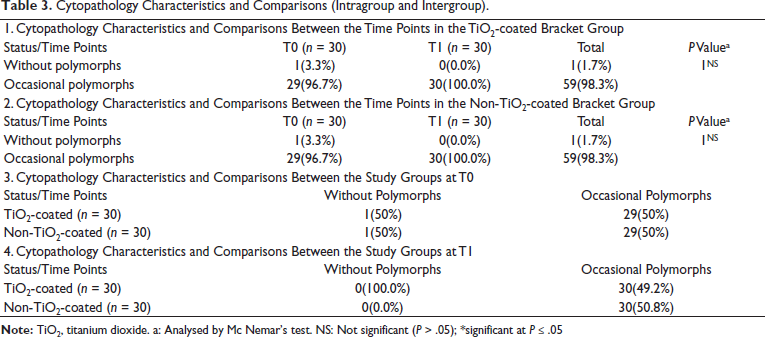
Between groups at baseline (T0), both TiO2-coated and non-coated brackets had identical distributions, with 1 (3.3%) sample without polymorphs and 29 (96.7%) with occasional polymorphs each. At T1, all samples in both groups exhibited occasional polymorphs, with no samples lacking polymorphs.
Discussion
Orthodontic therapy corrects malocclusions, but stainless-steel brackets, despite their strength and cost-effectiveness, can encourage biofilm formation, increasing the risk of enamel and periodontal problems. The accumulation of plaque around brackets alters the oral microbiome by encouraging the growth of pathogenic bacteria, making oral hygiene maintenance more challenging. This situation necessitates innovative antimicrobial strategies to mitigate these risks.
Nanotechnology presents promising solutions, particularly with TiO2 nanoparticles. TiO2 is known for its strong antibacterial and photocatalytic properties; when exposed to UV or visible light, it produces ROS that damage bacterial cell walls, preventing biofilm formation. Kumaravel et al. 23 reported that TiO2 coatings exhibit self-cleaning properties, which help minimize plaque accumulation and thereby enhance the durability of orthodontic brackets. Though TiO2 has shown antibacterial effectiveness in various dental applications, its clinical performance specifically on orthodontic brackets requires further investigation.
Our study employed a split-mouth RCT design, recognized for its methodological rigor. This approach reduced inter-individual variability by using each patient as their own control, which controls confounding factors such as genetics, oral hygiene habits, diet, and salivary composition—all of which influence microbial colonization and tissue response. This design enhanced statistical power and the reliability of results even with a modest sample size.
The primary aim was to assess whether TiO2-coated brackets reduce bacterial colonization and biofilm formation, which are the major contributors to white spot lesions, gingivitis, and periodontal disease in orthodontic patients. The secondary goal was to evaluate the cytological response of the buccal mucosa adjacent to the brackets to detect any inflammatory or degenerative tissue changes caused by mechanical irritation or microbial activity.
We enrolled 30 patients aged between 16 and 30 years with good systemic health and no prior orthodontic treatment. A split-mouth design was used by bonding TiO2-coated brackets on one maxillary quadrant and non-coated brackets on the opposite quadrant (Figures 4 and 5), allowing direct intra-subject comparison. The 60-day follow-up period was sufficient to observe biofilm development and tissue response while ensuring patient compliance.
Cytological analysis of buccal mucosa cells was conducted at baseline (T0) and after 1 month (T1) to identify histopathological changes indicative of inflammation or tissue stress related to bracket placement. Plaque samples collected at 30 and 60 days were analyzed via culture-based methods and microscopic grading, providing a comprehensive assessment of microbial colonization.
Plaque growth was assessed using a quadrant grading system on agar plates and OIF microscopic bacterial counts. This dual approach offered both qualitative and quantitative data, correlating visible bacterial colonies with actual cell density per unit area, and enhancing the robustness of microbial evaluation.
Results showed a significant reduction in bacterial load and bacterial counts on the TiO2-coated brackets when compared to non-coated ones (Table 1). The TiO2-coated side displayed only a slight but statistically significant increase in bacterial culture grade from T1 to T2, indicating sustained antimicrobial activity over 60 days. Conversely, the non-coated side exhibited a higher bacterial load with a non-significant increase, likely due to already elevated baseline levels.
Inter-group comparisons revealed significantly lower bacterial counts on TiO2-coated brackets at both time points. These findings align with previous studies, such as Rao et al.,
24
who reported a reduction in bacterial accumulation on TiO2-coated brackets. Avula and Padmanabhan
14
reported that nitrogen-doped TiO2-coated brackets demonstrated a significant reduction in
Microscopic bacterial counts remained stable over time on the TiO2-coated side, while the non-coated side maintained higher counts (Table 2), further confirming TiO2’s bactericidal effects, likely due to ROS generation under ambient light, damaging bacterial cell walls and DNA. These results are consistent with studies by Shah et al. 26 and Kumaravel et al., 23 who demonstrated TiO2’s broad-spectrum antibacterial efficacy against common oral pathogens implicated in plaque formation.
Cytological smears revealed a marginal, non-significant increase in polymorphonuclear leukocytes (PMNs) on the TiO2-coated side, suggesting no additional mucosal irritation or inflammation due to the coating (Table 3). No atypical or malignant changes were observed, supporting TiO2’s biocompatibility, corroborated by prior in vitro studies on gingival fibroblasts 27 and animal models, 28 which reported no significant tissue damage or apoptosis.
Scanning electron microscopy (SEM) analysis highlighted distinct surface differences between coated and non-coated brackets (Figure 9). TiO2-coated brackets showed smoother, more uniform surfaces, which reduced friction between the archwire and the bracket, potentially improving tooth movement efficiency, lowering the force required, and enhancing patient comfort (Figures 10 and 11). Smoother surfaces also minimize food debris retention and bacterial adherence, reducing plaque formation and associated risks of enamel decalcification and gingival inflammation. This aligned with a study by Suresh et al., 29 who demonstrated that TiO2 coatings reduce frictional forces on orthodontic wires, potentially improving treatment outcomes.
Scanning Electron Microscope (SEM).

Lateral Scanning Electron Microscope (SEM) View of Coated and Non-coated Brackets.

Front Scanning Electron Microscope (SEM) View of Coated and Non-coated Brackets.

Overall, the present study demonstrated the clinical potential of TiO2-coated orthodontic brackets in reducing bacterial colonization and promoting oral hygiene during orthodontic treatment. While bacterial growth was not completely eliminated, the significant reduction in microbial load and absence of increased mucosal inflammation provide a promising outlook for the application of nanotechnology in orthodontics.
By integrating cytological and microbiological analyses with culture-based quantification and microscopic grading, this study offers a comprehensive methodology for evaluating the impact of bracket coatings on oral health. The findings of the present study highlighted the advantages of TiO2-coated brackets, emphasizing their potential as an effective preventive strategy. These coated brackets helped to minimize plaque accumulation and lowered the risk of related complications, and ultimately contributed to improved treatment efficiency and greater patient comfort.
Comparisons of the Bacterial Culture Gradings Between the Study Groups at T1.
Comparisons of the Bacterial Culture Gradings Between the Study Groups at T2.
Comparisons of the Bacterial Cell Counts Between the Study Groups at T1.
Comparisons of the Bacterial Cell Counts Between the Study Groups at T2.
Limitations and Future Prospects
This study highlights the antibacterial efficacy and cytological safety of TiO2-coated orthodontic brackets, but has notable limitations. The small sample size (30 patients) and short follow-up period (60 days) restrict the ability to assess long-term effects and broader clinical relevance. Additional concerns include lack of blinding, omission of bond strength testing, and reliance on culture-based microbial analysis, which is limited to detecting only cultivable species and therefore overlooks the true complexity and diversity of the oral microbiota. Furthermore, unassessed patient compliance and the absence of hypersensitivity evaluation reduce the generalizability and clinical applicability of the findings.
Future research should address these limitations through long-term, multicenter clinical trials involving larger and more diverse populations to evaluate the sustained efficacy and biocompatibility of TiO2-coated brackets over the full course of orthodontic treatment. Advanced approaches could explore the development of smart, light-activated antimicrobial coatings or TiO2 composites doped with metals such as silver or zinc to enhance performance. Comparative studies with other nanoparticle coatings (e.g., silver, zinc oxide, graphene oxide) may also help identify the most effective and biocompatible materials. Moreover, the integration of such coatings with digital health systems could enable real-time monitoring of oral hygiene and personalized care strategies. Expanding the use of antimicrobial coatings to other dental devices like retainers, archwires, and aligners could pave the way for comprehensive antimicrobial solutions across multiple branches of dentistry.
Conclusion
TiO2 coating on stainless steel orthodontic brackets showed significantly reduced bacterial colonization and biofilm formation compared to non-coated brackets over 60 days.
TiO2 coatings consistently inhibited bacterial growth over time, as confirmed by both culture-based and microscopic OIF counts.
TiO2 coatings have been indicated to be biocompatible, non-toxic, and safe for long-term intraoral use through cytological analysis of the adjacent buccal mucosa, showing no signs of inflammation, degeneration, or malignancy.
TiO2 coating on brackets may enhance oral hygiene during orthodontic treatment by reducing plaque buildup and preventing issues like white spot lesions and gingivitis.
Footnotes
Acknowledgment
The authors sincerely thank Guide Professor Sanjeev Kumar Verma and other co-guides for their valuable contributions, insights, and support during this study.
Authors Contribution
Debdeep Kuila: Conceived the research idea, designed the study, conducted data analysis, and drafted the manuscript.
Sanjeev Kumar Verma: Project administration, supervision. Provided critical revisions.
Arbab Anjum: Contributed to methodology writing and reviewed literature. Provided supervision throughout the project.
Nazish Fatima: Analyzed data and performed formal analysis.
Pramod Kumar Yadav: Review and editing.
Aiswareya G.: Contributed to manuscript writing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The research protocol was approved by the Institutional Ethics Committee. The trial was registered in Clinical Trial Registry-India (ICMR-NIMS) with Ref No.: REF/2025/05/106684.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Informed consent was obtained from the patients and parents.
