Abstract
Ocular involvement in systemic diseases is frequent in cats; however, without concurrent clinical and ophthalmic examinations with gross and/or histologic analysis of the eye, these findings can be underdiagnosed. This article aims to provide gross, histologic, and immunohistochemical characteristics of ocular lesions from cats submitted to necropsy, focusing on those caused by systemic infectious agents. Cats that died due to a systemic infectious disease were selected based on necropsy diagnosis and presence of ocular lesions. Gross, histologic, and immunohistochemical findings were recorded. From April 2018 to September 2019, 849 eyes of 428 cats were evaluated. Histologic abnormalities were seen in 29% of cases, which were classified as inflammatory (41%), neoplastic (32%), degenerative (19%), and metabolic/vascular (8%). Macroscopic changes were present in one-third of eyes with histologic lesions. Of these, 40% were attributed to inflammatory or neoplastic diseases associated with infectious agents. The most important infectious agents causing ocular disease in this study were feline leukemia virus, feline infectious peritonitis virus, and Cryptococcus sp. The most common ocular abnormalities associated with infectious agents were uveitis (anterior, posterior, or panuveitis), optic neuritis, and meningitis of the optic nerve. Ocular lesions secondary to systemic infections in cats are frequent; however, these are not always diagnosed because gross lesions are less common than histologic lesions. Therefore, both gross and histologic evaluation of the eyes of cats is recommended, mainly for cases in which the clinical suspicion or necropsy diagnosis suggests that an infectious agent might be related to the cause of death.
Keywords
Ocular involvement in systemic diseases is frequent in cats; however, these findings can be underdiagnosed without concomitant clinical and ophthalmic examinations with macroscopic and histopathological examination of the eye.1,10 In addition, studies have demonstrated that a systemic cause may contribute to almost 70% of cases of uveitis in cats. 20 Several systemic diseases caused by infectious, vascular, metabolic, immune-mediated, and neoplastic etiologies can involve the eye, frequently resulting in uveitis. 10 Of the infectious agents that involve the eye and can cause systemic diseases, viruses, bacteria, fungi, and protozoa have been reported, and the importance of each agent varies according to the study.10,20,25
Retrospective studies characterizing ocular lesions in different domestic species can be found in the literature.6,14,15 However, these studies evaluated eyes with gross lesions and/or a previous history of ocular disease. Studies evaluating the ocular involvement in cats submitted for necropsy without evident macroscopic lesions to identify the true incidence of ocular lesions in cases of systemic infectious diseases are lacking in the literature. This study aims to characterize ocular lesions caused by infectious agents of cats submitted to necropsy through macroscopic, histological, and immunohistochemistry (IHC) evaluation. We hypothesize that the ocular lesions secondary to systemic infections are more frequent than what is described in the literature, and that significant histologic lesions are not necessarily apparent on gross examination.
Materials and Methods
The characterization of ocular involvement in systemic infectious diseases was based on the gross, histologic, and immunohistochemical examination of both eyes of all cats submitted for necropsy between April 2018 and September 2019. All cats from this study were housed in the metropolitan area of Porto Alegre, Rio Grande do Sul, Brazil. Exclusion criteria included advanced autolysis, putrefaction, or extensive trauma involving the head, which prevented the proper evaluation of both eyes.
Samples of multiple organs were fixed in 10% formalin during necropsy. The eyes were collected separately, identified as left and right, and placed in 10% formalin for ulterior gross evaluation. Depending on the size of the eye, 0.5 to 1 mL of 10% formalin was injected intravitreally with a 1 mL syringe and 26 g hypodermic needle positioned laterally to the optic nerve and parallel to the posterior ciliary arteries. The eyes were then fixed for 24 hours and, after that, a macroscopic evaluation was performed. First, we evaluated the eyes with the extraocular tissue to identify possible extraocular lesions. If lesions were absent, the extraocular muscles and palpebra were trimmed off. Then, through palpation and visual examination using a source of light, we examined the eye to identify possible intraocular masses. The eyes were sectioned through a vertical cut, perpendicular to the posterior ciliary arteries and adjacent to the optic nerve, on a vertical plane. The position of the lens, vitreous consistency, and presence of intraocular lesions were evaluated and, when present, gross lesions were photographed and described. Tissues were then placed in mega-cassettes (Tissue-Tek, Sakura Finetek USA, Torrance, CA, USA), routinely processed for histology, embedded in paraffin, cut in 3-µm thick sections, and stained with hematoxylin and eosin (HE).
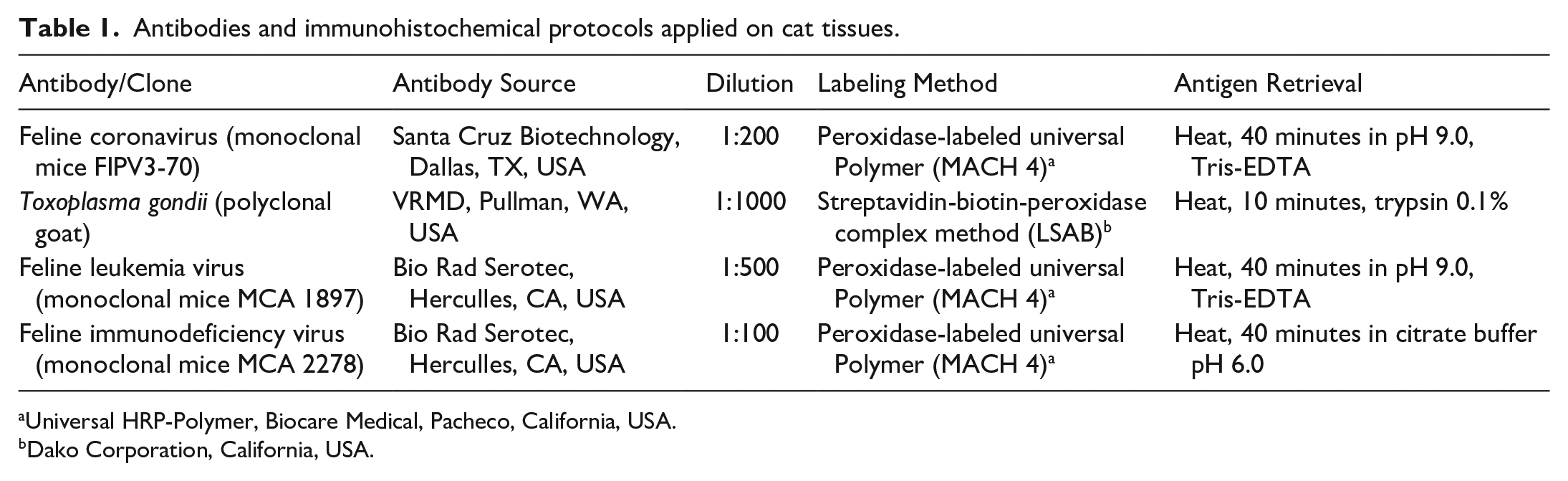
Clinical histories, when available, sex, age, viral status for feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV; SNAP FIV/FeLV Combo Test Product Insert, IDEXX Laboratories, Westbrook, ME, USA), necropsy diagnosis, macroscopic lesions, and histologic findings of all organs, including the eyes, were evaluated for all cats. In cases where FIV and FeLV status was not available per history, IHC was performed on bone marrow to investigate it (Table 1). Cases were grouped according to the etiological agent attributed to the cause of death and the presence of histologic lesions in the eye. The histologic slides from all eyes were reviewed by 2 pathologists (J.G.W. and L.S.).
Antibodies and immunohistochemical protocols applied on cat tissues.
Universal HRP-Polymer, Biocare Medical, Pacheco, California, USA.
Dako Corporation, California, USA.
Microscopic lesions of all collected eyes were described, and the etiological agent was characterized using histochemical and IHC techniques, when possible, in cases where the necropsy diagnosis involved an infectious agent. The intensity of the lesions was characterized as mild, moderate, or severe based on the number and extension of affected intraocular structures, the severity of inflammation, the invasiveness of neoplastic cells, the number of infectious agents when applicable, and the presence of secondary changes, such as the proliferation of fibrovascular membranes, synechia, edema, retinal detachment, secondary glaucoma, cataract, and other intraocular lesions.
Histological sections from the eyes with microscopic lesions possibly related to an infectious agent were subjected to IHC. Samples from cats with a necropsy diagnosis of leukemia or lymphoma that were positive for FeLV and/or FIV and showed inflammatory/neoplastic cells intraocularly were tested with an anti-FeLV and anti-FIV antibodies. Cats that were diagnosed with feline infectious peritonitis (FIP; based on the presence of protein-rich effusions in body cavities and/or small nodular lesions in multiple organs and histologic lesions characterized by fibrinous, granulomatous, or pyogranulomatous inflammation in multiple organs, mainly phlebitis and periphlebitis), and showed intraocular lesions characterized by inflammation in any portion of the eye were tested with anti-feline coronavirus (FeCO), anti-FeLV, and anti-FIV antibodies. Cats diagnosed with cryptococcosis (based on the presence of yeasts in the central nervous system (CNS), lungs, skin, and/or other organs, confirmed by Alcian Blue, and variable degree of inflammation) were tested with an anti-FeLV and anti-FIV antibodies. Cats diagnosed with systemic toxoplasmosis (based on the presence of the infectious agent associated with inflammation in multiple organs and confirmed by IHC) and lesions consistent with anterior uveitis were tested against anti-Toxoplasma gondii, anti-FeLV, and anti-FIV antibodies. IHC with FeLV- and FIV-antibodies was also performed in all cases of bacterial systemic disease with ocular involvement. The antibodies, dilution, and retrieval methods are presented in Table 1. All antibodies were incubated overnight. The chromogen 3-amino-9-tilcarbazoln (AEC Romulin, Biocare Medical, Pacheco, CA, USA) was applied in all IHC for 2 minutes at room temperature, and slides were counterstained with Harris hematoxylin for 15 seconds. Positive (infected tissues confirmed with a secondary assay) and negative (controls without primary antibody) controls were used for all IHC protocols.
When histological intraocular lesions suggested the involvement of bacteria or fungi, sections were submitted to the following histochemical techniques: Alcian Blue (pH 2.5), Periodic Acid-Schiff (PAS), and/or Grocott methenamine silver stains for fungi; and modified Brown and Hopps Gram stain and/or Ziehl-Neelsen stain when the lesions were suggestive of bacterial agents.
The interpretation of the collected data was based on number of cases.
Results
During the study period, 451 necropsies of cats were performed and the eyes of 428 cats were evaluated, totaling 849 eyes. Both eyes of 23 cats were excluded due to advanced postmortem changes or extensive trauma involving the head, precluding proper ocular evaluation. Only one eye was available for 7 cases due to previous enucleation or unilateral extensive trauma. A total of 132 cats were previously frozen when submitted to necropsy, and most of these were in mild to moderate degree of autolysis when necropsied.
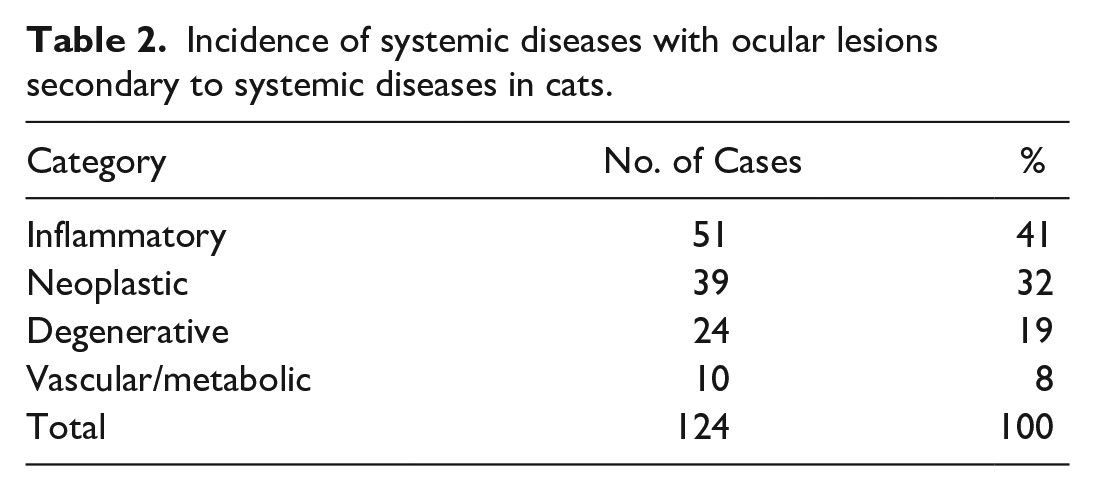
Microscopic lesions were observed in 124 of 428 evaluated cats (29%), and only a third of these cases had gross abnormal findings at necropsy (43 of 124). Microscopic lesions (Table 2) were observed in one or both eyes and were classified as inflammatory (51 of 124, 41%) and neoplastic (39 of 124, 32%), which included both infectious and noninfectious causes. In addition, lesions characterized as degenerative (24 of 124, 19%) and vascular/metabolic (10 of 124, 8%) were also observed, but not included in this article as they did not involve an infectious etiology.
Incidence of systemic diseases with ocular lesions secondary to systemic diseases in cats.
Infectious agents were involved in 49 of 124 (40%) cases with histologic lesions, and noninfectious lesions represented 75 of 124 (60%) cases. Among them, 22 of 49 (45%) cases were exclusively FeLV-induced, 10 of 49 (20%) were attributed to feline infectious peritonitis virus (FIPV), 5 of 49 (10%) to Cryptococcus sp., and 12 of 49 (24%) were caused by other infectious agents such as bacteria and T. gondii. In addition, coinfection of FeLV and/or FIV with another etiology leading to ocular lesions was observed for 15 cats. No ocular lesions were attributed to FIV alone.
The most frequently diagnosed infectious disease at necropsy with ocular involvement was hematopoietic neoplasia induced by FeLV. From all cats evaluated during the study period, 55 were diagnosed with leukemia or lymphoma induced by FeLV, and 22 of 55 of these cats (40%) presented with ocular lesions secondary to the systemic disease. The necropsy diagnosis, in addition to macroscopic and histologic lesions, is shown in Supplemental Table S1.
Most cats with ocular involvement were diagnosed with acute myeloid leukemia or multicentric lymphoma. Only one animal with leukemia or lymphoma had gross changes in the eye, and the main histological lesions in all cases were aggregates of lymphocytes (neoplastic or inflammatory) multifocally infiltrating the uvea, surrounding the scleral venous plexus (Fig. 1a) and/or the vessels around the optic nerve (Fig. 1b), and, more rarely, expanding the uvea. The inflammatory/neoplastic component was characterized based on cell morphology, and in cases of lymphocytic leukemia and lymphoma, the lymphocytic infiltrate could not be differentiated between inflammatory or neoplastic unless it formed nodules expanding the uvea (case 12). Both populations showed similar morphologic appearances (small lymphocytes) and variably immunolabeled with anti-FeLV IHC. In cases of acute myeloid leukemia, the differentiation from neoplastic and inflammatory cells was possible based on cell morphology, and although not all neoplastic cells were positive on IHC, the lesion was categorized as secondary to systemic infection if cells were positive.
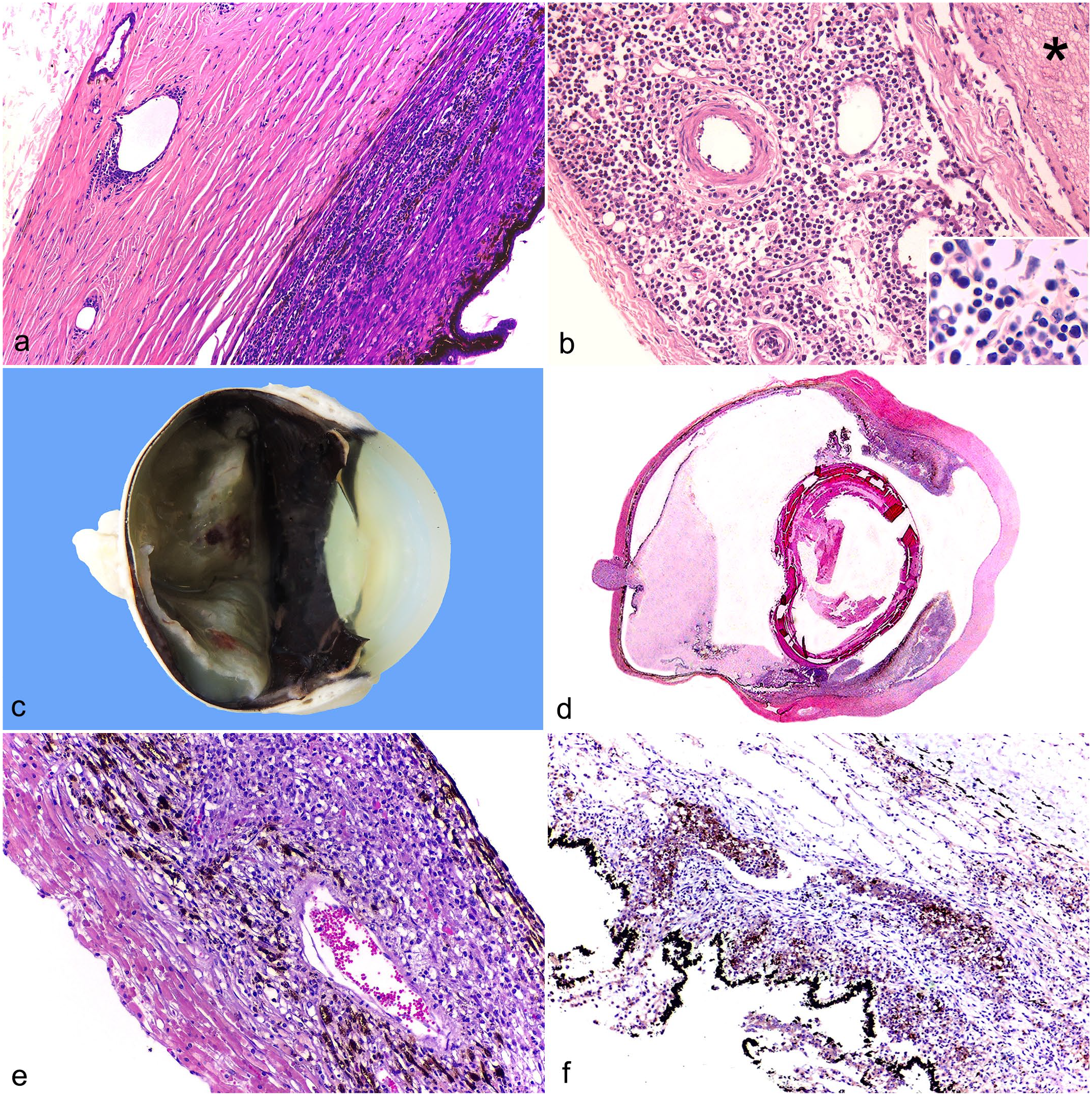
Ocular involvement of feline leukemia virus (FeLV) and feline infectious peritonitis (FIP). (a) Infiltration of small lymphocytes in the ciliary body and around the scleral venous plexus in a FeLV-positive cat diagnosed with lymphocytic leukemia, hematoxylin and eosin (HE). (b) Infiltration of neoplastic myeloid cells surrounding vessels in the meninges of the optic nerve (*) in a FeLV-positive cat with myeloid leukemia, HE. Inset: higher magnification of the neoplastic myeloid cells. (c) Deposition of white material in the vitreous and in the subretinal space (retinal detachment), as well as in the anterior chamber in a cat with FIP. (d) Submacroscopic image of Fig. 1c, showing deposition of proteinaceous material in the vitreous, in addition to a robust preiridal fibrovascular membrane, post-iridal, and cyclitic fibrovascular membranes associated with inflammatory infiltrate, HE. (e) Pyogranulomatous posterior uveitis, HE. (f) Labeling to feline infectious peritonitis virus in the inflammatory cells infiltrating the uveal tissue, immunohistochemistry anti-feline coronavirus, AEC Romulin, Harris hematoxylin.
During the research period, 21 cats were diagnosed with FIP, of which 10 (48%) had ocular lesions. Cases of effusive and noneffusive FIP were equally distributed. Only 4 of 10 (40%) of the cats with ocular involvement presented with gross lesions, which were always unilateral and were mainly characterized by deposition of solid, white material in the ocular chambers with retinal or choroidal detachment (Fig. 1c, d). Data on the macroscopic characterization of the systemic disease, the gross and histologic changes, immunohistochemical labeling in the eye, and FeLV and FIV status are shown in Table 3.
Cases of feline infectious peritonitis with ocular involvement.
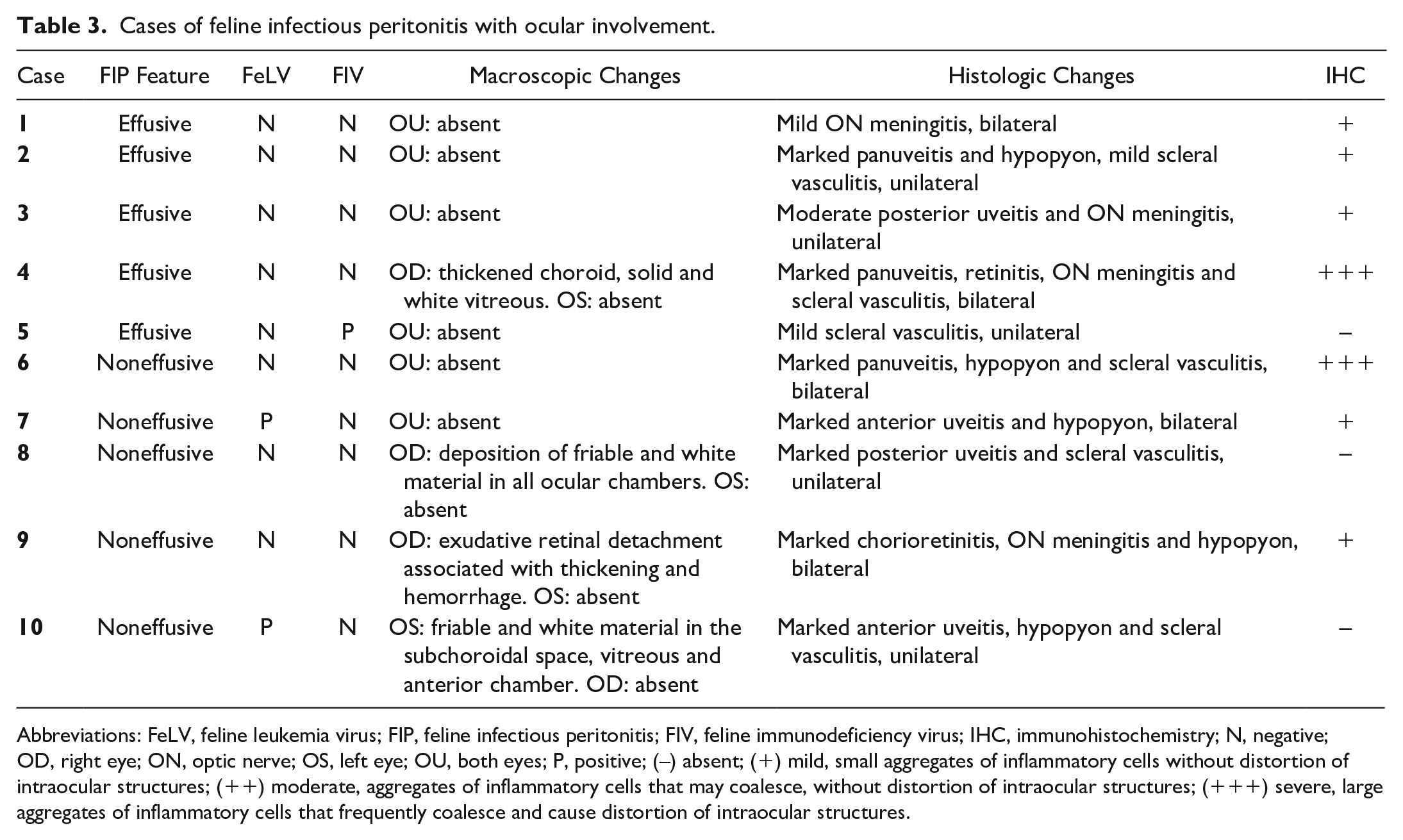
Abbreviations: FeLV, feline leukemia virus; FIP, feline infectious peritonitis; FIV, feline immunodeficiency virus; IHC, immunohistochemistry; N, negative; OD, right eye; ON, optic nerve; OS, left eye; OU, both eyes; P, positive; (–) absent; (+) mild, small aggregates of inflammatory cells without distortion of intraocular structures; (++) moderate, aggregates of inflammatory cells that may coalesce, without distortion of intraocular structures; (+++) severe, large aggregates of inflammatory cells that frequently coalesce and cause distortion of intraocular structures.
Histologically, the main lesion of ocular FIP was neutrophilic and histiocytic panuveitis (Fig. 1e), sometimes associated with meningitis of the optic nerve and fibrin in the ocular chambers (Fig. 1d). The immunohistochemical labeling varied from mild (scattered positive cells) to marked (numerous positive cells) and was mainly in the macrophages infiltrating the uvea (Fig. 1f). From all cats diagnosed with FIP, 8 showed involvement of the CNS (8 of 21), and the eye was involved in 7 of these cases. In addition, 3 of 10 (30%) cats diagnosed with FIP and with concomitant ocular involvement were positive for FeLV and/or FIV, although the severity of ocular lesions was not correlated with the positivity for either virus.
The third most common disease with ocular involvement diagnosed in this study was cryptococcosis. Information regarding systemic involvement, intraocular lesions, characteristics of the inflammatory infiltrate, and FeLV and FIV status is shown in Table 4. Six cases of cryptococcosis were diagnosed during the study period, and 5 of them (83%) exhibited microscopic ocular lesions. None of the eyes presented with gross lesions. All cats with the CNS involvement showed microscopic ocular lesions, characterized by mild (only a few neutrophils and macrophages) to moderate (moderate number of macrophages and neutrophils that multifocally effaced the affected structure) pyogranulomatous optic neuritis (Fig. 2a) and/or chorioretinitis associated with a variable amount of round to oval structures, which were morphologically compatible with Cryptococcus sp. (Fig. 2b). The polysaccharide capsule was highlighted by the Alcian blue stain (Fig. 2c) and PAS reaction. Furthermore, 2 of 5 (40%) of these cases were concomitantly positive for FeLV.
Cases of feline cryptococcosis diagnosed in the studied period.

Abbreviations: FeLV, feline leukemia virus; FIV, feline immunodeficiency virus; N, negative; P, positive; (–) absent, (+) mild, small aggregates of inflammatory cells without distortion of intraocular structures; (++) moderate, aggregates of inflammatory cells, that may coalesce, without distortion of intraocular structures; (+++) severe, large aggregates of inflammatory cells, that frequently coalesce, and cause distortion of intraocular structures.
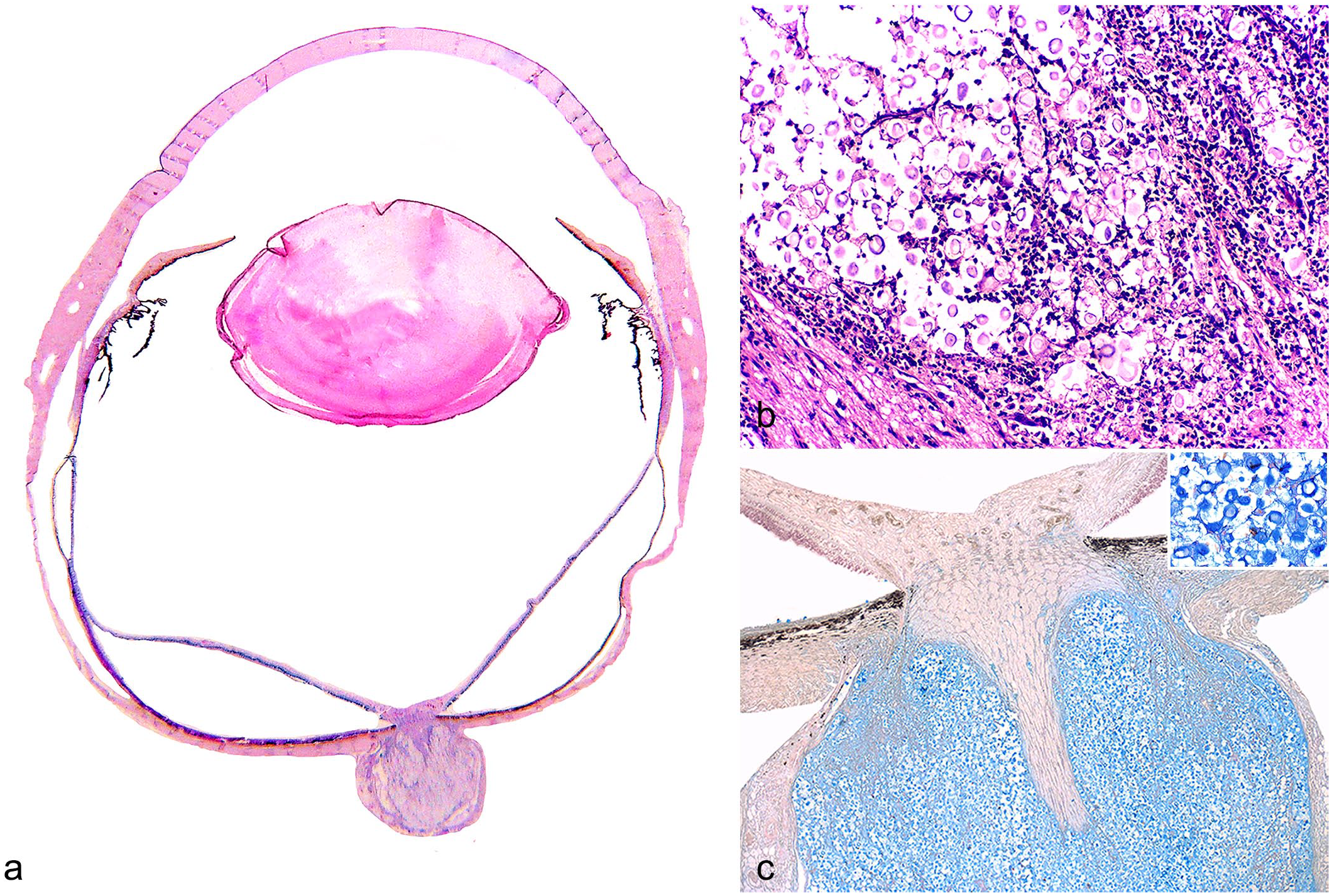
Feline ocular cryptococcosis. (a) Eye from a cat with cryptococcal lesion in the optic nerve, hematoxylin and eosin (HE). (b) Effacement of the optic nerve by fungal spores morphologically compatible with Cryptococcus sp. and pyogranulomatous inflammatory infiltrate, HE. (c) Multiple spores with a thick mucopolysaccharide capsule highlighted by the Alcian blue staining. Inset: higher magnification of the fungi.
Ocular lesions characterized by anterior uveitis and/or lymphocyte aggregates around the scleral venous plexus were observed in 12 cats that died of other systemic infections. Among these diseases, bacterial pneumonia (8 of 22 presented with ocular lesions), lymphoplasmacytic meningoencephalitis (2 of 3 presented with ocular lesions), bacterial colitis (1 of 3 presented with ocular lesions), and toxoplasmosis (1 of 1 presented with ocular lesions) were diagnosed. IHC for T. gondii was negative in the ocular tissue, but positive in systemic lesions in the case of systemic toxoplasmosis. In most cases of systemic bacterial infection, the etiological agent could not be identified. Gram and ZN stains were performed in the ocular tissue in cases of bacterial systemic infection, and the results were negative in the eye. Of the cats with ocular involvement, 10 of 12 (83%) were also positive for FeLV and/or FIV.
Discussion
Ocular lesions secondary to systemic disease are common, and sometimes ocular clinical signs precede systemic signs of these diseases. 10 In this study, 29% of the evaluated cats had histologic ocular lesions, classified as inflammatory, neoplastic, degenerative, or vascular. From these cases, 40% were due to inflammatory conditions, such as FIP or Cryptococcus sp., with or without associated immunosuppressive agents such as FeLV and/or FIV, or neoplastic processes of infectious origin caused by FeLV.
FeLV is a gamma-retrovirus associated with a variety of degenerative and proliferative diseases in cats and is highly prevalent in Brazil.3,11 The main ocular manifestation of FeLV infection is the development of lymphomas, predominantly involving the anterior uvea.8,26 In the cases described in the present study, the main ocular manifestations varied from inflammatory/neoplastic cells infiltrating the anterior uvea, to the formation of neoplastic nodules expanding the iris, as described in the literature.25,26 Furthermore, lymphocytic aggregates around the scleral venous plexus, and occasionally surrounding vessels from the meninges of the optic nerve, were frequently observed, which has not been described in the literature.
The differentiation of inflammatory and neoplastic infiltrates was possible in cases of acute myeloid leukemia due to the morphological features of the neoplastic cells. However, it was not possible to differentiate them in cases of lymphocytic leukemia and lymphoma with histology alone, unless there was obvious formation of a neoplastic nodule. Although IHC with anti-FeLV antibodies was performed in all cases with ocular involvement in this study, because FeLV inserts its DNA into the cellular genome, 23 neoplastic and non-neoplastic lymphocytes may be positive for the virus, which was observed in this study. Moreover, some authors suggest that the early stages of ocular lymphoma can manifest as uveitis, 21 which could be the explanation for these cases, although we could not draw a solid conclusion from these data. For this reason, further investigations are needed to differentiate inflammatory lymphocytic infiltrate from neoplastic lymphoid infiltrate in cases similar to these, as well as to clarify whether uveitis can progress and evolve into lymphoma.
Although not as widespread nor as severe as FeLV, 8 the lentivirus FIV can occasionally lead to the development of ocular lesions characterized by anterior uveitis and secondary lens luxation and glaucoma. In addition, immunosuppression promoted by this virus can lead to the development of secondary ocular infections.10,21 No ocular lesions that were exclusively due to FIV infections were identified in this study. However, coinfections were frequently observed with FeLV and with other infectious agents.
In the present study, most cats diagnosed with systemic bacterial diseases and ocular involvement were FeLV and/or FIV positive. In addition, coinfection with Cryptococcus spp. and one or both of these retroviruses led to at least moderate lesions in the eye in these cases. The most clinically important consequence of FeLV and FIV infection is severe immunosuppression. For this reason, the affected cats are prone to the development of systemic infections, or these infections can be exacerbated due to the retrovirus infection in both inflammatory cells and bone marrow.3,8,10,21,26 Thus, viral-mediated immunosuppression could have facilitated and/or aggravated the involvement of ocular tissues in these cats.
FIP is one of the most important infectious diseases of cats, with high prevalence and lethality. 9 In addition, FIP is an important cause of endogenous uveitis, 25 and it was equally relevant in this study. Of all the organs, the kidneys, brain, and eyes are the most commonly affected by the FIP virus.9,16 One study characterizing ocular lesions caused by FIP virus showed that 29% of FIP cases affected the eye, and these lesions were bilateral in 68% of the cases.9,16 Herein, the number of cats with histologic ocular involvement was higher, and most cases had bilateral changes, although the lesions were not always of the same severity in both eyes.
Macroscopic lesions were observed in less than half of FIP cases with histologic ocular lesions in this study, and they were always unilateral. Although there are no studies that compared the number of cases with gross lesions with those with histologic lesions, the results from this study corroborate the report by Pedersen, 16 who stated that ocular involvement in cases of FIP is usually underestimated. 16 Histologically, ocular FIP in the current study was characterized by anterior and/or posterior uveitis; vasculitis in the scleral venous plexus; inflammatory infiltrates associated with free fibrin in the anterior, posterior, and vitreous chamber; and meningitis, which sometimes extended to the nervous tunic, agreeing with what is described in the literature.9,10,16,21,25,27
In half of the cases with ocular FIP, the systemic disease was characterized as noneffusive. This information differs from what is described in the literature, which suggests that inflammatory ocular manifestations are more frequently associated with noneffusive cases.9,16,21,27 In almost all the cases with CNS involvement, the eyes were also affected, which is consistent with prior publications that suggest that the eye is almost always affected when there are lesions in the brain.9,16,21,27
Cryptococcosis is a fungal disease caused by basidiomycete yeasts belonging to the C. neoformans-C. gatii species complex and is considered the main systemic mycosis of cats.7,13,17 This disease has different clinical presentations, including the nasal, nervous, cutaneous, and systemic forms. In the eye, it results in uveitis, chorioretinitis, and more commonly, optic neuritis.7,10 Uveitis and chorioretinitis suggest hematogenous dissemination as the route of infection, and optic neuritis indicates extension from CNS.10,17,22 In this study, almost all cats diagnosed with cryptococcosis also presented with ocular lesions. More than half of them had meningitis and optic neuritis, and 1 had only chorioretinitis. The case with only chorioretinitis was the only one with ocular involvement without involvement of the CNS, corroborating the probable hematogenous dissemination resulting in the ocular lesions described in the literature.17,22 Similarly, all cats with CNS involvement developed ocular lesions, suggesting dissemination through the optic nerve.17,22 Contrary to the literature, which describes gross lesions characterized by an exudative retinal detachment, fibrin deposition, and hemorrhage on the anterior and posterior segment of the eye,10,17,22 no eye evaluated in this study presented with gross ocular lesions. This highlights the importance of histologic evaluation of the eye in cases of cryptococcosis, even if there are no gross lesions observed.
Bacterial diseases in several organs can result in ocular lesions secondary to septicemia in humans and animals, and this is often characterized by uveitis.2,6,12,19 In almost half of the cases of bacterial pneumonia diagnosed in this study, secondary ocular involvement was observed, with features similar to the lesions described in the literature.6,12,19
One neonatal cat diagnosed with systemic toxoplasmosis also presented with lymphoplasmacytic anterior uveitis. In cats, toxoplasmosis is generally subclinical, but it can cause fatal disease at any age and cause lesions in several organs, including the eye. 4 In the eye, the main lesions are granulomatous to lymphoplasmacytic chorioretinitis and iridocyclitis, which is usually multifocal.4,5,18 Several studies suggest that toxoplasmosis is one of the main causes of lymphoplasmacytic anterior uveitis in cats, even though the protozoan is not always seen within the ocular tissue. 5 In this case, the protozoan was identified by HE and IHC in several organs, although no organisms were identified in ocular tissues. The absence of positive labeling within the eye in cases of anterior uveitis due to toxoplasmosis is also described in the literature, and several theories have been proposed to explain the lack of identifiable organisms, such as cellular mimicry and ocular homing of effector immune cells. Other possible explanations are that the number of protozoa in the eye was too low or that the organisms are located in adjacent sections of the eye that were not evaluated. However, the role of Toxoplasma in the etiology of feline anterior uveitis is still unclear. 5
The main limitations of this study are related to the autolytic and putrefactive process and the quality of sampled eyes. Although cats with advanced postmortem changes were excluded, some eyes included in the study were from frozen animals and/or from animals presenting moderate autolysis, which hindered the evaluation of intraocular structures and the identification of subtle intraocular lesions. Furthermore, the process of autolysis may have limited the efficacy of some of the IHC analyses performed, such as for FeLV, FIV, and FIPV, as the degree of autolysis can alter tissue immunoreactivity even in formalin-fixed tissues. 24 When it is not possible to perform the necropsy right after the death of the animal, it is advised to remove both eyes and insert them into formalin or other fixative before storing the cadaver into a refrigerator or freezer, to better preserve the ocular architecture.
In this study, the most common infectious diseases with secondary ocular involvement were FeLV-induced hematopoietic neoplasia, FIP, and cryptococcosis, which mainly affected the uvea and optic nerve. This study further highlights the importance of FeLV and FIV coinfection in the development of ocular lesions, as these retroviruses are immunosuppressive agents that can predispose to and exacerbate systemic infections. Furthermore, most cases with ocular lesions did not show gross changes. Therefore, it is believed that ocular lesions secondary to systemic infectious processes are, in general, underdiagnosed, as gross lesions are not always present at the time of necropsy; hence, the eyes are not always histologically examined. For this reason, the evaluation of the eyes from cats, mainly in cases in which clinical and necropsy findings indicate the involvement of an infectious agent, is recommended. Finally, as not all ocular lesions are readily visible clinically, it is advised that any cat with systemic lesion should have their eyes examined for possible intraocular abnormal findings.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231158075 – Supplemental material for Ophthalmic and immunopathological characterization of systemic infectious diseases in cats
Supplemental material, sj-pdf-1-vet-10.1177_03009858231158075 for Ophthalmic and immunopathological characterization of systemic infectious diseases in cats by Júlia G. Wronski, Bianca S. de Cecco, Jacqueline Raiter, Luan C. Henker, Cíntia de Lorenzo, Marcele B. Bandinelli, David Driemeier, Saulo P. Pavarini and Luciana Sonne in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank to the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for scholarship funding.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
