Abstract
Humanized mice engrafted with human CD34+ hematopoietic stem cells (HSCs) are invaluable models for the preclinical evaluation of cancer immunotherapy drugs, helping to bridge the gap between in vitro studies and clinical trials. However, there are inherent challenges associated with these models, which require careful consideration, as they can obscure the distinction between treatment-related effects and model-specific background lesions. Here, we provide a comprehensive overview of common histopathological changes in such models, focusing on humanized nonobese diabetic (NOD) Cg-Prkdc scid Il2rg tm1Wjl/SzJ (huNSG) mice engrafted with human CD34+ HSCs. We characterize engraftment kinetics and histopathological changes at various timepoints post-engraftment and provide examples of huNSG mice utilized to evaluate immuno-oncology compounds and how these drugs influenced the development of huNSG-related background lesions. Flow cytometry and immunohistochemistry analyses reveal dynamic changes in human immune cell populations, with a predominance of B cells over T cells at early timepoints, shifting to T cell predominance at later stages. Notably, spontaneous granulomatous lesions, dominated by human macrophages, are observed in multiple organs, which increase in incidence and severity over time. These lesions are exacerbated by immunostimulatory treatments, highlighting the need for careful interpretation of drug-induced effects vs model-specific background pathology. Additionally, bone marrow necrosis that resembles ischemic lesions is identified in some huNSG mice, particularly following immune stimulation. Our findings underscore the critical importance of understanding the inherent pathological changes in huNSG mice to accurately assess the safety of cancer immunotherapy drugs.
Keywords
Humanized mice (huMice) refer to immunodeficient mice engrafted with human cells, including hematopoietic stem cells (HSCs), peripheral blood mononuclear cells (PBMCs), or fetal tissues such as thymus or liver. 1 Here we focus on huMice that have been engrafted with human CD34+ HSCs to generate a functional human immune system. HuMice represent a physiologically relevant in vivo platform for anticancer drug development by enabling the growth of human tumor xenografts in mice with a humanized immune system.49,54 They have become particularly relevant in the field of immuno-oncology drug development, including the development of monoclonal antibodies, 13 checkpoint inhibitors,16,56 targeted T cell therapies,4,15,18 and co-stimulator therapies, 44 where they help bridge the gap between in vitro studies and clinical trials.
A prerequisite for successful xenoengraftment of human HSCs and subsequent reconstitution of a functional human immune system in a murine host is the recipient’s immunodeficiency. Immunodeficiency is achieved by utilizing immunodeficient mouse models, often combined with preconditioning with sublethal γ-irradiation or radiomimetic drugs, such as busulfan, to further reduce the functionality of the host’s immune system. 1 Currently, there are numerous immunodeficient mouse strains available, each with its own characteristics, which, together with different engraftment protocols, have a direct impact on the ability of the xenotransplanted cells to engraft the host and establish a humanized immune system. 46 A commonly used humanized mouse model strain is the immunodeficient nonobese diabetic (NOD) Cg-Prkdcscid Il2rgtm1Wjl/SzJ (NSG) mouse that has been humanized by engraftment with CD34+ HSCs (huNSG). 53 NSG mice have a mutated Prkdc gene (also known as scid mutation) and complete deletion of the interleukin 2 receptor gamma chain. The scid mutation results in functionally deficient B and T cells, whereas the absence of the interleukin 2 receptor gamma chain blocks natural killer (NK) cell differentiation. After preconditioning, NSG mice can be reconstituted with CD34+ HSCs derived from human umbilical cord blood to develop a stable, multilineage engraftment of functional human immune cells, including the development of diverse human immune cell populations, such as T cells, B cells, monocytes, dendritic cells, and granulocytes.25,50,53 The human cell engraftment is maintained for up to 32 weeks; however, the composition of the humanized immune system undergoes constant dynamic changes. For instance, B cells typically predominate over T cells until 16–24 weeks post-engraftment (wpe), and at later timepoints, T cells predominate over B cells. 2
However, huNSG mice also present numerous challenges. For instance, significant inter-animal variability in engraftment levels is frequently observed due to differences between human HSC donors. 54 This heterogeneity has been confirmed by histology, revealing different levels of engraftment in immune cell organs and inconsistencies between tissue engraftment levels compared to peripheral blood engraftment assessed by flow cytometry. 6 Different NSG recipient strain variants and engraftment methodologies also significantly influence the establishment and functionality of the human immune system within these models.49,52 Furthermore, the resulting humanized immune system is not fully representative of human immunity; for example, humans exhibit a higher myeloid-to-lymphoid ratio compared to huNSG mice, where myeloid cells typically constitute only 5% to 10% of the immune population. 2 Additionally, huNSG mice are known to develop xenoreactive lesions characterized by the infiltration of human macrophages into organs such as the bone marrow, liver, spleen, and kidney.6,51 These model-related histopathological lesions may pose a significant challenge for pathologists evaluating the potential effects of drug candidates, as they can obscure the distinction between treatment-related effects and inherent model-specific lesions. Therefore, the goal of this manuscript is to provide a comprehensive overview of the most common histopathological background changes observed in huNSG mice at various timepoints following human umbilical cord CD34+ HSC engraftment. Additionally, as huMice can show differential responses to drug compounds based on their mechanism of action (MoA), 51 we provide examples where huNSG mice were utilized to evaluate immuno-oncology compounds with distinct mechanisms and how these drugs influenced the development of huNSG-related background lesions.
Materials and Methods
Animals
Experiments presented herein involved huNSG female mice and were conducted following strict ethical guidelines of the Association for Assessment and Accreditation of Laboratory Animal Care, an international-accredited animal facility of the Roche Innovation Center Zurich and Roche Innovation Center Basel. Experimental design and procedures were thoroughly reviewed and approved by the internal Institutional Animal Care and Use Committee. This approval process ensured that all efforts were made to minimize animal suffering and that the number of animals used was kept to the minimum necessary to achieve valid results. All animal housing, care, and experimental procedures were in compliance with the guidelines set by Gesellschaft für Versuchstierkunde/Society for Laboratory Animal Science, the Federation of European Laboratory Animal Science Associations, and the German Animal Welfare Act (Tierschutzgesetz).
NSG mice were bred at The Jackson Laboratory and were shipped to Roche at 5–6 weeks of age to start the humanization process. Animals were maintained under opportunistic-pathogen-free conditions in individually ventilated cages (GM500, Tecniplast, Italy) with 12-hour light/dark cycles at room temperature (20–24°C), with a relative humidity of 50–60%, in accordance with committed guidelines (Gesellschaft für Versuchstierkunde/Society for Laboratory Animal Science, Federation of European Laboratory Animal Science Associations, and Tierschutzgesetz). Health monitoring was performed on a daily basis.
NSG mice for the characterization study and studies 1, 2, and 3 were injected intraperitoneally with 15 mg/kg busulfan (prepared from a 6 mg/mL stock solution) to deplete murine HSCs, followed by an intraperitoneal injection of 1×105 human HSCs (CD34+ umbilical cord blood HSCs, STEMCELL Technologies, Canada) 24 hours later. For study 4, NSG mice received a single 100 cGy dose of γ-irradiation for preconditioning instead of busulfan to deplete murine HSCs and were injected with 5×104 human HSCs (CD34+ umbilical cord blood HSCs, STEMCELL Technologies, Canada) 24 hours later. Human T cell frequencies in blood from all these huNSG mice were assessed 13 weeks after the stem cell injection by flow cytometry. Only huNSG mice with a humanization rate greater than 20% (i.e., more than 20% of circulating human immune cells within all leukocytes) and up to 80% at 13 wpe were used in these studies, and a T cell count within the range of 30–300 cells/mL was mandatory.
Studies
The results presented herein include a pathology baseline characterization study of untreated huNSG mice at 4 different post-engraftment timepoints, as well as 4 different pharmacology studies with 4 different immune-oncology drugs targeting tumor-associated antigens and immune cell-associated antigens (study 1–4). For the baseline characterization study, 4 groups of ten animals each (40 animals in total) were evaluated at 15, 18, 26, and 35 wpe (weeks between the day of engraftment and necropsy), respectively. Further details of these studies are reported in the respective results sections below.
Flow Cytometry
Flow cytometry was used to assess leukocyte kinetics over time following CD34+ HSC engraftment. This analysis included the evaluation of human leukocytes, B cells, and T cells in peripheral blood at 4 distinct timepoints: 13, 16, 27, and 30 wpe. To further investigate the frequency of various human cell populations, specifically B and T cells, myeloid cells, and NK cells across different tissues (liver, spleen, bone marrow, lymph node, thymus, and blood), additional sampling was conducted at 15 wpe. Human blood samples from 3 healthy donors were also included for comparative purposes.
Blood from huNSG mice was collected in heparin tubes, and tissues were processed in fluorescence-activated cell sorting buffer (Dulbecco’s phosphate-buffered saline, 2% fetal bovine serum, 2 mM ethylenediaminetetraacetic acid). To obtain single-cell suspensions, spleen, bone marrow, thymus, and liver samples were minced through a cell strainer. Red blood cells were lysed by mixing with lysis buffer (BD Pharm/Lyse, 555899) for 5 minutes at room temperature to eliminate them from fresh blood, bone marrow, and spleen single-cell suspensions. The single-cell suspensions were then labeled with the flow cytometry panel of antibodies (Supplemental Table S1) to detect the following cellular subsets of human cells: leukocytes, T cells (including subsets of cytotoxic cells and T helpers), regulatory T cells, B cells, NK cells, myeloid cells, monocytes, and T cell receptors (TCRs) α/β and δ/γ. All antibodies (obtained from BioLegend, United States) were incubated with the single-cell suspensions at a 1:500 dilution (in fluorescence-activated cell sorting buffer) for 30 minutes at 4°C without light exposure. After 2 wash steps with fluorescence-activated cell sorting buffer, labeled cells were resuspended in 200 μL fluorescence-activated cell sorting buffer containing 4′,6-diamidino-2-phenylindole (1:10000) and acquired on a BD Fortessa cell analyzer. Information about the primary antbodies used can be found in Supplemental Table S1.
ELISA
Serum samples were collected 15 weeks after humanization and stored at −20°C. The samples were tested for the presence of human IgG antibodies by detecting the human Fc subunit (huFc). Biotinylated anti-huFc, test samples, digoxygenin-labeled anti-huFc (Dig-anti-Fc) antibody, and anti-digoxygenin detection antibody (horseradish peroxidase) were added stepwise into a 96-well streptavidin microtiter plate and incubated after every step for 1 hour at room temperature. The plate was washed 3 times after each step to remove unbound substances. Finally, the peroxidase-bound complex was visualized by adding 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) substrate solution to form a colored reaction product. The reaction product intensity, which was photometrically determined at 405 nm (with reference wavelength of 490 nm), was proportional to the analyte concentration in the serum sample. Materials and reagents used for this in-house ELISA are provided in Supplemental Table S2.
Pathological Assessment
huNSG mice were euthanized by deep isoflurane anesthesia followed by an intraperitoneal injection of pentobarbital (150 mg/kg) and cervical dislocation or bilateral pneumothorax. Animal organs and tissues were examined in situ, removed, and then screened for abnormalities. Macroscopic changes were documented. A comprehensive list of tissues (Supplemental Table S3) was sampled for phenotyping. For mice in pharmacology studies, only a few selected organs were harvested for histological purposes and are reported in detail within the individual studies below. Collected tissues were fixed in 10% neutral-buffered formalin for a maximum of 48 hours, transferred to phosphate-buffered saline with 0.1% w/v sodium azide, stored at 4°C for a maximum of 4 days before being trimmed according to the Registry of Industrial Toxicology Animal-data guidelines,32,38,43 and embedded in paraffin. Consecutive sections (3–5 µm thick) were prepared and routinely stained with hematoxylin and eosin or subjected to immunohistochemistry (IHC). To characterize specific findings, selected sections were additionally stained with Prussian blue to detect iron deposits and phosphotungstic acid-hematoxylin (PTAH) to visualize fibrin.
Immunohistochemistry
IHC was conducted on selected tissues to characterize cell populations and the origin of cells (human vs mouse) observed in the lymphoid organs and in the lesions identified via hematoxylin and eosin staining. Human leukocyte markers included CD3ε (T cells), CD20 (B cells), CD68 (macrophages), FOXP3 (regulatory T cells), while mouse leukocyte markers, included CD45 (general leukocyte marker) and F4/80 (macrophages). In addition, labeling for human leukocyte antigen (HLA)-A was utilized to detect the overall prevalence of human cells in the lymphoid organs of engrafted mice. Different combinations of duplex or multiplex immunolabeling for CD3, CD20, and CD68 were also conducted on selected samples to provide a more comprehensive view of cellular populations within a tissue. In addition, immunolabeling for fibroblast activation protein (FAP) was used to better characterize lesions observed in the bone marrow. The automated immunolabeling procedure for IHC was performed for all markers on a Discovery Ultra instrument (Ventana, Switzerland), with the exception of immunolabeling for HLA-A, which was conducted on a Leica Bond (Leica Biosystems, United Kingdom) instrument. Information about the primary antibodies can be found in Supplemental Table S4. As a negative control for nonspecific binding, serial sections were stained with an isotype control antibody (raised in the same species and matching the isotype of the primary antibody) at the same concentration as the primary antibody.
Histological Scoring
Histopathological lesions were scored following a semiquantitative grading scheme ranging from 1 to 5 (1—minimal; 2—mild; 3—moderate; 4—marked, and 5—severe). 45 Tissue protein expression via IHC was evaluated using a semiquantitative scoring system based on staining intensity. The scores were defined as negative (no discernible staining or staining intensity comparable to the isotype control), weak (very low intensity staining), slight (faint staining), moderate (definite staining, easily visible at low magnification), or strong (intense dark staining, may obscure cellular structure) immunolabeling. The location of the labeling was identified as membranous, cytoplasmic, or nuclear, and the frequency was judged as the percentage of labeled cells within the specific cell population.
Results
Flow Cytometry Analysis
For the baseline assessment of human immune cell development in huNSG mice, flow cytometry analysis was conducted in peripheral blood samples collected at 13, 16, 27, and 30 wpe to characterize the kinetics of humanization engraftment. Populations of human CD45+ leukocytes, as well as human CD3+ T cells and CD20+ B cells, were evaluated. The humanization process was confirmed over time by a gradual increase in the percentage of human leukocyte cells relative to the total number of white blood cells in the blood (Fig. 1a). Human B cell counts showed high numbers detected at 13 wpe, which remained stable over time (Fig. 1a). In contrast, human T cells were detected at low levels at 13 wpe, but their counts steadily increased over time thereafter (Fig. 1a).
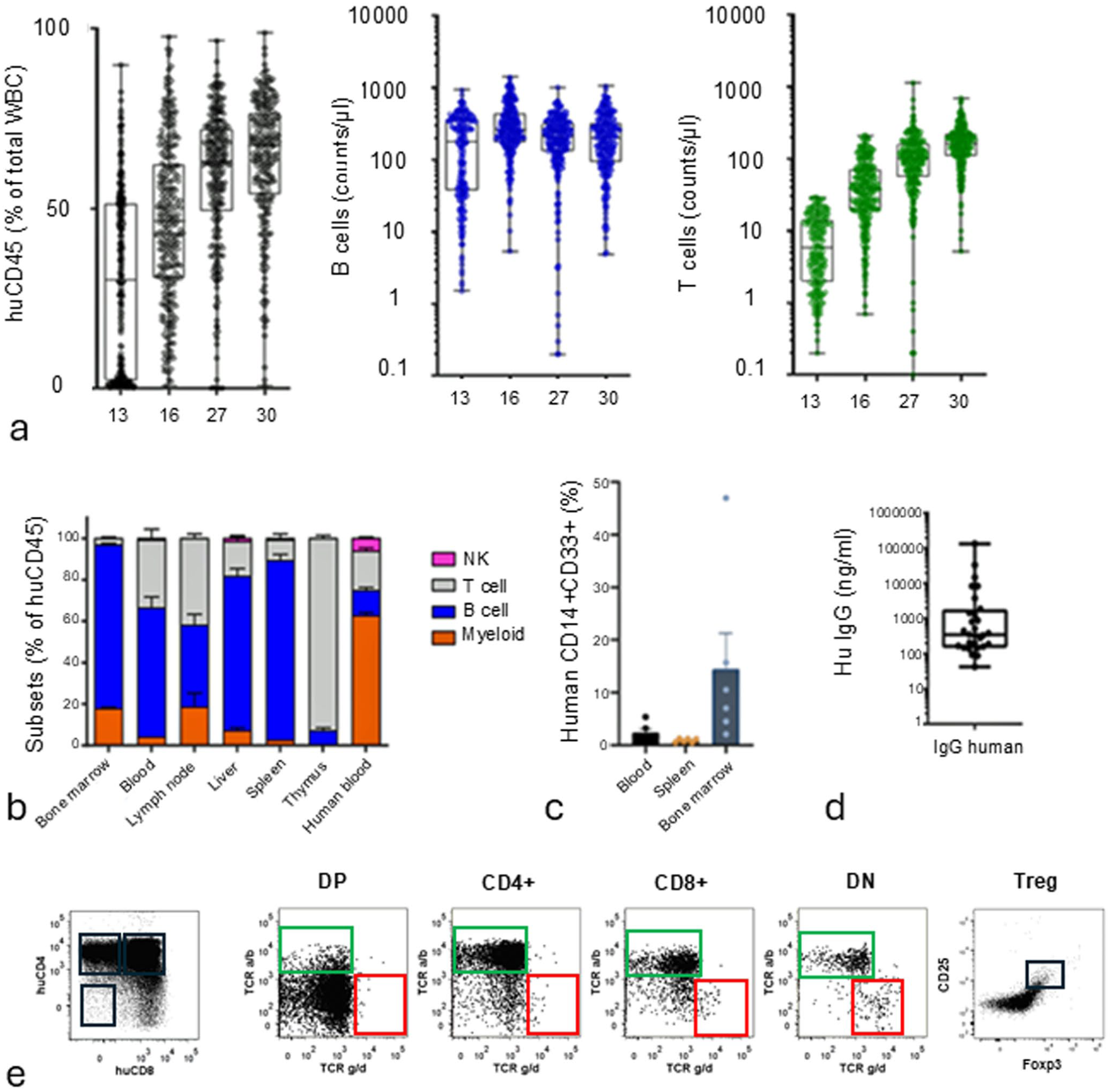
Flow cytometry baseline characterization of human immune cell development in humanized NSG (huNSG) mice. (a) Flow cytometry analysis of blood from huNSG mice at 13, 16, 27, and 30 weeks post-engraftment (wpe). Total of 209 animals/timepoint. Blood samples were labeled for total human leukocytes as a percentage of total white blood cells (WBCs), human B cells, and human T cells. The data presented shows the proportions of human leukocytes (huCD45), B cells, and T cells over time. Human cell engraftment, specifically T cell counts, increased over time, while B cell counts remained stable. (b) The percentages of natural killer (NK) cells, T cells, B cells, and myeloid cells within the human CD45+ population measured in the bone marrow, blood, lymph nodes, liver, thymus (6 mice), and spleen (7 mice) of huNSG mice at 15 wpe, and compared with human blood from 3 donors. Except for the thymus, which was dominated by T cells, all other organs showed a cell composition of predominantly B cells, followed by T cells, with low numbers of myeloid cells and very few NK cells. This differs from humans, where myeloid cells in blood are much more frequent. (c) The frequency of human CD14+CD33+ myeloid cells (monocytes/macrophages) at 16 wpe measured in blood, spleen, and bone marrow. The highest frequency of human CD14+ CD33+ myeloid cells is observed in the bone marrow, compared to blood and spleen. (d) Sera from 29 huNSG mice analyzed by an ELISA assay for the presence of human immunoglobulin G (IgG) at 16 wpe. Levels of human IgG were far below the typical human range of 7.5–22 mg/mL. (e) Single-cell suspensions from the thymus of huNSG mice at 15 wpe labeled with human CD3, CD4, CD8, CD45, T cell receptors (TCRs) α/β and δ/γ, and FOXP3 antibodies. Representative dot plots for thymocytes gated on CD3+ cells show (left to right) the presence of double-positive (DP; CD4+ and CD8+), CD4+ or CD8+ single positive (SP), as well as double-negative (DN; CD4− and CD8−) cells. Most of the DP cells show low expression of T cell receptors (TCR α/β and TCR δ/γ), suggesting they did not undergo TCR gene rearrangement and thus indicating a very early stage of T cell development. Conversely, SP cells, either CD4+ or CD8+, express a higher amount of TCRs (mainly TCR α/β), indicating that they had already undergone positive selection. TCR δ/γ T cells are found at low frequencies, the majority in CD8 and CD4 DN T cell progenitors. A representative dot plot for CD25 and FOXP3 shows the presence of regulatory T cells (gated on CD3+ cells) in these mice. The diverse cellular phenotypes in the thymus indicate a dynamic maturation process toward a functional immune system.
The percentage of different human cellular subsets relative to human CD45+ leukocytes, including B and T cells, NK cells, and myeloid cells, was evaluated in the blood, bone marrow, mesenteric lymph node, liver, spleen, and thymus at 15 wpe; blood from human donors was also included for comparison (Fig. 1b). Results showed low frequencies of human myeloid cells and very low to almost undetectable NK cells in the peripheral blood of huNSG mice, while it was confirmed that a prominent number of B cells and a low number of T cells were present at this early time of engraftment (Fig. 1b). This contrasted with the higher frequency of myeloid cells observed in human blood, which predominated over B and T cells (Fig. 1b). In human blood, NK cells also showed the lowest frequency, but the percentage was higher compared to huNSG mice (Fig. 1b).
In huNSG mouse tissues, with the exclusion of thymus, a similar predominance of B cells over T cells, as seen in blood, was present. The bone marrow and lymph nodes exhibited a higher percentage of myeloid cells compared to the blood, liver, and spleen (Fig. 1b). These data confirmed the frequency of CD14+ and CD33+ cells, which identify monocytes/macrophages, present in the blood, spleen, and bone marrow in mice at 16 wpe (Fig. 1c). Human IgG levels were also evaluated in blood samples from mice at 16 wpe (Fig. 1d). Excluding some outliers, the measured concentrations were found to be very low (50–1000 ng/mL) in comparison to expected human IgG levels, which typically range between 7.5 and 22 mg/mL. 19
The thymus showed higher frequencies of T cells compared to B cells, and almost undetectable myeloid and NK cells at 15 wpe (Fig. 1b). These observations reflect the physiological function of this organ, which plays an important role in the development of functional CD4+ and CD8+ T cells. Since T cells develop in the thymus, mice at 15 wpe were further evaluated to gain granularity on the different subsets of T cells present and to understand the evolution of the humanized immune system (Fig. 1e). Most T cells were both CD4+ and CD8+. The majority of these cells had not yet undergone TCR gene rearrangement and thus showed low expression of TCRs, indicating a very early stage of T cell development. CD4 and CD8 single-positive cells were also found in the thymus of huNSG mice. These cells had already undergone positive selection and expressed TCRs (mainly TCR α/β). TCR δ/γ T cells were found at low frequencies, and the majority were double negative for CD4 and CD8. Flow cytometry analysis also revealed a distinct population of human CD25+FOXP3+ T cells in the thymus of huNSG mice. These results confirmed that following engraftment, CD34+ HSCs are able to repopulate the murine thymus and generate human T cells. The different cellular phenotypes are also indicative of a dynamic maturation process toward a functional immune system.
Histological Evaluation of Cell Engraftment in Hematolymphoid Organs
Macroscopically, all thymuses were markedly smaller than those of standard laboratory mouse strains, making the organs difficult to identify during necropsy. The number of thymuses identified was 9/10 at 15 wpe, 8/10 at 18 wpe, 10/10 at 26 wpe, and 8/10 at 35 wpe. Histologically, thymuses were characterized by a fine connective meshwork with some inter-animal variability in terms of lymphoid cellularity at 15 wpe and uniformly abundant lymphoid cellularity starting from 18 wpe. In most cases, cortical and medullary landmarks were indistinguishable at 15 wpe, except in some samples there was a rough, uneven distinction between pale and denser areas resembling primitive corticomedullary differentiation (Fig. 2a). In all evaluated thymuses, most lymphoid cells were CD3+ T cells, with a low number (up to 5%) of CD20+ B cells scattered in the parenchyma (Fig. 2b).
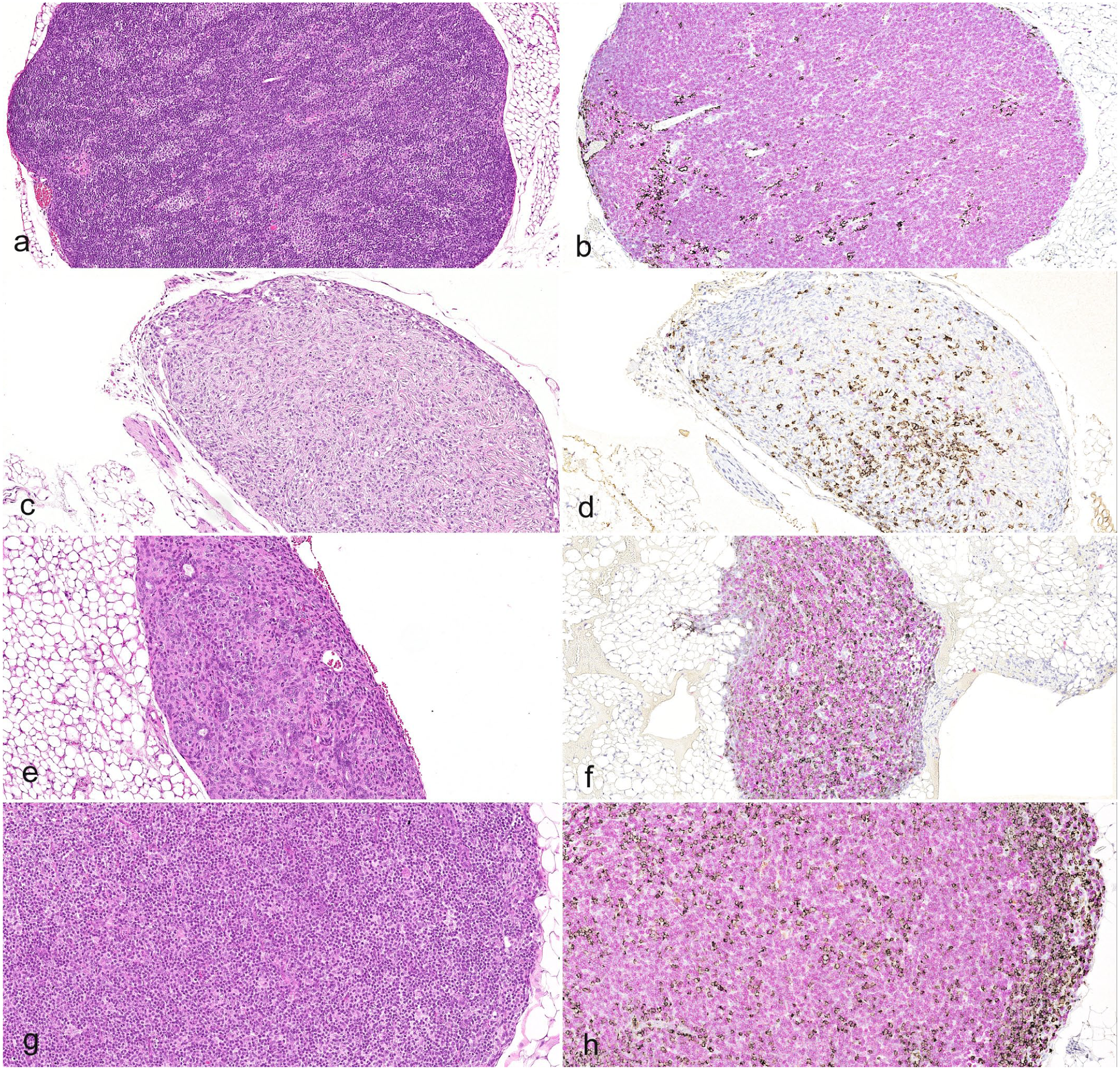
Thymus and mesenteric lymph node evaluation of humanized HSG mice. (a) Thymus of a mouse at 15 weeks post-engraftment (wpe) already showing abundant cellularity, with rough, uneven distinction between pale and denser areas resembling primitive corticomedullary differentiation. Hematoxylin and eosin (HE). (b) A serial section taken from the same block as the slide shown in (a) shows a predominance of CD3+ T cells (pink), with a low number of CD20+ B cells (brown). Multiplex immunohistochemistry (IHC) for CD3 and CD20. (c) Mesenteric lymph node of a mouse at 15 wpe, characterized by fibro-connective tissue infiltrated by a low number of lymphoid cells. HE. (d) A serial section taken from the same block as the slide shown in (c), showing scarce engraftment, with a prominent population of CD20+ B cells (brown) and a low number of CD3+ T cells (pink). Multiplex IHC for CD3 and CD20. (e) Mesenteric lymph node of a mouse at 15 wpe showing moderate numbers of lymphoid cells. Images (c) and (e) indicate there is a high inter-animal variability at 15 wpe. HE. (f) A serial section taken from the same block as the slide shown in (e), showing abundant lymphoid cellularity with a similar ratio between B (brown) and T (pink) cells. Multiplex IHC for CD3 and CD20. (g) Mesenteric lymph node of a mouse at 35 wpe showing a uniform, abundant lymphoid cellularity. HE. (h) A serial section taken from the same block as the slide shown in (g), showing how lymphocytes are evenly distributed, with a majority of CD3+ T cells (pink) and lower proportions of CD20+ B cells (brown). CD20+ cells are occasionally more abundant in the subcapsular space. Multiplex IHC for CD3 and CD20.
Similar to the thymus, the macroscopic evaluation of lymph nodes was also impaired by the small size of these organs. Mesenteric lymph nodes were identified in 5/10 animals at 15 wpe, 8/10 at 18 wpe, and 9/10 at both 26 and 35 wpe. Histologically, lymph nodes at all time points showed no recognizable compartmentalization into the cortex, paracortex, and sinuses, and no evidence of follicles or germinal centers. At 15 wpe, lymph nodes were characterized by fibro-connective tissue infiltrated by variable numbers (ranging from rare to moderate) of lymphoid cells (Fig. 2c, e), with a prominent population of CD20+ B cells and a variable number of T cells (5% to 50%; Fig. 2d, f). At 18 wpe, the lymph node stroma was engrafted with a highly variable amount (from minimal to abundant) of lymphoid cells, while at 26 and 35 wpe, lymph nodes showed a more uniform abundant cellularity of CD3+ T cells with lower proportions of CD20+ B cells and rare CD68+ macrophages. At 26 and 35 wpe, CD20+ B cells were occasionally more abundant in peripheral areas resembling subcapsular sinuses (Fig. 2g, h).
In the spleen, all animals displayed a clear distinction between the white and red pulp (Fig. 3a, b); however, the size of the white pulp varied across animals and groups, without correlation with post-engraftment time. In the white pulp, human HLA-A+ mononuclear leukocytes were arranged around vessels and resembled periarteriolar lymphoid sheaths (Fig. 3f), but no differentiated follicles or germinal centers were identified at any timepoint. At 15 wpe, 75% to 100% of the white pulp lymphoid cells were CD20+, with only a few CD3+ cells accounting for <5% of the lymphoid cells (Fig. 3c). At 18 wpe, the B cell population slightly decreased, representing >50% of the lymphoid cells, compared to the T cell populations, which increased from 5 to 25%. At 26 and 35 wpe, the white pulp was populated by a predominant population of CD3+ T cells in the central portions, with B cells more abundant at the periphery, suggestive of well-developed periarteriolar lymphoid sheaths (Fig. 3d).
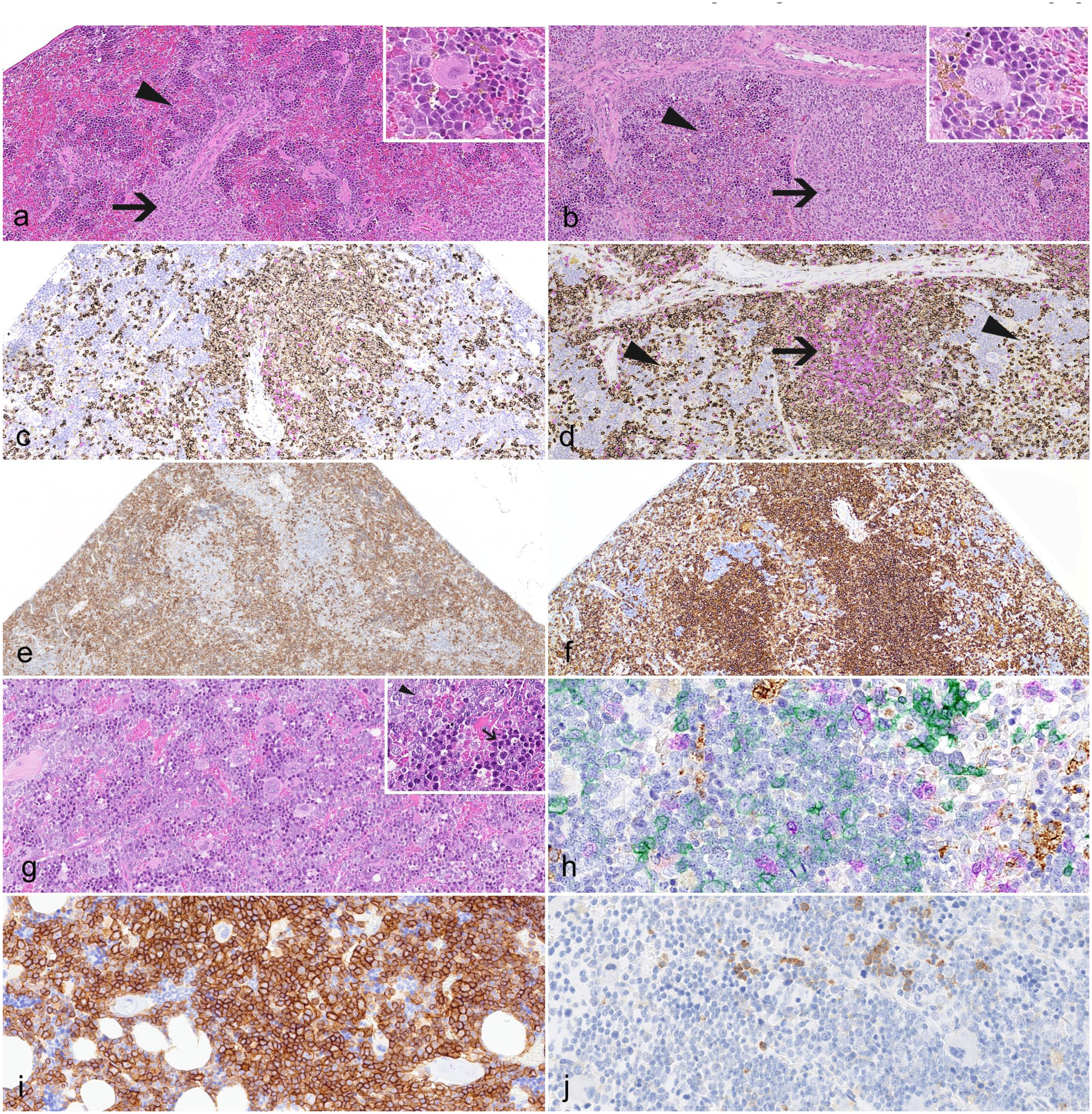
Spleen and bone marrow evaluation. (a) Spleen of a humanized NSG (huNSG) mouse at 15 weeks post-engraftment (wpe). There is a clear distinction between the white and red pulp, with well-developed red pulp (arrowhead) containing prominent extramedullary hematopoiesis (inset) and white pulp with mononuclear leukocytes surrounding vessels resembling periarteriolar lymphoid sheaths (PALS) (arrow). No differentiated follicles or germinal centers were identified at any time point. Hematoxylin and eosin (HE). (b) Spleen of a huNSG mouse at 35 wpe. Extramedullary hematopoiesis in the red pulp (arrowhead, inset) is reduced, indicating a progressive decrease of this compartment over time. The white pulp (arrow) remains unaltered. HE. (c) Spleen of a huNSG mouse at 15 wpe showing that most of the white pulp lymphoid cells are CD20+ (brown), with few CD3+ cells (pink). In the red pulp, occasional CD20+ B cells and very rare CD3+ cells are observed. Multiplex immunohistochemistry (IHC) for CD3 and CD20. (d) Spleen of a huNSG mouse at 35 wpe. The white pulp (arrow) is populated by a predominant population of CD3+ T cells (pink) in central areas, with B cells (brown) more abundant at the periphery, suggestive of well-developed PALS. In the red pulp (arrowheads), the percentage of CD20+ B cells remains stable, whereas CD3+ T cells are increased compared to 15 wpe. Multiplex IHC for CD3 and CD20. (e) Spleen of a huNSG mouse at 18 wpe. Murine CD45 immunolabeling shows that a large proportion of cells in the red pulp are of murine origin. Murine CD45 IHC. (f) Spleen of a huNSG mouse at 18 wpe. The white pulp consists of human leukocyte antigen (HLA)-A+ cells, which are also abundant in the red pulp. HLA-A IHC. (g) Bone marrow of a huNSG mouse at 35 wpe. The bone marrow shows abundant cellularity in all animals at all timepoints, and all cell lineages, namely the megakaryocytic, myeloid (arrowhead, inset), and erythroid (arrow, inset) lineages are represented. HE. (h) Bone marrow of a huNSG mouse at 18 wpe. Frequent CD20+ B cells (green), rare CD3+ T cells (purple), and rare medium to large-sized CD68+ macrophages (brown) are observed. Multiplex immunohistochemistry (IHC) for CD3, CD20, and CD68. (i) Bone marrow of a huNSG mouse at 18 wpe. The majority of cells in the bone marrow are of human origin; the low number of cells negative for HLA-A can frequently be identified based on morphology as granulocytes, megakaryocytes, or immature cells of myeloid or erythroid lineage. HLA-A IHC. (j) Bone marrow of a huNSG mouse at 18 wpe. Murine CD45 IHC confirms the murine origin of cells with myeloid morphology that are negative for the HLA-A marker. Murine CD45 IHC.
The red pulp in animals at 15 and 18 wpe was already well-developed, with prominent extramedullary hematopoiesis (Fig. 3a), a large part of the cells being murine CD45+ (Fig. 3e), together with occasional (25–50%) CD20+ B cells, and very rare CD3+ T cells accounting for less than 1% of the cells in the red pulp (Fig. 3c). In animals at later engraftment timepoints, the percentage of CD20+ B cells remained stable, and CD3+ T cells increased to 5%, while the extramedullary hematopoiesis in the red pulp progressively decreased over time (Fig. 3b, d).
The presence of developed Peyer’s patches was not observed in any of the intestinal samples examined.
The bone marrow of the sternum and femur showed abundant cellularity in all animals at all timepoints; all cell lineages (megakaryocyte, myeloid, and erythroid) were represented (Fig. 3g). HLA-A immunolabeling indicated that the majority (70 to 80%) of cells in the bone marrow were of human origin (Fig. 3i), and multiplex IHC showed rare to occasional CD20+ B cells and rare CD3+ T cells at all timepoints. CD68+ cells were rare and mainly small with scant cytoplasm at 15 wpe, while from 18 wpe, medium-to-large-sized CD68+ macrophages became progressively more prominent (Fig. 3h). To confirm whether the CD68+ macrophages were of murine origin, F4/80 immunolabeling was performed on serial sections. F4/80+ cells were frequent, but labeling was exclusively observed in small macrophages (ie, likely nonactivated cells). This pattern was comparable to that of a C57BL/6 mouse bone marrow, which was included as a positive control within the same run (data not shown). In all, 20% to 30% of cells negative for HLA-A in the bone marrow could be identified based on morphology as granulocytes, megakaryocytes, or immature cells of myeloid or erythroid lineages and were frequently positive for murine CD45 (Fig. 3i, j).
Spontaneous Lesions
Macrophage-dominated lesions developed in huNSG mice over time
A common histopathological pattern observed in this mouse model was the development of granulomatous lesions characterized by aggregates of large epithelioid-like macrophages with abundant pale eosinophilic cytoplasm and occasional multinucleated giant cells with features and nuclear arrangements of both foreign body and Langhans-type giant cells (Fig. 4a, e), surrounded by a small number of mononuclear cells and occasional fibroblasts. In some cases, large macrophages with abundant light to golden brown cytoplasm, consistent with hemosiderin, were present, particularly in the liver (Fig. 4j) and less frequently in the spleen and bone marrow. These granulomatous lesions were first detected at 18 wpe in 4/10 animals, and their incidence increased over time. By 26 and 35 wpe, 8/10 and 9/10 animals were affected, respectively. The spleen was the most frequently affected organ, followed by the liver and bone marrow. The mesenteric lymph node, lung, and salivary glands were moderately affected. In rare cases, and only at later timepoints, the skin, Harderian gland, adrenal, pituitary, and pancreas also contained granulomatous lesions. A summary of the incidence of granulomatous lesions identified in hematoxylin and eosin-stained sections is provided in Table 1.
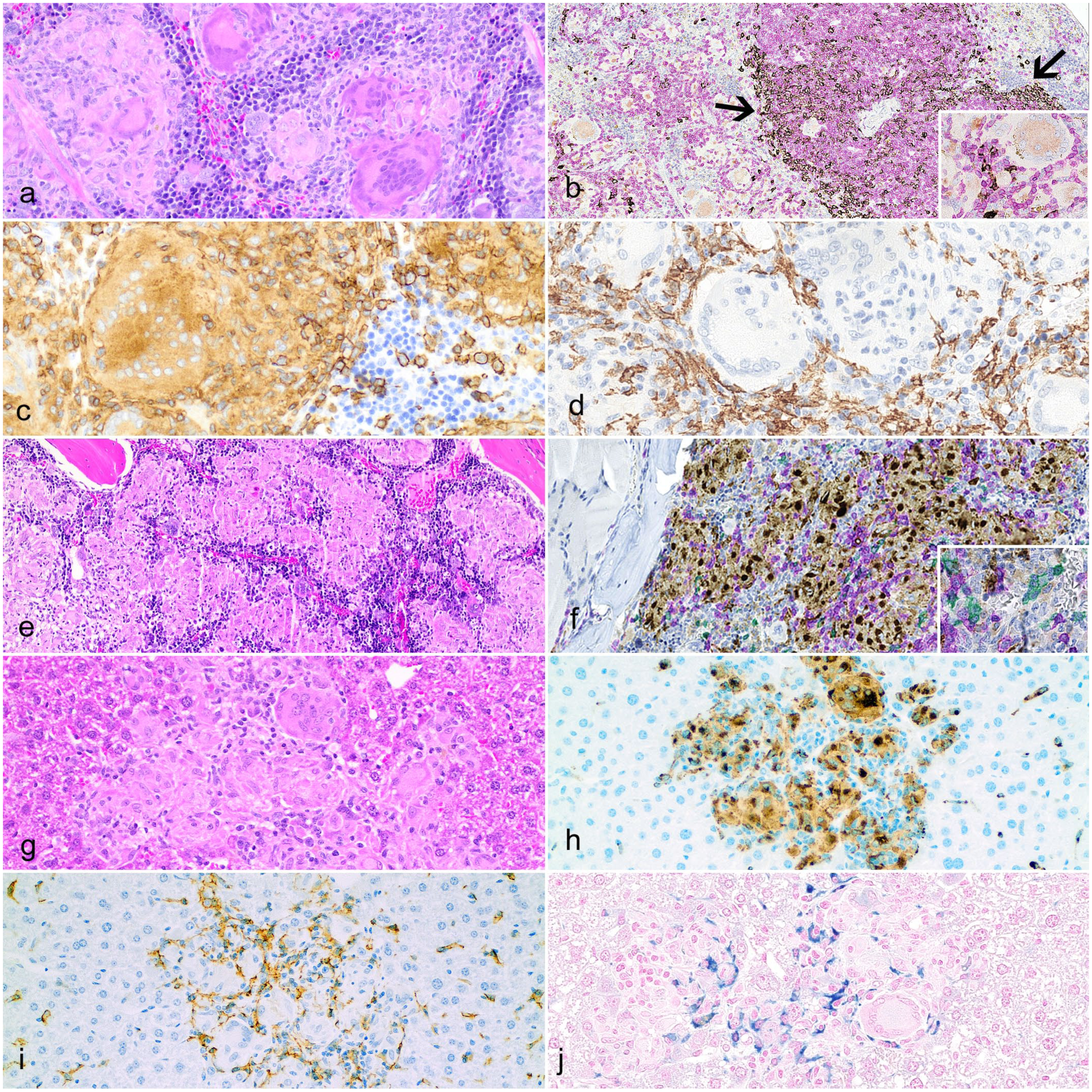
Spontaneous granulomatous lesions in the spleen, bone marrow, and liver of humanized NSG (huNSG) mice. (a) Spleen of a huNSG mouse at 26 weeks post-engraftment (wpe). Granulomatous lesions invading the red pulp are composed of multinucleated giant cells with the morphology of foreign body and Langhans-type giant cells. Hematoxylin and eosin (HE). (b) Spleen of a huNSG mouse at 26 wpe. Granulomas with CD68+ macrophages (brown) are surrounded by abundant T cells (purple) and rare B cells (black, inset); adjacent, the well-developed periarteriolar lymphoid sheath (PALS) is composed mostly of T cells intermixed with lower amounts of B cells, more frequent at the periphery of the PALS (arrows). Multiplex immunohistochemistry (IHC) for CD3, CD20, and CD68. (c) Spleen of a huNSG mouse at 35 wpe. High magnification of granulomatous lesions showing human leukocyte antigen (HLA)-A immunolabeling, confirming the human origin of these lesions. HLA-A IHC. (d) Spleen from the same mouse as in (c). Immunolabeling for murine CD45 showing the presence of murine cells surrounding the granulomatous lesions. Murine CD45 IHC. (e) Bone marrow in the femur of a huNSG mouse at 26 wpe with coalescing granulomatous lesions largely replacing the preexisting bone marrow population. HE. (f) Bone marrow from the same mouse as (e). Granulomas are CD68+ (brown) and are surrounded by CD3+ cells (purple), and rarely by CD20+ cells (green, inset). Multiplex IHC for CD3, CD20, and CD68. (g) Liver of a 26 wpe huNSG mouse with aggregates of large epithelioid-like macrophages with abundant, pale eosinophilic cytoplasm and multinucleated giant cells (with features of both foreign body giant cells and Langhans-type giant cells), surrounded by a small number of mononuclear cells and occasional fibroblasts. HE. (h) Liver from the same mouse as (g). Large CD68+ macrophages indicate their human origin. CD68 IHC. (i) Liver from the same mouse as (g). Reactive mouse F4/80+ macrophages surrounding granulomatous lesions. F4/80 IHC. (j) Liver from the same mouse as (g), showing abundant iron deposits in macrophages in the granulomatous lesions. Large multinucleated cells were generally negative. Prussian blue.
Incidence of granulomatous lesions detected via hematoxylin and eosin staining in humanized NSG mice.
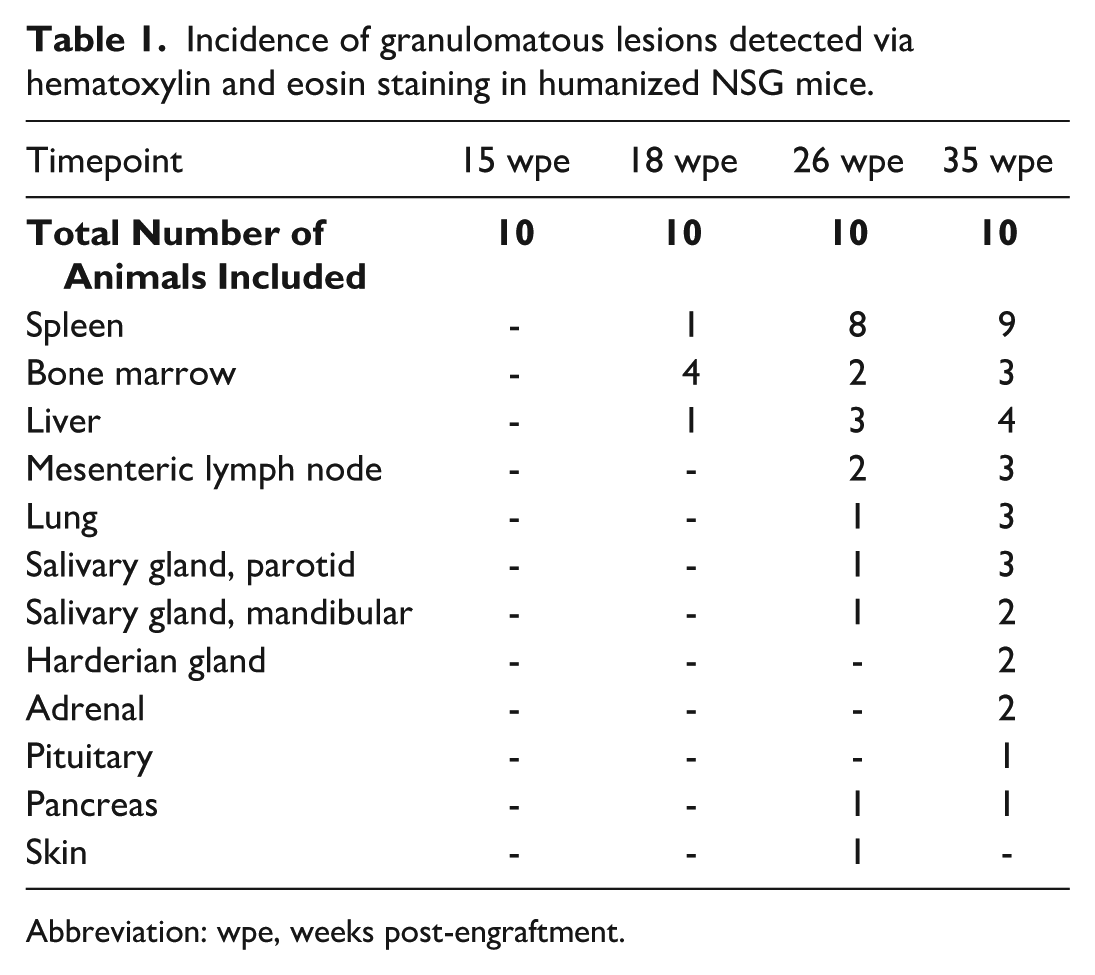
Abbreviation: wpe, weeks post-engraftment.
The severity of the granulomatous lesions increased over time. At 18 wpe, granulomatous lesions were dominated by small clusters of small to medium-sized macrophages; at later timepoints (ie, 26 and 35 wpe), both the size of the granulomatous clusters and the macrophages themselves increased, with the macrophages exhibiting more abundant cytoplasm, and more frequent multinucleated giant cells could be identified. In the spleen, granulomatous lesions were mainly occurring in the red pulp, but in more severe cases, granulomatous lesions almost completely replaced the red and white pulp, effacing the normal anatomy (Fig. 4a). At later timepoints, the presence of granulomatous lesions in the bone marrow was sometimes associated with decreased cellularity of preexisting hematopoietic compartments or, in the most severe cases, the granulomatous lesions largely replaced the preexisting bone marrow population (Fig. 4e). In addition, abundant spindle-shaped cells with elongated nuclei expressing FAP were noted in the bone marrow surrounding the granulomatous lesions, suggesting a heightened level of fibroblast activity, which is associated with the active remodeling of the bone marrow stroma, a consequence of the granulomatous inflammation and associated tissue damage.
In all affected organs, granulomatous lesions were HLA-A+ and CD68+, indicating their human origin and macrophage lineage. These lesions were surrounded by frequent CD3+ T cells, with a subset of FOXP3+ cells, and fewer CD20+ B cells (human-specific) (Fig. 4b, c, f), as well by varying numbers of reactive mouse macrophages that were positive for murine CD45 (Fig. 4d) and F4/80+ cells, which were particularly abundant in the most affected livers (Fig. 4h). It is important to note that the identification of the more subtle granulomatous lesions, which frequently occurred at earlier timepoints (ie, 15 and 18 wpe), could be easily overlooked with hematoxylin and eosin staining alone. IHC labeling for CD68 was essential for the accurate identification of early lesions of human-activated macrophages in those organs where a certain amount of macrophages/histocytes is expected, and to discriminate multinucleated giant cells from megakaryocytes in the spleen and bone marrow.
Spontaneous bone marrow necrosis
Two animals at 35 weeks developed diffuse necrosis of the bone marrow cavity of the femur, characterized by diffuse loss of hematopoietic cells and replacement with eosinophilic amorphous necrotic debris. The morphology of this necrosis resembled that observed in ischemic lesions. In the baseline characterization study, the necrotic lesions were not accompanied by any signs of underlying vascular lesions or thromboembolic events. The bone marrow in the sternum samples of the same animals showed abundant granulomatous lesions, but there was no necrosis.
Lesions After Treatment With Immunomodulatory Compounds: Case Studies
As huMice are widely used in oncology research due to their ability to support human HSC engraftment, enabling the study of human cancer development, progression, and response to therapies within a live animal model, we investigated spontaneous lesions following treatment with immunomodulatory compounds. The administration of immunostimulatory molecules to huNSG mice led to a rapid worsening of existing lesions or accelerated the formation of new lesions.
Immune stimulation with cytokine-based biologic treatments accelerates the incidence and severity of spontaneous lesions
Study 1 was a pharmacological study investigating an immunocytokine therapeutic drug using huNSG mice at 15 wpe that were engrafted subcutaneously with cell line-derived xenograft tumor cells. The intended immunocytokine MoA was to boost the immune system within the tumor microenvironment to kill neoplastic cells. The main goals of the study were to compare efficacy and evaluate the pharmacodynamic responses of various compound candidates. The animals evaluated were divided into 4 groups: a vehicle-only control group (group A, n = 4) and 3 groups testing different test-item formats (group B = targeted cytokine format 1, n = 4; group C = targeted cytokine format 2, n = 4; group D = digoxigenin-labeled cytokine, n = 3). In the 2 targeted-cytokine-treated groups (B and C), the molecules targeted an immune checkpoint inhibitor. The digoxigenin-labeled cytokine (group D) did not have the checkpoint inhibitor binding. Animals received 2 doses of the control/immunocytokine separated by 7 days. Histological evaluation, conducted 7 days after the second dose, was performed on a selected list of tissues suspected to develop potential toxicological liabilities, including the spleen, bone marrow, lung, liver, and kidney. The evaluation was performed on 4 animals per group, apart from group D, which contained 3 animals (15 animals in total were evaluated). Details on the lesion incidence in each group of study 1 are provided in Supplemental Table S5. All treated animal groups showed minimal-to-severe granulomatous inflammation in the bone marrow, liver, spleen, or kidney, which was not observed in the vehicle-only control group. The bone marrow showed severe necrosis of the hematopoietic compartment in the 2 groups receiving the checkpoint inhibitor-targeting molecules. The occurrence of these lesions only in animals receiving the targeted or untargeted cytokine was interpreted as an exacerbation of the expected mouse model-related changes induced by the treatment, and not as direct test-item toxicities. Additionally, the lungs contained moderate to severe perivascular infiltration of mononuclear cells with lymphoid morphologies. The incidence and severity were increased in the treated animals compared to controls. These changes, together with the minimal to moderately increased cellularity of the white pulp observed only in the spleen of treated animals, were considered to be part of the direct pharmacological effect caused by the test item and not as mouse model-related. Immune activation in the spleen resulted in effacement of normal white pulp architecture, affecting the periarteriolar lymphoid sheaths (PALS), follicles with germinal centers, and the marginal zone, and was characterized by an expansion of mononuclear cells that were larger and with paler basophilic cytoplasm compared to normal lymphocytes.
Study 2 was a pharmacology study in which huNSG mice at 16 wpe were injected subcutaneously with a human tumor cell line and used to study inhibition of tumor cell growth. Two groups of 12 animals each were subjected to 4 intravenous weekly treatments with an antibody conjugated to a modified cytokine, either targeting a tumor antigen (group B) or untargeted (group C), in which the tumor antigen binding domain was replaced with a nonbinding germline sequence (DP47). A group of 12 additional animals injected with vehicle only (group A) was used as a negative control. In total, 36 animals were included in study 2. Microscopic examination of the spleen, liver, and lung was conducted 5 days after the last treatment and revealed that mice treated with antibodies conjugated with the modified cytokine, whether targeted or untargeted, exhibited a marked increase in both the incidence and severity of activated macrophages (characterized by an increase in size and abundant eosinophilic or vacuolated cytoplasm), and granulomatous lesions in the liver and spleen, indicating an exacerbation of background lesions triggered by a systemic inflammatory response. The lung showed no relevant microscopic lesions, and the bone marrow was not evaluated microscopically in this study. Details of the lesion incidence in each group of study 2 are provided in Supplemental Table S6. These findings strongly implied that the presence of the cytokine moiety, rather than the targeting capability of the antibody, was the primary cause of the accelerated development of granulomatous lesions.
Bone marrow lesions are specific to huNSG mice
Study 3 involved huNSG mice at 26 wpe treated with a monoclonal antibody targeting FAP and 4-1BB ligand (4-1BBL) fusion protein. The FAP portion of the tested molecule cross-reacted with murine FAP, and 4-1BBL targeted human 4-1BB expressed on the human immune cells present in the huNSG mice. In this study, bone marrow necrosis was hypothesized based on changes observed macroscopically in the femur (paleness of the bone at necropsy) and on a marked decrease in the total cell count in the bone marrow from the femur in flow cytometry, as previously reported in Claus et al. 14 Here, we report additional comprehensive microscopic investigations conducted in these animals at 2 timepoints. Ten animals per group received the test molecule (FAP 4-1BBL) intravenously (group B); an additional 10 animals per group (group C) received another molecule where FAP was replaced by a nonbinding germline sequence (DP47 4-1BBL), which was used as an untargeted negative control, and 10 animals per group received vehicle only (group A). Five animals from each group were sacrificed 72 hours after 1 dose (day 3) and 5 animals after 2 doses (day 10). In total, 30 animals were included in study 3. No changes were noted in animals from group B that were sacrificed 3 days after the first dose of FAP 4-1BBL, but diffuse severe bone marrow necrosis involving all cells within the bone marrow cavity of the femur was observed in 3 out of the 4 mice surviving to the scheduled sacrifice 3 days after the second dose (1 animal was not available for microscopic examination). In the fourth animal, necrosis of the bone marrow was present only focally in the diaphysis and was accompanied by a moderate amount of activated fibroblasts, granulomas, and decreased cellularity of the erythroid lineage. Mice receiving the nontargeted molecule (DP47 4-1BBL; group C) and those treated with vehicle only (group A) did not show necrotic lesions in the bone marrow. Several animals had granulomatous lesions in the spleen, liver, or bone marrow, and these were considered spontaneous since they occurred with similar incidence and severity in all animals within the study, including controls, and were expected due to the late time post-engraftment. Details of the lesion incidence in each group of study 3 are provided in Supplemental Table S7.
Study 4 was conducted to further clarify the changes previously observed across multiple research investigations conducted in our facility, where huNSG mice administered a single dose of a T-cell-bispecific (TCB) antibody targeting CD20 and CD33 displayed bone marrow necrosis in the femur and/or sternum. Necrosis typically affected all bone marrow cell populations in the damaged areas, exhibiting features consistent with ischemic necrosis. The bones evaluated, including the knee joint, with portions of the femur and tibia, and the sternum, exhibited a variable distribution of bone marrow necrosis within individual animals. A cohort of six 26-week-old huNSG mice engrafted with a modified protocol employing a reduced number of CD34+ HSCs (5×104 cells) was treated with a single dose of CD20 CD3 TCB, and 10 animals were treated with vehicle only. A total of 16 animals were included in this study. Bone marrow necrosis was observed in the femur of 4 animals 24 hours following CD20 CD3 TCB administration and in the sternum of 2 of these animals (Fig. 5a–c). Notably, no bone marrow lesions were detected in 2 treated animals and in the 10 animals of the control group. Thrombosis was noted only in 2 animals exhibiting bone marrow necrosis, in rare small-to-medium-sized vessels within the bone marrow cavity or close to the periosteal surface of the sternum and femur (Fig. 5c–f). No thrombi were observed in the other tissues evaluated microscopically (lung, liver, heart, kidney, and spleen). Notably, no granulomatous lesions were observed in this study in any organ of the control animals or treated animals, in contrast to the frequent occurrence in huNSG mice at 26 weeks after engraftment with 1×105 human HSCs. Details of the lesion incidence in each group in study 4 are provided in Supplemental Table S8.
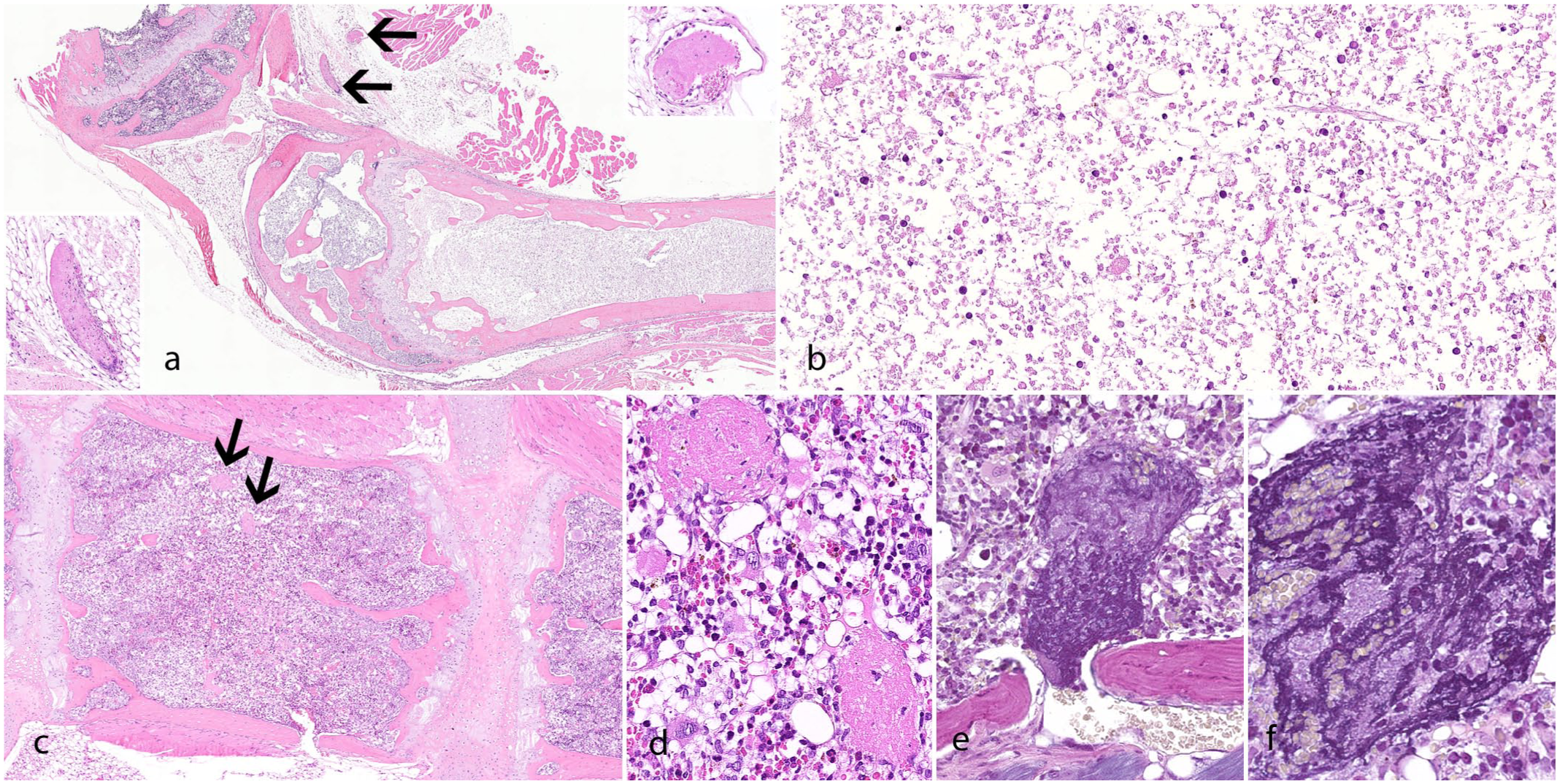
Bone marrow necrosis in the femur and sternum of humanized NSG (huNSG) mice. (a) Bone marrow in the femur of a huNSG mouse at 26 weeks post-engraftment, sacrificed 24 hours after receiving a single dose of a CD20 CD3 T-cell-bispecific antibody. Most of the marrow in the femoral diaphysis and epiphysis is necrotic, with pale eosinophilic ghost cells with blurred outlines. Note that the bony trabeculae in the femur, as well as the bone marrow in the proximal tibia, are spared. Thrombi are present in the adjacent tissue (arrows, insets). Hematoxylin and eosin (HE). (b) Higher magnification of bone marrow necrosis shown in (a). HE. (c) Sternum from an age- and treatment-matched animal showing initial necrosis of the bone marrow (cell rarefaction, shrinkage of cells, and reduced nuclear staining) surrounding thrombotic vessels (arrows). HE. (d) Higher magnification of thrombi in (c). HE. (e, f) Higher magnification of thrombi in vessels from the same animal depicted in (c). Phosphotungstic acid-hematoxylin stain.
Discussion
Humanized mouse models have become indispensable preclinical tools for drug discovery, particularly in the field of cancer immuno-oncology, 30 and there are currently approximately 50 different humanized mouse models available. 49 HuNSG mice are a common in vivo model used in pharmacology studies of cancer immunotherapeutic drugs due to their capacity to grow human tumors in an in vivo model that has a humanized immune system. 41 While offering significant advantages, the intrinsic chimeric nature of huMice (human immune cells within a murine host) presents inherent challenges. For example, there is a high inter-animal variability influenced by factors such as the human donors, engraftment protocols, and the specific host strain used, all of which can have an impact on the level of human immune cell reconstitution.5,49 For pathologists, a critical consideration is the spontaneous development of xenoreactive lesions arising from the interaction between engrafted human immune cells and murine host tissues.6,51 This is particularly problematic in drug safety studies, where accurate differentiation between compound-specific effects and background pathology is paramount. Consequently, a thorough understanding of the specific characteristics of each humanized mouse model is essential for accurate data interpretation. As engraftment kinetics are nonlinear, a comprehensive assessment at different timepoints after human cell engraftment is needed.2,47 Furthermore, beyond the development of spontaneous lesions, huMice might also show differences in the way that they respond to various immunostimulatory treatments. 58 This manuscript provides a comprehensive overview of the microscopic and flow cytometry findings observed in huNSG mice following engraftment with human umbilical cord CD34+ HSCs across different timepoints post-engraftment, and the progressive development of pathological features. Moreover, it describes key findings obtained in preclinical studies evaluating diverse immuno-oncology therapeutic drugs, highlighting both the model’s strengths and the critical considerations for accurate pathology interpretation.
Flow cytometry immunophenotyping of the blood showed continuous changes of the newly evolving immune system in huNSG mice, with a gradual increase in human CD45+ leukocytes. Among human leukocytes, T cell counts were lower at earlier timepoints and increased over time, whereas B cell counts were already high at earlier timepoints and remained stable throughout the evaluation period. These findings underscore the need to have a thorough understanding of the engraftment kinetics in the model prior to its use in drug development studies, as these kinetics can significantly impact study outcomes, depending on the therapeutic MoA. For example, when evaluating T cell immunomodulators, a low number of human T cells at earlier timepoints post-engraftment could directly influence the observed results. A significant finding when comparing human and huNSG blood was the notably low representation of innate immune cells, such as myeloid cells and NK cells, compared to lymphocyte populations. In human blood, myeloid cell populations consisted of approximately 75% peripheral blood, whereas these populations were <5% in huNSG mice. These findings are in line with published research 2 and suggest that lymphocyte predominance over myeloid cells is not a unique characteristic of huMice but rather reflects the nature of the murine immune system, which shows lymphocyte predominance over myeloid cells. 40 This observation contrasts with the human immune system, where myeloid cells are more abundant than lymphocytes. 2
Flow cytometry immunophenotyping was also performed at 15 wpe in the bone marrow, spleen, liver, thymus, and mesenteric lymph nodes. Similar to results reported in blood, at 15 wpe, a low frequency of human myeloid cells and monocytes was observed in the spleen, lymph nodes, bone marrow, and thymus. Regarding B and T cell frequencies, the same pattern was observed in the spleen, lymph nodes, and bone marrow, which was characterized by the predominance of B cells over T cells. This contrasted with the thymus, which exhibited a clear predominance of T cells, even at earlier timepoints. Given the observed increase in T cell counts in the blood over time, the low T cell frequencies at 15 wpe likely represent an early phase of the humanization process, with a potential shift toward higher T cell frequencies at later timepoints, correlating with increased circulating T cells. This trend was supported by IHC analysis of the spleen and lymph nodes, which revealed an increasing number of CD3+ cells over time. Macroscopic evaluation across the different timepoints showed a notable reduction in thymus and lymph node size compared with wild-type mice, often making them challenging to identify during necropsy. This is a recognized characteristic of huMice that necessitates careful consideration during necropsies, especially for less experienced personnel. In the current study, most thymuses and lymph nodes were successfully identified at necropsy and sampled for histology. High inter-animal variability in the cellularity was observed in both the thymus and lymph nodes, ranging from minimal to abundant. However, the inherent limitations of histology must be acknowledged, considering that a single section may not fully represent the engraftment status of the animal and, therefore, special care should be taken for result interpretation. When possible, the authors suggest a comparison between histological and flow cytometry data to confirm findings. In any case, a trend toward a positive correlation between cellularity and time post-engraftment in both the thymus and lymph nodes was noted in the current study. The normal microscopic architecture of the thymus and lymph nodes was largely absent; the typical corticomedullary differentiation of the thymus was lost, and the lymph nodes lacked discernible follicles and germinal centers, which were replaced by a uniform sheet of mononuclear cells. The thymus occasionally displayed small, paler areas interspersed with denser, more basophilic regions, suggestive of a rudimentary medulla/cortex organization. More detailed analysis of the thymus with flow cytometry revealed a mixed T cell population with evidence of various maturation stages, supporting the ability of CD34+ HSCs to migrate to the thymus, develop into functional T cells, and undergo negative selection against murine self-antigens. 22
The spleen retained discernible histological landmarks of the white and red pulp, with mononuclear cells resembling PALS but lacking distinct follicular structures. The red pulp was well-developed and exhibited extramedullary hematopoiesis. The bone marrow showed abundant cellularity with representation of all hematopoietic lineages across all animals, and a predominance of human cells. Overall, these findings align with previously published reports in huNSG mice. 6 These results collectively indicate that huNSG mice develop a humanized immune system, characterized by dynamic changes over time. This underscores the importance of understanding the engraftment kinetics and characteristics of this model prior to being used in drug testing studies. Furthermore, while the humanized immune system in this model reflects several aspects of the human system, significant morphological differences exist, and pathologists evaluating these animal models should be familiar with their key histological features.
Histological evaluations in our huNSG mice revealed the presence of spontaneous inflammatory lesions dominated by macrophages, with increasing incidence over time. The main affected organs were the spleen, liver, and bone marrow, although in more severe cases, other organs were sometimes involved. Similar lesions have also been described in huMice under various other names, such as graft vs host disease (GvHD), macrophage activation syndrome (MAS), or more generically as autoimmune disease.24,26,31,34,48,51 Confirming previous findings, we observed that the spontaneous inflammatory lesions were composed of and dominated by human macrophages surrounded by a mix of human and mouse B and T cell populations, together with mouse activated macrophages and fibroblasts.
In humans, GvHD refers to a clinical condition occurring after allogeneic hematopoietic cell transplantation, where donor immune cells attack recipient antigens. 22 The pathogenesis of GvHD is complex and, in many aspects, not fully understood. The disease can be subclassified as acute GvHD (aGvHD) or chronic GvHD, based on the timing of disease onset (aGvHD typically appears before 100 days post-engraftment) and the primary organs affected (aGvHD primarily affects the skin, liver, and gastrointestinal tract, whereas chronic GvHD can affect any organ). 35 The pathogenesis of aGvHD is typically described to occur in 3 phases: (1) an initiation phase involving antigen-presenting cells activated by tissue damage from conditioning treatments; (2) the activation of donor T cells by host antigen-presenting cells; and (3) an effector phase where donor T cells attack host cells. 35 In huNSG mice described here, xenoreaction lesions differed from typical aGvHD in humans by the fact that inflammatory lesions were dominated by macrophages, not lymphocytes. Why lymphocytes are not the main cell driver of these lesions could be explained by comparing our model with huMice engrafted with PBMCs. In contrast to HSC huMice, mice engrafted with PBMCs have been shown to better mimic the lymphocyte-dominated aGvHD described in humans.9,17 The main difference between HSC and PBMC engraftment is that more mature PBMCs from the donor trigger a xenoreactive response against murine host tissues due to major histocompatibility complex mismatch, with human T cells recognizing murine major histocompatibility complexes as foreign.9,17 This mechanism, however, is not expected to be the primary driver of pathology in the current huNSG mice engrafted with CD34+ HSCs due to differences in the humanization process. Following transplantation, HSCs migrate to the murine thymus, where they differentiate into mature human T cells. In the thymus, these newly formed T cells undergo selection to recognize murine antigens presented by major histocompatibility complex cells as “self.”22,37 By flow cytometry of the thymus of animals at 16 wpe, we showed a variety of T cell phenotypes at different stages of maturation that support this process of humanization within the murine recipient thymus.
Due to the macrophage-dominated nature of the spontaneous inflammatory lesions, some studies refer to this as chronic GvHD34,48 or MAS.26,51 Similar to aGvHD, chronic GvHD is also presented in 3 phases of initiation, activation, and effector phases; however, the activation phase involves damage in the thymus, leading to dysregulation of the immune system, resulting in chronic inflammation where macrophages play an important role, and there is aberrant tissue repair leading to scleroderma or bronchiolitis obliterans. 21 This pathological presentation is not reflected in the current huNSG model. On the other hand, MAS is an acute and severe inflammatory condition dominated by macrophages, often triggered by preexisting inflammatory conditions such as infections, autoimmune diseases, or neoplasia, leading to uncontrolled activation of antigen-presenting cells (including macrophages) and lymphocytes due to dysfunctional inhibitory pathways that normally resolve such activation. 39 A typical histopathological pattern of MAS is the presence of hemophagocytosis; this is why MAS is often classified within hemophagocytic lymphohistiocytosis diseases. 39 While hemophagocytosis was a feature observed in huNSG due to the presence of hemosiderin within human macrophages, a key difference in huMice is that the triggering factor seems to be an xenorejection process where human macrophages attack murine tissues, suggesting at least a partially different pathogenesis. 7 A potential hypothesis involves a mismatch in intrinsic self-recognition mechanisms involving macrophages, such as the CD47-SIRPα axis. CD47, expressed on many normal cells, is a ligand for SIRPα, which is highly expressed on macrophages. This interaction typically acts as a “don’t eat me” signal. A human-mouse mismatch in this axis could potentially explain the underlying cause of the observed macrophage-dominated lesions.28,33 In conclusion, while these macrophage-dominated GvHD-like lesions share features of both GvHD and MAS, their precise pathogenesis remains unclear. The underlying mechanisms likely involve a complex repertoire of different factors influenced by inter-species interactions and the characteristics of the cell donor.
Despite the fact that we observed a rising number of animals presenting with granulomatous lesions over time from engraftment, a subset of animals remained unaffected by these lesions. This uneven distribution in the development of granulomatous lesions within the huNSG mouse model could be attributed to various factors. The literature describes how intrinsic donor characteristics can influence the predisposition to develop GvHD; specifically, mouse cohorts engrafted with donors sharing HLA haplotypes predisposed to autoimmune diseases are more susceptible to developing GvHD.20,48 However, in our experience, some animals engrafted from the same donor developed granulomatous lesions while others remained lesion-free, suggesting that multiple factors are involved. Myeloablation before engraftment represents another critical factor in the pathogenesis of GvHD, which induces cytokine activation and increases susceptibility to the disease. 22 In the current study, most animals were pre-conditioned using busulfan (15 mg/kg) and only a few underwent irradiation; however, the possibility of varying degrees of tissue damage cannot be excluded. The number of engrafted cells appeared to play a role in the current studies. Generally, a range of 1×104–105 isolated HSCs is administered per mouse in order to enable efficient human hematopoietic engraftment and long-term reconstitution. 49 Herein, we report that most animals injected with 1×105 cells presented with spontaneous macrophage-dominated GvHD lesions at 26 wpe. However, in our experience, in a study where animals were injected with 5×104 cells, no GvHD lesions were observed after 26 weeks. These findings contrast with a previous study that did not find any correlation between the number of HSCs injected and the incidence of GvHD observed. 10 Considering that different donors show different engraftment characteristics, 8 not only the predisposition to autoimmunity, but also the engraftment kinetics could perhaps play a role in the development of these lesions.
The treatment of huNSG mice with immunostimulatory compounds resulted in a rapid increase in the number of granulomatous lesions in several organs compared to age-matched untreated animals. This suggests that, while immunostimulatory treatment was intended to enhance immune responses, it may have inadvertently promoted the development or progression of the pathological process underlying lesion formation. This observation aligns with the current understanding of GvHD pathogenesis, where various stimulatory signals are central to disease development and can exacerbate the condition. 23 While there is limited literature directly supporting this, some evidence suggests inflammatory stimuli can exacerbate GvHD lesions; for instance, studies in humans and mice transplanted with HSCs have shown that mucositis-induced oral dysbiosis in the oral cavity exacerbated GvHD. 29 This outcome underscores the potential for unintended consequences when modulating the immune system, particularly in mouse models with potential immune deficiencies/dysregulation. Furthermore, it is important to note that not all huMice exhibit a similar response to immunostimulatory therapies. This has been demonstrated in a study comparing 2 different models and in vitro evaluations to predict cytokine release syndrome induced by TGN1412, an anti-CD28 drug known to cause severe cytokine release syndrome in humans. The results showed that bone marrow-thymus-liver huMice and in vitro systems could predict cytokine release syndrome, but CD34+ HSC huMice could not. 58 This indicates that both the drug candidate’s MoA and the intrinsic characteristics of the model need to be accurately considered for proper interpretation.
The spontaneous occurrence of bone marrow necrosis was rarely observed in older nontreated huNSG mice. Necrosis affected all bone marrow cell populations and occurred randomly throughout different bones, and even within the same bone (for example, affecting the diaphysis and not the epiphysis). The morphological appearance of these lesions suggests that they were not specifically targeted toward a single bone marrow cell population but rather resembled ischemic necrosis. Treatment with different drugs, each with a distinct MoA but all stimulating the immune system, significantly enhanced the incidence and accelerated the occurrence of the condition, even at earlier timepoints post-engraftment. For example, in study 4 with animals treated with CD20 CD3 TCB, bone marrow necrosis was already observed in animals euthanized 24 hours post-dosing, indicating an acute onset of the lesions following immune stimulation. In this study several thrombi were identified within periosteal and marrow vessels in a few animals, and these were suspected to be the main underlying cause of the bone marrow necrosis. The rapid onset of necrosis aligns with the drug’s MoA, whereupon binding of the antibody to the target and immediate activation of surrounding T cells, it induces apoptosis and releases pro-inflammatory cytokines. 3 The expression of both targets (CD3 and CD20) in the bone marrow may have resulted in the release of high levels of cytokines locally and in the circulation. During such inflammatory responses, pro-inflammatory cytokines like interleukin-6, tumor necrosis factor-alpha, and interferon-gamma are elevated. 55 These cytokines can lead to an overwhelming inflammatory response, which can damage the endothelium and promote activation of coagulation pathways. 55 However, no clear correlation between cytokine release and bone marrow necrosis could be established in the current study, which leaves the definitive confirmation of this pathogenesis challenging, as the absence of thrombi in other tissues of the affected animals remains unexplained. It is important to consider that cytokine increases are difficult to detect using single serum samples; the transient nature of cytokine release means they may be overlooked due to the brief interval between their elevation and subsequent decline. We also hypothesize that the absence of thrombi in other studies of huNSG mice treated with CD20 CD3 TCB may be due to the delayed observation period, during which thrombi could have potentially resolved in the affected regions.
It is important to note that bone marrow necrosis was not reported in toxicological studies with cynomolgus monkeys treated with CD20 CD3 TCB, nor in human patients. 42 This indicates model-specific pathogenesis of the bone marrow lesions observed in huNSG mice. Similar morphological changes were also seen in the bone marrow of huNSG mice after treatment with FAP 4-1BBL, which is a costimulation drug that enhances T cell proliferation, cytotoxicity, T helper cell polarization, and cytokine secretion. 12 In this case, the presence of lesions in the bone marrow suggests a FAP on-target effect. This is supported by the observation that animals showing a high level of granulomatous lesions in the bone marrow also showed local remodeling with FAP upregulation. Accordingly, lesions developed only in animals receiving the FAP-targeted molecule and not the untargeted DP47 4-1BBL. Interestingly, the lesions only developed after the second treatment, suggesting that initial immune system activation may be necessary to induce this damage. Bone marrow necrosis was also not observed in immunocompetent C57BL/6J mice treated with 3 weekly doses of a surrogate murine bispecific antibody (ie, an immunoglobulin with equivalent functionality as the therapeutic antibody but directed against the orthologous murine antigen), featuring the anti-FAP binder 28H1, which has high affinity for murine FAP, and an anti-mouse 4-1BB binder, when sacrificed 8–13 days after the last treatment (data not shown). Similarly, when the therapeutic antibody FAP 4-1BBL was administered to cynomolgus monkeys in single- and repeat-dosing safety studies or human patients in clinical trials, no evidence of microscopic or functional changes was observed in bone marrow. 36 In both of the cases presented, the observed effect, although challenging to elucidate, has been shown to be exclusive to this particular mouse model and not translatable to humans. This highlights the potential limitations of using animal models to predict human responses, particularly in safety studies with potential bone marrow liabilities. Bone marrow necrosis is a clinical entity that is rarely described in humans; however, it is mostly associated with underlying hematological malignancies, and alterations in the microcirculation of the bone marrow are suspected.11,27,57 These findings indicate that huMice are prone to bone marrow necrosis, a condition worsened by immune stimulation treatments. Although the exact cause is unclear, thrombotic events in bone blood vessels are the most likely explanation; however, the unknown mechanism highlights the need for further research into this phenomenon.
The pathology characterization conducted over time also allowed us to optimize the experimental design of pharmacology studies, with the identification of a critical interval between effective engraftment, enabling the use of these animals for cancer immunotherapy research, and the increased frequency of granulomatous lesion development, which could introduce confounding variables in the studies. We also identified that immuno-oncology therapies that elicit strong immune stimulation can accelerate spontaneous lesion incidence and severity, which is not translatable to humans. The identification and understanding of these peculiar features occurring in these mice and their identification as being strain-related is of paramount importance for proper histopathological interpretation and to distinguish them from potential experimentally induced lesions within the investigated models.
In summary, huNSG mice engrafted with human CD34+ HSCs serve as a crucial model for cancer immunotherapy research, despite inherent challenges such as spontaneous granulomatous lesions and bone marrow necrosis. The findings reported herein emphasize the necessity for thorough characterization and careful interpretation of pathological changes in huNSG mice to distinguish between model-specific background lesions and drug-induced effects. This comprehensive understanding is essential for the accurate safety assessment of novel immunotherapeutic agents.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858261423132 – Supplemental material for Pathological insights of human CD34+ hematopoietic stem cell-engrafted NSG mice
Supplemental material, sj-pdf-1-vet-10.1177_03009858261423132 for Pathological insights of human CD34+ hematopoietic stem cell-engrafted NSG mice by Anna Maria Giusti, Johannes Sam, Sara Colombetti, Inês Berenguer Veiga and Josep M. Monné Rodríguez in Veterinary Pathology
Footnotes
Acknowledgements
We would like to thank José Galván, Bettina Amberg, Rachel Neff, Virgine Ott, and Gabrielle Branellec from the PASS Chapter Pharmaceutical Sciences, Roche Innovation Center Basel, for IHC staining, and Ben McDermott of the Bioscript Group (Macclesfield, UK), who provided medical writing assistance in accordance with Good Publication Practice guidelines, which was funded by F. Hoffmann-La Roche Ltd.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees and shareholders of F. Hoffmann-La Roche Ltd.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Roche Pharma Research and Early Development, F. Hoffmann-La Roche Ltd., Basel, Switzerland.
