Abstract
Coiled-coil domain containing 85c (Ccdc85c) is a causative gene for genetic hydrocephalus found in hemorrhagic hydrocephalus (hhy) mice. The Ccdc85c knockout (KO) rat has subcortical heterotopia with frequent brain hemorrhage as seen in hhy mice. In this study, we report aberrant alpha-smooth muscle actin (α-SMA) expression in the wall of lateral ventricle of the Ccdc85c KO rats. The α-SMA-positive cells were distributed at the dorsal, medial, and lateral regions of the lateral ventricle of KO rats. The expression of α-SMA was first observed on postnatal day 20 (P20) and became noticeably stronger at P26 when hydrocephalus was prominent. Double immunofluorescence showed co-expression of α-SMA with nestin, vimentin, and glial fibrillary acidic protein in the ventricular lining of KO rats. Therefore, we conclude that α-SMA-positive cells may represent an immature subpopulation of cells at adult age around the lateral ventricle of Ccdc85c KO rats.
Hydrocephalus is a neurological disorder caused by abnormal cerebrospinal fluid accumulation in the brain ventricles due to an imbalance between the production and absorption of this fluid. This excess fluid increases the size of the ventricles and puts harmful pressure on the brain tissue. 22 According to the conventional definition, hydrocephalus refers to 2 types: communicating and non-communicating/obstructive hydrocephalus.10,25 Typically, it is divided into congenital and acquired forms. Congenital hydrocephalus is mostly present at birth and is caused by genetic abnormalities. Its incidence is nearly 1–3 per 1000 live births in humans. 23 Conversely, acquired hydrocephalus is caused due to craniocerebral complications such as hemorrhage, inflammation, or neoplasm. In congenital or acquired hydrocephalus, high intraventricular fluid pressure causes harmful effects on the cerebral parenchyma, leading to edema, oxidative stress, inflammation, ependymal cell dysfunction, and reactive modifications in glial cells. 23 Moreover, autosomal recessive mutations are also responsible for some genetic forms of hydrocephalus in animal models including rats and mice. 27
Coiled-coil domain containing 85c (Ccdc85c) is a causative gene for non-obstructive hydrocephalus and subcortical band heterotopia with frequent brain hemorrhage, which was originally identified from hemorrhagic hydrocephalus (hhy) mice.12,14 Most hemorrhagic hydrocephalus (hhy) mice showed multiple hemorrhages in the meninges and brain parenchyma between 3.5 and 19 weeks of age, following ventricular dilatation. 12 Deficiency of the Ccdc85c gene causes radial glial demise and ependymal agenesis in the developing lateral ventricle (LV) of the hhy mice. 14 To investigate the pathophysiological role of the CCDC85C protein, we established a Ccdc85c knockout (KO) rat by genome editing using transcription activator-like effector nuclease (TALEN) that displayed pathological characteristics resembling those of hhy mice. 11 CCDC85C is expressed in several simple epithelia but not in stratified epithelia, and its expression is specifically found in cell-cell and apical junctions of these simple epithelia of rats. 26 Moreover, deficiency of CCDC85C causes disrupted neurogenesis, gliogenesis, and ependymal cell maldevelopment in the ventricular lining from the early to the late postnatal period. 5 Ccdc85c KO rat also shows brain hemorrhage; however, the pathogenesis of hemorrhage remains to be clarified.
Alpha-smooth muscle actin (α-SMA) is the actin isoform that polymerizes in a helical fashion to form an actin filament. This filament constitutes the cytoskeletal protein found predominantly in vascular smooth muscle cells and is crucial for regulating blood flow by causing vascular constriction.2,9
In Ccdc85c KO rats, maldevelopment of ependymal cells was evident, and this abnormality is associated with the development of hydrocephalus. Thus, this study aimed to investigate the role of α-SMA in the disrupted ependymal layer of the LV of Ccdc85c KO rats. Interestingly, we discovered the aberrant expression of α-SMA in the wall of the LV of Ccdc85c KO rats at postnatal day 20 (P20). There is convincing evidence that hydrocephalus causes the death of brain cells, and it is also linked to the creation of new neurons from 2 regions of the adult brain namely, the subgranular zone of the dentate gyrus of the hippocampus and the ventricular-subventricular zone lining the LV. 21 Hence, we hypothesized that α-SMA-positive cells may play an essential role in the development of the LV of Ccdc85c KO rats.
Materials and Methods
Animals
Ccdc85c KO rats were maintained in a specific pathogen-free room with a controlled temperature and a 12:12-h light-dark cycle at the Animal Facility of Osaka Metropolitan University with sufficient access to food and water. All animal care was followed under the Guidelines for Animal Experimentation of Osaka Metropolitan University. Heterozygous rats were mated, and brain samples were collected from the homozygous offspring and their age-matched wild-type controls. Rats were euthanized with isoflurane on postnatal days 6 (P6), P13, P20, and P26. Brains were collected on each of the respective days for histologic examination. Immunohistochemistry (IHC) and immunofluorescence (IF) studies were conducted on 5 homozygous KO and 5 age-matched wild-type rats per postnatal day. One P26 KO and one age-matched wild-type rat brain were used for scanning electron microscopy.
Immunohistochemistry
IHC was conducted on coronally sliced brain sections at the mammillary body level from P6 to P26 rats. Brains were cut and fixed using SUPER FIX (KURABO, Osaka, Japan) for 24 hours and then embedded in paraffin. Paraffin-embedded tissues were cut at 4 µm in thickness and deparaffinized. Following antigen retrieval by autoclaving in 0.01 M citrate buffer (pH 6.0) for 10 minutes at 121°C, tissue sections were processed in a Histostainer (Nichirei Biosciences, Tokyo, Japan). Briefly, tissue sections were first treated with 5% skimmed milk in phosphate-buffered saline for 15 minutes. Then they were incubated with primary antibodies (α-SMA, nestin, vimentin, and glial fibrillary acidic protein [GFAP]) for 1 hour at room temperature (Table 1). After the incubation, sections were treated with 3% H2O2 in phosphate-buffered saline for 15 minutes and incubated again with a horseradish peroxidase–conjugated secondary antibody (Histofine Simple Stain MAX PO, Nichirei Biosciences) for 1 hour at room temperature. Positive reactions were visualized with 3,3’-diaminobenzidine (Nichirei Biosciences) for 10 minutes. Sections were lightly counterstained by hematoxylin. Images were scanned using an Olympus VS-120 virtual slide system (Olympus, Tokyo, Japan).
Primary antibodies used for immunohistochemistry and immunofluorescence.
Abbreviations: α-SMA, alpha-smooth muscle actin; GFAP, glial fibrillary acidic protein.
Immunofluorescence
Double IF was performed to investigate the origin of α-SMA-positive cells in the LV. For single IF, α-tubulin antibody was used to examine the ependymal cilia. Double IF was conducted with a combination of α-SMA with nestin, vimentin, and GFAP. SUPER FIX-fixed, paraffin-embedded brain tissues from P20 and P26 rats were used for this study. The fixed tissues were sectioned at 4 µm thickness and deparaffinized. For antigen retrieval, sections were autoclaved with 0.01 M citrate buffer (pH 6.0) at 121°C for 10 minutes. After autoclaving, they were treated with 10% normal goat serum in phosphate-buffered saline (Thermo Fisher Scientific, Waltham, MA, USA) for 30 minutes. Sequentially, brain sections were incubated with primary antibodies against α-SMA, nestin, vimentin, GFAP, and α-tubulin at 4°C overnight (Table 1). Following washing in phosphate-buffered saline, the sections were incubated with an Alexa 488-labeled or 568-labeled secondary antibodies against mouse IgG2a, mouse IgG2b, and mouse IgG1 (1:500, Thermo Fisher Scientific) at room temperature for 45 minutes. The sections were covered with Fluoro-KEEPER Antifade mounting medium with 4,6′-diamidino-2-phenylindole (DAPI) (Nacalai tesque, Kyoto, Japan). Images were captured using a virtual slide scanner (VS-120, Olympus).
Scanning Electron Microscopy
Ccdc85c KO and age-matched wild-type control rats at P26 were used for this study. Rats were euthanized with isoflurane, and the brains were removed. Coronally sliced brain sections were immersed in fixation solution (2.5% glutaraldehyde [FUJIFILM Wako, Japan], 2% paraformaldehyde [FUJIFILM Wako], and 0.1 M sodium cacodylate buffer [FUJIFILM Wako]) at 4°C overnight. Furthermore, brain samples were sliced using a microtome, washed with 0.1 M sodium cacodylate buffer solution (FUJIFILM Wako), and then post-fixed in 1% osmium oxide solution (OsO4; FUJIFILM Wako) in 0.2 M sodium cacodylate buffer at 4°C for 1 hour. The brain samples were dehydrated with a stepwise ethanol gradient and finally immersed in absolute ethanol at room temperature overnight. The samples were immersed in isopropyl alcohol (FUJIFILM Wako) for 20 minutes and then dried using a critical point dryer (HCP-2; Hitachi, Tokyo, Japan) with carbon dioxide. After drying, the tissue samples were mounted on an aluminum stub, coated with platinum (E-1030, Hitachi), and analyzed under scanning electron microscope (JEOL JSM-6510LV, Tokyo, Japan).
Results
Aberrant Expression of α-SMA in the Wall of the LV of Ccdc85c KO Rats
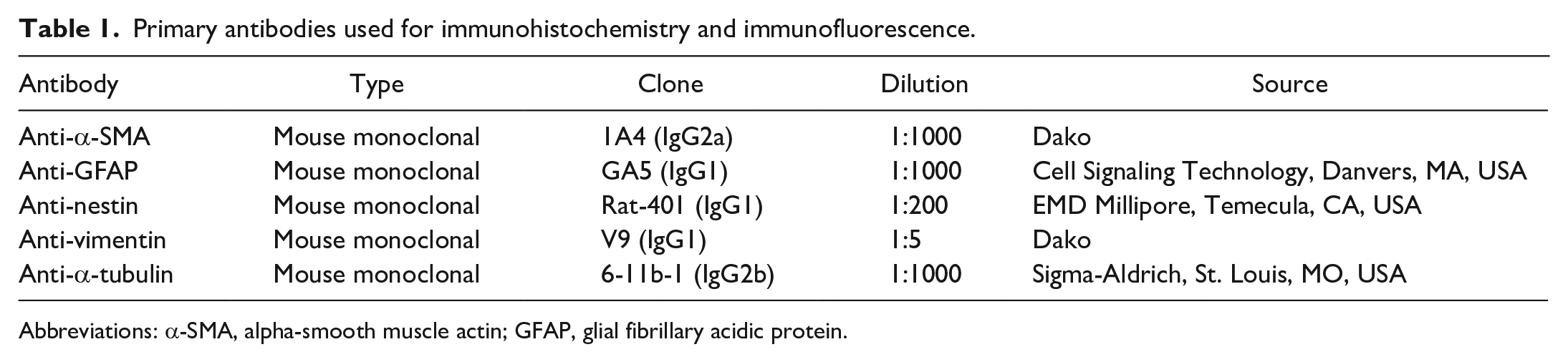
The Ccdc85c KO rats displayed abnormal development of the LV, which resulted in congenital hydrocephalus. 11 We performed α-SMA IHC in Ccdc85c KO and age-matched wild-type rats at P26 when hydrocephalus was prominent. In wild-type rats, no α-SMA expression was observed in the wall of the LV at P26, only the cortical and meningeal vessels were immunolabeled with α-SMA (Fig. 1a). However, aberrant expression of α-SMA was observed in the wall of the LV of Ccdc85c KO rats (Fig. 1b). The majority of the α-SMA-positive cells were seen in the dorsal and medial aspect of the ventricular wall; a few positive cells were also observed in the lateral side. The α-SMA-positive cells were spindle-shaped with an elongated nucleus (Fig. 1b).
Expression of alpha-smooth muscle actin (α-SMA) in the lateral ventricle (LV) of rats at postnatal day 26 (P26). (a) Immunohistochemistry (IHC) for α-SMA in wild-type (WT) rats show an absence of labeling in the lining of the LV; only cortical blood vessels are immunolabeled for α-SMA (black arrow). (b) α-SMA IHC shows expression in the lining of the LV (red arrowheads) in Ccdc85c knockout (KO) rats. Inset: higher magnification demonstrates spindle-shaped cells.
Aberrant α-SMA Expression Starts Around 3 Weeks of Age
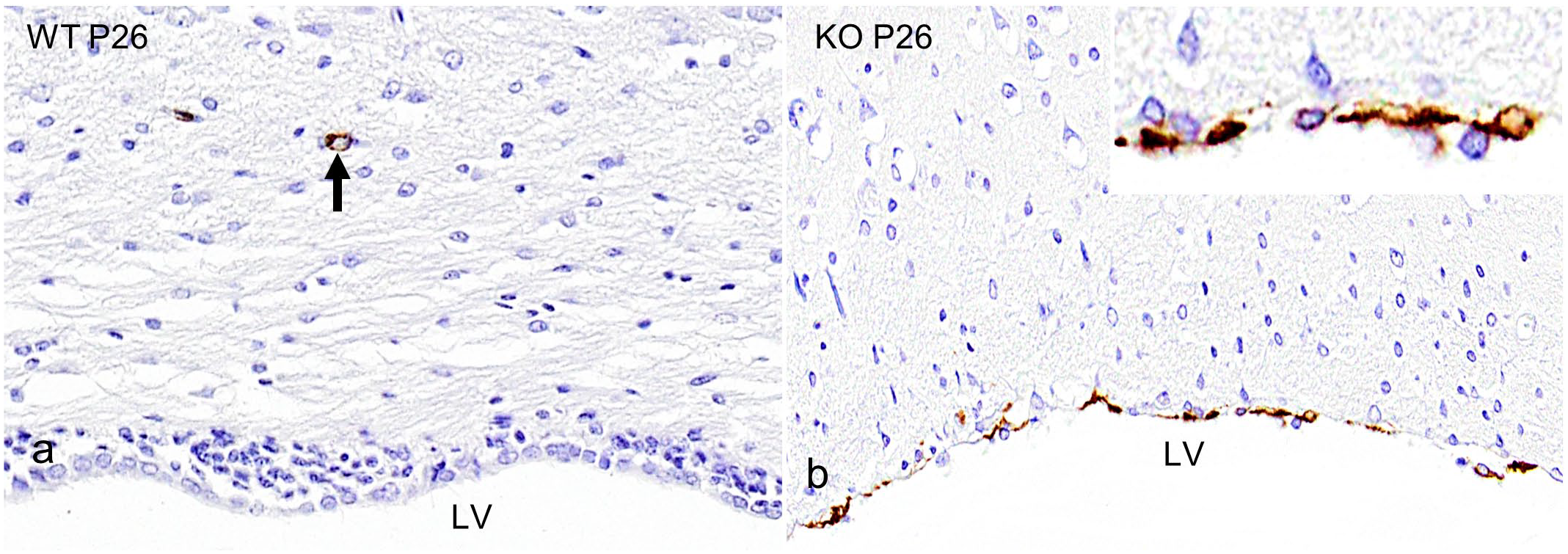
To explore the temporal changes of the aberrant α-SMA expression, Ccdc85c KO and age-matched wild-type rats were immunolabeled with α-SMA at P6, P13, P20, and P26 (Fig. 2a-h). KO rats started to show aberrant expression of α-SMA at P20 (Fig. 2g). Before this age, expression patterns were similar in KO and wild-type rats. An increased number of aberrant expression of α-SMA was observed at P26 (Fig. 2h).
Lateral ventricle (LV). Immunohistochemistry (IHC) of alpha-smooth muscle actin (α-SMA) in wild-type (WT) and Ccdc85c knockout (KO) rats at postnatal days 6 (P6), P13, P20, and P26. (a-d) WT rats from P6 to P26 show an absence of α-SMA expression in the lining of the LV at all ages. (e-h) KO rats from P20 to P26 show α-SMA expression in the lining of the LV (red arrowheads). The black arrows indicate normal vascular expression.
Origin of Aberrant α-SMA Expression in the Wall of the LV of Ccdc85c KO Rats
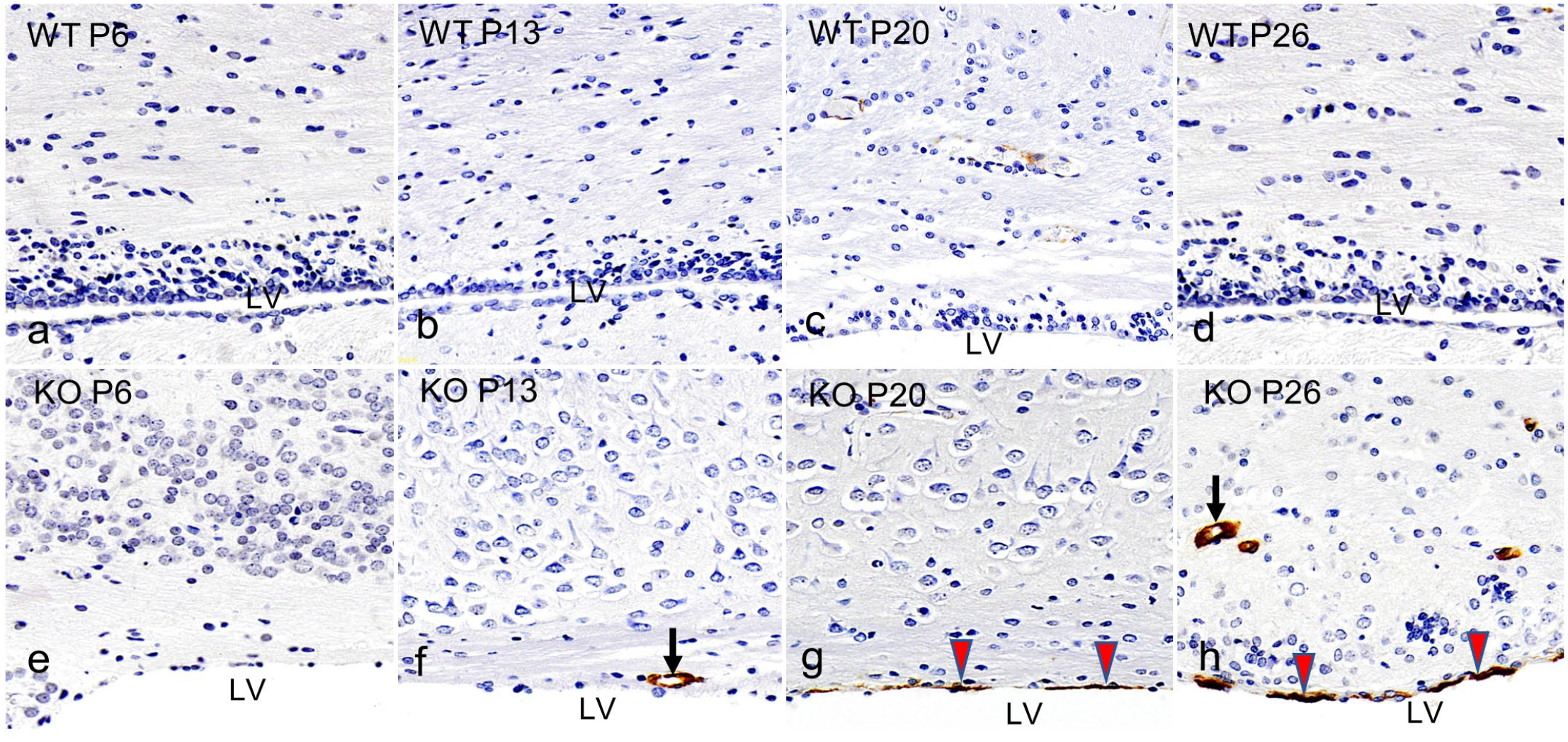
To investigate the persistence of α-SMA-positive cells in the wall of the LV at P26, the Ccdc85c KO rats were subjected to serial section IHC with neuro-glial markers such as nestin, vimentin, and GFAP after α-SMA expression. When comparing the aberrant expression of α-SMA (Fig. 3a) with neuro-glial markers, each of these neuro-glial markers showed distinct expression patterns in the wall of the LV (Fig. 3b-d).
Lateral ventricle (LV), Ccdc85c knockout rats, postnatal day 26, immunohistochemistry (IHC). IHC for (a) alpha-smooth muscle actin (α-SMA), (b) nestin, (c) vimentin, and (d) glial fibrillary acidic protein (GFAP) show distinct expression patterns in the lining of the LV (black arrow).
Furthermore, the origin of aberrant α-SMA-expressing cells was then investigated using double IF with nestin, vimentin, or GFAP at P20 and P26. We found α-SMA was not co-expressed with nestin, vimentin, and GFAP (Fig. 4a, c, e) in wild-type rats at P20 and P26 (Supplemental Figure S1). However, α-SMA was co-expressed with nestin, vimentin, and GFAP in Ccdc85c KO rats at P20. Among the co-expressed cells, nestin co-expression was more conspicuous (Fig. 4b) than vimentin and GFAP (Fig. 4d, f) at P20. The ventricular lining of Ccdc85c KO rats at P26 also showed similar co-expression at P20 (Supplemental Figure S1). Thus, these results suggest that the lining of the LV of adult Ccdc85c KO rats contains immature cells derived from GFAP, nestin, and vimentin-expressing cells.
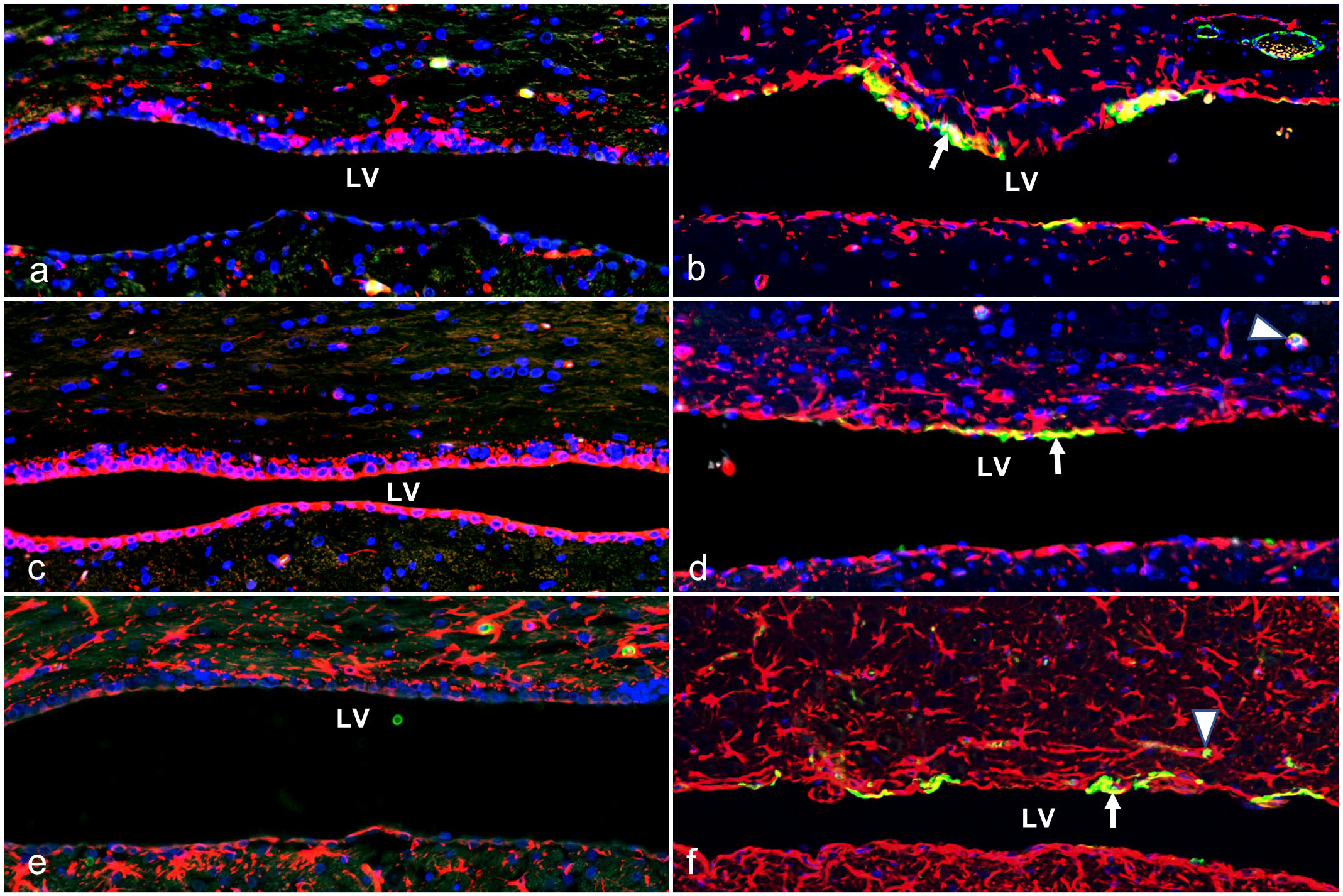
Lateral ventricle (LV). Double immunofluorescence (IF) for alpha-smooth muscle actin (α-SMA) with nestin, vimentin, and glial fibrillary acidic protein (GFAP) in wild-type (WT) and Ccdc85c knockout (KO) rats at postnatal day 20 (P20). (a, c, e) WT rats show an absence of co-expression of α-SMA with nestin (a), vimentin (c), and GFAP (e) in the lining of the LV. (b, d, f) KO rats show co-expression of α-SMA with nestin (b), vimentin (d), and GFAP (f) in the lining of the LV (white arrow). A box in the top right corner highlights meningeal vascular expression, and the white arrowheads indicate cortical vascular expression by α-SMA. Green, α-SMA; red, nestin (a, b), vimentin (c, d), GFAP (e, f); blue, DAPI; yellow, co-localization with α-SMA.
Loss of Ependymal Cilia in the Ccdc85c KO Rats
Previous research showed ependymal cell maldevelopment in the lining of the LV in Ccdc85c KO rats. 5 To find out the condition of ependymal cilia in KO and age-matched wild-type rats at P26, α-tubulin IF was performed. In the wild-type rats, ependymal cilia were immunolabeled with α-tubulin (Fig. 5a). No cilia were immunolabeled with α-tubulin in KO rats (Fig. 5b), indicating the loss of ependymal cilia in the dorsal lining of the LV. Furthermore, scanning electron microscopic study revealed undamaged cilia in the ventricular surface of wild-type rats (Fig. 5c), but the KO rats showed a large number of round cell bodies covering the wall of the LV (Fig. 5d).
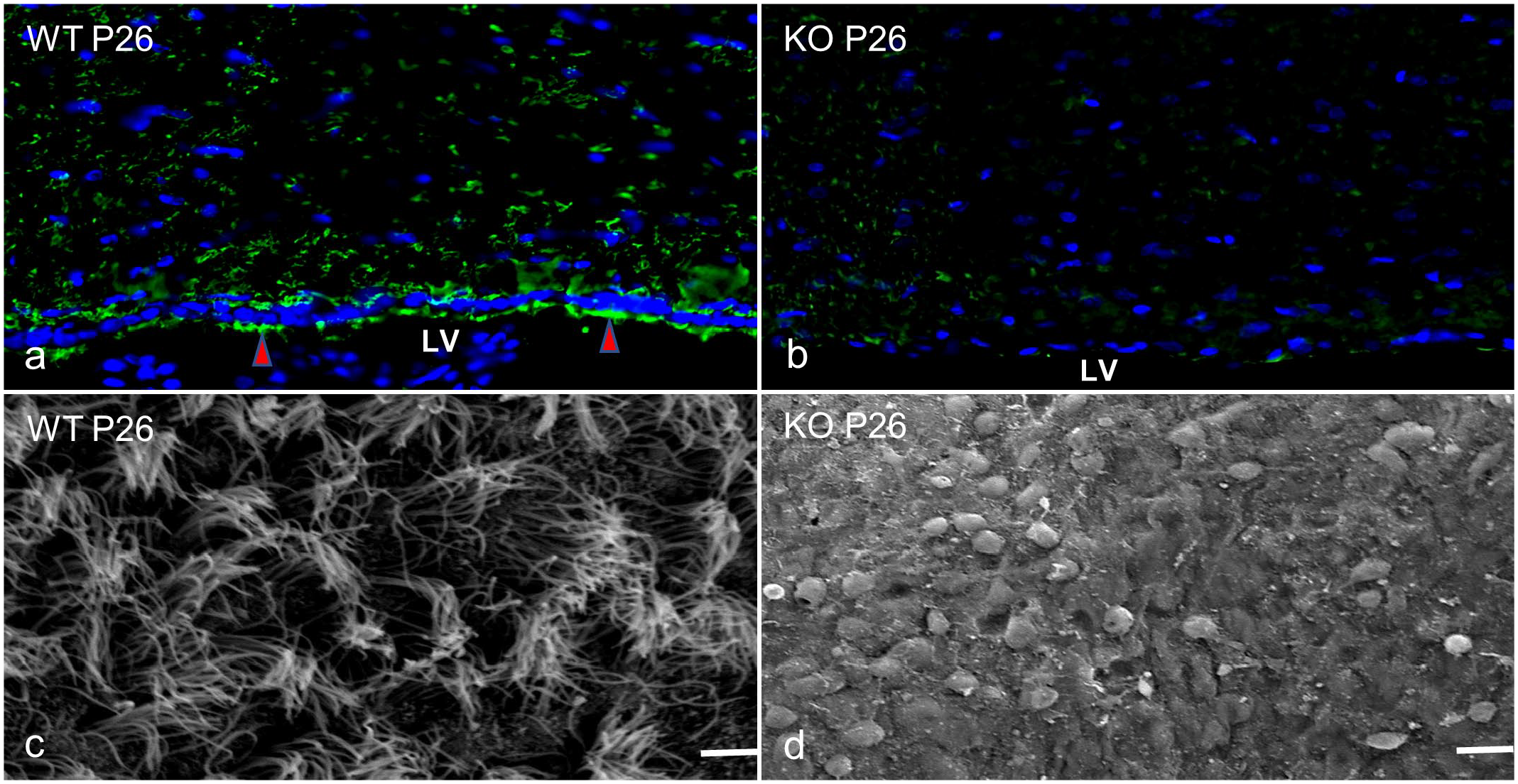
Lateral ventricle (LV). Wild-type (WT) and Ccdc85c knockout (KO) rats at postnatal day 26 (P26). (a) Immunofluorescence (IF) for α-tubulin (green) shows positive expression by the ependymal cilia of WT rats (red arrowheads). Blue, DAPI. (b) Positive expression of α-tubulin by the ependymal cilia is absent in the KO rats. IF. Blue, DAPI. (c) Intact cilia in the ventricular surface of the WT rats. Scale bar = 10 µm. Scanning electron microscopy (SEM). (d) The KO rats show round cell bodies in the ventricular wall. Scale bar = 20 µm. SEM.
Discussion
The Ccdc85c KO rats exhibited disrupted neurogenesis and gliogenesis in the region of the LV. 5 In this study, we performed α-SMA IHC and interestingly elucidated aberrant expression of α-SMA in the wall of the LV of Ccdc85c KO rats.
The mammalian actin protein is comprised of 6 different isoforms which are responsible for several essential cell functions, including tight junction formation, cell migration and division, and transcriptional regulation. 20 Among them, the alpha isoform of SMA is a contractile protein mainly found in adult vascular smooth muscle cells. 24 In our present study, α-SMA was expressed normally in the cortical and meningeal vessels of both KO and wild-type rats. However, in the Ccdc85c KO rats, we observed a special population of cells lining the wall of the LV expressed by α-SMA at P20. Furthermore, immunohistochemical study of α-SMA from P20 to P26 revealed increased α-SMA expression after 3 weeks of age (P26). In vitro research shows that neural stem cells isolated from rat embryonic cerebral cortex can differentiate into smooth muscle-like cells that express α-SMA.15,16,19
To determine the origin of aberrant α-SMA-expressing cells, GFAP, nestin, and vimentin immunohistochemical investigations were performed. This study observed significant GFAP expression in the ventricular wall of KO rats at P26. In addition, at this age, double immunofluorescence for α-SMA and GFAP showed substantial co-expression in the wall of the LV. From previous research in the adult mammalian brain, some GFAP-positive fusiform type cells with few processes were identified in the ventricular-subventricular zone, known as type B cells bearing the characteristics of astrocytes, which serve as neural progenitor cells. 4 Recently, a group also observed the colocalization of α-SMA and GFAP on the ventricular surface of a mouse model of communicating hydrocephalus with FOXJ1 haploinsufficiency and ependymal motile cilia dysfunction, indicating incomplete differentiation of ependymal cells and the origin of α-SMA-positive cells from GFAP-positive cells. 8
Nestin is a class VI intermediate filament protein expressed by neural progenitor cells 7 and also supports the proliferation, differentiation, and migration of neural stem cells in the ventricular-subventricular zone during the neocortex development. 1 In the present study, according to the immunohistochemical results, nestin expression was visible in the wall of the LV of KO rats. IF investigation also revealed co-expression of α-SMA and nestin in the ventricular wall. Nestin-positive cells can generate differentiated cells of neuronal and astrocytic lineages in the adult brain. 1
Vimentin is a class III intermediate filament protein and is expressed by radial glial cells in the ventricular wall during development. 17 As development progresses, radial glial cells decrease vimentin expression and gradually begin to express GFAP. 13 Previous research has shown intense vimentin expression in the radial processes around perinatal periods, but limited vimentin expression was observed in the dorsal wall of the LV in Ccdc85c KO rats during postnatal development.5,6 In this study, we found strong vimentin immunoreactivity in the ventricular wall during the postnatal period (P26), indicating the persistence of radial glia-like cells in this region. A recent work showed that CCDC85C interacts with polarity protein PAR3, which is located in the adherens junction at the ventricular zone surface, regulating the proliferation of radial glial cells via phase separation coupled to percolation. 29 According to the aforementioned findings, it is indicated that the ventricular wall of the LV of Ccdc85c KO rat is a proliferating zone, containing immature cells that express GFAP, nestin, and vimentin around 3 weeks of age.
It is known that actin and other cytoskeletal proteins are crucial for the formation of planar cell polarity and maturation of ventricular zone ependymal cells. 18 Most of the ependymal populations are multiciliate and maintain cerebrospinal fluid flow, regulate the clearance of metabolic substances, and mediate the adhesions of inflammatory cells.23,28 The ependyma is also known to undergo significant stretching and lose its surface specialization and cilia in hydrocephalus. 3 Our previous research in Ccdc85c KO rats has shown ependymal cell maldevelopment in the wall of the LV. 5 In this study in Ccdc85c KO rats, we performed α-tubulin IHC and scanning electron microscopy to know the fate of ependymal cilia at P26. In the KO rat, only round cell bodies remained on the ventricular surface upon the loss of ependymal cilia. Thus, it is conceivable to state that the Ccdc85c gene mutation causes variable degrees of loss of ependymal cilia and also contributes to the proliferation of smooth muscle-like immature cells in the ventricular wall of KO rats.
In conclusion, we serendipitously discover a novel population of α-SMA-positive cells in the ventricular wall of the LV of Ccdc85c KO rats. The co-expression of α-SMA with neuro-glial markers indicates that these α-SMA-positive cells may act as stem/progenitor cells, which might be needed further for the development of lateral ventricle abnormalities.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241312611 – Supplemental material for Aberrant alpha-smooth muscle actin expression in the lateral ventricle of Ccdc85C knockout rats
Supplemental material, sj-pdf-1-vet-10.1177_03009858241312611 for Aberrant alpha-smooth muscle actin expression in the lateral ventricle of Ccdc85C knockout rats by Nure Jannat, Md. Mehedi Hasan, Hisaki Tokuno, Miyuu Tanaka, Takeshi Izawa, Jyoji Yamate and Mitsuru Kuwamura in Veterinary Pathology
Footnotes
Acknowledgements
The Ccdc85c KO (F344-Ccdc85cem1kyo) rat stain was deposited into the National BioResource Project (NBRP)-Rat (NBRP Rat No. 0824).
Author Contributions
MK designed the experiment; NJ and MMH performed the experiments and data curation; MT, TI, and JY performed formal analysis, writing review, and editing; MK and HT performed scanning electron microscopical evaluations; the manuscript was written by NJ, MMH, and MK.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Society for the Promotion of Science (JSPS; grant number 21H02368).
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
