Abstract
Immunodeficient mice engrafted with human immune cells represent an innovative tool to improve translatability of animal models for the study of human diseases. Immunophenotyping in these mice focuses on engraftment rates and cellular differentiation in blood and secondary lymphoid organs, and is predominantly carried out by FACS (fluorescent activated cell sorting) analysis; information on the morphological aspects of engraftment and the prevalence of histologic lesions is limited. We histologically examined 3- to 6-month-old NSG mice, naïve or engrafted with CD34+ human hemopoietic stem cells (HSC), and employed a quantitative immunohistochemical approach to identify human and murine cell compartments, comparing the results with the FACS data. NSG mice mainly exhibited incidental findings in lungs, kidneys, testes, and adrenal glands. A 6-month-old NSG mouse had a mediastinal lymphoblastic lymphoma. The lymphoid organs of NSG mice lacked typical lymphoid tissue architecture but frequently exhibited small periarteriolar leukocyte clusters in the spleen. Mice engrafted with human HSC frequently showed nephropathy, ovarian atrophy, cataract, and abnormal retinal development, lesions considered secondary to irradiation. In addition, 20% exhibited multisystemic granulomatous inflammatory infiltrates, dominated by human macrophages and T cells, leading to the observed 7% mortality and morbidity. Immunophenotypic data revealed variable repopulation of lymphoid organs with hCD45+ human cells, which did not always parallel the engraftment levels measured via FACS. The study describes the most common pathological features in young NSG mice after human HSC engraftment. As some of these lesions contribute to morbidity, morphological assessment of the engraftment at tissue level might help improve immunophenotypic evaluations of this animal model.
Keywords
Recent advances in the generation of humanized murine models, featuring immunodeficient mice engrafted with human immune system cells, have brought tangible breakthroughs in numerous research fields, such as the preclinical development of immunotherapeutic agents and the study of infectious, metabolic, and autoimmune diseases. Progress has been facilitated by the optimization of immunodeficient mouse models, which, being deprived of acquired and innate immunity, can be transplanted with human cells that eventually develop into a variably functional immune system in the model. 9,45,57
Among the numerous immunodeficient mouse strains available, the NOD-scid IL-2Rγnull (NSG) mouse is currently the most widely used for successful humanization of the immune system, showing one of the highest engraftment rates among the various strains. 29,58,64 NSG mice are nonobese diabetic (NOD) mice bearing the Prkdc scid mutation and a genetic deletion of the common γ chain of the IL-2 receptor. Accordingly, they exhibit defects in lymphoid cell development, lacking both innate and adaptive lymphocytes, and have diminished complement activity due to C5 deficiency. 9,24,52 In contrast to their NOD/scid predecessors, NSG mice are not prone to develop thymic lymphoma; they have a long life span of about 2 years. 9,23 Functional immune system abnormalities in immunodeficient mice are associated with abnormal development of the primary and secondary lymphoid organs; however, the morphological equivalents of the latter vary across the available models. 49 NSG mice, for example, exhibit a small spleen, thymus, and lymph nodes that lack lymphoid structures and are composed almost exclusively of reticular stromal cells. 47, 51,52
Immunodeficient mice can be transiently or stably humanized by administration of human mature lymphoid or CD34+ hematopoietic stem cells (HSC), respectively. 61 Most models allow multilineage human immune cell engraftment, and both the acquired and innate human immune system components develop to variable degrees. The present study focuses on CD34+ HSC humanized NSG (hu-NSG) mice, a model in which T and B cell engraftment is generally successful, though without consistent immunoglobulin (Ig) class switching and mostly IgM production by B cells. 19,45,56 Hu-NSG mice have human dendritic cells, NK cells, and myeloid cells including neutrophils. 25,35,60 However, the latter two are generally seen at low numbers, and seem to be defective in their maturation and function. 19,23,45,57 It has been shown that the transplanted human leukocytes form variably sized aggregates in the lymphoid organs of these mice, which in general do not exhibit distinctive secondary lymphoid structures such as lymphoid follicles in spleen and lymph nodes. 23,57,60 In the thymus, repopulation appears to be more efficient in immunodeficient mice transplanted shortly after birth, while in mice engrafted at adult age the thymus is less prone to support human cell engraftment. 7,62 In the bone marrow the proportion of lymphoid and myeloid cells varies, as different published studies indicate. 60,62
Effective engraftment in hu-NSG mice is generally determined by fluorescent activated cell sorting (FACS) analysis of the blood, usually at 12 to 16 weeks postengraftment. The FACS data suggest that the extent of engraftment can vary substantially depending on the age of the mice, the source and manipulation of the HSC, and the administration route. 23,26 So far, most of the immunophenotyping data that are available for these mice are based on FACS analyses, carried out on circulating and organ-specific immune cells at different endpoints. 7,26,39,56 Accordingly, there is very limited information concerning the in situ aspects of engraftment, in particular in the hemolymphatic tissues and, specifically, how these correlate with the engraftment rates and cellular differentiation determined by FACS. Furthermore, little is known about the incidence of spontaneous diseases and common histopathological lesions in hu-NSG mice, and even in NSG mice. Information about the spectrum of histopathological lesions occurring spontaneously in NSG mice is mainly limited to the prevalence of neoplastic findings in the NOD background strain 22 or a few reports on the most common causes of mortality and morbidity and age-related lesions in old NSG mice housed in conventional mouse facilities. 16,47 In the few reports available about hu-NSG mice, spontaneous changes were generally mentioned alongside the findings relevant to the study instead of being a focus of investigation. 8,32,47,62 However, a recent study reported histological lesions that provide evidence of the occurrence of graft versus host disease (GvHD) in HSC hu-NSG mice. 11 Considering the increasing use of humanized mice in efficacy and safety studies, including regulatory safety assessment, this knowledge gap needs to be addressed and better characterization of the model is warranted to consolidate its translatability.
The present study aimed to characterize the morphological phenotype of NSG mice and their humanized counterpart, the CD34+ hu-NSG murine model generated via perinatal intrahepatic injection of human CD34+ fetal liver cells into NSG mice after preconditioning irradiation. We found high variability in the levels of human cell engraftment in the blood of hu-NSG mice within the same experiment and hypothesized that this is reflected by a similarly variable reconstitution in the lymphoid organs. Another purpose of this study was to assess the spectrum of histopathological lesions and identify the main causes of morbidity and mortality in young NSG and CD34+ HSC hu-NSG mice, since, despite the increased popularity of these animal models in nonclinical testing, the published data on this topic is very sparse.
Materials and Methods
Animals
The study was undertaken on NOD-scid IL-2Rγnull mice from a breeding colony established in a research facility at the Institute of Experimental Immunology, University of Zurich, Switzerland, from animals originally purchased from Jackson Laboratories (Bar Harbor, ME). Animals were housed in groups of up to 5 males or females respectively and fed ad libitum with conventional diet (M/R Haltung Extrudat, Alleinfuttermittel für Mäuse und Ratten, 3436; Granovit AG). Water was provided ad libitum, the day and night cycle included 12 hours of light, from 6
The study cohort comprised a total of 109 mice. Among these were 48 NSG mice (31 males, 17 females) that did not receive any treatment. Of these, 10 (6 males, 4 females) were culled at 1 to 3 months of age, representing animals bred for, but not used in humanization experiments. Thirty-eight (25 males, 13 females) were breeding animals that were culled at 6 months of age, at the end of their breeding career. The second cohort of 61 hu-NSG mice (31 males, 30 females) were subjected to human cell engraftment (inefficiently humanized mice at 3 months of age [n = 22; <15% hCD45+ cells in peripheral blood after reconstitution], and efficiently humanized mice at 4–5 months of age [n = 39]). These animals had served as the noninfected control group in an experiment to investigate the effects of Epstein-Barr virus infection on the immune system and were culled at the age of 3 to 5 months, at termination of the experiment. As part of the above-mentioned experiment on Epstein-Barr virus infection, the animals were injected intraperitoneally with 100 µl phosphate-buffered saline (PBS) at 12 to 14 weeks after irradiation. At the age of 4 months, 4 of these animals, 2 males and 2 females, developed progressive weight loss and signs of unease such as hunched posture, rough coat, and reddened skin, and had to be euthanized.
After reconstitution, mice were clinically checked daily for 3 days, and then 3 times a week until weaning. A weekly check was performed on mice 4 weeks and older, including naïve NSG mice.
Human Tissue Engraftment
CD34+ HSC were isolated from fetal livers (Advanced Bioscience Resources) following established protocols. 54 Cells originated from 13 different donors and were frozen in liquid nitrogen prior to their use in the engraftment experiments. At the age of 1 to 6 days, NSG mice (n = 61) were each irradiated with 1 Gy (preconditioning), using a “Rad Source 2000” equipped with an X-ray source, and injected intrahepatically 5 to 7 hours later with 1 to 3 × 105 CD34+ human HSC 31 After 10 to 12 weeks, blood was collected via the tail vein and analyzed by FACS. In addition to the above reported morbidity after 4 months, 6% of irradiated and reconstituted hu-NSG pups were lost prior to weaning due to neglect or cannibalism by the mother. It remains unclear if this was connected to irradiation and/or reconstitution.
Ethics Statement
All animal protocols were in accordance with the Swiss Animal Welfare Act, Tierschutzgesetz (TSchG), and were approved by the veterinary office of the canton of Zurich, Switzerland (Protocols 148/2011, 209/2014, and 159/17). The studies involving human samples were reviewed and approved by the cantonal ethics committee of Zurich, Switzerland (Protocols KEK-StV-Nr.19/08, KEK-ZH-Nr. 2010-0057, and 2019-00837).
Macroscopic and Histological Examination
Mice were euthanized by 100% carbon dioxide asphyxiation, followed by exsanguination and creation of a pneumothorax. A full necropsy was conducted on each mouse and a standard selection of organs, hemolymphatic tissues, as well as bones and joints were sampled and fixed in 10% neutral buffered formalin for 48 to 120 hours, then trimmed and routinely embedded in paraffin wax following a predefined blocking pattern. Consecutive sections (3–5 μm) were prepared and routinely stained with hematoxylin and eosin (HE), special stains (when appropriate), or subjected to immunohistochemistry. Examined tissues were adrenal glands, brain, eyes, female genital tract (mammary gland, ovaries, uterus, vagina), gastrointestinal tract (stomach, duodenum, jejunum, ileum, cecum, colon, and rectum), Harderian gland, heart, kidneys, liver with gall bladder, lungs, male genital tract (epididymis, prostate, testes, seminal vesicles), pancreas, salivary glands, skeletal muscle (Musculus quadriceps femoris), skin, spinal cord, tongue, and urinary bladder. Examined hemolymphatic tissues were bone marrow from several locations (see bones), lymph nodes (cervical [one lymph node of the cervical chain: mandibular, accessory mandibular or superficial parotid], mesenteric [colic or jejunal], proper axillary) (in most animals, not all 3 lymph nodes were successfully sampled as these could not be identified grossly), spleen (1–2 cross sections), and thymus. Since thymus and lymph nodes were difficult to grossly identify especially in NSG mice, tissue in the corresponding area was collected and included in the paraffin block. Bones (sternum, femur, and tibia with femorotibial joint, spinal column) as well as the head with nasal and oral cavities and teeth were also collected. After fixation, these were decalcified for 3 to 5 days in a mild decalcifying solution (RDF, Mild Decalcifier, CellPath), followed by paraffin embedding. Special stains were applied when considered necessary and comprised the Gram, Giemsa, Grocott, and Ziehl-Neelsen stains as well as the periodic acid–Schiff reaction.
Immunohistochemistry
Immunohistochemistry was applied to detect cells of human origin (Ku80+), human and mouse leukocytes (hCD45+, mCD45+), T cells (CD3+, CD4+, CD8+), human and mouse B cells (CD20+ and CD45R-B220+, respectively), monocytes/macrophages (calprotectin+, Iba1+, F4/80+, lysozyme+), and MHC II expression. Antibodies and detection methods are listed in Tables 1 and 2.
Antibodies and Detection Methods Used for Immunohistochemistry.
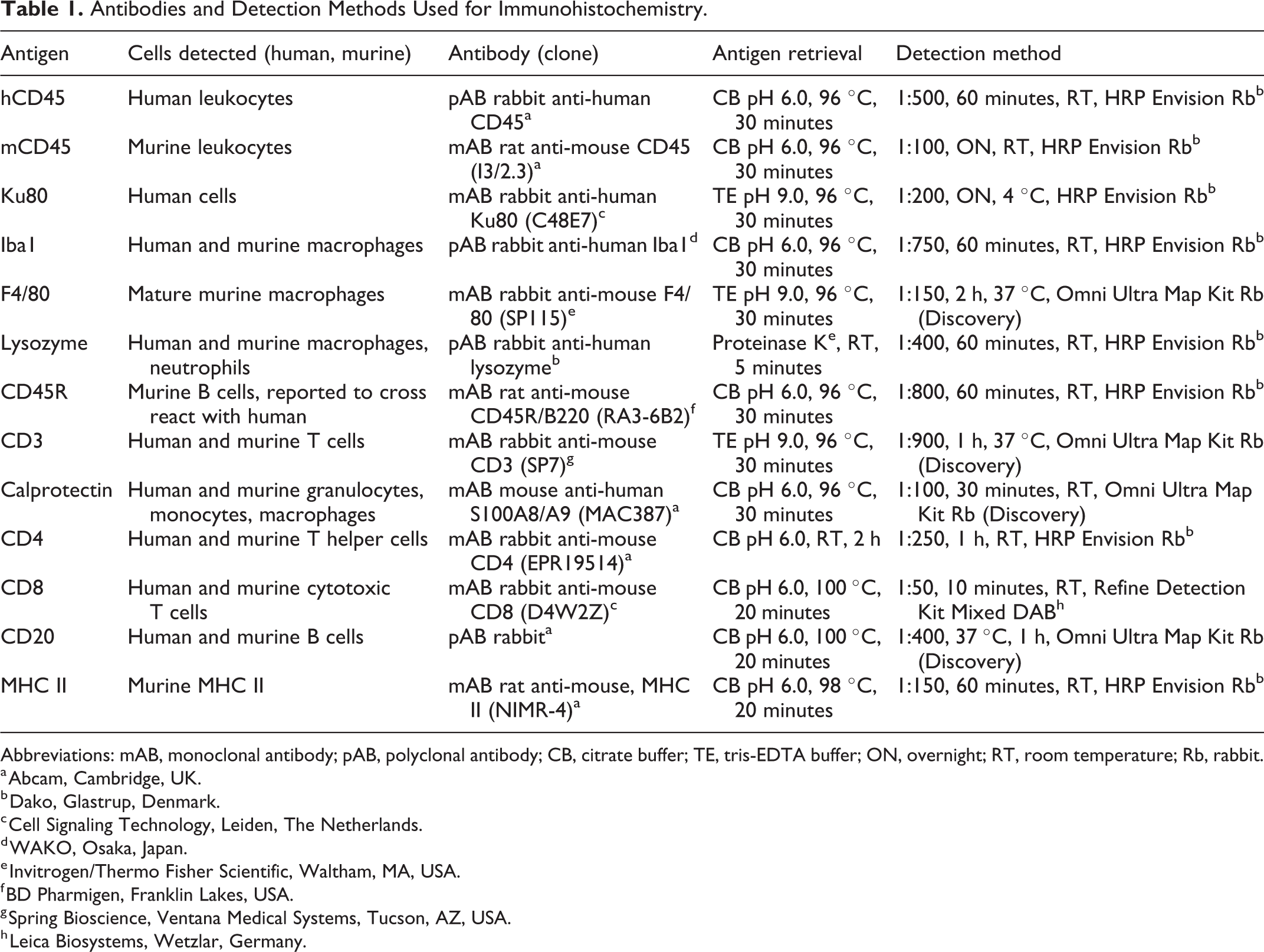
Abbreviations: mAB, monoclonal antibody; pAB, polyclonal antibody; CB, citrate buffer; TE, tris-EDTA buffer; ON, overnight; RT, room temperature; Rb, rabbit.
a Abcam, Cambridge, UK.
b Dako, Glastrup, Denmark.
c Cell Signaling Technology, Leiden, The Netherlands.
d WAKO, Osaka, Japan.
e Invitrogen/Thermo Fisher Scientific, Waltham, MA, USA.
f BD Pharmigen, Franklin Lakes, USA.
g Spring Bioscience, Ventana Medical Systems, Tucson, AZ, USA.
h Leica Biosystems, Wetzlar, Germany.
Antibodies Applied in the FACS Analysis to Detect Human Cells.
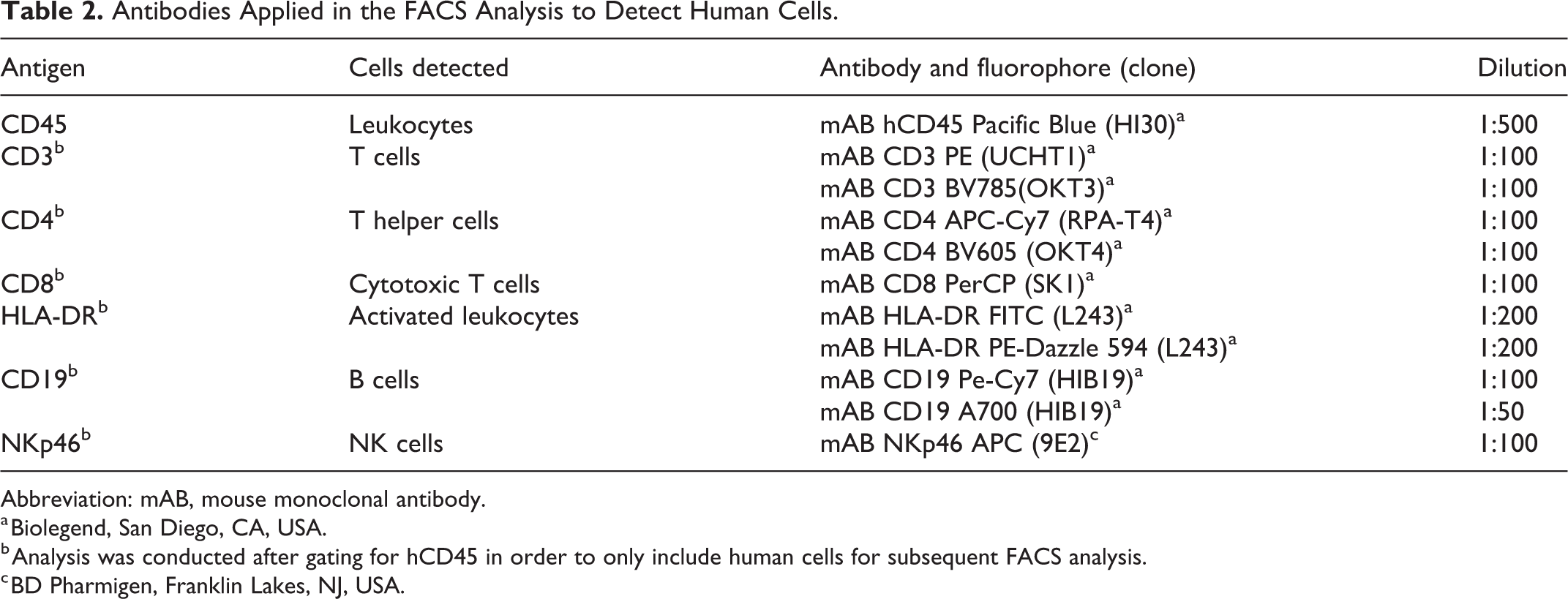
Abbreviation: mAB, mouse monoclonal antibody.
a Biolegend, San Diego, CA, USA.
b Analysis was conducted after gating for hCD45 in order to only include human cells for subsequent FACS analysis.
c BD Pharmigen, Franklin Lakes, NJ, USA.
Histological Scoring and Histomorphometry
All tissues/organs were examined for histopathological changes which were diagnosed following the International Harmonization of Nomenclature and Diagnostic Criteria (INHAND), Society of Toxicological Pathology (www.toxpath.org/inhand.asp). When appropriate, lesions were graded semiquantitatively using a score from 1 to 5. Granulomatous inflammation scores were primarily determined by the amount of tissue involvement (1 = minimal [<1%]; 2 = mild [1% to 25%]; 3 = moderate [26% to 50%]; 4 = marked [51% to 75%]; 5 = severe [76% to 100%]) as described. 50
For morphometric evaluation, slides with hemolymphatic tissue sections stained by immunohistochemistry were scanned using a digital slide scanner (NanoZoomer-XR C12000, Hamamatsu Photonics K.K.), and the Visiopharm Integrator System (Visiopharm) was used for the subsequent quantitative analysis. The mCD45 and hCD45 positive cell fraction areas and the number of Ku80 (nuclear marker) positive nuclei per high power field were calculated on consecutive sections. The mCD45 positive cell fraction area in hemolymphatic organs repopulated by human cells was calculated following subtraction of the hCD45 positive area from the total tissue area.
FACS Analysis
FACS served to identify hCD45+, CD3+, CD4+, CD8+, CD19+, NKp46+, CD4/DR+, CD8/DR+ populations and followed previously described protocols. 2
Peripheral blood was collected from hu-NSG mice 3 months after human cell engraftment by tail vein bleeding into heparin-containing tubes (Heparin-Na, Braun). After 2 rounds of red blood cell lysis, peripheral blood mononuclear cells (PBMCs) were washed with PBS and stained with fluorescence coupled antibodies for 20 minutes at 4 °C. Antibodies are listed in Table 2. Subsequently, cells were fixed in 4% paraformaldehyde diluted in PBS for 0 to 24 hours. Data were acquired on the BD FACSCanto II using DIVA software and analyzed using FlowJo software Version 10.2. The cells were first gated for singlets, followed by hCD45.
Statistical Analysis
Statistical analysis was conducted using a Statistics Software (SPSS, 23). Correlation between FACS results and abundance of hCD45 positive cells in the lymphoid organ tissue sections was tested using Pearson test. Spearman’s correlation analysis was used to study the relationship between granulomatous inflammation and activated T cells. For this analysis, granulomatous inflammation was divided into 3 categories as 0 = not present; 1 = mild, occurring with low severity and limited distribution; and 2 = severe, occurring with high severity and broad distribution. Statistical significance was evaluated using Mann-Whitney U test and t test. P values <.05 were considered statistically significant.
Results
Background Lesions and Radiation-Induced Changes
The NSG mouse cohort (n = 48) remained healthy during the examination period and exhibited only a limited selection of histological lesions, with generally low severity (Table 3). The most frequently encountered changes were minimal or mild multifocal alveolar macrophage accumulation with pleural thickening in the lungs; minimal focal tubular basophilia and tubular dilation with epithelial cell hypertrophy in the kidneys; minimal or mild patchy degeneration and dystrophic mineralization of seminiferous tubules in the testes; and minimal to moderate multifocal subcapsular cell hyperplasia in the adrenal glands. 50 The latter occurred with higher frequency in females. Sporadically, minimal or mild focal degenerative changes in the intersternebral and intervertebral joints as well as cortical thickening of the femoral diaphysis were noted. Individual animals exhibited inflammatory processes, such as minimal focal neutrophilic myositis in retro-orbital and masticatory muscles, focal neutrophilic folliculitis in the skin of the muzzle, and a moderate diffuse chronic sialoadenitis. The latter appeared to be the consequence of an obstruction of the main excretory duct by a hair. Also, one mouse showed marked chronic pancreatitis, characterized by diffuse fibrosis, moderate mononuclear cell inflammation, and lobular acinar atrophy. Apart from occasional dilation of endometrial glands, the female reproductive tract was free of histopathological changes. Ovaries exhibited numerous follicles at different stages of maturation and corpora lutea. The vagina showed normal cyclic changes consistent with all 4 phases of the rodent reproductive cycle.
Spectrum of Histological Lesions in Male and Female NSG and hu-NSG Mice.
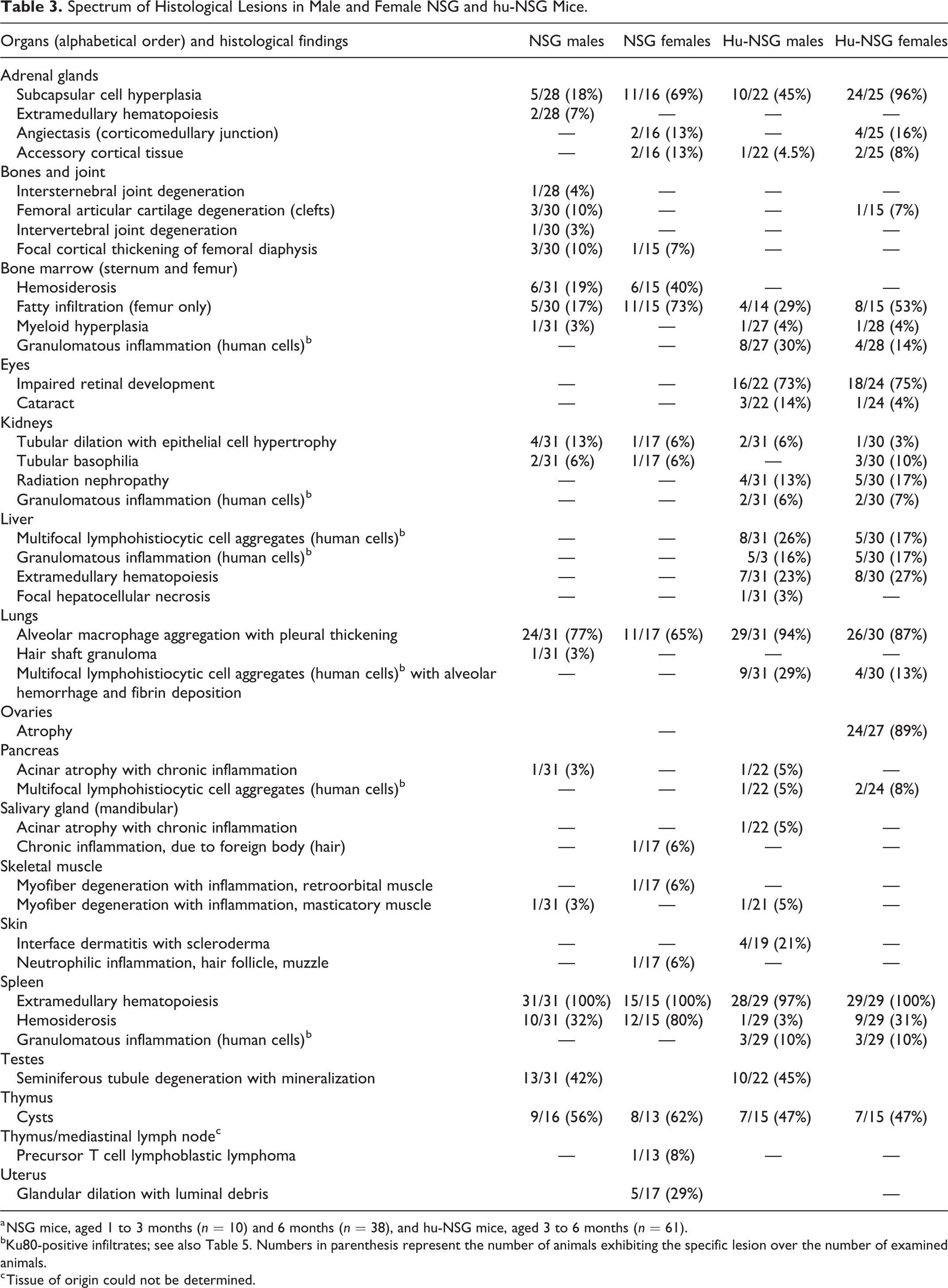
a NSG mice, aged 1 to 3 months (n = 10) and 6 months (n = 38), and hu-NSG mice, aged 3 to 6 months (n = 61).
bKu80-positive infiltrates; see also Table 5. Numbers in parenthesis represent the number of animals exhibiting the specific lesion over the number of examined animals.
c Tissue of origin could not be determined.
The hu-NSG mice exhibited the same type of background lesions as the NSG mice, and with similar frequency and severity (Table 3). In addition, hu-NSG mice showed histological changes consistent with exposure to radiation in the early postnatal period, that is, radiation nephropathy, cataract and impaired retinal development, 4 and ovarian atrophy. 1,6 Radiation nephropathy was characterized as deposition of proteinaceous material and reduced number of capillaries in glomeruli, cortical tubular collapse, 40 and was observed in relatively few mice (13% of males, 17% of females) and with minimal severity (Fig. 1). Impaired retinal development was more frequent (more than 70% of animals), and ranged from a thinning of the outer nuclear layer and the outer segment layer, to a lack of distinction between outer and inner nuclear layer, leading to a very thin retina composed of a single row of nuclei (Fig. 2). Changes were more severe in the peripheral portion of the retina. Cataract occurred in fewer mice (Table 3) with low severity and consisted of rupture of the posterior lens capsule with lens fiber fragmentation and focal liquefaction. Almost all female animals exhibited ovarian atrophy of varying degree. In more severe cases, the ovaries were reduced in size and exhibited a marked reduction to complete absence of oocytes, follicles and corpora lutea (Fig. 3). Such animals appeared to be in persistent anestrus, as there was no evidence of estrus cycling in uterus and vagina.
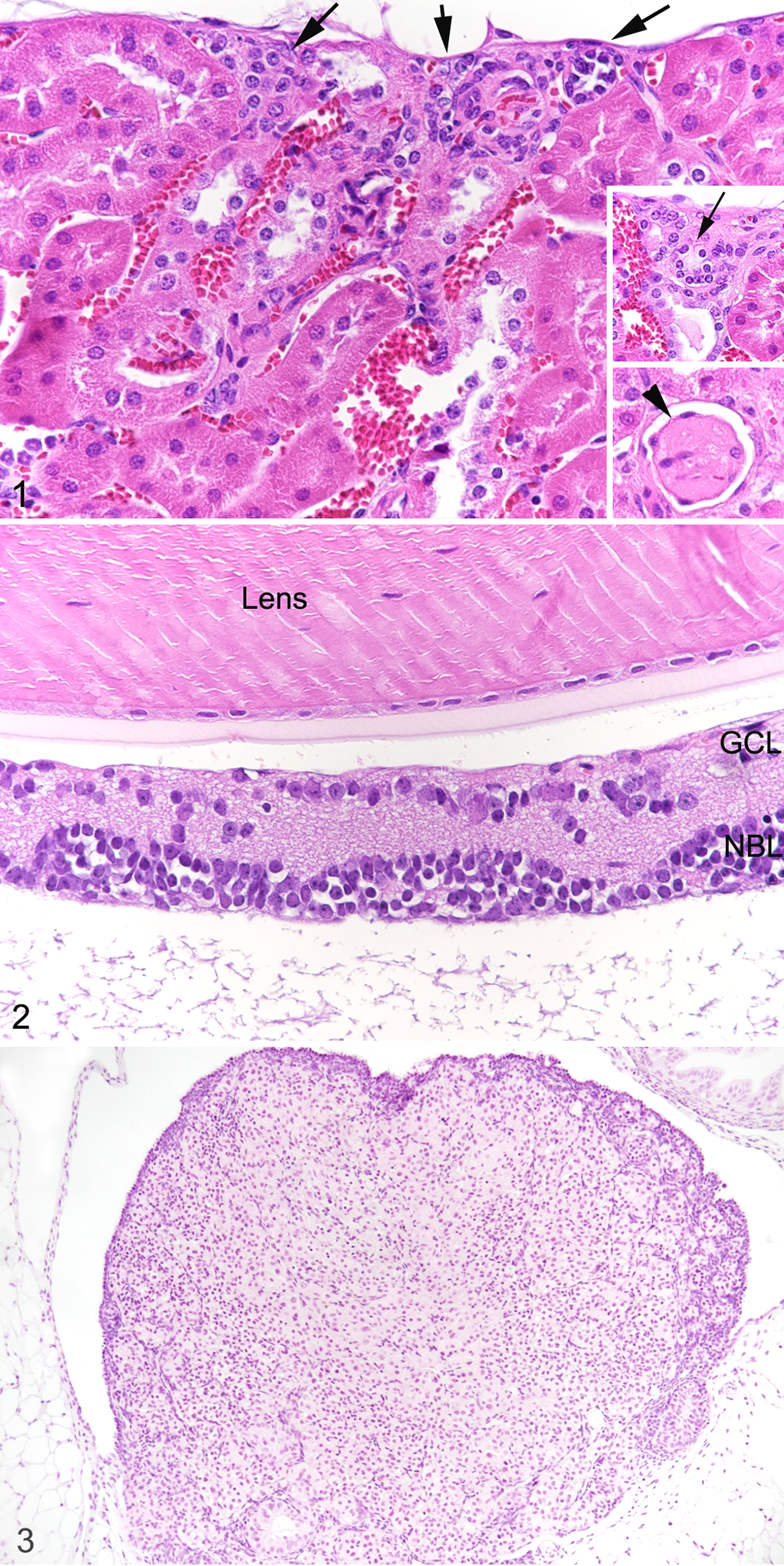
Radiation exposure (one-time irradiation with 1 Gy at the age of 1–6 days), hu-NSG mouse. Hematoxylin and eosin.
Hemolymphatic Tissues
Spleens, lymph node, thymus, and bone marrow were compared in both groups of mice, in particular to comparatively assess the murine and human leukocyte component in hu-NSG mice. Based on previous experience of the group, hu-NSG mice were classified as inefficiently humanized (hu-NSG(i) mice), when they exhibited less than 15% hCD45+ cells in the blood (determined by FACS). This applied to 22/61 (40%) of the animals in the present study.
Spleen
In the NSG mice, spleens were generally small, less than 12 × 2 × 1 mm in size, with rare, randomly distributed leukocytes (mCD45+) and no morphological evidence of lymphocytes populating follicles and T cell zones. In 27/48 of the NSG mice, small aggregates of mCD45+ cells were found around 1 or 2 arterioles, reminiscent of periarteriolar lymphoid sheaths (PALS; Fig. 4a, b). The aggregates were composed of round to polygonal cells of approximately 15 µm diameter, with scant cytoplasm and a large round to slightly elongated nucleus with finely stippled chromatin and no evident nucleolus. These cells were consistently negative for T cell (CD3), B cell (CD45R-B220), and macrophage (F4/80, calprotectin and lysozyme) markers. The red pulp was mainly populated by erythroid progenitors and some megakaryocytes, representing minimal to moderate extramedullary hematopoiesis (Fig. 4a), and scattered leukocytes; some animals also exhibited minimal hemosiderosis.
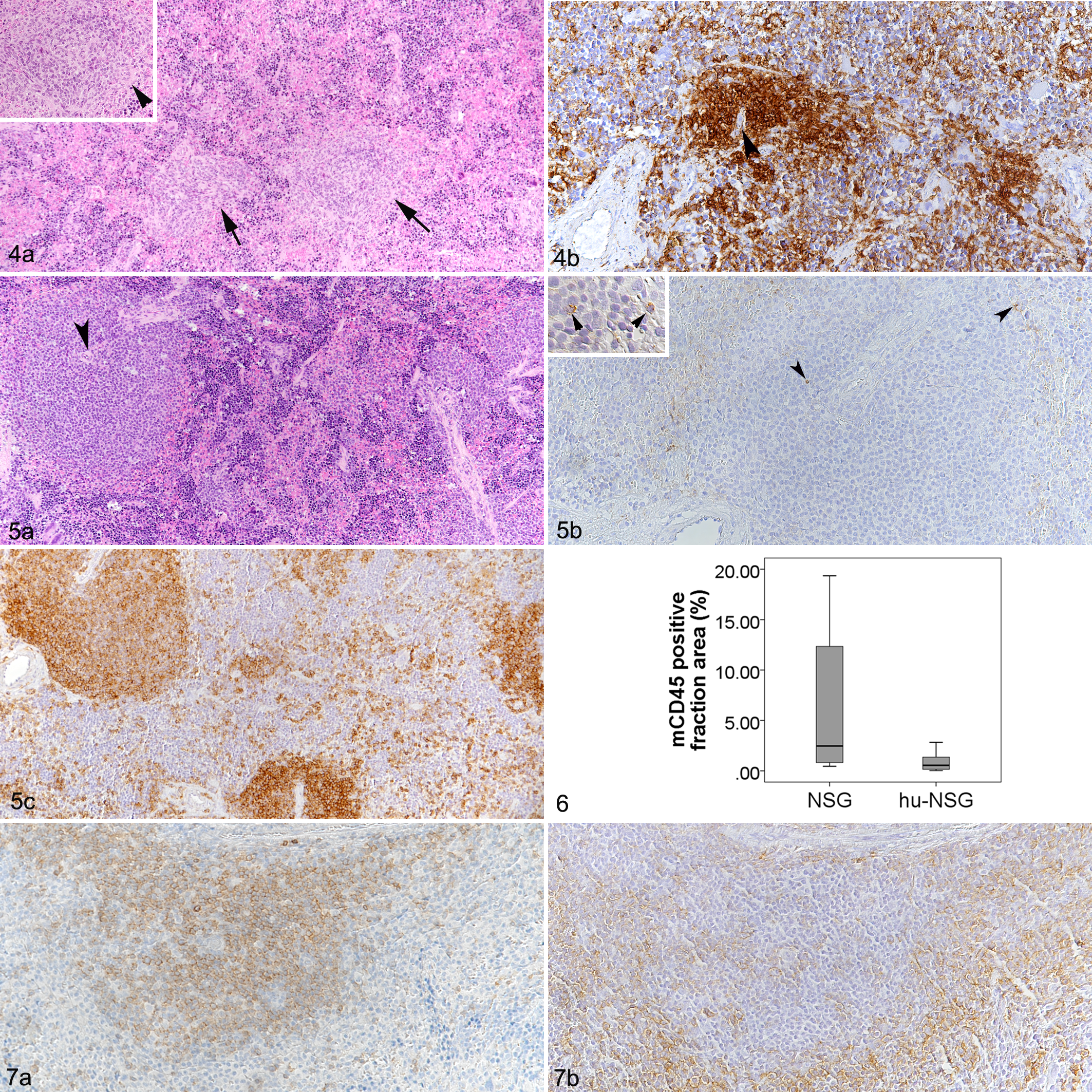
Spleen.
In all hu-NSG mice, the spleens were substantially larger, approximately 15 × 5 × 2 mm on average. Histologically, this was due to the much higher cellularity of the organ secondary to human cell repopulation. While there was no evidence of follicular structures, human cells tended to cluster around splenic arterioles, forming PALS-like structures (Fig. 5) similar to but generally much larger than those formed occasionally by mCD45+ cells in the spleen of NSG mice. In the latter, due to the PALS-like aggregates, the proportion of the area in the spleen covered by mCD45+ cells averaged 6% (n = 6; mean = 6.3; SD = ±7.8), but exceeded 10% in individual animals (Fig. 6), whereas murine leukocytes (mCD45+) were extremely rare in hu-NSG mice (Figs. 5b, 6). In contrast, they carried numerous hCD45+ cells (Fig. 5c). In efficiently humanized (hu-NSG(e)) mice, the hCD45-positive cell fraction area was 21.0% on average, though the proportion varied considerably between animals (Table 4). In hu-NSG(i) mice, the hCD45 positive cell fraction area had a mean of 8.8% (Table 4); however, the difference between the 2 groups was not significant. The white pulp in hu-NSG mice consisted of B cells (CD20+) and T cells (CD3+) in equal proportions. The T cells were mostly found in the center of the reconstituted white pulp, in the PALS-like structures around the arteries (Fig. 7a). B cells tended to occupy the periphery of the white pulp but did not arrange in clear follicular structures (Fig. 7b). Immunolabeling for CD4 and CD8 identified mainly CD4+ T cells whereas only random individual cells scattered throughout the parenchyma were CD8+. In all animals, the remaining area (red pulp) was mainly occupied by erythroid progenitor cells, intermingled with megakaryocytes (moderate extramedullary hematopoiesis) and scattered hCD45+ leukocytes with the morphology of lymphocytes; these appeared to be represented by B cells and T cells in equal proportions (Fig. 7).
Human Cells in the Lymphoid Organs of Efficiently and Inefficiently Humanized NSG Mice.a,b
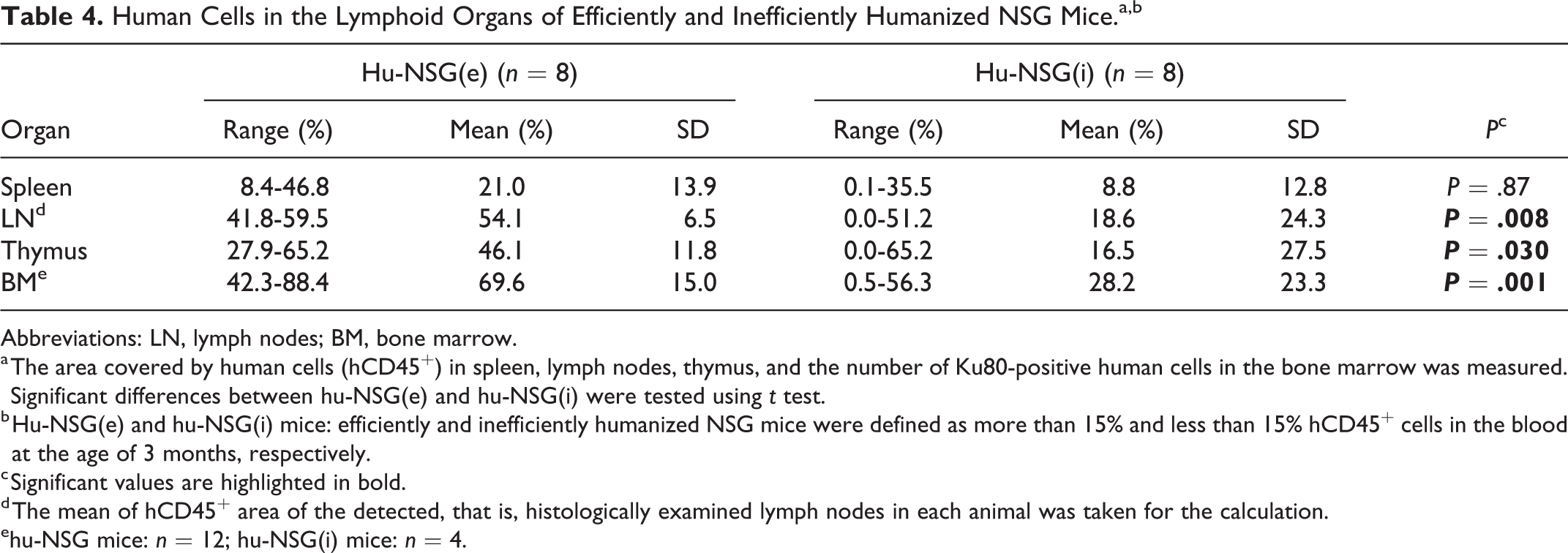
Abbreviations: LN, lymph nodes; BM, bone marrow.
a The area covered by human cells (hCD45+) in spleen, lymph nodes, thymus, and the number of Ku80-positive human cells in the bone marrow was measured. Significant differences between hu-NSG(e) and hu-NSG(i) were tested using t test.
b Hu-NSG(e) and hu-NSG(i) mice: efficiently and inefficiently humanized NSG mice were defined as more than 15% and less than 15% hCD45+ cells in the blood at the age of 3 months, respectively.
c Significant values are highlighted in bold.
d The mean of hCD45+ area of the detected, that is, histologically examined lymph nodes in each animal was taken for the calculation.
ehu-NSG mice: n = 12; hu-NSG(i) mice: n = 4.
Lymph Nodes
In NSG mice, lymph nodes were generally very small and difficult to identify grossly. However, in the majority of animals, the mesenteric and mandibular lymph nodes were successfully sampled. They were composed predominantly of connective tissue, that is, the reticular stroma (Fig. 8a). Leukocytes (mCD45+) were present in low numbers, scattered throughout the fibrous scaffold (Fig. 8b). When sampled successfully, axillary and inguinal lymph nodes exhibited the same morphological features.
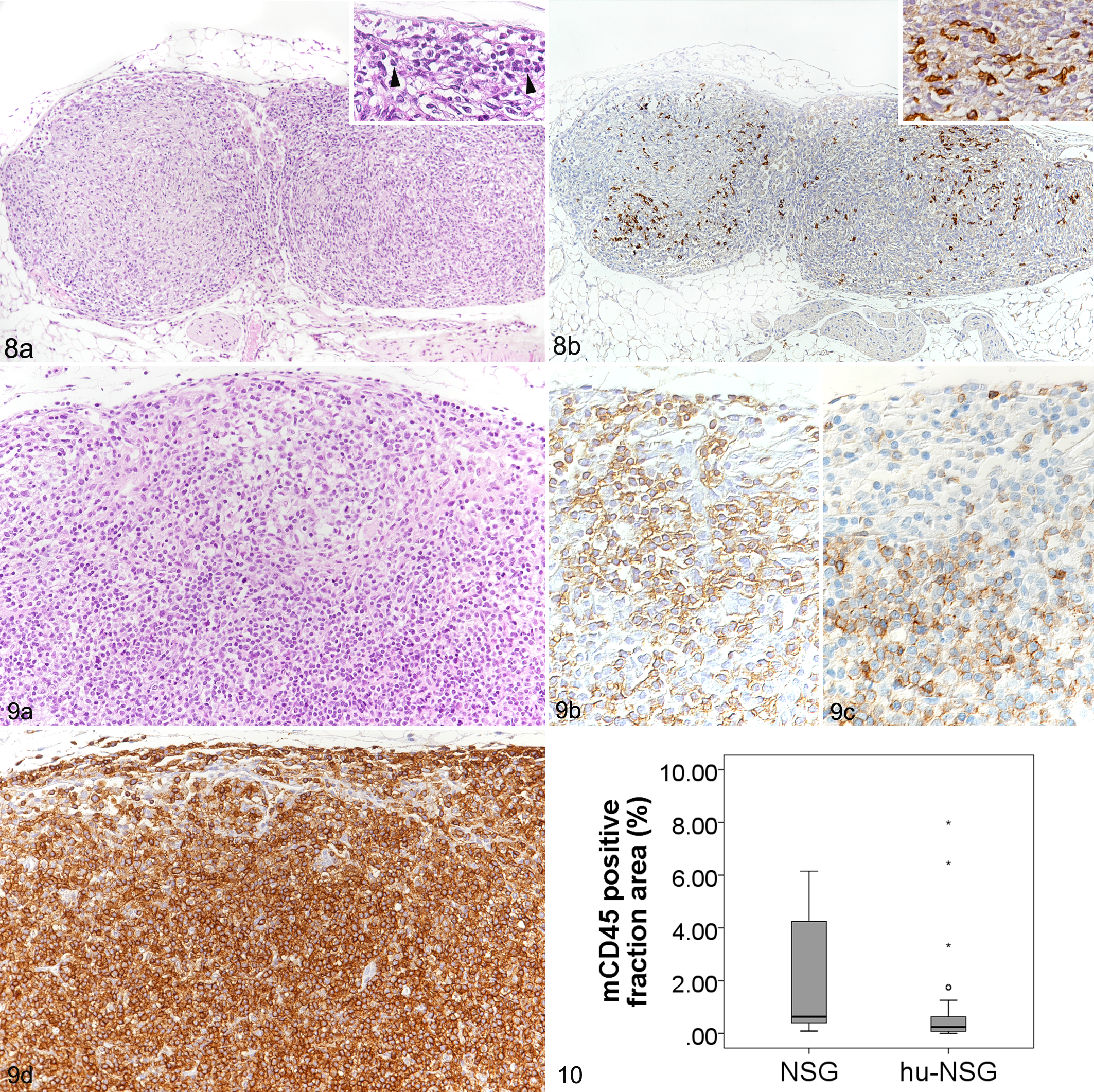
Lymph nodes.
In hu-NSG mice, lymph nodes were in general larger and therefore easier to identify grossly. Histologically, most lymph nodes were abundantly populated by lymphoid round cells (Fig. 9). They contained only few murine leukocytes (mCD45+; n = 33, mean ± SD = 0.86 ± 1.8%), and on average less than the lymph nodes of NSG mice (n = 11; 2.8 ± 4.1%; Fig. 10). Instead, human leukocytes (hCD45+) repopulated the fibrous stroma (n = 33; 42.1 ± 22.4%; Fig. 9d). In efficiently humanized mice, the hCD45-positive cell fraction area had a mean of 54.1%, whereas in inefficiently humanized mice, the mean was 18.6% (Table 4). In some of the latter animals, no human cells were identified at all; these lymph nodes had morphological features identical to those of the NSG mice. However, also in the efficiently humanized mice, repopulating hCD45+ cells appeared to distribute randomly across the lymph node, did not aggregate to form appreciable lymphoid follicles or paracortical structures, and did not allow distinction of the cortex and medulla. Accordingly, CD20+ B cells which were present in moderate to high numbers, and CD3+ T cells which were slightly less numerous, were seen randomly distributed throughout the stroma. Overall, the periphery had a higher proportion of B cells (Fig. 9b) while T cells tended to be more numerous in the center (Fig. 9c). Most T cells appeared to be CD4+, whereas CD8+ cells were only present in very small numbers.
Thymus
In NSG mice, the thymus was generally barely visible and composed of hypoplastic lobules that lacked a defined cortex and medulla and often contained cysts, as previously reported (Fig. 11). 7 Again, leukocytes (mCD45+) were rare (Fig. 12).
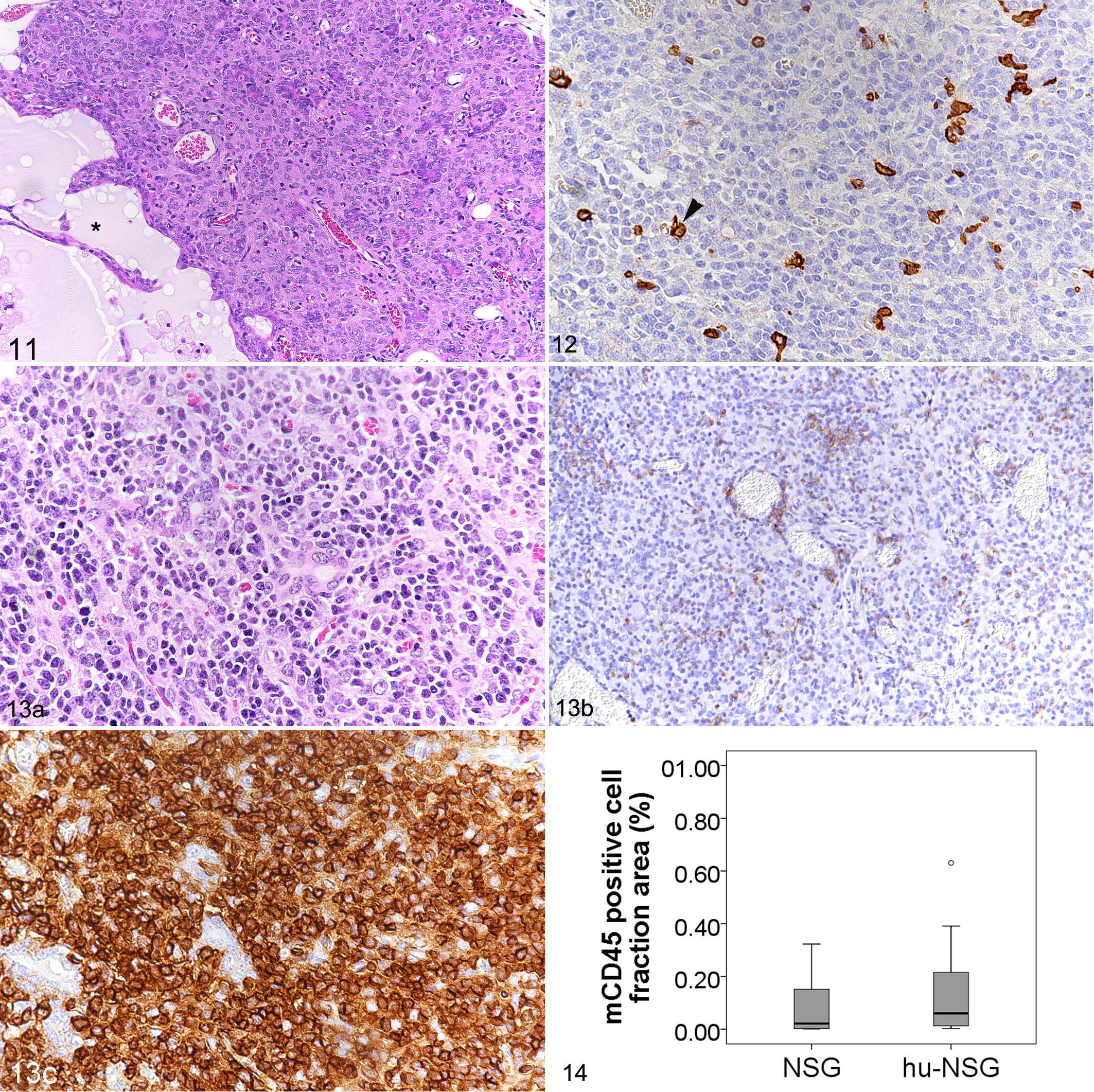
Thymus.
In hu-NSG mice, the thymus generally appeared slightly larger, with an approximate size of 2 × 1 × 1 mm. Lobules were more cellular and exhibited moderate numbers of lymphoid cells (Fig. 13), but still lacked the typical corticomedullary distinction. Murine leukocytes were rare (Fig. 13b): the mCD45-positive cell fraction area had a mean of 0.4% (Fig. 14). Still, mCD45+ cells were sometimes more numerous than in NSG mice, where the area covered by mCD45+ cells did not exceed 0.3% (n = 9; mean ± SD = 0.09 ± 0.1%; Fig. 14). Repopulation with hCD45+ leukocytes was variable in hu-NSG mice. In efficiently humanized mice (Fig. 13c), the mean of the hCD45 positive cell fraction area reached 46.1%, whereas it was 16.5% in inefficiently humanized mice (Table 3). The majority of these cells were CD3+. Of this subpopulation, most cells were CD4+ whereas CD8+ cells were only visible in very small numbers. CD20+ cells were barely present.
One 6-month-old female breeding NSG mouse exhibited a white-tan, firm mass of approximately 0.4 cm in diameter in the thoracic cavity. Histologically, it was composed of dense sheets of large round neoplastic cells which also infiltrated the adjacent adipose tissue (Supplemental Fig. S1). The cells exhibited scant amphophilic cytoplasm and round nuclei with coarsely arranged chromatin. There was moderate anisocytosis and a mitotic rate of 2 to 4/HPF; a moderate number of apoptotic cells was observed (Supplemental Fig. S2a). The vast majority of cells were found to express CD3 and CD4 (Supplemental Fig. S2b, c), whereas B cells (CD45R+) were seen in low numbers and randomly distributed. This led to the diagnosis of a precursor T cell lymphoblastic lymphoma. 38 It was not possible to determine whether the neoplasia originated from the thymus or the mediastinal lymph nodes. The mouse did not exhibit any additional histopathological changes.
Bone Marrow
In NSG mice, the bone marrow (sternal and femoral) generally exhibited a high cellularity, represented by erythroid, mCD45+ myeloid, and megakaryocytic hematopoietic precursors (Fig. 15). Mild to moderate hemosiderosis as well as fatty infiltration (femur only) was occasionally seen (Table 2).
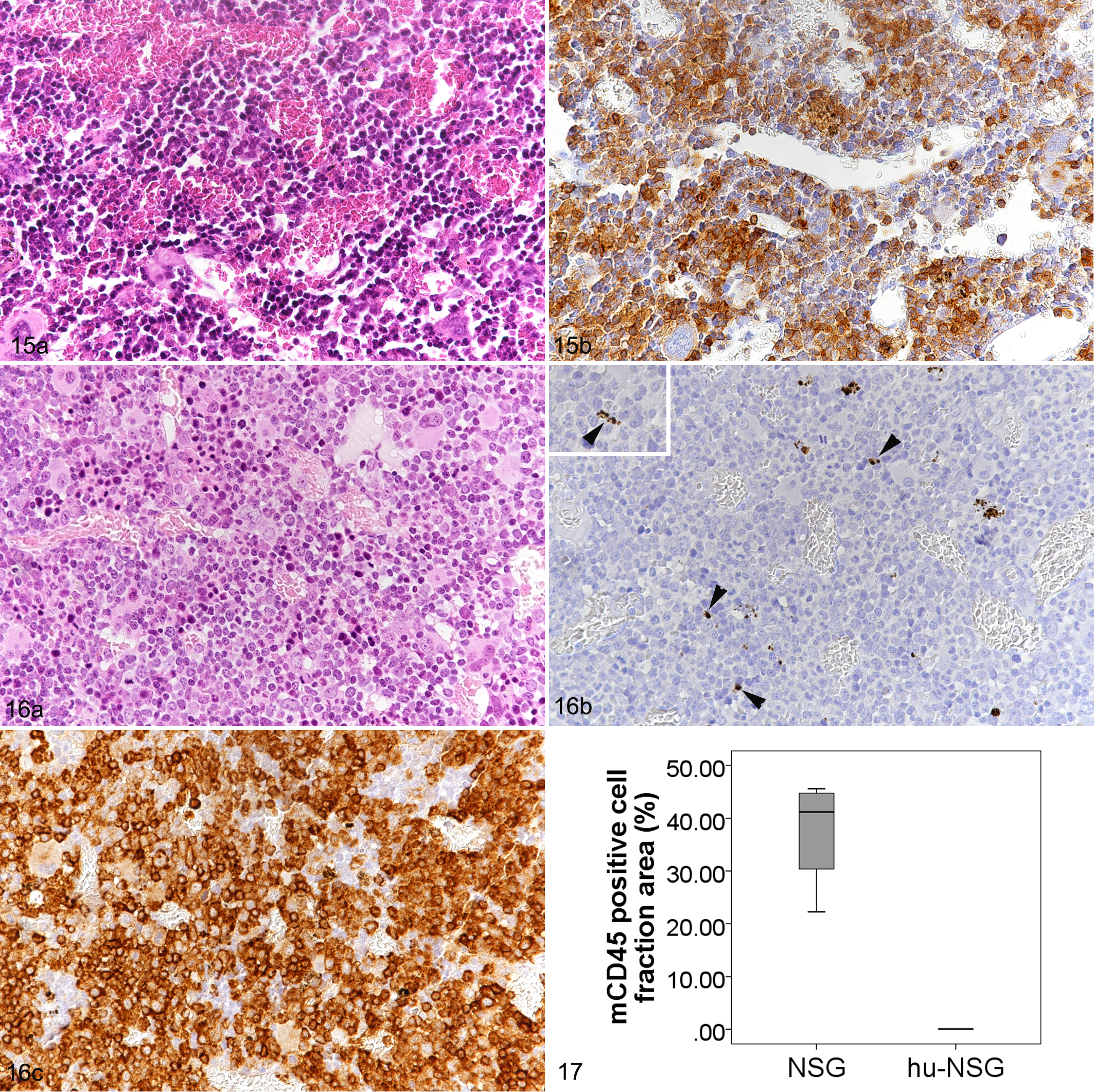
Femoral bone marrow.
In hu-NSG mice, the morphology of the bone marrow was similar, with high to very high cellularity (Fig. 16). While mCD45+ cells were very rare, making up a significantly lower proportion of area than in the NSG mice (Fig. 16b, 17), hCD45+ cells were the dominant cell population (Figs. 16c). The number of human cells (Ku80+) was significantly higher in efficiently humanized vs inefficiently humanized mice (Table 4).
Granulomatous Inflammation in CD34+ hu-NSG Mice
Two male and 2 female hu-NSG mice developed poor health before study completion and were electively euthanized, the females at 28 weeks after HSC transplantation due to severe progressive weight loss, and the males at 30 weeks. Both males were thin and exhibited clinical signs, that is, hunched posture, rough coat, and reddened skin. All 4 mice exhibited grossly enlarged lymph nodes and spleen, diffusely pale bone marrow, and a red and white mottled liver. Histological examination revealed extensive granulomatous inflammatory infiltrates in these tissues as well as in the kidneys (Figs. 18–24). There was no evidence of bacterial or fungal organisms within the inflammatory infiltrates, as confirmed by various special stains (Gram, Giemsa, periodic acid–Schiff, Grocott, and Ziehl-Neelsen). 11,53 Granulomatous infiltrates were represented by multifocal to coalescing, partly nodular aggregates of macrophages and epithelioid cells, admixed with numerous lymphocytes and multinucleated giant cells (Figs. 18–24). In the infiltrates, cells were often found to proliferate, which was reflected by the presence of 1 to 3 mitotic figures per high power field. The vast majority of cells were human leukocytes (Ku80+ [Fig. 19b], hCD45+), with only very few intermingled murine (mCD45+) leukocytes. Macrophages and T cells (CD3+) dominated in the infiltrates which also contained a variable portion of B cells and a few viable neutrophils (Fig. 19c, d). Combined immunohistochemistry for CD4 and CD8 antigens revealed a striking predominance of CD4+ T cells, with only rare CD8 or double-positive cells. In the liver, lesions consisted of lymphohistiocytic or granulomatous infiltrates that were mainly located in periportal areas (Figs. 18, 19a) and were associated with minimal to moderate deposition of fibrous connective tissue as well as scattered hemosiderin-laden macrophages. Bile ducts were often embedded within the inflammatory reaction, but were neither altered nor infiltrated, suggesting that they were not targeted by the inflammatory cells at this stage. Bone marrow changes presented as scattered nodules randomly distributed in the hematopoietic tissue (Fig. 20a) to subtotal replacement of the hematopoietic cell component by, in the majority, MHC II positive cells (Fig. 20b), with concurrent bone marrow necrosis and hemorrhage as well as trabecular bone resorption (data not shown). Affected spleens and lymph nodes (Figs. 21–24) showed scattered focal infiltrates or severe disruption of the architecture by the inflammatory infiltrates. When affected, kidneys exhibited only mild changes, represented by scattered granulomatous foci randomly distributed throughout the cortical interstitium. In individual cases, pancreas, ovaries, serosae, Harderian glands, nose, and/or oral cavity also showed granulomatous infiltrates.
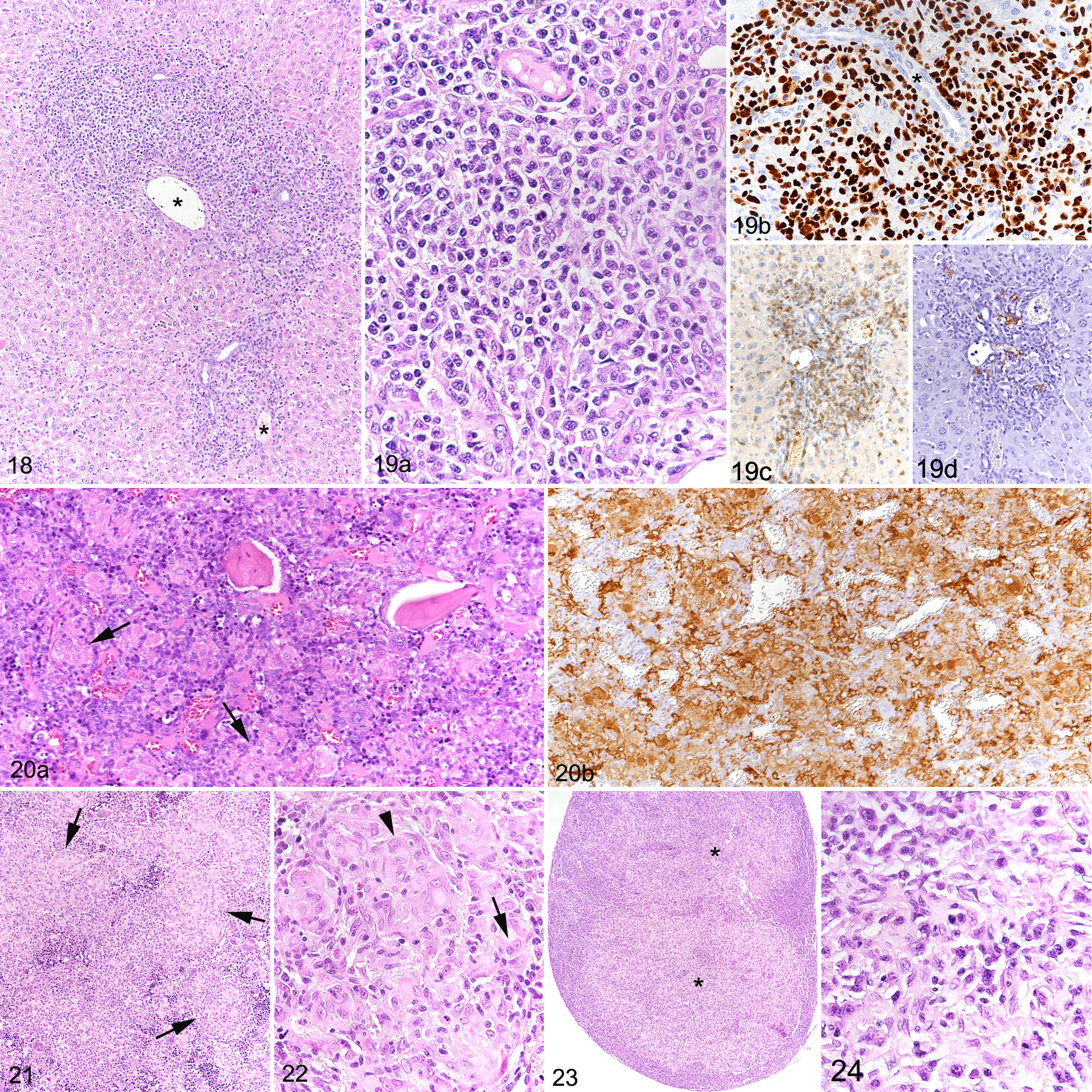
Granulomatous inflammation, hu-NSG mouse.
Some animals with the above-described lesions also exhibited inflammatory infiltrates in lungs and skin. In the lungs, these represented peribronchiolar and perivascular aggregates of Ku80-positive cells, which were predominantly T cells and B cells, with fewer macrophages, occasionally containing golden brown pigment, and no evident giant cells. Infiltrates occasionally extended into the bronchiolar and alveolar lumina. In the skin, minimal or mild infiltration of Ku80-positive, predominantly CD4+ and fewer CD8+ T cells and rare B cells was seen at the dermo-epidermal junction, predominantly along the follicular dermo-epidermal junction, associated with vacuolar change and occasional apoptosis in the basal follicular keratinocytes and, to a lesser extent, the basal epidermal keratinocytes.
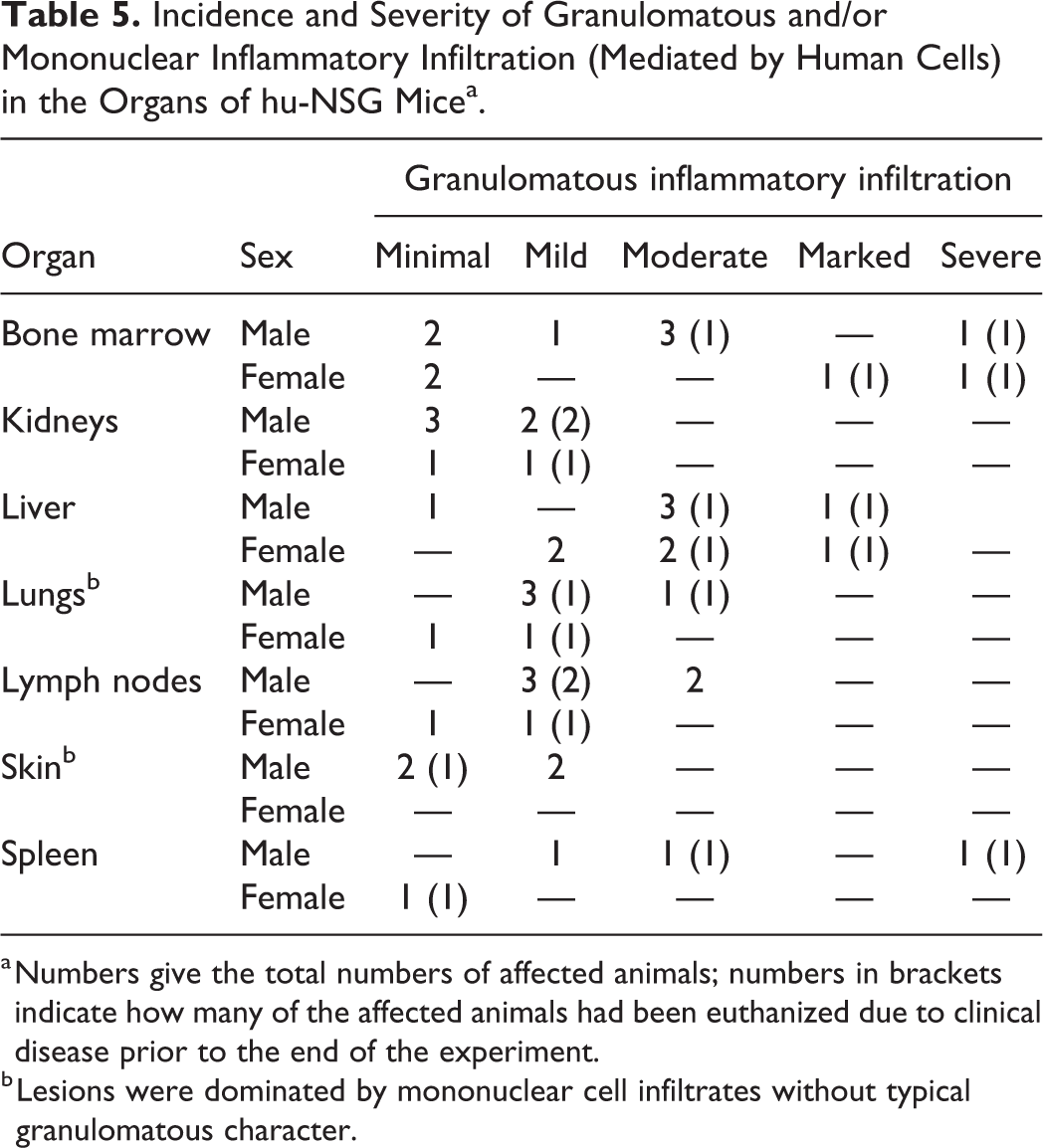
Granulomatous inflammatory infiltrates were also observed in mice without clinical signs, with lower severity and higher incidence in males (21%; 6/29) than in females (11%; 3/28). Liver, spleen, lymph nodes, bone marrow at different locations (sternum, femur and tibia, vertebrae and frontal bones in the skull) and kidneys were most consistently affected (Table 5). Prevalence and severity of the inflammatory infiltrates was comparable across all engrafted mice, with no obvious differences between the reconstitutions from different donors; however, this could not be specifically addressed due to the low number of mice reconstituted with the same donor.
Incidence and Severity of Granulomatous and/or Mononuclear Inflammatory Infiltration (Mediated by Human Cells) in the Organs of hu-NSG Micea.
a Numbers give the total numbers of affected animals; numbers in brackets indicate how many of the affected animals had been euthanized due to clinical disease prior to the end of the experiment.
b Lesions were dominated by mononuclear cell infiltrates without typical granulomatous character.
Comparison of Blood and Lymphoid Tissues
The FACS analysis performed on the peripheral blood of the hu-NSG mice revealed a large variability in the proportion of human leukocytes (hCD45+), ranging from 0.14% to 66.5% (Figs. 25, 26). As mentioned above, mice with more than 15% hCD45+ cells in the blood were considered as efficiently humanized (Figs. 25, 27), and below this threshold as inefficiently humanized (Figs. 26, 28). Immunohistochemistry against hCD45 was performed on the lymphoid organs of a random selection of efficiently and inefficiently humanized mice (each n = 8) to correlate with the FACS data. As expected, this grouping of mice matched to some extent with the degree of repopulation of the hemolymphatic organs, as overall the percentage of human cells was significantly higher in lymph nodes, thymus, and bone marrow of the efficiently compared to the inefficiently humanized mice (Table 4). Interestingly, there was no substantial difference in the spleen. This may be due to the overall low relative extent of repopulation of this organ, which still contained a significant proportion of murine hematopoietic cells; even in efficiently humanized mice, the mean percentage area covered by human leukocytes (hCD45+) was 21.0%, whereas it was 46.1% and 54.1% in thymus and lymph nodes respectively (Table 3).
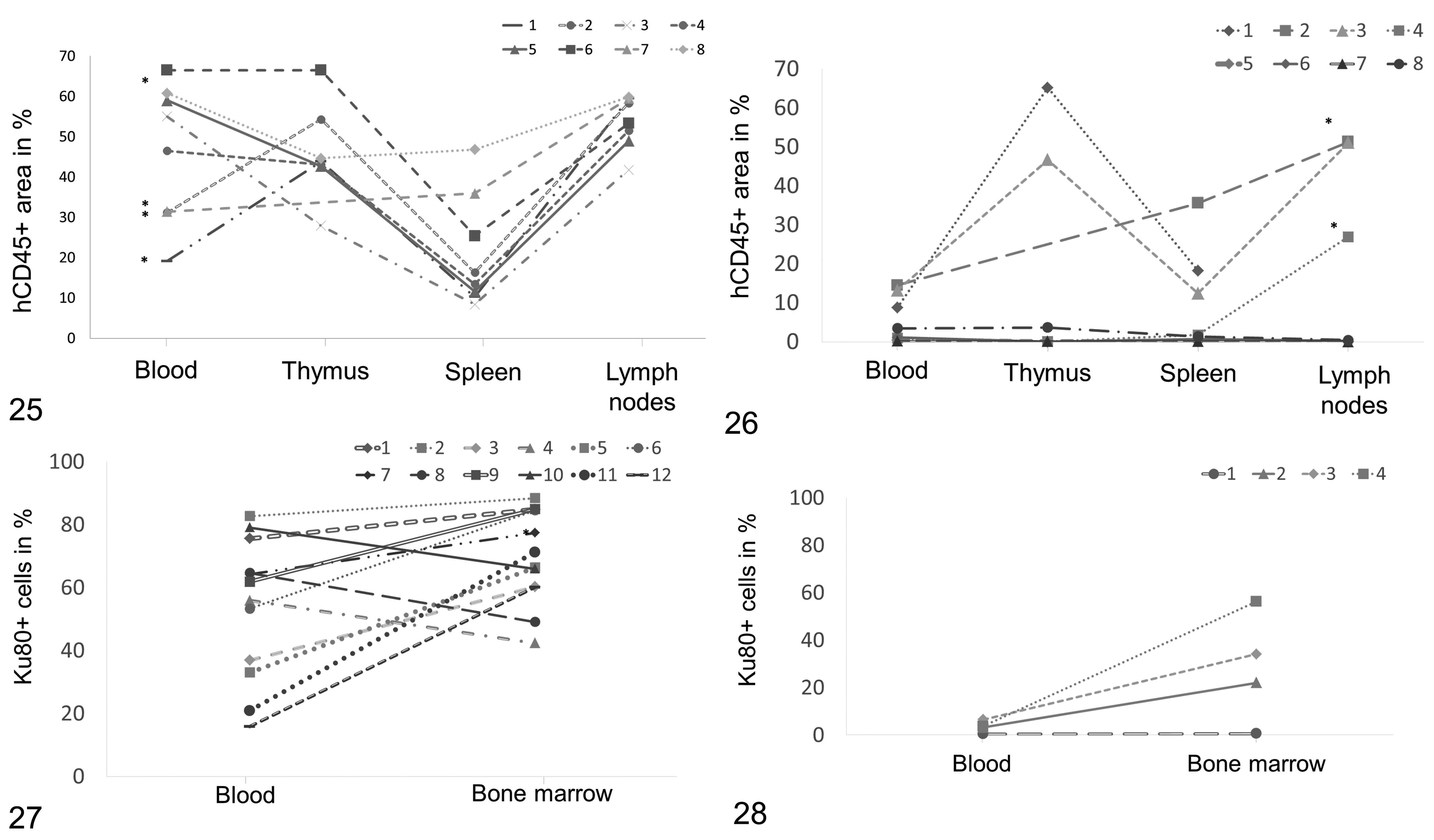
Comparison of efficiently humanized mice (hu-NSG(e), Figs. 25, 27) and inefficiently humanized mice (hu-NSG(i), Figs. 26, 28).
However, looking at repopulation levels in the different organs of individual animals it became obvious that there was substantial variation. The bone marrow was specifically analyzed because it can be expected to be most relevant for the production of cells that later appear in the blood stream. However, a mouse classified as inefficiently humanized could have a proportion of human cells in the bone marrow that was as high as that seen in efficiently humanized mice (Figs. 26, 28).
The FACS results of animals with evidence of granulomatous inflammation (n = 13) were compared with those of mice without such lesions (n = 48). Interestingly, animals with granulomatous inflammation were found to have significantly lower proportions of human leukocytes (Table 6). In the latter, the human leukocyte population contained significantly lower total lymphocyte proportions and B cells, but significantly higher proportions of T cells and monocytes. There were also significantly more activated T cells, activated T helper cells and activated cytotoxic T cells (Table 6; correlation coefficient of 0.630 for CD4DR+ and of 0.661 for CD8DR+ cells, Spearman’s correlation analysis).
Cellular Composition of Peripheral Blood in Animals With and Without Granulomatous Inflammation (GI)*.
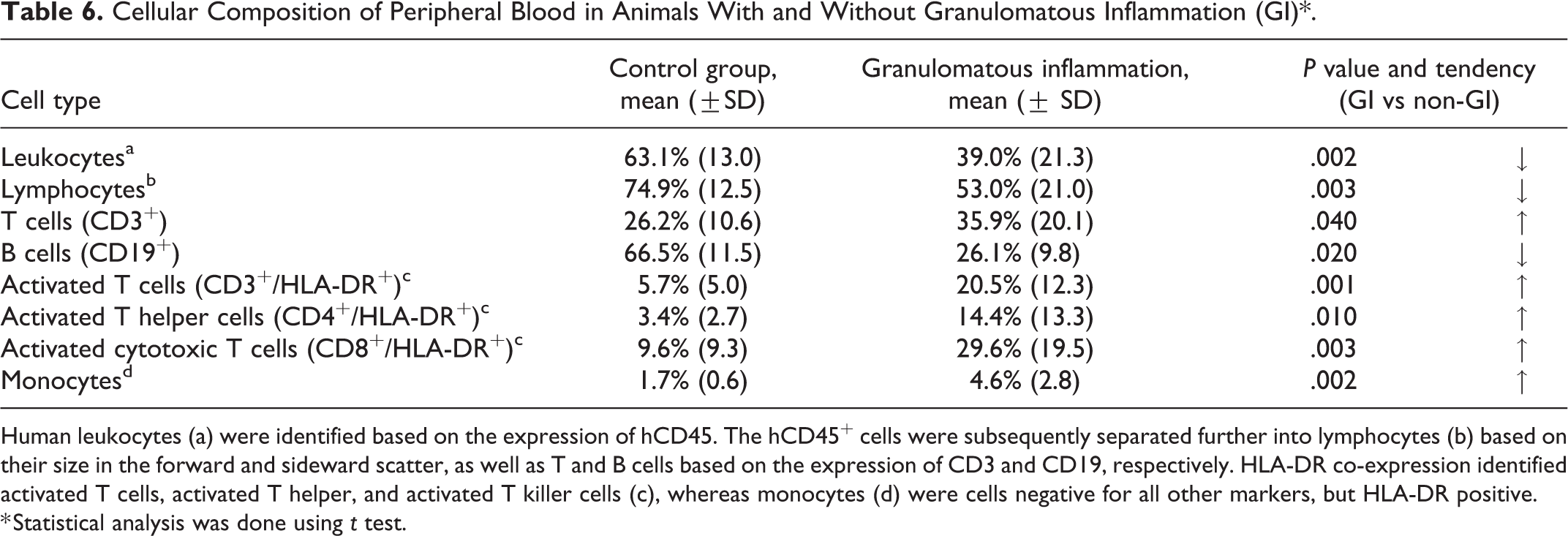
Human leukocytes (a) were identified based on the expression of hCD45. The hCD45+ cells were subsequently separated further into lymphocytes (b) based on their size in the forward and sideward scatter, as well as T and B cells based on the expression of CD3 and CD19, respectively. HLA-DR co-expression identified activated T cells, activated T helper, and activated T killer cells (c), whereas monocytes (d) were cells negative for all other markers, but HLA-DR positive.
* Statistical analysis was done using t test.
Discussion
Data illustrating the spectrum of spontaneous and experimentally induced diseases in humanized mice are very limited, and the situation is similar for the recipient NSG immunodeficient mice. Besides, detailed characterization of the morphological aspects of repopulation at tissue level is highly desirable to allow accurate correlation with the engraftment features obtained via FACS analysis. This study aimed to characterize the morphological phenotype of NSG mice and their humanized counterpart, the CD34+ hu-NSG murine model, and to gather data on the most common background lesions in both types of mice. In addition, a comparative quantitative assessment was undertaken in the attempt to relate levels of human repopulation in the hemolymphatic organs of hu-NSG mice to FACS results obtained from the peripheral blood, as the latter is commonly used as a tool to determine the extent of humanization.
Full histological examination of a cohort of 48 NSG mice aged 3 to 6 months revealed a limited spectrum of background lesions, occurring with low severity. Salient changes were observed in lungs, kidneys, testes, and adrenal glands and were all among those commonly encountered in other strains. 13,17,44 In addition, there was a very low prevalence of incidental inflammatory changes, without evidence of infectious conditions. This can likely be ascribed to housing of the mice under specific pathogen free conditions as in general NSG mice are prone to infections because their immune system is compromised. 16
Since the present study focused on young animals, it cannot provide data on potential spontaneous neoplastic processes that might develop with age. 47 Interestingly though, lymphoma was found in the thoracic cavity of a 6-month-old non-irradiated female NSG mouse, which could have originated from the thymus or the mediastinal lymph nodes. The tumor was predominantly composed of CD3+, CD4+ T cells and further characterized as a T cell lymphoblastic lymphoma. Hematopoietic neoplasms are reported to occur with low prevalence in NSG mice, in subjects older than 1 year, 16,47 due to the inactivation of IL-2Rγ, which plays an important role in the signaling pathways for B and T cell growth. 26 In the present case, it could be a spontaneous event that might have compensated for IL-2Rγ deficiency, and led to full manifestation of the NOD-scid background phenotype, which has been reported to frequently develop lymphoma. 41 Lymphoma, especially thymic lymphoma might also occur secondarily to irradiation, which is used to deplete mouse hematopoetic cells prior to human cell engraftment, or insertional mutagenesis resulting from gene editing therapeutic products. Hence, it is crucial to report on the occurrence of such tumors in untreated NSG mice at an age when they are used in transplantation experiments, to build an accurate historical database.
All NSG mice in the present study showed poorly developed lymphoid organs, lacking the typical lymphoid tissue architecture. This has previously been reported as a feature in the lymphoid organs of several immunodeficient murine lines, including NSG mice, whose genetic manipulation leads to severe impairment of immune functions. 7,47,49,52 However, we observed small or moderately sized periarteriolar clusters of mCD45+ cells in the spleen of numerous NSG mice, a feature that has to our knowledge not been reported yet. Cells in these clusters did not express more specific lymphocyte or macrophage markers, so further work is needed to assess their exact nature and whether they represent early lymphoid precursors, dendritic or myeloid subsets. According to the experience of the authors, these aggregates can complicate the histologic interpretation of NSG mice engrafted with human hematopoietic cells or administered cell-based therapy products, which might have morphological features similar to these native cells in HE-stained sections. Another interesting observation was that, like the bone marrow in scid and other immunodeficient mouse lines, the bone marrow of NSG mice did not show major histological abnormalities in the hematopoetic cell component. 14
The comprehensive histological screening of hu-NSG mice, that is, NSG mice engrafted with human CD34+ HSC, revealed a spectrum of spontaneous lesions similar to that observed in naïve NSG mice. However, the mice additionally exhibited changes that can be ascribed to either the preconditioning irradiation and/or the engraftment of human hematopoietic cells.
Among the lesions potentially related to perinatal total body irradiation, to which the scid mutation of NSG mice renders them particularly sensitive, 5 were nephropathy, ovarian atrophy, and ocular changes. All three are well-known complications of radiation exposure in humans. 3,12 Radiation nephropathy has also been described in different preclinical species including mice. 12 In both humans and laboratory animals it represents a late-onset, adverse event following external beam irradiation or radionuclide therapy, characterized by concurrent injury to glomerular and tubular compartments. 12,40 In the present study it was observed with low frequency and severity, which suggests an early stage of the disease in the still-young hu-NSG mice. At this early stage, radiation-induced lesions can be hard to differentiate from tubular basophilia and the early changes that can occur in the context of chronic progressive nephropathy, a common degenerative spontaneous disease in the kidneys of mice and rats. 17
So far, both ovarian atrophy and retinal changes have only been sporadically reported as radiation-induced alterations in animal models, and the present report is the first of such lesions in hu-NSG mice undergoing perinatal preconditioning. Interestingly, both types of changes occurred with high prevalence and severity, indicating a high sensitivity of ovaries and retina of these mice to irradiation. It is known that the germline cells in the ovary are highly susceptible to radiation damage and that accelerated reproductive aging can occur when the follicle pool, which is finite and nonrenewable, is damaged due to irradiation. 1 From the present findings it could be speculated that radiation-induced damage of the female germ cells at an early prepubertal stage might cause loss of ovarian reserve, which then manifests as atrophy of the gonads and lack of cyclical changes in uterus and vagina. Interestingly, there were no substantial microscopic changes in the testes of hu-NSG mice, suggesting that the irradiation procedure did not affect the male germline. This difference might be due to different radiation exposure or reduced sensitivity of the male gonadal tissues in the postnatal period, as previously reported. 15
Radiation retinopathy in humans encompasses a number of retinal changes following exposure to radiation from any source, typically external beam radiation exposure in patients affected by tumors of the nasopharynx, sinus, or orbita. 3 In animal models, complications of ocular radiation have been reported as rare, most frequently manifesting as cataracts, which also occurred in this study with low prevalence and low severity. 43 The retinal lesions observed in our mouse cohort suggest a perinatal arrest of the retinal development, allowing only the neuroblastic layer and the ganglion cell layer to be identified. 59 To our knowledge, these retinal lesions have not been previously reported in mice that have undergone total body irradiation. Extensive photoreceptor loss and impaired retinal development in the hu-NSG mice might therefore be linked to radiation exposure in the perinatal period, when cell proliferation is still ongoing in the retina as part of the postnatal development process. Nonetheless, a more thorough investigation of the retinal changes was beyond the scope of the present study, as it would require dedicated long-term studies with multiple endpoints and multiple radiation quantities. Notably though, researchers should be aware of potential impaired vision resulting from the retinal lesions. Impaired retinal development affects predominantly the outer layers, hence resembling outer retinal atrophy, from which it needs to be differentiated. The latter is characterized by loss of photoreceptors and is caused by a number of factors, including aging and phototoxicity. 42
The changes that can be ascribed to the engraftment of human hematopoietic cells are represented by granulomatous inflammatory infiltrates of almost exclusively human origin that were found in multiple organs of around 20% of hu-NSG mice. The absence of microorganisms and foreign material and the dominance of macrophages and CD4+ T cells suggest either an uncontrolled proliferation of activated human immune cells and/or an underlying human-anti-mouse immunopathological process. Granulomatous infiltrates containing hemosiderin-laden macrophages are reminiscent of post-transplant hemophagocytic lymphohistiocytosis (HLH), an hyperinflammatory condition driven by hypercytokinemia and triggered, for example, by transplantation of HSC. 46 The disease has been reproduced in perforin-deficient mice infected with lymphocytic choriomeningitis virus 27 and in humanized mice following experimental infection with EBV. 48 More recently, it has also been described in NSG-SGM3 mice without human thymus, engrafted with human CD34+ fetal liver cells. 63 The infiltrates in these mice were similar to the lymphohistiocytic infiltrates observed in hu-NSG mice in our study and were not present in NSG mice. However, in general, the inflammatory infiltrates in CD34+ HSC humanized NSG mice as in our study exhibit only occasional hemosiderin-laden macrophages and, in the lungs, have a predominantly lymphocytic rather than histiocytic/granulomatous character. In the liver, inflammatory foci were predominantly present in portal areas, but seemed not to target the biliary ducts; the latter is a striking feature of GvHD in humans. To date, a number of studies have reported multiorgan infiltrates of lymphocytes and macrophages, occasionally with epithelioid macrophages and giant cells, as a manifestation of GvHD in humanized mice, 11,18,28,34,53 and report GvHD as a major cause of morbidity in studies employing humanized mice. 21,28 Considering that Yoshihara and coauthors consider lymphohistiocytic infiltrates with a prominent hemophagocytic component as indicative of HLH rather than GvHD it is also possible that both conditions manifest concurrently in the same model. This seems likely in our murine model, where the inflammatory processes observed in the skin of a few mice, that is, an interface dermatitis with vacuolation and apoptosis of keratinocytes in the basal layer of hair follicles, closely resemble the typical manifestation of chronic GvHD in human patients, while the multisystemic granulomatous infiltration would rather suggest an uncontrolled proliferation of human macrophages driven by an excess in cytokines, as in HLH. 27,46 In the absence of additional evidence pointing toward one entity or the other, we feel that other methodological approaches, such as T cell receptor repertoire analysis and quantification of plasma and tissue cytokine levels are needed to provide more insight into the exact nature and pathogenesis of these inflammatory lesions. 11,21,51
Since mice affected by granulomatous inflammation do not reach the experimental end-point they are often excluded from the analysis. Thus, the frequency of the respective pathological changes, whose underlying cause is ascribed to GvHD or HLH, is rarely reported. 3 In the present study, 13 mice showed weight reduction or clinical signs and all but one of them exhibited granulomatous inflammation, with more males found to be affected (21%) than females (11%). These data indicate that monitoring of weight loss and clinical signs can identify the majority of mice with these specific pathological changes. Still, some mice with only few and mild lesions that can only be detected by histological examination may not be identified based on clinical signs, yet these may show an aberrant immunological response of the engrafted human cells in different types of experiments. It is important to highlight though that the prevalence and features of granulomatous inflammation reported here might solely apply to the model employed in our study, as there is growing evidence that the incidence and severity of inflammatory lesions varies extensively across experiments depending on the source, number, and manipulation of human donor cells employed and the features of the recipient host system (eg, strain susceptibility, environmental features, and preconditioning protocols involving irradiation or chemotherapy). 20,36
In the present study, the in situ detection of human and murine cells in the hemolymphatic tissues provided information on the extent and distribution of homing of graft and host leukocytes after humanization with CD34+ HSC. We observed that the mouse component (mCD45+ cells) generally decreased with humanization of the animal, even though the difference was not statistically significant. This phenomenon cannot be explained without further studies employing larger numbers of animals. It could simply be a consequence of irradiation; alternatively, it is possible that the introduction of human cells leads to a reduced proliferation of the murine leukocytes.
Both FACS data and the image analysis of tissue sections of hemolymphatic organs suggested that the extent of hCD45+ repopulation was highly variable among the engrafted animals. The reason for this is not clear, but pretreatment, injection route, and source of HSCs will inevitably have an impact on the inter- and intra-experimental variability of engraftment. This makes comparisons across different models difficult, even when experimental conditions vary only slightly. Human cells showed a variable distribution in the different organs. In the spleen, hCD45+ cells formed aggregates reminiscent of the white pulp of immunocompetent mice, despite the absence of discernible PALS and lymphoid follicle organization. The thymus of hu-NSG mice was variably repopulated by human T cells, providing further evidence of some functional homing of the lymphocytes; however, the typical architecture was not observed. In the lymph nodes, however, there was no evidence of a cell type-specific arrangement. It has previously been suggested that the genetic background of the mice is responsible for the lack of the typical architecture in the lymphoid organs, as γc deficiency compromises IL-7-dependent lymphoid tissue inducer cell development. 33 The architecture is crucial for the proper functionality of the immune system, 39 and it has been suggested that the poorly organized lymphoid organs are a limiting factor in the humoral immune response of humanized mice. 61 Repopulation of the bone marrow seems to be less complicated; in bone marrows free of inflammatory lesions, all cell types appeared to home effectively.
The present study also investigated the correlation between the pattern and extent of reconstitution in the lymphoid organs by human leukocytes and the results of the FACS analysis on peripheral blood. The FACS analysis revealed a high variability in reconstitution between different animals, and similarly the actual distribution of hCD45+ in the lymphoid organs of an individual can be quite variable. Upon pairing the obtained FACS data of the reconstitution 3 to 6 months after human HSC injection with the morphological analysis of the lymphoid organs it became evident that a high reconstitution based on analysis of blood does not necessarily correlate with a high repopulation at the tissue level. In the efficiently humanized mice there was no significant correlation between the level of circulating hCD45+ cells and the number of hCD45+ cells in the organs, whereas a significant correlation was seen in the inefficiently humanized animals. This significance may be due to the low hCD45+ cell levels in all the compartments and thus a lower variability.
In the animals affected by granulomatous infiltrates the proportion of human leukocytes and lymphocytes in the blood were significantly lower compared to mice that did not exhibit evidence of such inflammatory changes. This might result from migration of human leukocytes from the blood stream to the sites of injury, similarly to what occurs in infections and sepsis, 55 and by compromised hematopoietic function due to granulomatous lesions in the bone marrow, as was visible in the histological evaluation of some of the animals. However, compared to the cohort not affected by granulomatous inflammation, those affected by this process had a significantly higher proportion of T cells in the blood including higher numbers of activated T cells, activated T helper cells, and activated cytotoxic T cells. The increased numbers of T cells fit with the central role of effector graft donor T cells in driving these posttransplant pathological processes. 10,36 Moreover, the granulomatous lesions in the bone marrow of affected animals were dominated by MHC II-positive cells, and also activated T cells are known to express this surface antigen. 30 This hypothesis is supported by a study that shows an increase in the proportion and extent of fluctuation of CD3+CD4+CD8β+ T cells. 10
Identification of an increase in T cells, primarily activated helper T cells in the FACS data might help spot individual mice affected by subclinical inflammatory lesions, which could then be confirmed via the histological examination of typically affected organs such as the bone marrow, liver, and kidneys.
Supplemental Material
Supplemental Material, Combined_supplemental_materials-Blumich_et_al - Human CD34+ Hematopoietic Stem Cell–Engrafted NSG Mice: Morphological and Immunophenotypic Features
Supplemental Material, Combined_supplemental_materials-Blumich_et_al for Human CD34+ Hematopoietic Stem Cell–Engrafted NSG Mice: Morphological and Immunophenotypic Features by Sandra Blümich, Hana Zdimerova, Christian Münz, Anja Kipar and Giovanni Pellegrini in Veterinary Pathology
Footnotes
Acknowledgements
The authors wish to thank the technical staff of the Histology Laboratory, Laboratory for Animal Model Pathology, Institute of Veterinary Pathology, Vetsuisse Faculty, University of Zurich, for excellent technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CM is supported by Cancer Research Switzerland (KFS-4091-02-2017), KFSP-PrecisionMS of the University of Zurich, the Vontobel Foundation, the Baugarten Foundation, the Sobek Foundation, the Swiss Vaccine Research Institute, Roche, ReiThera, and the Swiss National Science Foundation (310030B_182827 and CRSII5_180323).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
