Abstract
Feline leukemia virus (FeLV) infection is associated with distinct neoplastic, hematologic, and immunosuppressive diseases. Here we report on a novel neurologic syndrome in 16 cats infected with FeLV for more than 2 years. Clinical signs consisted of abnormal vocalization, hyperesthesia, and paresis progressing to paralysis. The clinical course of affected cats involved gradually progressive neurologic dysfunction invariably resulting in euthanasia. Microscopically, white-matter degeneration with dilation of myelin sheaths and swollen axons was identified in the spinal cord and brain stem of affected animals. Neither neoplastic nor hematologic diseases commonly associated with FeLV infection were present. Fungal and protozoal infection in one animal was suggestive of impaired immune competence. Immunohistochemical staining of affected tissues revealed consistent expression of FeLV p27 antigens in neurons, endothelial cells, and glial cells. Furthermore, proviral DNA was amplified from multiple sections of spinal cord as well as intestine, spleen, and lymph nodes. These findings suggest that in a proportion of chronically FeLV-infected cats, a virus evolved with cytopathic potential for cells in the central nervous system.
The feline leukemia virus (FeLV) is a common virus of domestic cats. Vaccines against the virus are widely used; however, these appear to protect predominantly against tumor formation and not against infection.20 FeLV belongs to the oncogenic Retroviridae, a family of viruses that inserts a double-stranded DNA copy of its single-stranded RNA genome into the host cells' chromosomes. Once stably inserted, the viral genes behave like cellular genes and may be transcribed into functional protein products or may remain latent for long periods of time.10 Depending on the infecting viral strain and on the host response, infection with FeLV may result in early viral clearance because of an effective immune response or in viremia.10,20 In cats that are viremic, multiple tissues, including bone marrow, are infected and may retain the latent virus indefinitely. A proportion of viremic cats subsequently mounts an immune response that results in viral clearance. Cats that remain persistently viremic may be clinically healthy or, more commonly, develop immunosuppressive, hematologic, intestinal, or reproductive disorders, neoplasms such as lymphoma or leukemia, or autoimmune diseases.19 Thus, FeLV infection can result in a wide range of potential disease manifestations.
The specificity of FeLV in infecting cells resides in the glycoprotein envelope.10 Recombination of exogenous FeLV subgroup A env genes with complementary endogenous retroviral sequences can result in FeLV viruses typed as subgroup B or C.6 Opportunities for recombination are expanded during long periods of active viral replication and may result in viruses infecting endothelial or epithelial cells.20 We have identified a neurologic syndrome in FeLV-positive cats that appears to be associated with chronic FeLV infection (FeLV-associated myelopathy, FAM). The present study seeks to understand more completely the pathomechanistic spectrum of this important feline pathogen through characterization of the clinical, morphologic, and immunohistochemical features of FAM in cats.
Materials and Methods
Animals
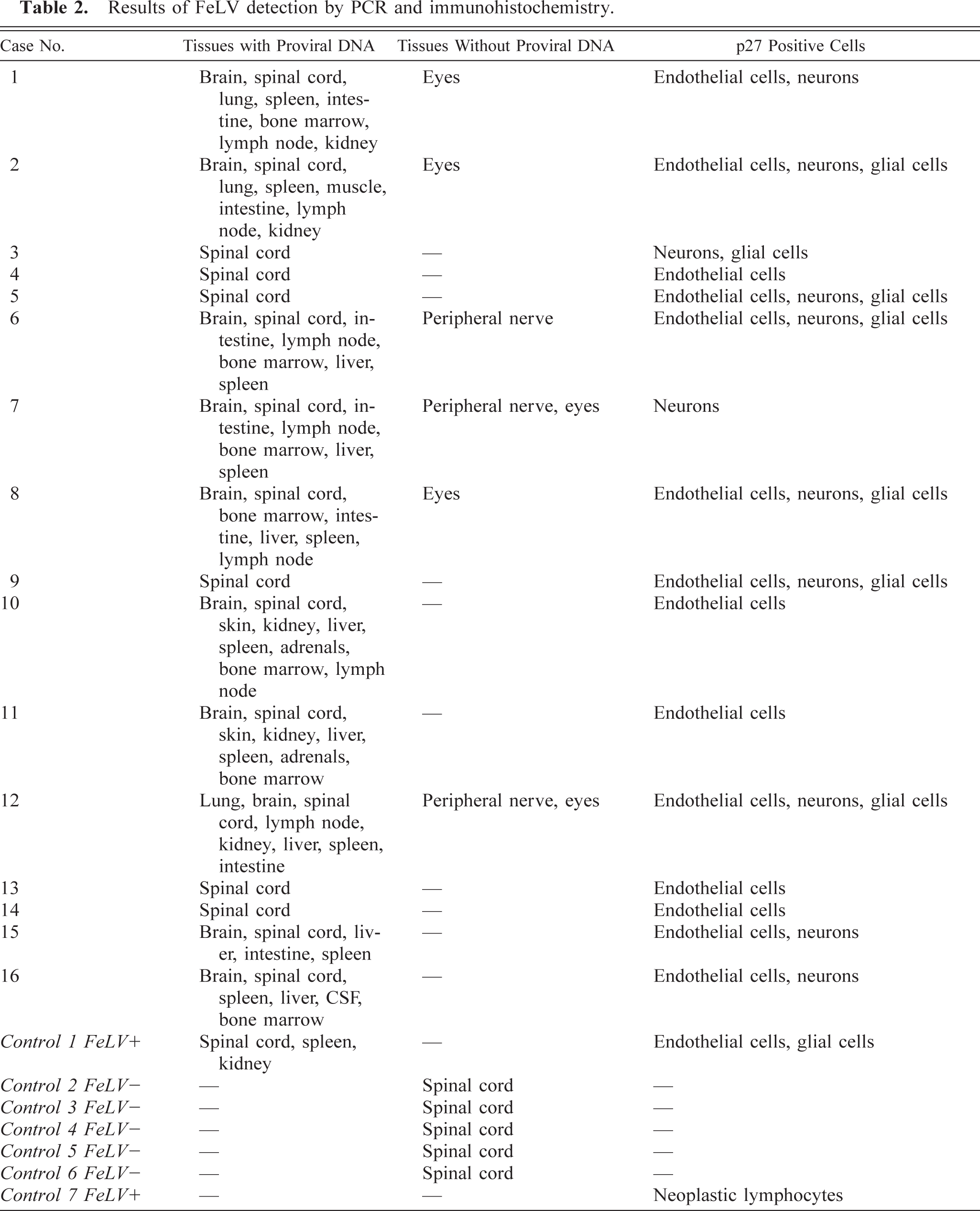
Tissues from 16 cats with a neurologic syndrome characterized mostly by progressive ataxia were available for study. These cats were FeLV antigenemic (p27 antigen detection, IDEXX, Westbrook, ME). Clinical and history data were obtained from the submitting clinicians or from the necropsy reports. Formalin-fixed tissues were obtained from eight animals (cat Nos. 1–8) submitted for necropsy examination from 1994 through 2001, and archived histologic sections from eight additional cats (cat Nos. 9–16) with a similar neurologic syndrome. The clinical history and pathology of these cases are summarized in Table 1.
Summary of clinical data from FeLV positive cats with degenerative myelopathy.
No abnormal findings.
Urinalysis.
Not available.
Bone marrow.
Tissues from six control animals were used in the study. Four of these animals were FeLV-seronegative specific pathogen free cats with no clinical signs of myelopathy (control cat Nos. 2–5). Two were FeLV-seropositive cats that had no clinical signs of neurologic disease (control cat Nos. 1 and 6). Spleen and spinal cord from a FeLV-positive cat with spinal lymphoma (control cat No. 7) and preparations from a persistently FeLV-infected cell line (3,201 cells stably transfected with the FeLV-61E plasmid by J. I. Mullins, AIDS Research and Reference Reagent Program) were used as positive controls.
Pathology
Samples from 16 cats euthanatized because of progressive neurologic dysfunction were examined. The tissues were immersion-fixed in 10% neutral buffered formalin for less than 24 hours and routinely processed for microscopic examination. Sections of cervical, thoracic, and lumbar spinal cord, brain stem, cerebellum, and cerebrum from all cases were routinely stained with hematoxylin and eosin (HE). Selected replicate sections were stained with Luxol fast blue-cresyl violet and Bodian's silver stain.
Immunohistochemistry
Five-micrometer sections of spinal cord and spleen from all the cases were deparaffinized at −70 C for 35 minutes and immersed in Hemo-De (Fisher Scientific, Pittsburgh, PA) three times for 3 minutes each. Sections were rehydrated in alcohol, endogenous peroxidases were quenched with 3% hydrogen peroxide (Sigma, St. Louis, MO), and antigenic sites were unmasked through incubation with citrate buffer (1:3 dilution of Antigen Unmasking Solution, Vector Laboratories, Burlingame, CA) for 10 minutes at 96 C. Nonspecific reactivity was blocked with 10% horse serum (Atlanta Biologicals, Norcross, GA). Slides were incubated overnight with goat anti-FeLVp27 antibody (ViroStat Laboratories, Portland, ME) diluted 1:100 in a mixture of 10% goat serum in PBS/0.005% Tween 20 (PBST).11 After extensive washing, the slides were incubated at 37 C for 1 hour with a 1:250 dilution of a mixture of biotinylated rabbit anti-goat antibody in 10% goat serum/PBST (Vector Laboratories). After further washing and 1 hour exposure to Vectastain Elite ABC solution (Vectastain Elite ABC Kit, Vector Laboratories), specific staining was visualized with DAB (Vector). The sections were counterstained with hematoxylin and coverslipped. Immunohistochemical staining was graded as +++, ++, +, or 0 according to the intensity of color development and the number of cells stained. Negative control tissues (from control cat Nos. 1–6, FeLV-seronegative cats and FeLV-seropositive cats lacking neurologic signs), positive control tissues (cytospin preparations of FeLV-infected lymphocytes and sections from an FeLV-positive cat [control cat No. 7] with spinal cord lymphoma), and reagent controls (nonimmune goat serum at the same dilution as the primary antibody and phosphate buffered saline as negative controls for the primary antibody) were included in all assays. The identity of cells immunoreactive for FeLV antigens was verified by staining replicate sections of p27-positive spinal cord for glial fibrillary acidic protein (DAKO, Carpenteria, CA) and anti-galactosylceramidase (Chemicon, Temecula, CA).
Polymerase chain reaction assay
Genomic DNA was extracted from 10-μm sections of paraffin-embedded sections of brain, spinal cord, peripheral nerve, lung, spleen, intestine, bone marrow, lymph node, muscle, kidney, and eye of all cats. In addition, DNA was extracted from liver in six cats (cat Nos. 6, 7, 8, 10, 11, and 12), skeletal muscle in one cat (cat No. 2), and cerebrospinal fluid in another (cat No. 16). Sections were cut using sterile blades to prevent sample carryover. The samples were deparaffinized through two extractions with xylene and washed with absolute ethanol. The tissue pellet was incubated overnight at 37 C in 50 mM Tris/1 mM ethylenediamine-tetraacetic acid (EDTA)/0.5% Tween-20 with 1 µg/µl proteinase K. DNA was extracted with phenol/chloroform/isoamyl alcohol, precipitated with sodium acetate, and resuspended in 10 mM Tris/1 mM EDTA and the concentration determined. FeLV-specific proviral sequences were amplified from approximately 0.5 µg of DNA in a nested polymerase chain reaction (PCR).11 The outer primers were 5′ TTA CTC AAG TAT GTT CCC ATG 3′ and 5′ CTG GGG AGC CTG GAG ACT GCT 3′; they amplified a 166-bp segment in the viral long terminal repeat in a 50-µl reaction. Cycling parameters consisted of 30 rounds of 94 C for 30 seconds, 55 C for 30 seconds, and 72 C for 30 seconds. Five microliters of the first-round product was subjected to a second round of amplification with primers 5′ GGT TAA GCA CCT GGG CCC CGG 3′ and 5′ GCA GCG GCC TTG AAA CTT CTG 3′ with identical cycling parameters except for an annealing temperature of 58 C. The final amplicon had a size of 85 bp, and sequence determination confirmed complementarity with published FeLV sequences. Positive and negative controls, as well as all appropriate reagent controls, consisted of genomic DNA from FeLV-seropositive and -seronegative cats, respectively.
Results
Clinical findings
The clinical signs of FeLV-seropositive cats consisted predominately of ataxia, hyperesthesia, and paresis progressing to paralysis (Table 1). Four cats (cat Nos. 3, 4, 7, and 11) had a history of weakness, lethargy, abnormal behavior, and disorientation. Two cats (cat Nos. 6 and 12) had anisocoria with a diminished papillary light reflex, whereas another two (cat Nos. 1 and 10) had no deep pain on neurologic examination. Urinary incontinence was seen in three cats (cat Nos. 3, 10, and 11), and one cat (cat No. 11) had recurrent constipation. All cats were FeLV-seropositive for more than 4 years, except one that was FeLV-seropositive for 2 years. Four cats had mild anemia (hematocrit 0.20–0.30 L/L), one had macrocytosis (mean corpuscular volume > 55 fL) without anemia, 10 cats had lymphopenia (<1.5 × 109/L), and three of 12 cats had mild neutropenia (2.0–2.5 × 109/L). Thrombocytopenia was not detected, and mean platelet volumes were inconsistently reported because of frequent platelet clumping. Changes on the biochemistry panel in affected cats were nonspecific. Findings from cerebrospinal fluid examination were unremarkable in all of seven cases examined.
Gross pathologic findings
Neither typical FeLV-associated hematologic abnormalities nor tumors were identified grossly or microscopically in any of the cats. One cat (No. 8) had multiple nodular masses in the mesentery and lungs that were gray-yellow and firm. There was no gross evidence of central nervous system (CNS) involvement in this cat.
Histopathology
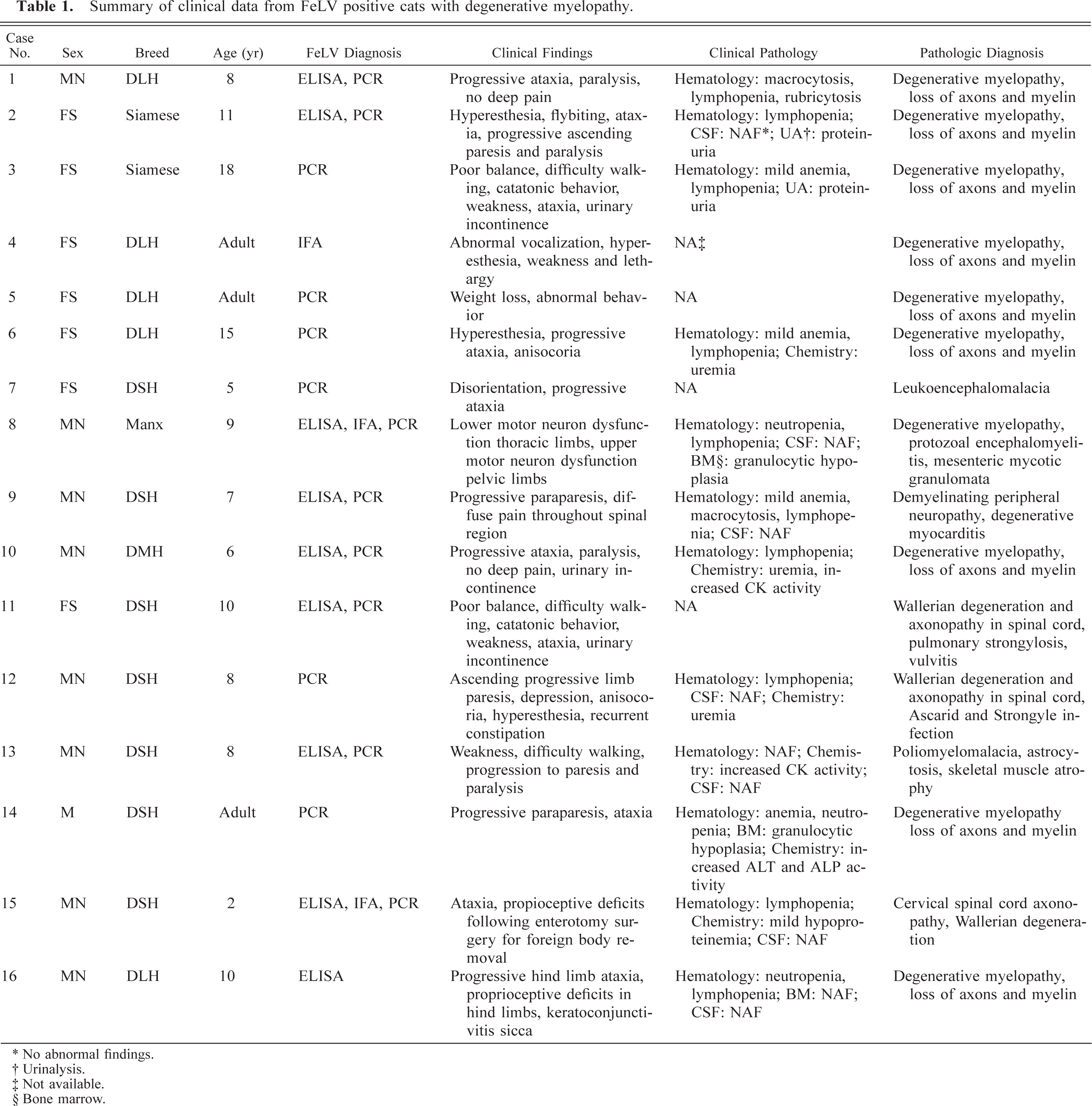
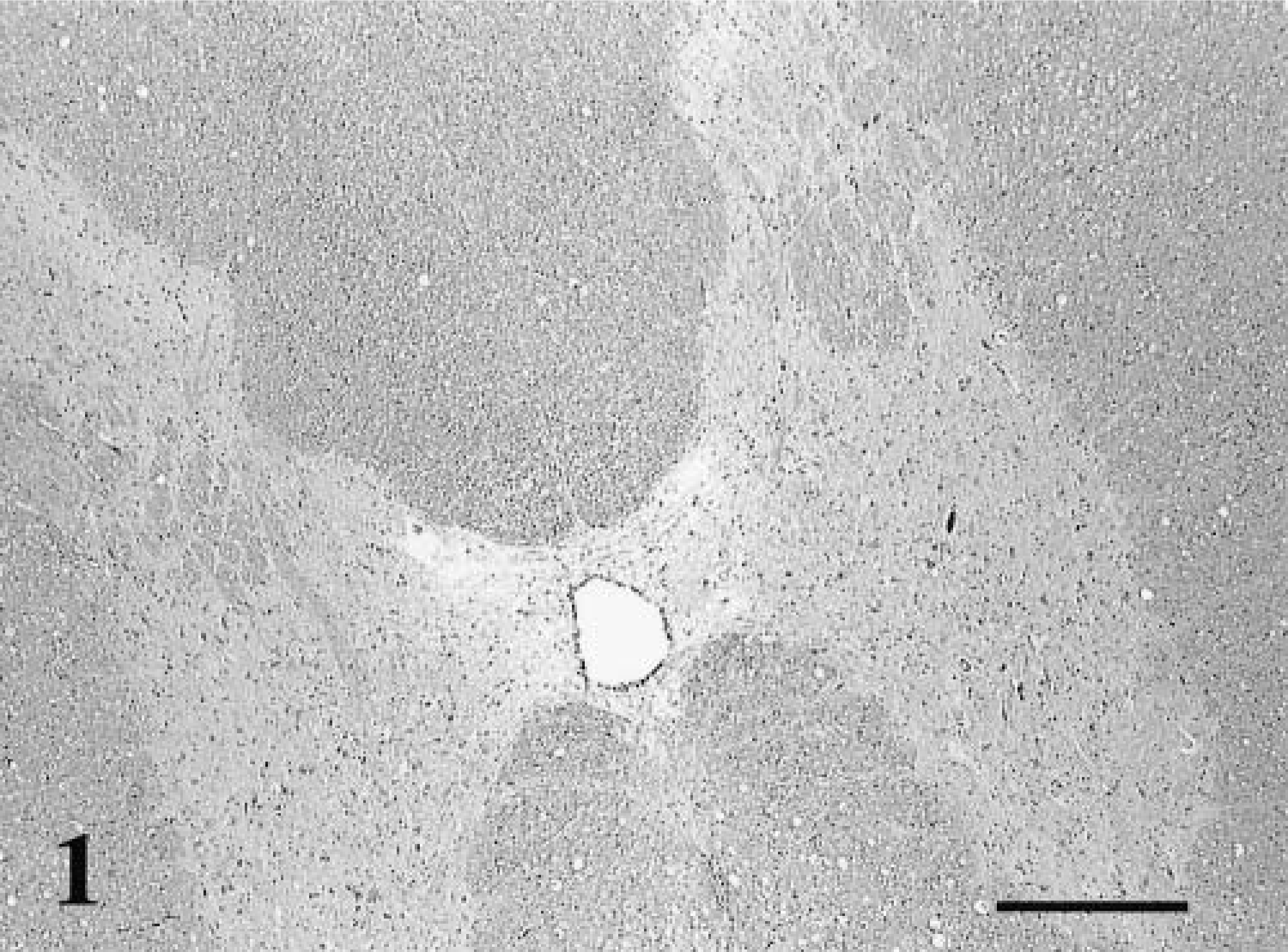
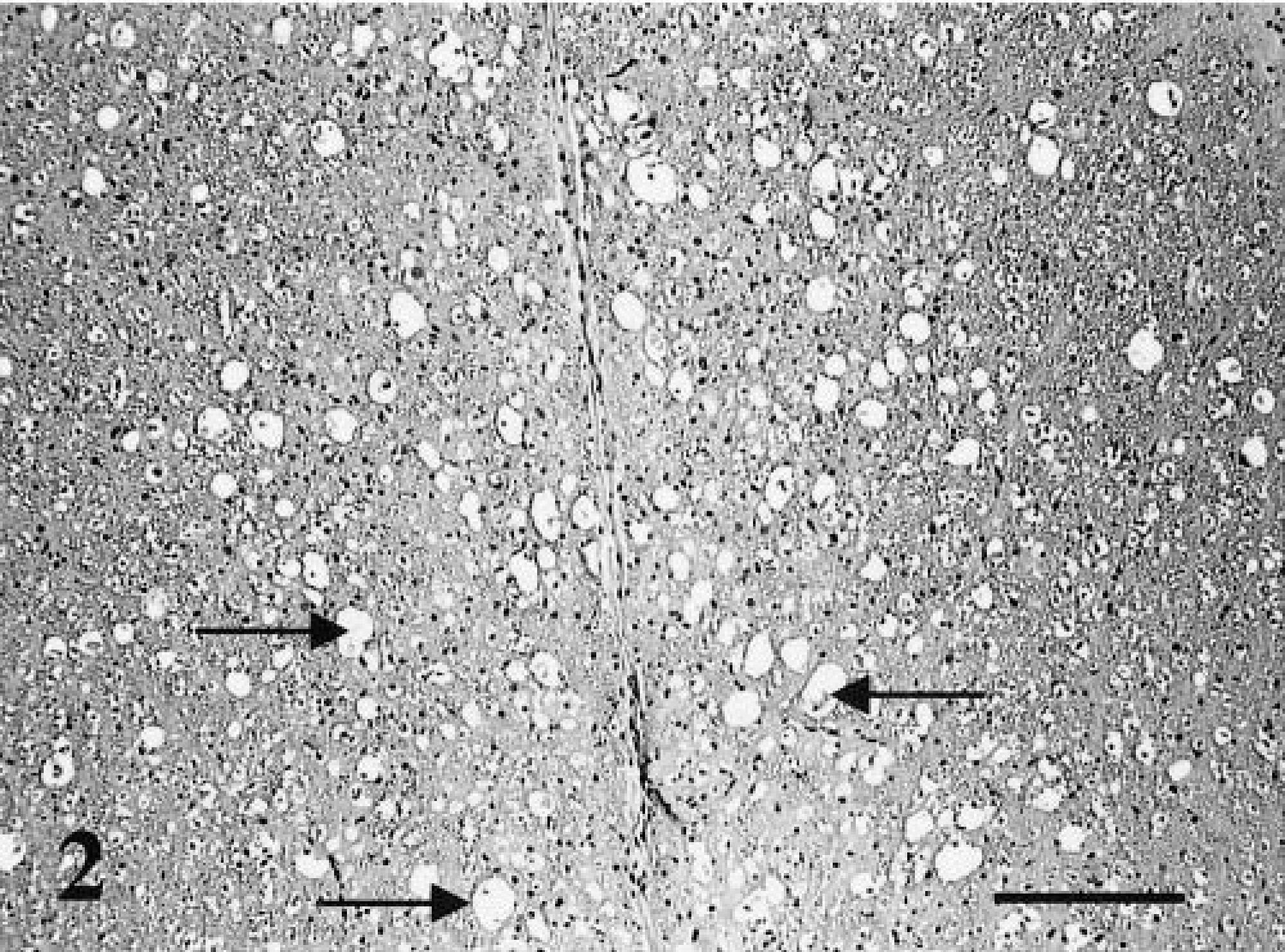
Histologically, all affected cats had widespread lesions in the spinal cord that most often involved the ventromedial funiculus, the superficial lateral funiculi, and the deep ventromedial funiculus (Fig. 1). The lesions consisted of diffuse white-matter degeneration characterized by dilated myelin sheaths (Fig. 2). Some of these dilated myelin sheaths were devoid of axons, whereas others contained either intact or swollen axons (Fig. 3). In some areas, myelin sheaths devoid of central axons and containing infiltrating gitter cells were prominent (Fig. 3). There appeared to be a mild decrease in the numbers of neurons present in the ventral horns of some affected cats when compared with control animals. Luxol fast blue-cresyl stain showed diffuse staining, and there was a decrease in staining especially in the peripheral spinal cord white matter. Bodian's silver stain revealed axonal material in gitter cells. In three cases, the thoracic spinal cord appeared to be more severely affected, whereas in the other cases no difference in the severity of lesions in different segments of the cord was noted. Sections of peripheral nerves did not contain lesions. Cat No. 8 had multiple granulomas in the mesentery and the lungs associated with the pigmented fungus Cladosporium sp. as identified by culture. This cat also had disseminated Toxoplasma infection involving the spinal cord.
Spinal cord; FeLV-associated myelopathy–affected cat. This low-power photomicrograph shows the distribution of the vacuolar change in the spinal cord. HE. Bar = 300 μm.
Spinal cord; FeLV-associated myelopathy–affected cat. The ventral funiculus of the thoracic cord has diffuse vacuolar change (arrows). HE. Bar = 145 μm.
Spinal cord; FeLV-associated myelopathy–affected cat. Dilated myelin sheaths contain both gitter cells (large arrows) and intact axons (small arrows). HE. Bar = 85 μm.
Immunohistochemistry
Positive FeLV p27 immunostaining was present in the sections of spinal cord examined (Table 2). The cells stained were glial cells, neurons, and endothelial cells (Table 3). In these cells, diffuse, intense staining was present involving the entire cell. In neurons, the cytoplasm of both cell bodies and processes were immunoreactive for p27 (Fig. 4). Staining of neurons was not seen in nonimmune serum tissue controls (Fig. 5) or in p27-stained controls that were FeLV-negative (Fig. 6). Endothelial cell staining was seen predominately in smaller blood vessels in both the gray and white matter (Fig. 7). The larger blood vessels of the meninges and ventral median fissure stained less consistently. Glial cells stained intensely, and in many cases, the immunoreactivity obliterated the nucleus (Fig. 7 inset). Control slides of a lymphoma (spinal) and spleen from an FeLV-positive cat showed similar staining. Glial cell neurons and endothelial cells of the negative controls did not express p27 antigen.
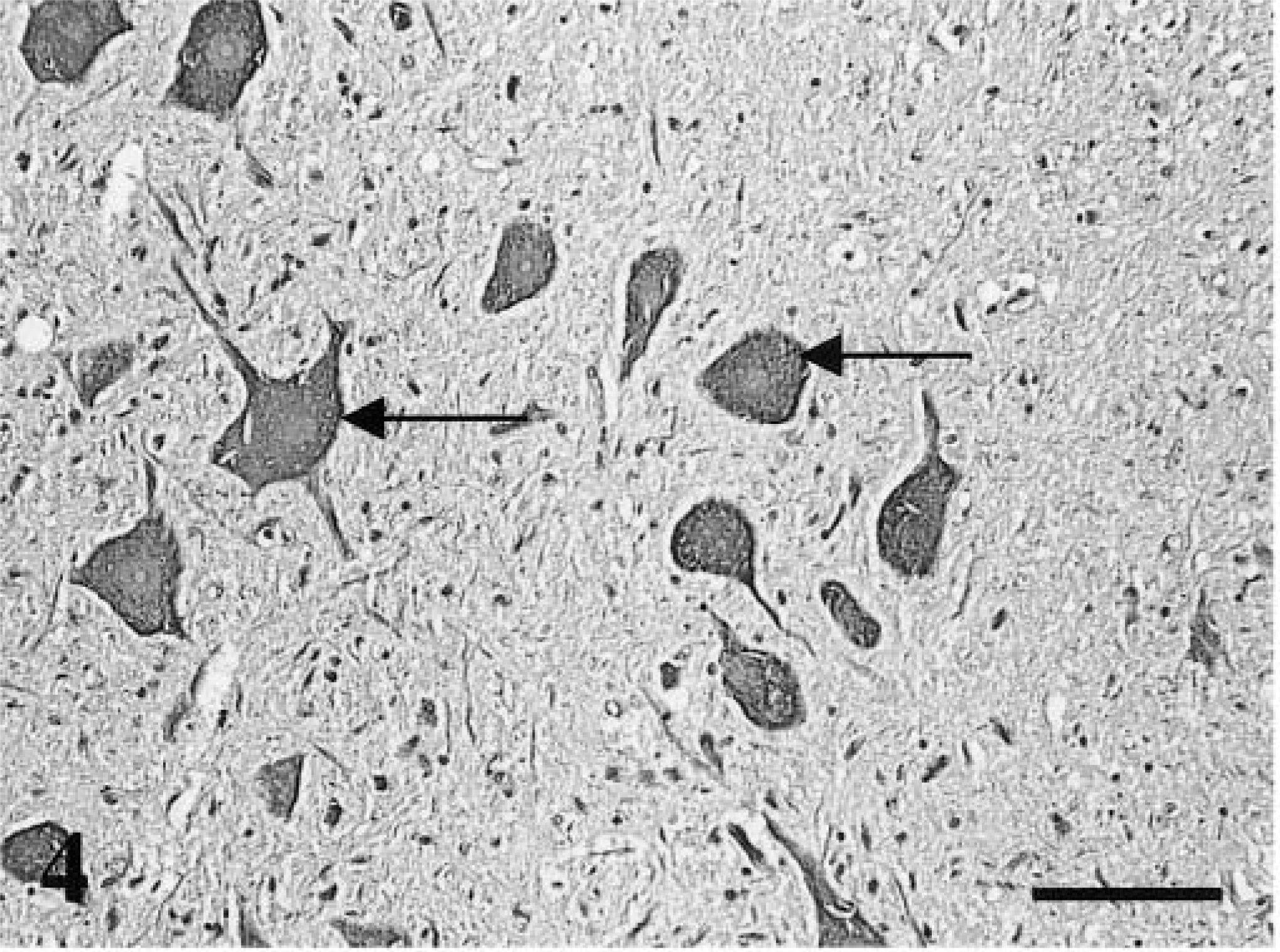
Spinal cord; FeLV-associated myelopathy–affected cat. Positive staining of neurons with anti-p27 antibody (arrows); DAB chromogen. Hematoxylin counterstain. Bar = 85 μm.
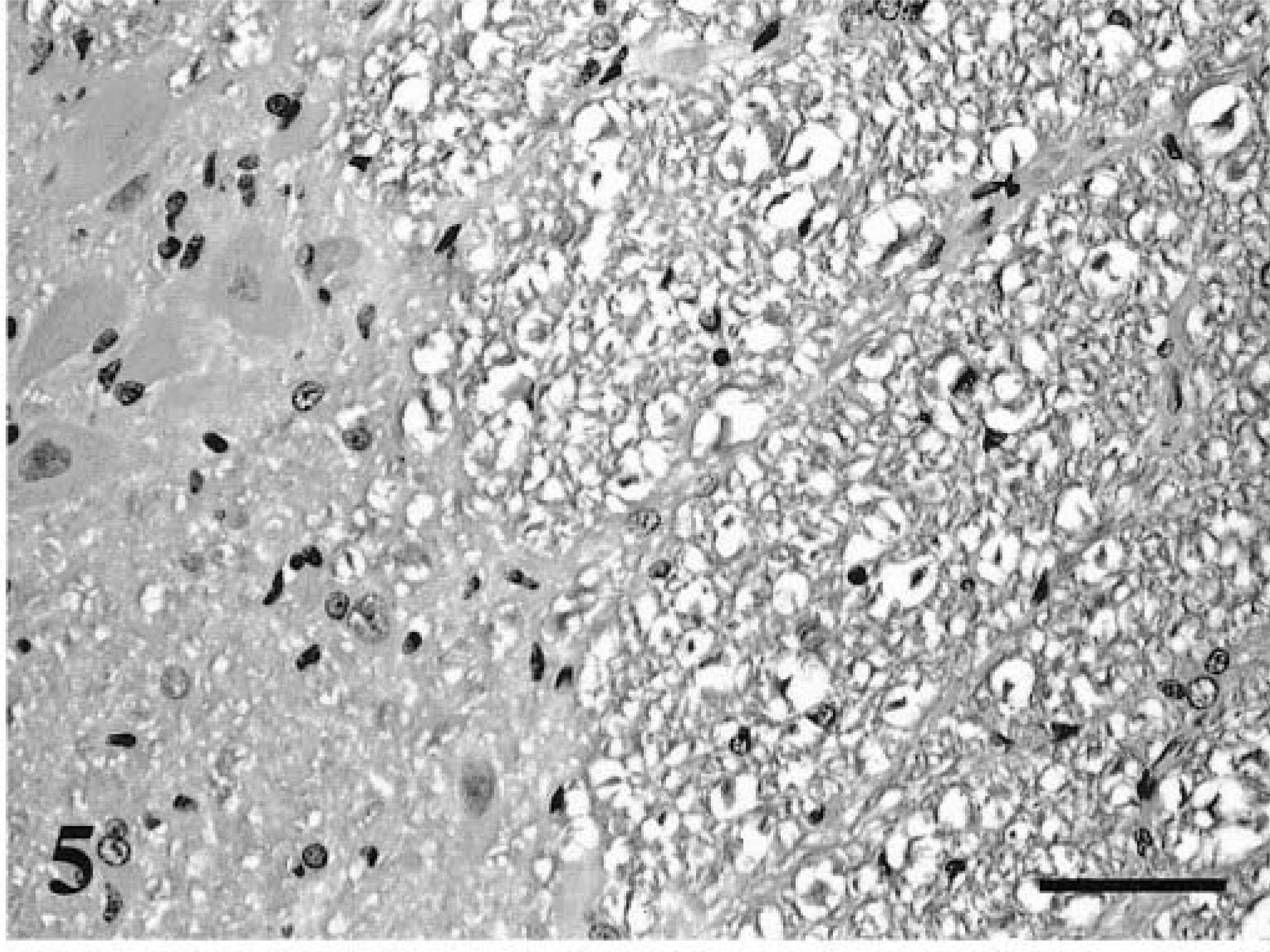
Spinal cord; FeLV-associated myelopathy–affected cat. No staining of neurons is noted with nonimmune serum. DAB chromogen. Hematoxylin counterstain. Bar = 85 μm.
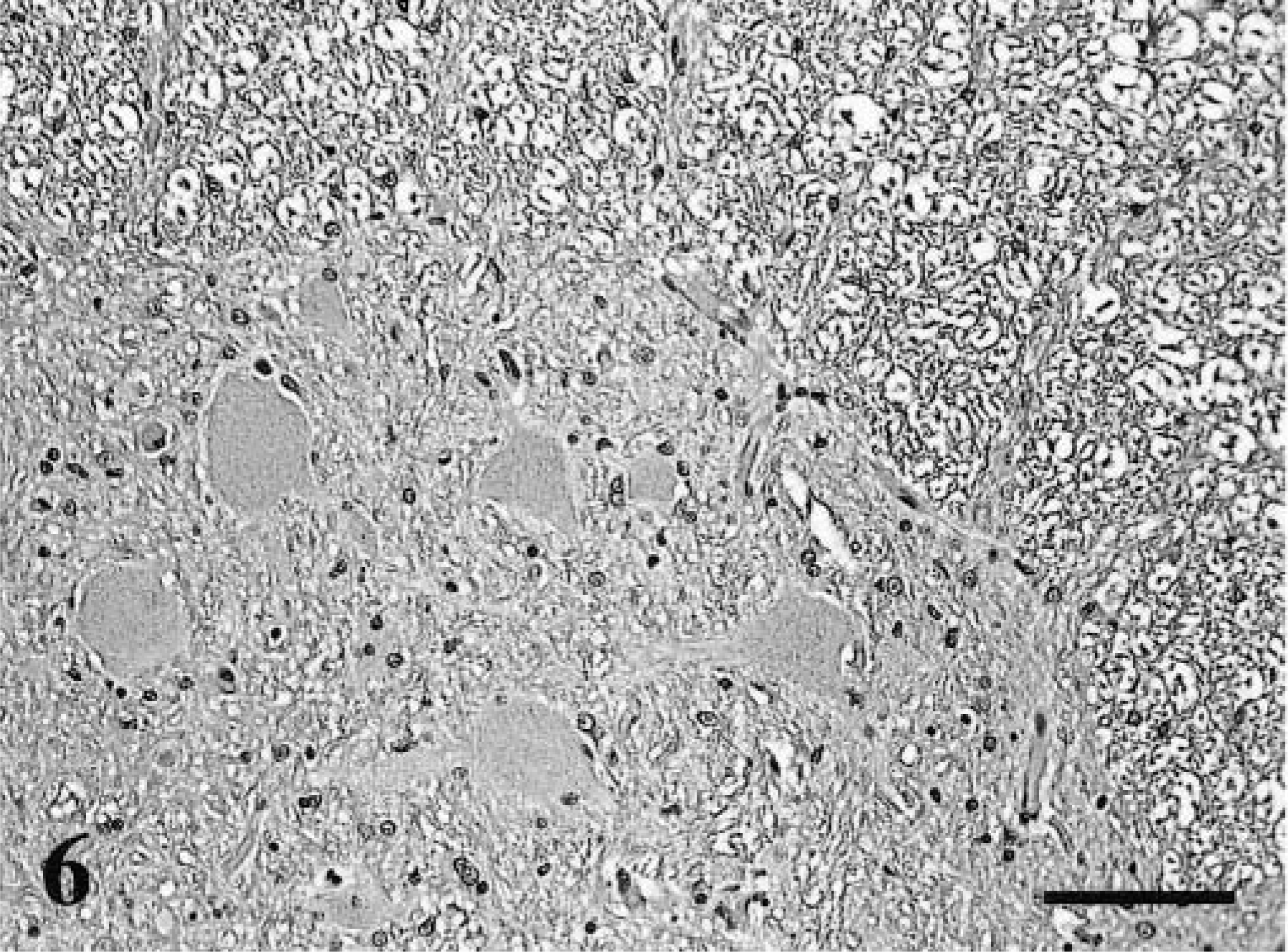
Spinal cord; FeLV-negative cat. There is no staining of neurons with anti-p27 antibody. DAB chromogen. Hematoxylin counterstain. Bar = 85 μm.
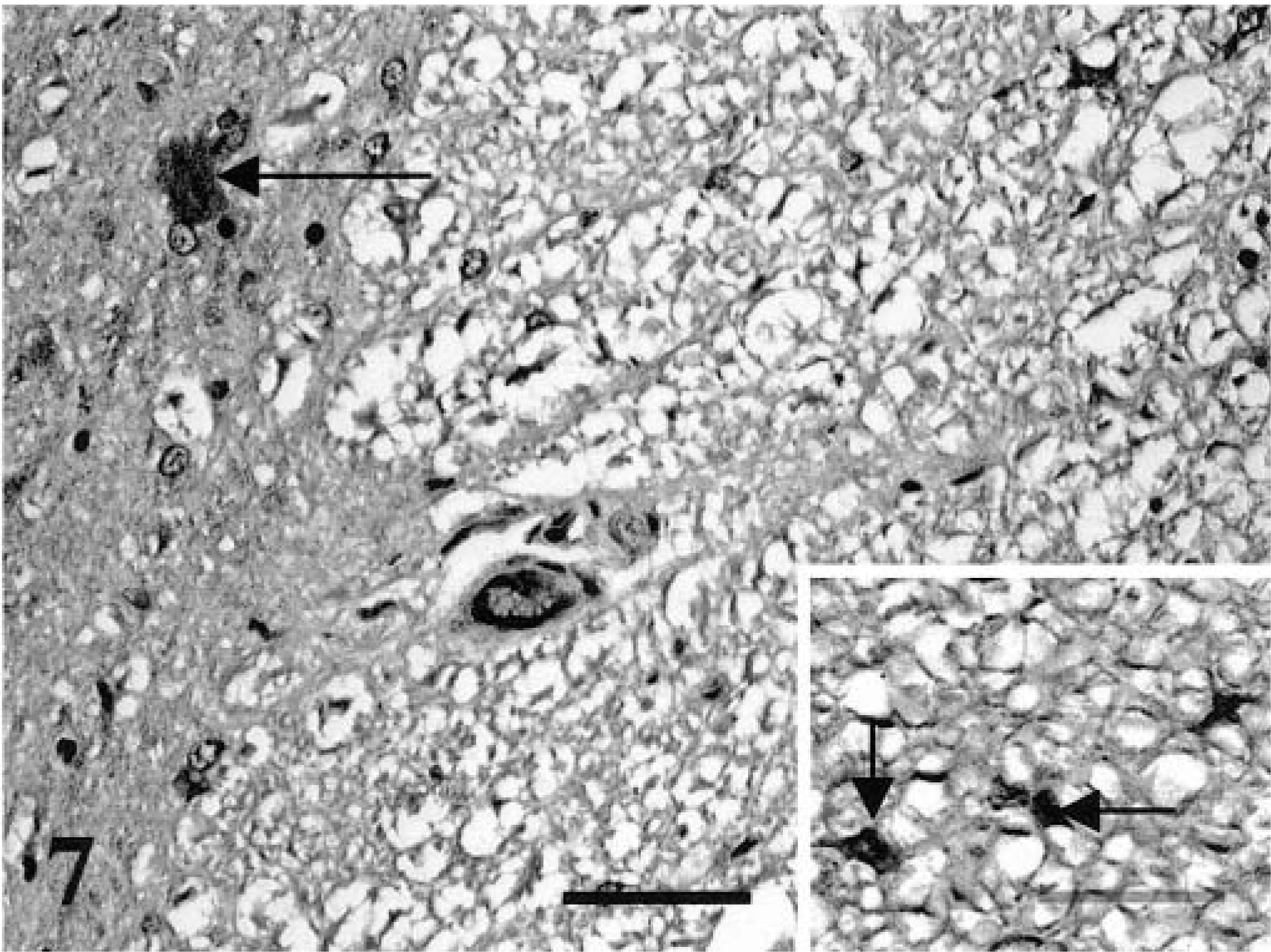
Spinal cord; FeLV-associated myelopathy–affected cat. Positive staining of endothelial cell (center), neuron (arrow), and glial cells (inset) with anti-p27 antibody. DAB chromogen. Hematoxylin counterstain. Bar = 85 μm. Inset: Bar = 40μm.
Results of FeLV detection by PCR and immunohistochemistry.
Relative staining of spinal cord cells with p27 immunohistochemistry.

+ = mild staining; ++ = moderate staining; +++ = intense staining; – = no staining.
Neurons and endothelial cells were readily identifiable on routine immunohistopathology. The identity of glial cells immunoreactive for anti-FeLV antigens was verified by staining replicate sections for glial fibrillary acidic protein (GFAP; astrocytes) and galactosylceramidase (GALC; oligodendroglia). Although both oligodendroglial cells and astrocytes expressed p27 protein, in general, sections of spinal cord stained with GFAP and GALC revealed that there were relatively fewer immunoreactive astrocytes than oligodendroglial cells.
PCR assay
Multiple samples including spinal cord, spleen, and bone marrow sections from affected cats contained amplifiable proviral sequences (Table 2). FeLV proviral DNA was extracted from bone marrow of seven cats (cat Nos. 1, 2, 6, 7, 8, 11, and 14). In five cats (cat Nos. 1, 2, 6, 7, 8, and 12), brain, spinal cord, spleen, and intestine were all positive, and in five cats (cat Nos. 3, 4, 5, 13, and 14) proviral DNA was found in spinal cord only. Lymph node was positive for proviral DNA in six cats (cat Nos. 1, 2, 6, 7, 8, and 12), and kidney was positive in only three cats (cat Nos. 1, 2, and 12). Cerebrospinal fluid was positive in only one cat (cat No. 16). DNA extracted from eyes consistently yielded negative results (Fig. 8).
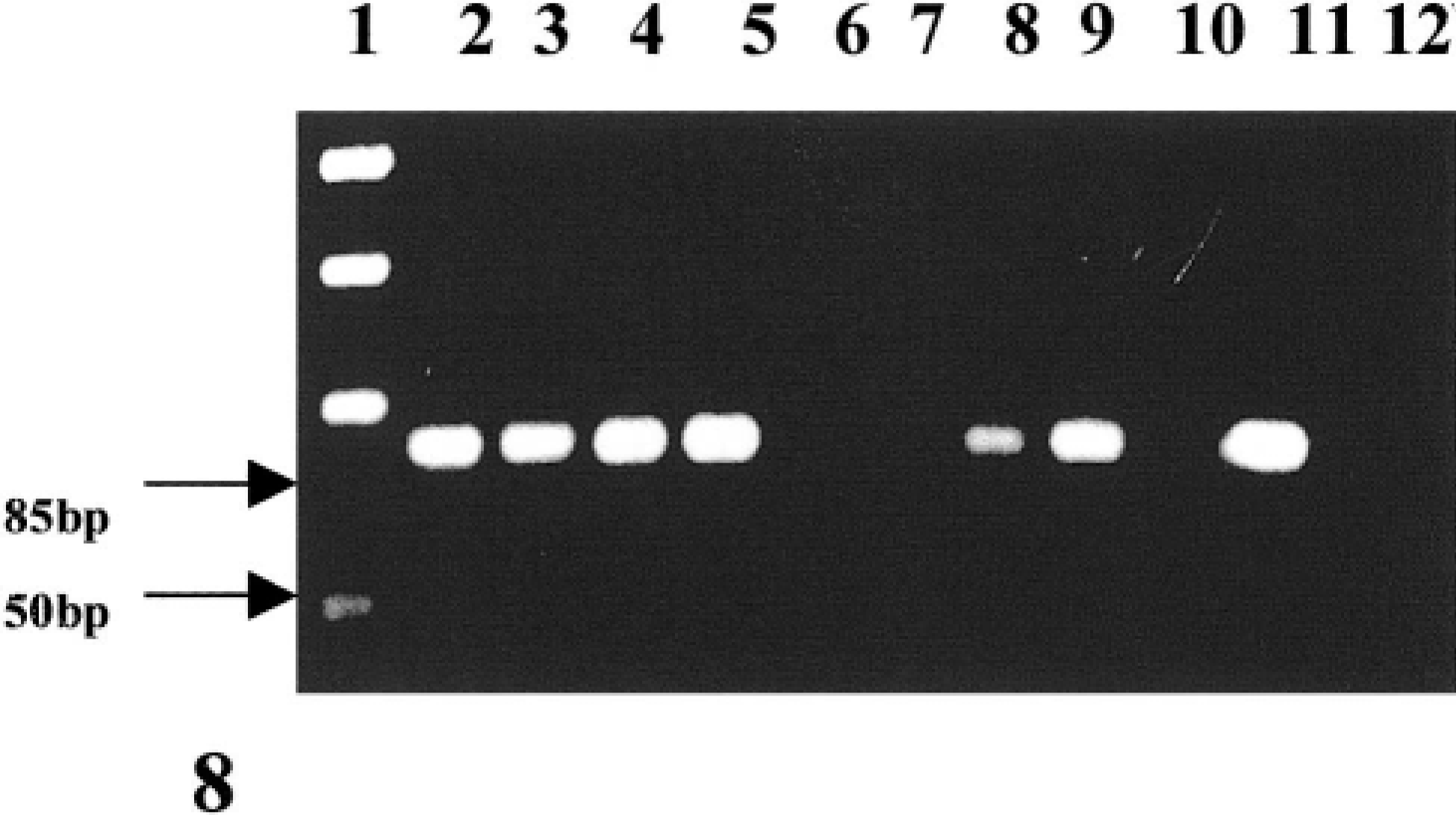
Electrophoretogram of PCR-amplified FeLV DNA. Lane 1, markers (50-bp ladder); 2–5, spinal cord; 6, 7, eye; 8, 9, intestine; 10, muscle; 11, positive control; and 12, negative control.
Discussion
In this report, we have described the clinical and pathologic features of a novel neurologic syndrome in FeLV-infected cats. FeLV is an oncogenic retrovirus associated with a wide range of diseases. Strong associations have been reported for T-cell lymphomas in young cats, pure red-cell aplasia in cats infected with subtype C, erythroid myeloproliferative disease, and a rapidly progressive immunodeficiency syndrome.20 Less well characterized are associations of FeLV infection with chronic diarrhea, infertility, and hematologic abnormalities such as neutropenia, macrocytic anemia, or both.19 Abnormal neurologic findings have been reported as a common feature in a retrospective survey of lesions observed at necropsy of FeLV-seropositive cats and in one case of degenerative myelopathy in a FeLV-seropositive cat.14,19 In addition, descriptions of behavioral and nerve conduction abnormalities in experimentally infected cats suggest that neurologic lesions are not an entirely new sequela of FeLV infection.4,9 However, detailed descriptions of either the clinical or pathologic characteristics have been lacking. Hind limb paresis progressing to paralysis within approximately 1 year was the most common clinical abnormality in affected cats. There was evidence of neither peripheral nerve deficits nor cortical or brain stem lesions. Lower motor neuron dysfunction manifested with urinary incontinence in two cats. Hematologic abnormalities typically associated with FeLV infection (anemia, macrocytosis, rubricytosis, neutropenia, macrothrombocytes, and thrombocytopenia) were noted in four of 12 cats.5,21 The hematologic changes were mild but likely a consequence of chronic FeLV infection of hematopoietic precursor cells. Lymphopenia noted in most of the cats in this study (10 of 12 cats examined) was attributed to steroid-induced leukocyte kinetic changes or immunosuppression resulting from cytopathic FeLV infection. Overt immunodeficiency, supported by the lymphopenia, may have resulted in systemic mycotic as well as protozoal infections in cat No. 8 and may have contributed to the extensive metazoan infections in cat Nos. 11 and 12.
In cases in which serologic testing was done, the time from the first seropositive test result until the appearance of neurologic signs ranged from 1 to 6 years with a mean of 3 years. This time span is probably an underestimate because the majority of cats were tested for FeLV infection only after clinical signs became apparent. Further, the average age of cats with known birth dates was 9 years, and FeLV infection is most commonly acquired at a young age.10 Tumors were not noted in any of the cats reported here. In summary, although clinical pathology abnormalities in this group of cats were either nonspecific or absent, the neurologic findings were striking.
Although there are etiologic agents that may induce myelitis and could result in the clinical signs observed, degenerative myelopathy in cats is a rare histopathologic diagnosis, typically of unknown etiology. The association of degenerative myelopathy with long-standing FeLV infection in all 16 cats reported here suggests that the lesions result from the viral infection.
In the cats of this study, glial cells (oligodendroglia and astrocytes) were frequently involved. In vivo infection of these cells with FeLV has not been previously reported; however, experimental infection of brain cultures with FeLV has been described.9 It is thus not surprising that these cells were frequently found to contain immunoreactive viral antigen. Endothelial cells were also commonly affected, and this has previously been described in vitro with a strain of recombinant FeLV.6 This immunoreactivity is possibly a reflection of the systemic viremia observed and may indicate one possible route of entry into the CNS. In the CNS, there is close association between capillary endothelial cells and astrocytes both physically and functionally in the blood-brain barrier, and it is possible that viral infection of the endothelial cells may have been transferred to astrocytes because of this relationship.7,12 It was somewhat surprising to identify viral antigen in neurons because direct in vivo infection of these cells has not previously been speculated to occur. But, in vitro experiments have shown FeLV-associated neuronal infection and damage.8,15 It is possible that the supportive role of the astrocytes to neurons and oligodendroglia in the CNS may have facilitated neuronal infection.
Viral infection of the CNS may have also occurred subsequent to an earlier episode of lymphocyte infiltration that receded before the onset of clinical signs. This is a common route of viral dissemination in many retroviral infections, and a recent histologic observation of perivascular lymphocytic cuffing in a euthanatized nonneurologic FeLV-positive cat by one of the authors (K. P. Carmichael) suggests that this is a possibility. The reason for the increased numbers of immunoreactive oligodendroglia as compared with astrocytes in this study is not known, but oligodendroglial infection could be responsible for the degeneration of axonal sheaths. Conversely, neuronal infection may directly result in a diffuse axonopathy or in calcium imbalances leading to neuronal dysfunction and death.
FeLV subtype A is the prototype of FeLV that is transmitted among cats through salivary contact. Other viral subtypes evolve from subtype A and are defined by interference with superinfection in indicator cell lines and interaction with different cellular receptors. FeLV subtype A will infect epithelial cells, lymphocytes, and a range of other cells and is minimally cytopathic. Infection restricted to the oronasal cavity with immune clearance of virus localized to cranial lymphoid tissues is thought to be the most common scenario subsequent to salivary exposure with FeLV. Cats infected by this route rapidly clear the virus and are presumed to never become antigenemic and thus to never test FeLV-antigen positive by commonly used serum enzyme-linked immunosorbent assay tests.14 The second most common sequel to oronasal infection results in viremia followed by a systemic immune response and clearance of virus after a more protracted time period. Cats in which this occurs will test antigen-positive in serum, and viral antigens can be detected by immunofluorescent labeling of bone marrow cells. It is thought that cell-associated virus is cleared before the disappearance of antigenemia.10 Finally, some FeLV-infected cats fail to mount an effective immune response and become persistently viremic or, on rare occasions, have persistent latent infection in select cell populations. Considering the time span between initial seropositivity, the development of clinical disease, and retesting, it is likely that the cats in this study were persistently viremic. Persistent replication of FeLV offers opportunities for the development of viral diversity through the acquisition of nucleotide changes or through recombination within the envelope gene of FeLV. The surface unit of the envelope gene defines receptor interaction, and therefore cell tropism. Pathogenic properties of the virus within specific target cell populations appear to result from initial infection with FeLV subtype A and subsequent evolution of viruses with enhanced ability to use an alternate receptor that is expressed in a cell-restricted manner.16 This concept was illustrated by the recent identification of an accessory cellular protein termed FeLIX, which is expressed and secreted by feline T-cells.1 FeLIX supports replication of a variant of FeLV subtype A that is T-cell–tropic and results in fatal immunodeficiency.1 Similarly, use of a cell-restricted receptor results in the development of pure red-cell aplasia in cats with a prorubricyte-tropic FeLV variant broadly classified as FeLV subtype C.17 This virus results in impairment of the transition of erythroid precursors from the burst-forming unit–erythroid (BFU-E) to the colony-forming unit–erythroid (CFU-E).17 The receptor used by this virus encodes an organic anion transporter protein that is speculated to be involved in the accumulation of heme compounds necessary for the synthesis of hemoglobin at the BFU-E to CFU-E transition stage.17 Another membrane transporter protein, Pit1, involved in the movement of phosphate across cell membranes, is the receptor used by FeLV subtype B.18 FeLV-B is a variant of FeLV subtype A that arises because of recombination with portions of endogenous cat env genes, and it has the unique ability to infect many different mammalian cells.3 This virus variant containing truncations or mutations in the Env protein is present in many FeLV-associated malignancies affecting lymphoid cells.13 The syndrome described in this report does not bear resemblance to typical diseases associated with FeLV subtypes B, C, or T. Thus, presumptively, the virus may still be of subtype A, or alternatively, a new variant with particular neurotropism may be involved. Studies are underway to characterize the viral variant involved in this feline degenerative myelopathy. The histopathologic changes observed are similar to those described in human T-cell leukemia virus (HTLV)-1–associated myelopathy or tropical spastic paresis.2 Infection with this human retrovirus induces T-cell leukemia/lymphoma in 20–50% and myelopathy in approximately 5% of infected individuals.2 Inflammatory cells in the spinal cord are present transiently during the active/chronic stage, and lesions during the chronic stage of the disease consist predominantly of degeneration of the lateral and dorsal spinal tracts and endothelial fibrosis.2 Whether the pathogenesis or epidemiology of the feline myelopathy described in this report is similar to HTLV-associated myelopathy in humans is currently unknown.
Footnotes
Acknowledgements
We would like to thank Roberta Mihut, Amanda Hobe, and Dr. James Stanton for technical assistance. We also appreciate feline spinal cord samples sent to us by Dr. Patricia Blanchard and Dr. Thomas Van Winkle. This study was funded by a grant from the University of Georgia Veterinary Medical Experiment Station.
