Abstract
ΔNp63 is an isoform of p63 that plays an essential role in the development and growth of some epithelial tissues. In this study, we investigated the expression of ΔNp63 in normal and neoplastic tissues of cats and compared the results with the expression of pan-p63. Immunohistochemistry for ΔNp63 and pan-p63 was performed in normal tissues from 2 adult cats and in 10 cases each of 22 different types of feline neoplasms. In normal tissues, there was nuclear ΔNp63 immunolabeling in basal cells of stratified squamous, transitional, and pseudostratified columnar epithelia; basal cells of sebaceous glands; trophoblasts; and myoepithelial cells. Of the neoplasms, 10/10 apocrine ductal adenomas, 10/10 mammary ductal carcinomas, 10/10 pulmonary adenosquamous carcinomas, 10/10 squamous cell carcinomas, 10/10 trichoblastomas, and 10/10 urothelial carcinomas immunolabeled for ΔNp63. The ΔNp63 immunolabeling was diffuse in almost all neoplastic cells with squamous, basal, and urothelial origins. In the neoplasms with ductal differentiation, only the neoplastic suprabasal myoepithelial cells immunolabeled. Application of pan-p63 to the same set of neoplasms revealed positivity not only in the same neoplasms, but also in several unexpected tumor types (3/10 exocrine pancreatic carcinomas, 3/10 fibrosarcomas, 3/10 pulmonary adenocarcinomas, 2/10 lymphomas, 1/10 cholangiocarcinomas, 1/10 hemangiosarcomas, 1/10 mast cell tumors, and 1/10 meningiomas). Both ΔNp63 and pan-p63 antibodies demonstrated 100% diagnostic sensitivity and negative predictive value for diagnosing feline neoplasms with squamous, basal, and urothelial epithelia or myoepithelial cells. However, ΔNp63 showed higher diagnostic specificity (100% vs. 90.6%), positive predictive value (100% vs. 80%), and overall accuracy (100% vs. 93.1%) compared with pan-p63.
Keywords
Identified in the late 1990s, p63 belongs to the p53 family of transcription factors and plays a crucial role in the development and growth of some epithelial tissues.9,12,48 The TP63 gene encodes different isoforms using 2 promoters and alternative C-terminus splicing events. 29 TAp63 and ΔNp63 are the main isoforms and exhibit intricate putative functions with diverse transactivation (TA) capacities.28,29 The TAp63 isoform contains an N-terminal TA domain with homology to p53, which regulates the expression of growth-inhibitory genes. 7 Conversely, the ΔNp63 isoform lacks the TA domain and opposes the activity of TAp63 and p53. 29 Immunohistochemical expression of pan-p63 in postnatal human tissues is predominantly in the nuclei of basal cell layers of stratified squamous and transitional epithelia, basal cells of certain glandular epithelia, myoepithelial cells, cytotrophoblasts, and thymic epithelial cells. 7 ΔNp63 is the predominant isoform in these cells and serves to maintain the self-renewing capacity of the progenitor cells.22,29,43 Limited studies in domestic animals have demonstrated similar nuclear ΔNp63 expression in normal tissue, including cutaneous epidermal and adnexal basal cells in the skin of cats, myoepithelial cells in the mammary glands of dogs, and bronchial basal cells in the lung of cattle and cats.6,41,46,52
Pan-p63 expression in specific tissues has been extensively researched for diagnosing human neoplasms but often reveals conflicting data regarding positive neoplasms. 45 In the past decade, antibodies targeting only the ΔN of ΔNp63 (also called p40) emerged as powerful immunohistochemical markers.44,47 Human neoplasms consistently positive for ΔNp63 include squamous cell carcinomas (SCCs) of any site, pulmonary adenosquamous carcinomas, and urothelial carcinomas.1,7,48 This potential diagnostic marker is particularly useful in pulmonary carcinomas, where ΔNp63 has greater specificity than other squamous markers. 7 Despite its significance in human pathology, ΔNp63 remains practically unknown in routine veterinary pathology and has only been tested in certain feline cutaneous carcinomas, feline and bovine pulmonary carcinomas, canine mammary tumors, and a salivary neoplasm in a wild rodent.6,41,46,50,52 Comprehensive studies evaluating the applicability of ΔNp63 as a diagnostic marker for neoplasms in domestic animals are currently lacking. Therefore, this study aimed to (1) investigate the expression of ΔNp63 in normal and neoplastic tissues of cats and (2) compare the immunolabeling of ΔNp63 with that of pan-p63 within the same set of neoplasms. We hypothesized that ΔNp63 is highly valuable for diagnosing feline neoplasms with squamous, basal, and urothelial epithelia or myoepithelial cells, and that it demonstrates greater specificity for these neoplasms than pan-p63.
Materials and Methods
Normal Tissues Collection
Two adult cats submitted for postmortem examinations in August and September 2023 to the Department of Veterinary Pathology at the Universidade Federal do Rio Grande do Sul were used to generate a systematic collection of normal tissues. These cats had causes of death unrelated to neoplasms. Collected tissues included adrenal gland, cerebellum, cerebrum, ceruminous gland, esophagus, eye, haired skin, heart, kidney, large intestine, liver, lung, mammary gland, mesenteric lymph node, nasal mucosa, oral mucosa, pancreas, parathyroid gland, salivary gland, skeletal muscle, small intestine, spinal cord, stomach, thyroid gland, trachea, and urinary bladder. In addition, feline uterus with placenta and ovary samples from a biopsy case were also selected. The tissues were fixed in 10% neutral-buffered formalin for 48 hours at room temperature and processed routinely for histology. To evaluate the effect of prolonged fixation on ΔNp63 immunolabeling in normal-haired skin, which is a well-documented positive tissue control for ΔNp63 in cats, 41 additional samples from the same tissue specimen were fixed for 48 hours and 1, 2, 3, 4, and 5 weeks. These normal cutaneous samples were collected from the dorsal trunk.
Neoplasm Selection
The electronic records of the feline biopsies and cats submitted for necropsy between January 2013 and December 2022 to our department were searched for primary neoplastic lesions. During this period, 4251 feline cases (2003 necropsies and 2248 biopsies) were evaluated, and 1840 (43.2%) had a conclusive diagnosis of at least one primary neoplasm (666 from necropsies and 1174 from biopsies) diagnosed histologically. Neoplasms (n = 22) with samples available from at least 10 distinct cases were included in this study. For each neoplastic type, formalin-fixed paraffin-embedded tissue blocks from 10 distinct cases were retrieved, resulting in 220 samples. Samples from more recent years were prioritized, and those with autolysis were avoided. Cats with more than one type of neoplasm in which at least one was metastatic were excluded. While the standard procedure in our laboratory is to trim the tissues within 72 hours after the receipt/collection, the exact fixation time of each sample in 10% neutral-buffered formalin was not recorded.
The selected neoplasms included apocrine ductal adenoma, ceruminous adenoma, cholangiocarcinoma, exocrine pancreatic carcinoma, fibrosarcoma (8 cutaneous/subcutaneous and 2 oral), hemangiosarcoma (9 cutaneous and 1 intestinal), intestinal adenocarcinoma (6 from small intestine and 4 from colon), leukemia, lipoma (all subcutaneous), lymphoma (9 intestinal and 1 nodal), mammary ductal carcinoma, mammary simple carcinoma, mast cell tumor (all cutaneous), melanoma (8 ocular and 2 cutaneous), meningioma, osteosarcoma, pulmonary adenocarcinoma, pulmonary adenosquamous carcinoma, SCC (9 cutaneous and 1 oral), thyroid adenoma, trichoblastoma, and urothelial carcinoma (all from the urinary bladder). Hematoxylin and eosin-stained sections of each selected case were reevaluated by 3 authors to confirm the diagnoses, and a consensus was reached for all cases. Current literature on the classification of tumors in domestic animals was used to standardize the diagnoses;10,14,18,26,27,30,33–35,37,38,41,51,58 the key histologic features for the diagnosis of each neoplasm type are summarized in Supplemental Table S1. Some cases were used in previous studies.5,8,41
Immunohistochemistry Protocol and Evaluation
Immunohistochemistry (IHC) for ΔNp63 and pan-p63 was performed manually on formalin-fixed paraffin-embedded sections of the selected tissue samples. Briefly, the sections were deparaffinized in xylenes and rehydrated in alcohols. Endogenous peroxidase activity was inhibited with Novocastra Peroxidase Block (Leica Microsystems, Illinois) at room temperature for 5 minutes. Pretreatment with heat-induced epitope retrieval was performed using citrate buffer (pH 6.0) in a pressure cooker at 96°C for 40 minutes (for ΔNp63) or in a microwave oven using three 5-minute cycles (for pan-p63). Nonspecific tissue-antibody interactions were blocked with Novocastra Protein Block (Leica Microsystems) at room temperature for 5 minutes. The sections were then incubated with a mouse monoclonal anti-ΔNp63 (also called p40; clone BC28; 1:300 dilution; Biocare Medical, California) and mouse monoclonal anti-pan-p63 (clone A4A; 1:800 dilution; Abcam Limited, Cambridge) antibodies at room temperature overnight. Cross-reactivity of these ΔNp63 and pan-p63 clones with feline tissues was previously demonstrated.41,42 Detection was performed using the NovoLink HRP-Polymer Detection System (Leica Microsystems); the Romulin AEC Chromogen Kit (Biocare Medical; for ΔNp63) or the DAB Chromogen System (DakoCytomation, California; for pan-p63) as the chromogens. The slides were counterstained with Harris hematoxylin. Normal feline skin was used as the external positive control; for the negative control, replicate sections of the positive control tissue were incubated with Polymer Negative Control Serum (Biocare Medical).
The expected nuclear immunolabeling for both antibodies was evaluated based on the intensity, 41 percentage of positive cells, distribution pattern, and type of positive cells in all neoplastic tissues. This evaluation was done simultaneously by 2 authors and discrepancies were resolved by a consensus. The immunolabeling intensity was scored as 0 (no labeling), 1 (weak labeling), 2 (patchy labeling), or 3 (intense labeling), based on the majority labeling intensity. The percentage of positive cells was scored as 0 (<1% of the cells), 1 (1%–15%), 2 (16%–50%), or 3 (>50%). The immunolabeling distribution was classified as 0 (no labeling), 1 (focal, individual immunolabeled cells), 2 (multifocal, pockets of immunolabeled cells), or 3 (uniform, immunolabeled cells present diffusely). Three final mean scores, one for percentage of immunolabeled cells, one for immunolabeling intensity, and one for immunolabeling distribution, for each antibody were calculated for each positive tumor type. The assessment of melanomas was made in areas with little or no intracytoplasmic melanin pigment. The percentage of positive cells and immunolabeling intensity were also assessed in the normal-haired skin that was used to evaluate the effect of prolonged fixation.
Statistical Analysis
We evaluated the diagnostic performance of both ΔNp63 and pan-p63 antibodies in detecting feline neoplasms with squamous, basal, or urothelial epithelia and myoepithelial cells. For that, diagnostic sensitivity, diagnostic specificity, negative predictive value, positive predictive value, and overall accuracy were assessed. The diagnostic sensitivity was determined by the antibody’s capability to label apocrine ductal adenomas, mammary ductal carcinomas, pulmonary adenosquamous carcinomas, SCCs, trichoblastomas, and urothelial carcinomas (n = 60). Histologic evaluation served as the gold standard for diagnosis (Supplemental Table S1). The diagnostic specificity of these antibodies for accurately diagnosing these neoplasms was determined by the absence of immunolabeling in other types of tumors evaluated (n = 160). Cases were considered positive if nuclear immunolabeling was identified in neoplastic cells, regardless of percentage, intensity, or distribution. Ninety-five percent confidence intervals (95% CIs) were calculated using the Wilson score method in OpenEpi, version 3.01 (www.openepi.com). All data analyzed in this study are available upon request from the authors.
Results
ΔNp63 Expression in Normal Tissues
Intense nuclear ΔNp63 immunolabeling was detected only in epithelia and myoepithelial cells. All tested tissues containing stratified squamous epithelium (cornea, esophagus, haired skin, and oral mucosa) and transitional epithelium (urinary bladder, renal pelvis, and nasal mucosa) showed immunolabeling in the basal layer, with a gradual decrease in labeling in the more differentiated layers (Fig. 1a, b). No ΔNp63 expression was observed in the most superficial layers. In the skin, the distribution of positive cells was consistent and independent of epidermal pigmentation. Specific structures of the hair follicles (hair matrix and outer root sheath) and basal germinative epithelial cells of the sebaceous lobules also exhibited immunolabeling (Fig. 1c). Other epithelia with immunolabeling included trophoblasts in the labyrinth zone of the placenta (Fig. 1d) and basal cells of the bronchial tree and tracheal mucosa.
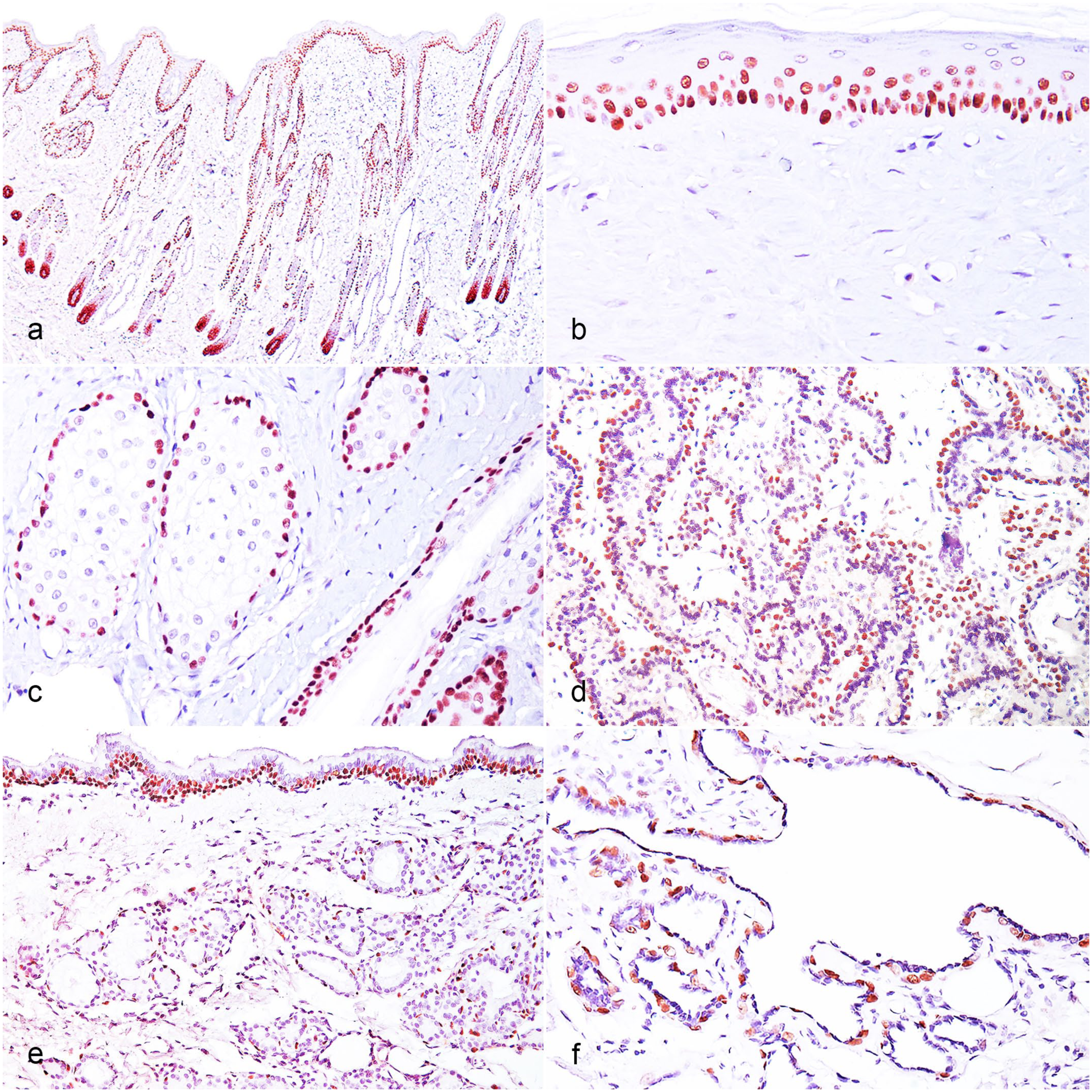
Immunohistochemistry for ΔNp63 in feline normal tissues, cat. (a) Haired skin. Nuclear immunolabeling in the basal layers of the epidermis and hair follicles. (b) Haired skin. Nuclear immunolabeling in the cells of the basal layer of the epidermis, whereas the most superficial layers are negative. (c) Haired skin. Nuclear immunolabeling in the basal germinative epithelial cells of the sebaceous lobules and basal layer of the hair follicle. (d) Placenta. Nuclear immunolabeling in the trophoblasts of the labyrinth zone. (e) Trachea. Nuclear immunolabeling in the basal cells of the mucosa and myoepithelial cells around acini and ducts of the tracheobronchial submucosal glands. (f) Mammary gland. Nuclear immunolabeling in myoepithelial cells around secretory and ductal cells.
Myoepithelial cells around acini and ducts of salivary glands and tracheobronchial submucosal glands (Fig. 1e) immunolabeled for ΔNp63. Myoepithelial cells on the basement membrane of the secretory and ductal cells of the apocrine, mammary (Fig. 1f), and ceruminous glands also had immunolabeling. All other feline tissues subjected to IHC for ΔNp63 were negative. Nonspecific immunolabeling and background were absent. When investigating the effects of prolonged fixation, there was no reduction in the intensity and number of positive cells in normal-haired skin fixed in 10% neutral-buffered formalin for up to 5 weeks compared with tissues fixed for 48 hours.
ΔNp63 Expression in Feline Neoplasms
Nuclear immunolabeling for ΔNp63 was observed in neoplastic cells of 60 of the 220 cases, specifically in 10/10 mammary ductal carcinomas, 10/10 SCCs, 10/10 pulmonary adenosquamous carcinomas, 10/10 trichoblastomas, 10/10 urothelial carcinomas, and 10/10 apocrine ductal adenomas (Table 1). All neoplastic cells in the SCCs immunolabeled (Fig. 2a) except for areas with evidence of keratinocyte maturation and formation of keratin pearls in the center of certain trabeculae. Only the nests formed by the squamous epithelial component of the pulmonary adenosquamous carcinomas were positive. Almost all neoplastic cells of the trichoblastomas (Fig. 2b) and the urothelial carcinomas (Fig. 2c) immunolabeled. In addition, there was ΔNp63 immunolabeling in the neoplastic suprabasal myoepithelial cells located on the basement membrane of the ducts in the apocrine ductal adenomas (Fig. 2d) and mammary ductal carcinomas (Fig. 2e). The neoplastic ductal epithelia of these neoplasms were negative. The immunolabeling mean scores (intensity, percentage of positive cells, and distribution) for ΔNp63 expression in these positive cases are shown in Table 1.
Immunolabeling mean scores of the ΔNp63 and pan-p63 expression in 14 types of feline neoplasms.
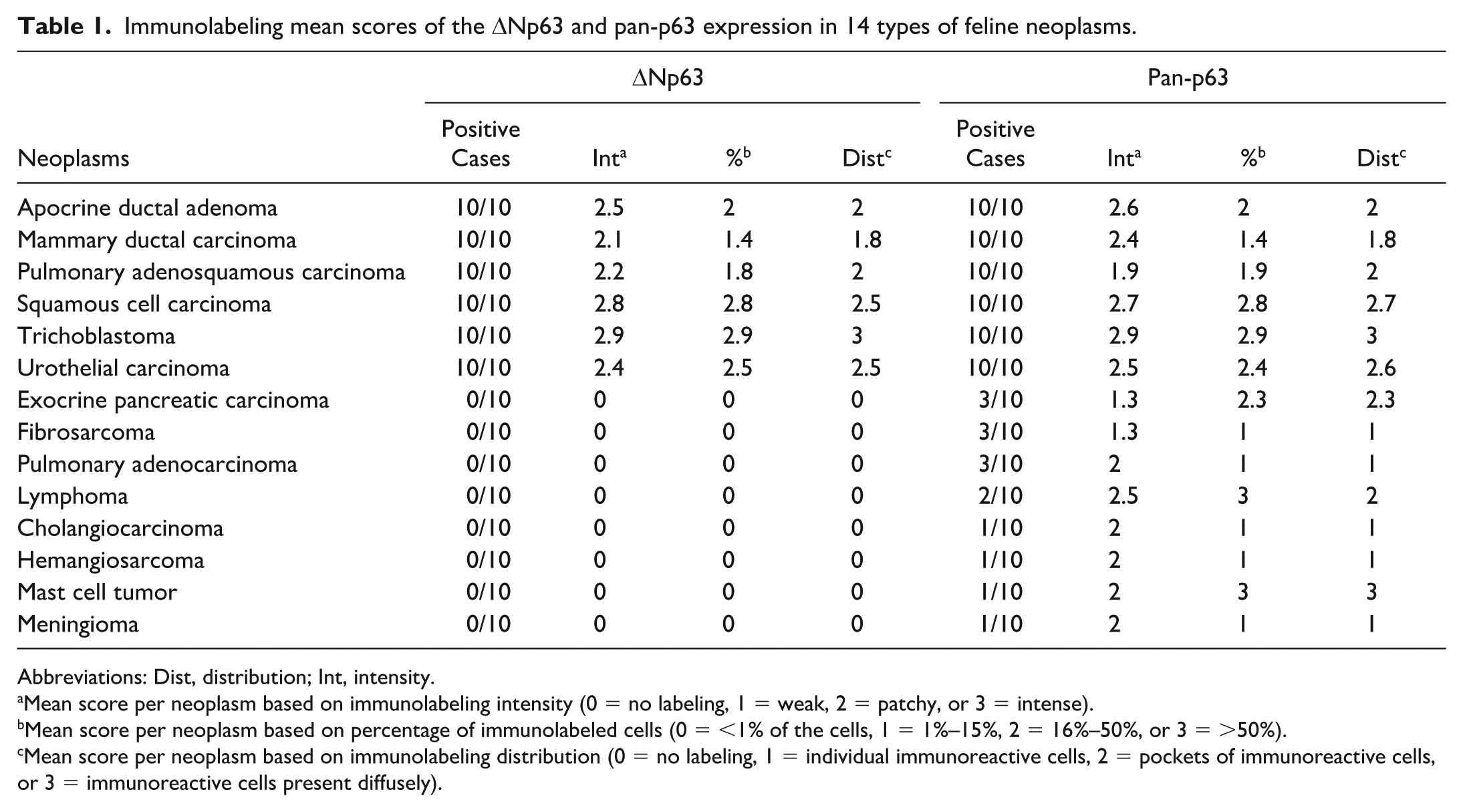
Abbreviations: Dist, distribution; Int, intensity.
Mean score per neoplasm based on immunolabeling intensity (0 = no labeling, 1 = weak, 2 = patchy, or 3 = intense).
Mean score per neoplasm based on percentage of immunolabeled cells (0 = <1% of the cells, 1 = 1%–15%, 2 = 16%–50%, or 3 = >50%).
Mean score per neoplasm based on immunolabeling distribution (0 = no labeling, 1 = individual immunoreactive cells, 2 = pockets of immunoreactive cells, or 3 = immunoreactive cells present diffusely).
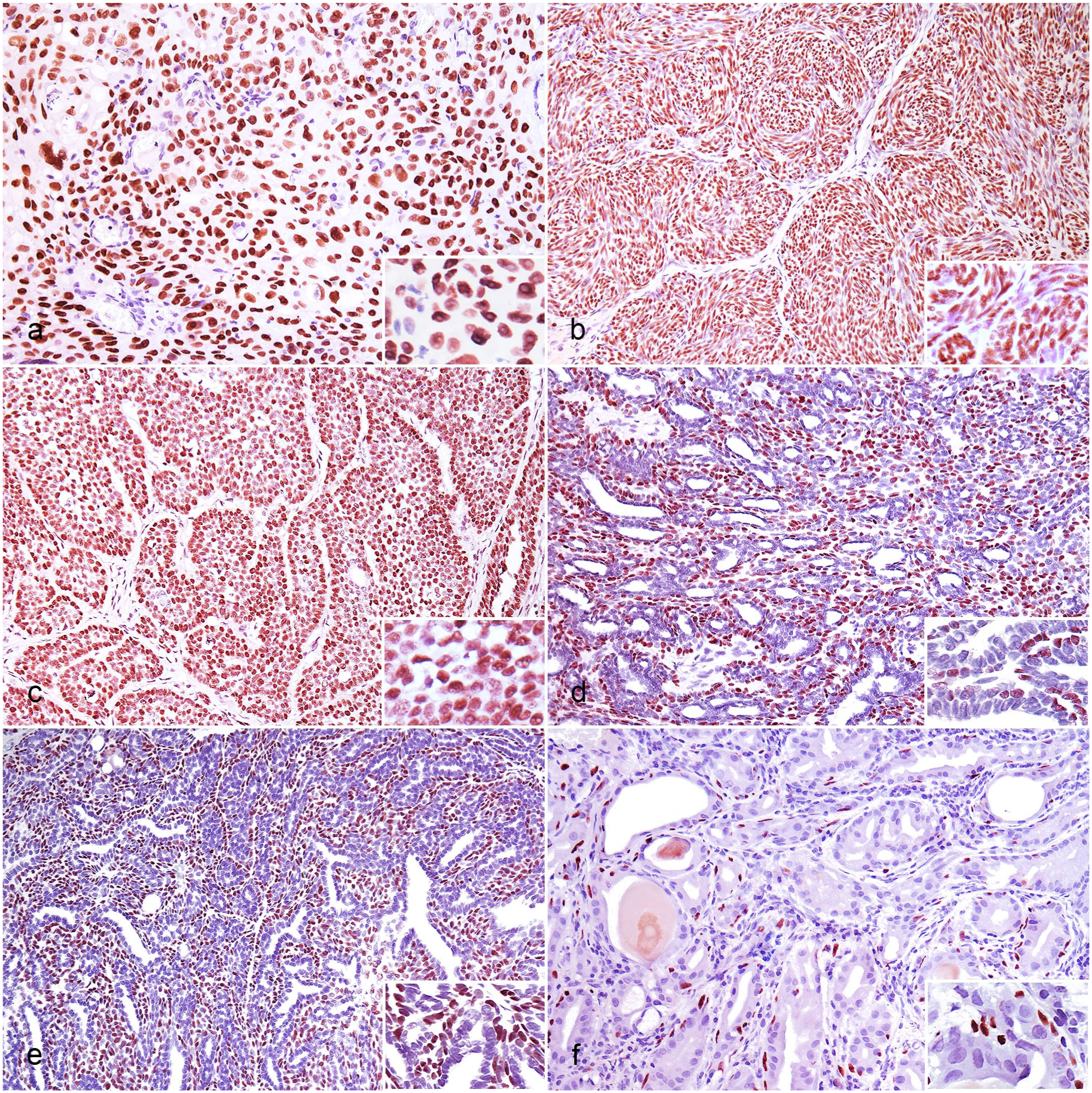
Immunohistochemistry for ΔNp63 in feline neoplasms, cat. (a) Squamous cell carcinoma. Nuclear immunolabeling (inset) in the nests of squamous neoplastic cells. (b) Trichoblastoma. Nuclear immunolabeling (inset) in the cords of neoplastic cells. (c) Urothelial carcinoma. Nuclear immunolabeling (inset) in the nests of neoplastic urothelial cells. (d) Apocrine ductal adenoma. Nuclear immunolabeling (inset) in the neoplastic suprabasal myoepithelial cells, whereas the ductal epithelia are negative. (e) Mammary ductal carcinoma. Nuclear immunolabeling (inset) in the neoplastic suprabasal myoepithelial cells, whereas the ductal epithelia are negative. Malignancy criteria used during the diagnosis (e.g., mitotic count, necrosis foci, and infiltration) are not present in this field. (f) Ceruminous adenoma. Nuclear immunolabeling (inset) in the resting myoepithelial cells, whereas the ductal epithelia are negative.
The neoplastic cells from all other tested neoplasms were negative (Supplemental Figs. S1a–f, S2a–f, and S3a–b), whereas the resting myoepithelial cells of the ceruminous adenomas (Fig. 2f) and mammary simple carcinomas immunolabeled. Notably, there was no difference in the ΔNp63 immunolabeling pattern between well and poorly differentiated positive neoplasms. The individual results of each case are detailed in Supplemental Table S2.
Pan-p63 Expression in Feline Neoplasms
Nuclear immunolabeling for pan-p63 was observed in neoplastic cells in 75 of the 220 cases, including 10/10 mammary ductal carcinomas, 10/10 SCCs, 10/10 pulmonary adenosquamous carcinomas, 10/10 trichoblastomas, 10/10 urothelial carcinomas, 10/10 apocrine ductal adenomas, 3/10 exocrine pancreatic carcinomas, 3/10 fibrosarcomas, 3/10 pulmonary adenocarcinomas, 2/10 lymphomas, 1/10 cholangiocarcinomas, 1/10 hemangiosarcomas, 1/10 mast cell tumors, and 1/10 meningiomas. The types of positive cells and the immunolabeling mean scores (intensity, percentage of positive cells, and distribution) in SCCs (Fig. 3a), pulmonary adenosquamous carcinomas, trichoblastomas (Fig. 3b), mammary ductal carcinomas, urothelial carcinomas (Fig. 3c), and apocrine ductal adenomas (Fig. 3d) were similar to those observed for ΔNp63 (Table 1). In the lymphoma and mast cell tumor cases, pan-p63 immunolabeling was diffusely present in the neoplastic cells (Fig. 3e, f). By contrast, in the other positive neoplasms (fibrosarcoma, pulmonary adenocarcinoma, cholangiocarcinoma, hemangiosarcoma, and meningioma), only a few individual and small pockets of immunolabeled cells were detected (Fig. 3h, i).
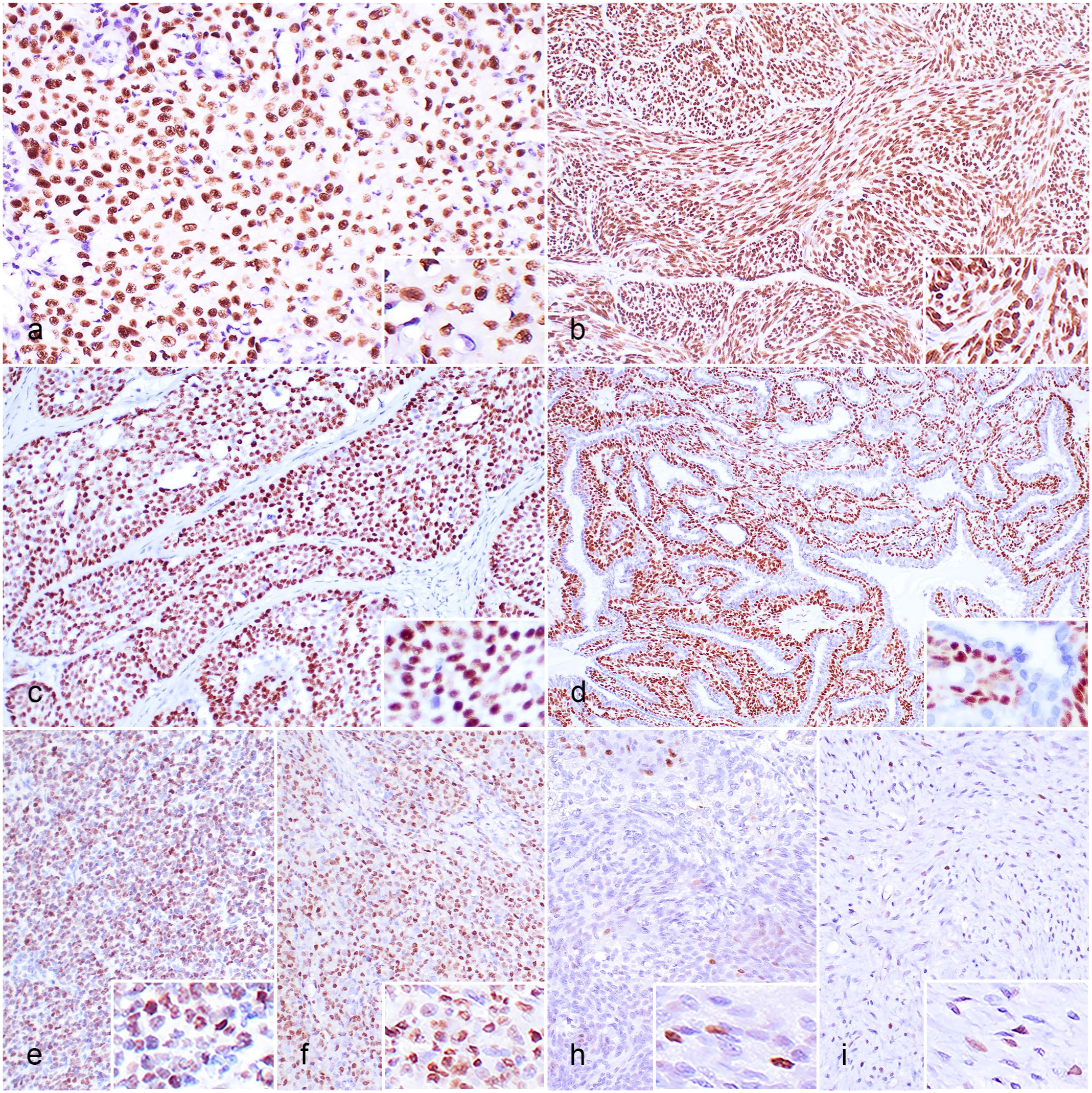
Immunohistochemistry for pan-p63 in feline neoplasms, cat. (a) Squamous cell carcinoma. Nuclear immunolabeling (inset) in the nests of squamous neoplastic cells. (b) Trichoblastoma. Nuclear immunolabeling (inset) in the cords of neoplastic cells. (c) Urothelial carcinoma. Nuclear immunolabeling (inset) in the nests of neoplastic urothelial cells. (d) Apocrine ductal adenoma. Nuclear immunolabeling (inset) in the neoplastic suprabasal myoepithelial cells, whereas the ductal epithelia are negative. (e) Lymphoma. Nuclear immunolabeling (inset) in the neoplastic lymphocytes. (f) Mast cell tumor. Nuclear immunolabeling (inset) in the neoplastic mast cells. (h) Meningioma. Occasional nuclear immunolabeling (inset) in the neoplastic meningeal cells. (i) Fibrosarcoma. Occasional nuclear immunolabeling (inset) in the neoplastic fibroblasts.
The neoplastic cells from all the other tested neoplasms were negative for pan-p63. Only the resting myoepithelial cells of the ceruminous adenomas and mammary simple carcinomas were positive. Details of the results for individual cases are provided in Supplemental Table S2.
Diagnostic Performance Values
All samples of the neoplasms of interest for our hypothesis (apocrine ductal adenomas, mammary ductal carcinomas, pulmonary adenosquamous carcinomas, SCCs, trichoblastomas, and urothelial carcinomas) were positive for both ΔNp63 and pan-p63 antibodies. Consequently, both antibodies achieved 100% diagnostic sensitivity (95% CI: 93.9%–100%) and 100% negative predictive value (95% CI: 97.6%–100% for ΔNp63; 97.4%–100% for pan-p63). For ΔNp63, the diagnostic specificity was 100% (95% CI: 97.6%–100%), the positive predictive value was 100% (95% CI: 93.9%–100%), and the overall accuracy was 100% (95% CI: 98.2%–100%). For pan-p63, the diagnostic specificity was 90.6% (95% CI: 85.1%–94.2%), the positive predictive value was 80% (95% CI: 69.5%–87.4%), and the overall accuracy was 93.1% (95% CI: 89%–95.8%).
Discussion
We investigated the immunohistochemical expression and potential diagnostic utility of the ΔNp63 in feline tissues. The initial 14 amino acids of the sequences from the ΔNp63 isoform are highly conserved among different species (Fig. 4a). 25 According to the manufacturer’s datasheet, the antibody against ΔNp63 used in our cases is designed to recognize a specific human epitope mapped between amino acids 5–17 at the ΔN. The corresponding residues in the p63 sequence of domestic cats presumed to represent a ΔNp63 isoform (A0A337S7C7_FELCA, www.uniprot.org) demonstrate 76.9% identity with the target human epitope (Q9H3D4-2, P63_HUMAN) (Fig. 4b). Despite this relatively modest sequence identity of the residues, the results regarding expression in normal tissues in this study provide evidence supporting the cross-reactivity of ΔNp63 in cats. Our findings on the restricted tissue-specific distribution of ΔNp63 in normal adult tissues complement existing knowledge on this marker in the feline literature41,46 and are identical to those observed in humans.19,29,53 The positive cells were derived from the ectodermal origin and included basal cells of stratified squamous, transitional, and pseudostratified columnar epithelia; basal cells of the sebaceous glands; trophoblasts; and myoepithelial cells. While ΔNp63 is relatively understudied in cats, these results underscore its crucial function in maintaining ectoderm-derived tissues in this species. 22
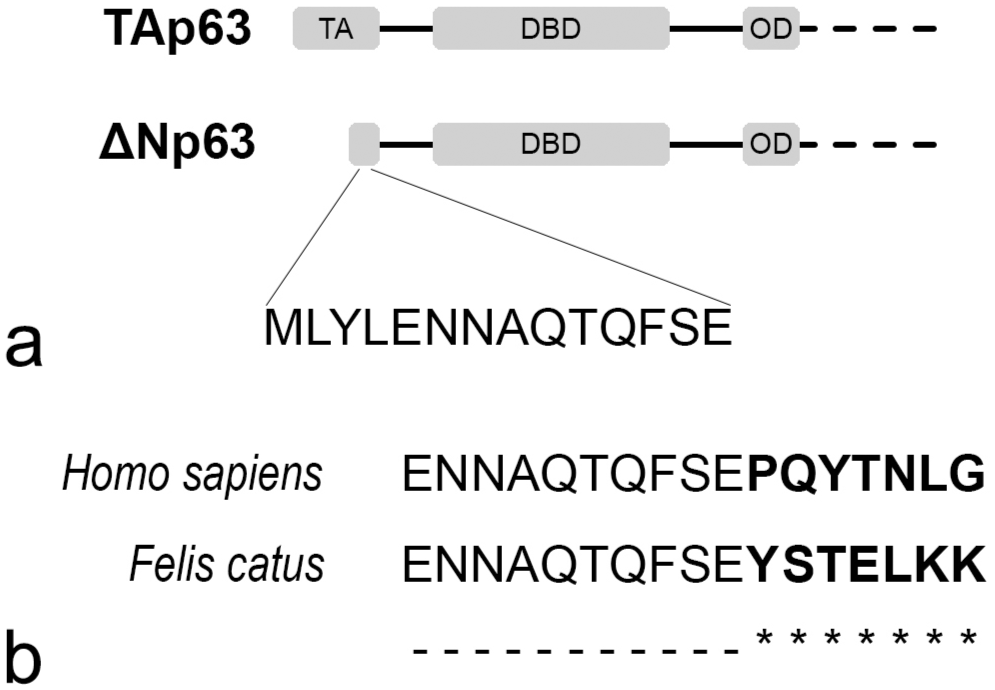
Structure of p63 isoform. (a) TAp63 contains the conserved N-terminal transactivation domain (TA), whereas ΔNp63 is a truncated protein. Both isoforms include a DNA-binding domain (DBD) and an oligomerization domain (OD). Alternative splicing at the C-terminus (dashed lines) generates at least 5 variants. The sequence of the initial 14 amino acids of ΔNp63 is highly conserved among different species. (b) Sequence alignment of amino acids 5–21 of the human (P63_HUMAN) and domestic cat (A0A337S7C7_FELCA) ΔNp63 isoforms. Residues 5-17 are the target of the ΔNp63 antibody used in our study.
Our results reaffirm that ΔNp63 is a strong immunohistochemical marker for diagnosing feline neoplasms with squamous components, consistent with previous reports in humans and some domestic animals.7,41,46,52 SCCs comprise a large part of neoplastic processes in cats, and predominantly arise from cutaneous and oral squamous epithelium.16,20 Some other epithelial neoplasms can also exhibit squamous differentiation, such as mammary, pulmonary, and urothelial carcinomas.11,26,41 All our conventional SCCs and other neoplasms with squamous differentiation were positive for ΔNp63, irrespective of their anatomic location or level of differentiation. This high diagnostic sensitivity is comparable with other commonly used squamous cell markers in feline tumors, such as 94% for cytokeratin (CK) 5/6 and 100% for pan-p63. 36 In humans, ΔNp63 is considered more specific for SCCs with poor differentiation and spindle cell morphologies. 2 Regrettably, unconventional subtype cases were unavailable for inclusion in this study. Moreover, recent studies have explored the protumorigenic or antitumorigenic role of ΔNp63 in human SCCs, which may involve the regulation of tumor initiation, promotion, or suppression. 39 Despite its strong expression in our cases, the role of ΔNp63 in feline SCCs remains unknown.
Cutaneous basal cell neoplasms are commonly diagnosed in cats. 20 Following current recommendations, 18 during our case selection, the review of the cases previously classified as basal cell tumors resulted in the reclassification of the majority as either trichoblastomas or apocrine ductal adenomas (data not shown). Applying IHC for ΔNp63 in these 2 types of epithelial tumors revealed positivity in all cases and an expression pattern that allows easy differentiation between them. In our experience, the distinction between trichoblastoma and apocrine ductal adenoma in small biopsy specimens or tumors with few areas of ductal differentiation can be difficult based on histology alone. All neoplastic cells in the trichoblastomas immunolabeled for ΔNp63, while only the suprabasal myoepithelial cells immunolabeled in the apocrine ductal adenomas. As trichoblastomas derive from the primitive hair germ of embryonal follicular development, 55 a ΔNp63-positive structure, the immunolabeling of neoplastic cells in all our cases was expected. The diagnostic sensitivity of ΔNp63 to detect its human counterpart is up to 100%. 4 Even though IHC is rarely necessary for diagnosing trichoblastomas, 55 the observed positivity in these tumors reaffirmed ΔNp63 expression in basal epithelial neoplasms. Unfortunately, the number of other basal neoplasms diagnosed in the period evaluated was insufficient for inclusion in this study.
Feline urothelial carcinomas are uncommon and can originate from the urinary bladder, urethra, or renal pelvis. 56 These tumors are relatively well described in cats,31,54 but there are few comprehensive studies focused on immunohistochemical aspects. To our knowledge, only one survey established the positivity for CK7 and CK20 in 3/4 feline urothelial carcinomas. 15 The utility of additional markers for urothelial epithelium applied in humans and other domestic animals, such as uroplakin III and pan-p63,3,24,26,32 has not been documented in cats. In our study, all urothelial carcinomas immunolabeled for ΔNp63 in almost all neoplastic cells. This high percentage of positive cases was anticipated based on the expression of ΔNp63 in the normal urinary bladder of cats and is comparable with that observed in human bladder urothelial carcinomas, where the diagnostic sensitivity can reach up to 87%. 24 Hence, while these tumors are relatively easy to diagnose histologically, there is a potential role for ΔNp63 in diagnosing and differentiating urothelial carcinomas from other neoplasms in cats. A possible pitfall is areas of squamous metaplasia in some cases of urothelial carcinomas, 26 which can also show immunolabeling and may be difficult to distinguish from neoplastic urothelial cells.
Markers for myoepithelial cells are widely applied in the diagnostic routine of human and animal mammary tumors.13,17 Classical myoepithelial cell markers, such as pan-CK, CK5/6, CK14, pan-p63, vimentin, calponin, and α-smooth muscle actin, can achieve positive results in up to 100% of feline mammary tumors with ductal differentiation. 57 In our study, all mammary ductal carcinomas had immunolabeling for ΔNp63 in the neoplastic suprabasal myoepithelial cells. The absence of ΔNp63 expression in myofibroblasts, pericytes, and luminal cells is a great advantage in comparison with some other myoepithelial cell markers. 13 Unfortunately, our archive lacked sufficient samples of neoplasms containing pleomorphic interstitial myoepithelial cells, which can occasionally lose the suprabasal myoepithelial cell expression. 40 In human mammary ductal carcinomas, ΔNp63 positivity may lead to misidentification of luminal epithelial cells as myoepithelial cells, 21 a finding that was not observed in our cases. In addition to identifying the myoepithelial phenotype, which can be difficult through routine histology, myoepithelial cell markers may help to distinguish invasive from non-invasive mammary carcinomas. 17 Notably, ΔNp63 highlighted the resting myoepithelium in all our simple mammary carcinomas. These results show the potential of ΔNp63 as a valuable component of the IHC panel for diagnosing mammary carcinomas in cats.
IHC for pan-p63 is widely employed to classify various human tumors, particularly those containing squamous, urothelial, and myoepithelial components. 45 Nonetheless, concerns have emerged regarding the specificity of pan-p63 compared with ΔNp637,23 and the potential pitfalls of pan-p63 in diagnosis, such as large cell lymphomas. 45 In domestic animals, available data comparing the diagnostic utility of pan-p63 and ΔNp63 are sparse, though similar expression has been reported in canine mixed mammary tumors and feline basal/squamous cutaneous neoplasms.6,46 In our study, the mean immunolabeling scores were similar for both antibodies in apocrine ductal adenomas, mammary ductal carcinomas, pulmonary adenosquamous carcinomas, SCCs, trichoblastomas, and urothelial carcinomas. Since all cases within these tumor types were positive, diagnostic sensitivity and negative predictive value were similar (100%). However, pan-p63 also immunolabeled a small fraction of other neoplasm types (exocrine pancreatic carcinoma, fibrosarcoma, lymphoma, pulmonary adenocarcinoma, cholangiocarcinoma, hemangiosarcoma, mast cell tumor, and meningioma). The positivity in these unexpected neoplasms reduced the diagnostic specificity and overall accuracy of pan-p63 for detecting feline neoplasms of squamous, basal, urothelial, or myoepithelial origin. Notably, with the exception of the lymphoma and mast cell tumor, the immunolabeling in these additional neoplastic types was mostly sparse.
This study has limitations that are inherent to its nature. First, based on our examination of ΔNp63 expression in normal tissues of cats and human oncology studies,7,23,49,53 several other tumors could potentially have ΔNp63 immunolabeling and were not included due to an insufficient number of cases in our archive. Thymic neoplasms, other mammary epithelial neoplasms, and other cutaneous neoplasms from the epidermis, hair follicles, sebaceous glands, and apocrine ducts are notable examples. Second, although the sample size for each tumor category was defined to include as many cases as possible, financial constraints limited our selection and might have impacted the study’s statistical power. This limitation could be mitigated using a larger number of samples in future studies. Third, we are unable to establish the clinical significance of ΔNp63 expression in feline tumors because follow-up data for the included cases were not available. Finally, despite the efforts to meticulously select cases, the immunohistochemical analysis may have been affected by fixation or autolytic artifacts. Of note, there were no changes in ΔNp63 expression in our investigation of the effects of prolonged fixation nor between biopsy and necropsy samples in positive neoplasms.
In conclusion, this study provides significant insights into the expression of ΔNp63 in normal and neoplastic tissues of cats. Our results demonstrate the utility of ΔNp63 for diagnosing feline neoplasms with squamous, basal, and urothelial differentiation, including SCCs, pulmonary adenosquamous carcinomas, trichoblastomas, and urothelial carcinomas. This marker can also highlight the neoplastic suprabasal myoepithelial cells of apocrine ductal adenomas and mammary ductal carcinomas. ΔNp63 exhibited high diagnostic sensitivity for detecting these neoplasms, and achieved higher diagnostic specificity and overall accuracy than pan-p63.
Supplemental Material
sj-docx-1-vet-10.1177_03009858251409220 – Supplemental material for Immunohistochemical expression of ΔNp63 versus pan-p63 in normal and neoplastic feline tissues
Supplemental material, sj-docx-1-vet-10.1177_03009858251409220 for Immunohistochemical expression of ΔNp63 versus pan-p63 in normal and neoplastic feline tissues by Igor R. Santos, Carolina B. Brunner, Milena C. Paz, Gabriela Hartmann, Gabrielle Z. Tres, Luciana Sonne, Welden Panziera, David Driemeier, Marcele B. Bandinelli and Saulo P. Pavarini in Veterinary Pathology
Supplemental Material
sj-docx-2-vet-10.1177_03009858251409220 – Supplemental material for Immunohistochemical expression of ΔNp63 versus pan-p63 in normal and neoplastic feline tissues
Supplemental material, sj-docx-2-vet-10.1177_03009858251409220 for Immunohistochemical expression of ΔNp63 versus pan-p63 in normal and neoplastic feline tissues by Igor R. Santos, Carolina B. Brunner, Milena C. Paz, Gabriela Hartmann, Gabrielle Z. Tres, Luciana Sonne, Welden Panziera, David Driemeier, Marcele B. Bandinelli and Saulo P. Pavarini in Veterinary Pathology
Supplemental Material
sj-pdf-3-vet-10.1177_03009858251409220 – Supplemental material for Immunohistochemical expression of ΔNp63 versus pan-p63 in normal and neoplastic feline tissues
Supplemental material, sj-pdf-3-vet-10.1177_03009858251409220 for Immunohistochemical expression of ΔNp63 versus pan-p63 in normal and neoplastic feline tissues by Igor R. Santos, Carolina B. Brunner, Milena C. Paz, Gabriela Hartmann, Gabrielle Z. Tres, Luciana Sonne, Welden Panziera, David Driemeier, Marcele B. Bandinelli and Saulo P. Pavarini in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge Dr Leonardo Susta of University of Guelph for his collaboration and for providing the antibodies.
Supplemental material for this article is available online.
Author Contributions
IRS and SPP designed the study and performed evaluations; CBB, MCP, GH, GZT, and MBB contributed to the experimental designs; LS, WP, DD, and SPP made the initial diagnoses of the cases; and the manuscript was written by IRS with contribution from the other authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) supported this study—Finance Code 001.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
