Abstract
Cancer-associated fibroblasts (CAFs) are fibroblastic cells that express α–smooth muscle actin and have been identified in the stroma of numerous epithelial tumors. The presence of CAFs within the tumor stroma has been associated with a poorer prognosis in some human cancers, including oral squamous cell carcinomas (SCCs). Cats frequently develop oral SCCs, and although these are generally highly aggressive neoplasms, there is currently a lack of prognostic markers for these tumors. The authors investigated the prognostic value of the presence of CAFs within the stroma of oral SCC biopsy specimens from 47 cats. In addition, several epidemiologic, clinical, and histologic variables were also assessed for prognostic significance. A CAF-positive stroma was identified in 35 of 47 SCCs (74.5%), and the median survival time (ST) of cats with CAF-positive SCCs (35 days) was significantly shorter than that of cats with CAF-negative SCCs (48.5 days) (P = .031). ST was also associated with the location of the primary tumor (P = .0018): the median ST for oropharyngeal SCCs (179 days) was significantly longer than for maxillary (43.5 days; P = .047), mandibular (42 days; P = .022), and sublingual SCCs (22.5 days; P = .0005). The median ST of sublingual SCCs was also shorter compared with maxillary SCCs (P = .0017). Furthermore, a significant association was identified between site and the presence of stromal CAFs (P = .025). On the basis of this retrospective study, evaluating the tumor stroma for CAFs in feline oral SCC biopsy specimens may be of potential prognostic value.
Keywords
Oral squamous cell carcinomas (SCCs) are the most common tumor of the oral cavity in cats. 30 They are locally aggressive and have a grave prognosis, with a median survival time (ST) of just 33 days. 13 The median age at diagnosis is 12 years, and there is no reported breed or sex predilection. 25,34 Oral SCCs can arise from any site, but the 2 most commonly reported locations are sublingual and gingival, with less frequently reported areas being the tonsil, caudal pharynx, soft and hard palates, and buccal mucosa. 13,19,30 In cats, there is a paucity of reliable and accessible prognostic markers that determine outcomes in oral SCC. Anatomic staging, histologic grading, and epidermal growth factors receptor expression have not been shown to predict survival of cats, and the value of cyclooxygenase-2 expression as a prognosticator is yet to be determined. 5,37,38
In humans, oral and pharyngeal cancer, in which SCCs account for the majority of cases, is the sixth most common cancer in the world. 36 Prognosis in human oral SCCs is determined largely by the World Health Organization–recommended tumor, node, and metastasis classification system. The tumor stroma, which has traditionally been seen as a supporting structure for neoplastic cells, is now considered to modulate some of the complex interactions that occur between cancer cells and the stroma, and it may even promote a malignant phenotype. 9 Cancer-associated fibroblasts (CAFs), which are also called activated fibroblasts or myofibroblasts, are a major component of the tumor stroma and have been demonstrated to increase cancer cell invasion, proliferation and growth rate, promote angiogenesis, modulate inflammatory cell infiltrates and the expression of extracellular matrix proteases, and decrease cancer cell death. 10,22,23,27 Studies have shown significant correlations between a CAF-rich stroma and adverse survival prognostic factors in many human cancer types, including oral SCCs. 16,18 Their value as a molecular marker is further supported by the provision of new, adjunctive anticancer therapies that target CAFs within the stroma. 6,26
The aim of the present study was to evaluate whether CAFs were present within the stroma of feline oral SCCs and whether they could be used as predictors for survival. Additionally, several epidemiologic, clinical, and histologic characteristics were assessed for their effect on survival. To our knowledge, this is the first time the presence of CAFs within the tumor stroma has been investigated as a possible prognostic indicator within a nonhuman species.
Materials and Methods
Case Selection and Recruitment of Study Material
For this retrospective study, oral SCCs were identified by searching the databases of New Zealand Veterinary Pathology Ltd (Palmerston North, New Zealand) and Gribbles Veterinary Pathology, New Zealand, between 2008 and 2014. For a total of 112 cases, surveys were distributed to 44 referring veterinary clinics to obtain information regarding the patient signalment (age, sex, reproductive status, and breed), primary site of the oral SCC, cytologic or histologic evidence of regional nodal metastasis, clinical or radiographic evidence of bony involvement, treatment following the initial biopsy, and the date and cause of death. Complete survey information was gathered for 59 cases from 27 veterinary clinics.
Inclusion and Exclusion Criteria
The study included histologically confirmed oral SCCs from cases with complete survey data that received only palliative care and had incisional biopsy specimens of adequate quality. Palliative care was defined as cats that received either no treatment or treatment without a curative intent that included the use of analgesic, anti-inflammatory, and antimicrobial medications, as well as the surgical debulking of the SCC (n = 2). Outcome categories included euthanasia because of the oral SCC and loss to follow-up. Cases that were excluded included cats that were not euthanatized because of the oral SCC (n = 1), cats that underwent curative surgery (n = 2), and cases in which histologic samples did not contain adequate stroma surrounding the tumor (n = 9). ST was defined as the time from initial biopsy until euthanasia. Cats were censored at the point of the last clinical visit if lost to follow-up.
Histologic Assessment
A diagnosis of oral SCC was confirmed in all cases by examination of hematoxylin and eosin–stained sections. The SCCs were initially graded according to the invasive front grading (IFG) system, as described for human oral SCCs and outlined in Supplemental Table S1. 2,7 With this grading system, oral SCCs were scored according to 5 criteria that assess the neoplastic cells (degree of keratinization, nuclear pleomorphism, and mitotic count), the pattern of invasion (POI), and the presence of lymphoplasmacytic infiltrates. All features were assessed at the most anaplastic area within the most invasive sites of the SCCs. All 5 individual scores for each category were then totaled to produce a total malignancy score, and the median malignancy score was used to stratify the oral SCCs into low- and high-grade scores. The POI, a criterion of the IFG system, reflects the cohesiveness of tumor cells, and oral SCCs were classified as having either a high or a low POI. A high POI defined tumor cells that infiltrated individually or as small clusters and strands where the clusters contained <15 cells each. A low POI defined tumor cells that infiltrated in a more cohesive manner where the clusters contained ≥15 cells each. 2
Tumor budding was assessed as the presence of isolated single cancer cells or clusters composed of fewer than 5 cells scattered in the stroma of the invasive front. 32 Oral SCCs were initially examined under low magnification to select the area with the highest density of budding, and then tumor budding was counted using the 20× objective lens. The intensity of budding was defined as low if the total number of buds was <5 buds/field and high if the total number of buds was ≥5 buds/field. 1,35
Immunohistochemical Analysis
Immunohistochemistry with primary antibodies for α–smooth muscle actin (α-SMA) (clone 1A4; DAKO, Glostrup, Denmark) was performed on 3- to 5-μm sections using a commercial staining platform (BenchMark ULTRA; Ventana Medical Systems, Tucson, AZ) and a biotin-free indirect detection system (ultraView Universal DAB; Ventana Medical Systems). The primary antibody was applied for 28 minutes at a dilution of 1:5000. Tissue sections processed simultaneously with the omission of the primary antibody were used as negative controls, and the smooth muscle surrounding blood vessels within the sections was used as internal positive controls. α-SMA specificity was demonstrated by the absence of stromal cell staining in the adjacent, non-neoplastic stroma of 7 oral SCC cases. For CAF evaluation, the percentage of positive α-SMA stromal cells was semiquantitatively evaluated in 10 random low-power fields; if these were not available, then the entire tumor stroma was evaluated. A SCC was considered to be CAF positive if >5% of cells within the stroma contained α-SMA positivity, whereas SCCs with ≤5% of cells with α-SMA immunostaining were classified as CAF negative. 14
Statistical Analysis
Analyses were conducted using SAS version 9.4 (SAS Institute, Inc, Cary, NC). Kaplan-Meier analysis was used to assess the associations between epidemiologic, clinical, histologic, and immunohistochemical variables and ST, and differences between strata were assessed using the log-rank test. Hazard ratios were calculated using the Cox proportional hazards regression model, and a forward selection process was used to identify statistically significant model parameters. The Fisher exact test was used to assess associations between categorical variables. Two cases were lost to follow-up and were considered censored events. In all cases, statistical significance was set at a P value ≤ .05.
Results
Forty-seven feline oral SCCs were included in the study. The overall median ST for cats diagnosed with oral SCC was 37 days; the 30-, 60-, 180-, and 365-day survival rates were 67.4%, 28.3%, 4.3%, and 0% respectively. The median age at time of biopsy was 13 years (range, 6–18.6 years), and there were 30 males (6 entire) and 17 females (1 entire). Breeds included 32 domestic short haired, 10 domestic long haired, 2 domestic, and 1 domestic medium haired, 1 Chinchilla, and 1 Maine Coon. The most commonly reported site was the gingiva overlying the maxilla in 23 cases (48.9%), followed by sublingual in 14 cases (29.8%), the mandibular gingiva in 7 cases (14.9%), and oropharyngeal in 3 cases (6.4%). The oropharyngeal site consisted of SCCs located in the tonsil (n = 1), caudal fauces (n = 1), and caudal pharynx (n = 1). Forty-six cases were not assessed for cytologic or histologic involvement of regional lymph nodes, and only 1 case had confirmed histologic cervical lymph node involvement; as such, these tumors were not staged, and no analysis of regional lymph node involvement on survival could be performed. Of the 30 maxillary and mandibular SCCs, invasion of the underlying bone was detected by radiographic or computed tomographic imaging or histologic evidence of neoplastic epithelial cells infiltrating between bony trabeculae in 28 tumors (93.3%). Because of the high proportion of mandibular and maxillary SCCs with bone invasion, this variable was excluded from the survival analysis. Treatment with nonsteroidal anti-inflammatory drugs (meloxicam, n = 21; piroxicam, n = 1) was reported in 22 of 47 cases (46.8%).
Analysis of slides stained with α-SMA revealed expected immunostaining of both positive and negative controls. CAFs within the stroma were identified as spindle-shaped, fibroblast-like cells with cytoplasmic immunoreactivity for α-SMA. These CAFs were most frequently visible immediately adjacent to the neoplastic cells. Twelve SCCs (25.5%) were assessed as CAF negative. Thirty-five SCCs (74.5%) were classified as CAF positive (Figures 1 and 2). Of the CAF-positive SCCs, 11 (23.4%) exhibited focal to multifocal immunopositivity, where between 6% and 50% of the stroma was immunopositive for α-SMA, and 24 SCCs (51.1%) were found to have greater than 50% stromal immunopositivity.
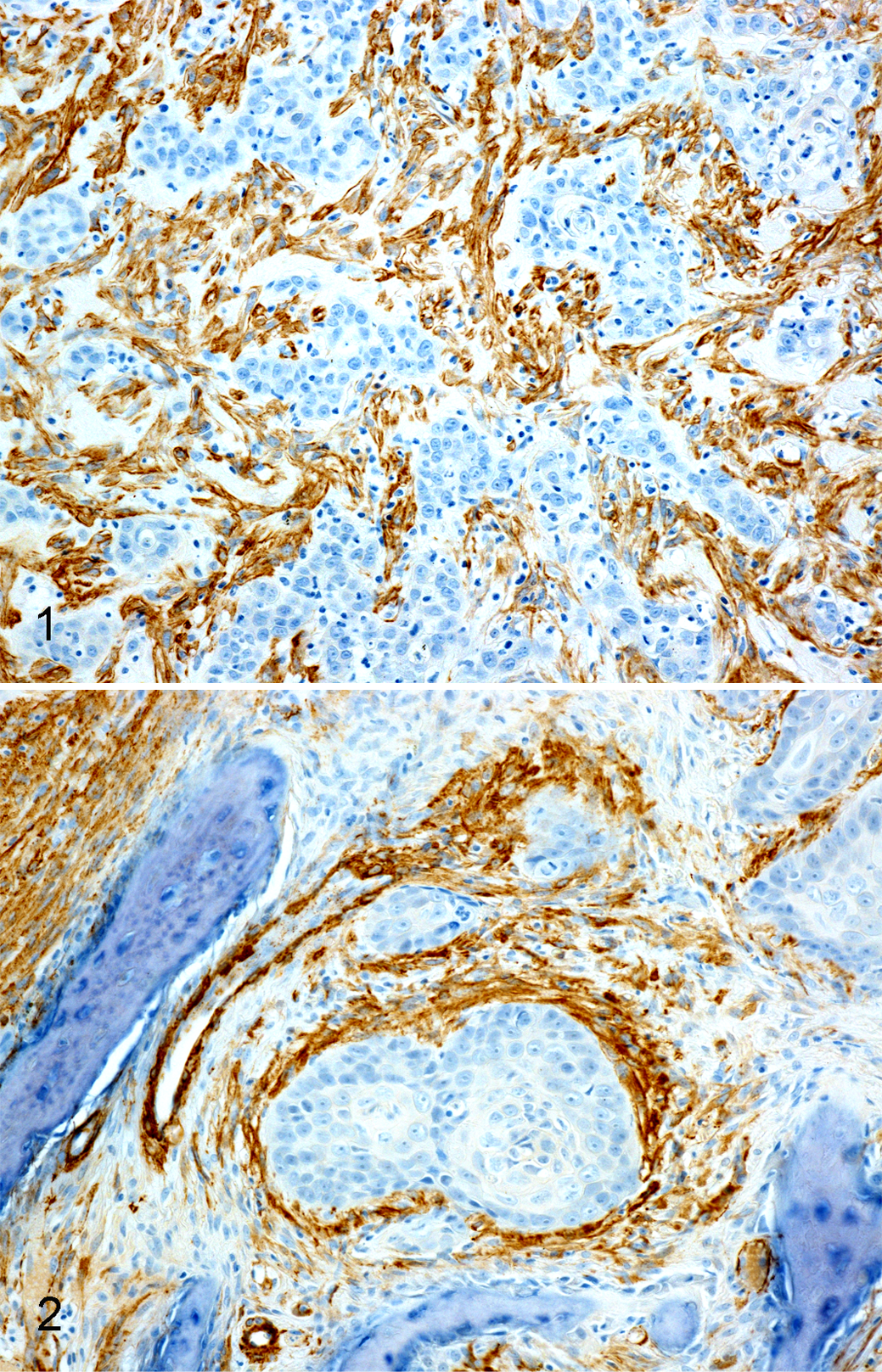
Oral squamous cell carcinoma (SCC) in the cat, immunohistochemistry for α–smooth muscle actin (α-SMA).
Survival Outcomes
Kaplan-Meier survival analysis (Table 1) revealed that the presence of stromal CAFs and the site of the oral SCC were significant predictors for ST (Figures 3 and 4; log-rank P = .031 and P = .0018, respectively), whereas a strong association was not found between ST and age (P = .53), sex (P = .58), reproductive status (P = .26), or nonsteroidal anti-inflammatory drug use (P = .32). Cats with CAF-positive SCCs had a statistically significantly shorter median ST of 35 days compared with cats with CAF-negative SCCs, which had a median ST of 48.5 days (P = .031). The median ST for oropharyngeal, maxillary, mandibular, and sublingual SCCs was 179, 43.5, 42, and 22.5 days, respectively. Site was found to retain its prognostic significance in the multivariate model, where the median ST for oropharyngeal SCCs was significantly longer compared with maxillary (P = .047), mandibular (P = .022), and sublingual SCCs (P = .0005). Furthermore, the median ST of sublingual SCCs was significantly shorter compared with that of maxillary SCCs (P = .0017). There was no significant difference in median ST between cats with sublingual SCCs and those with mandibular SCCs or between cats with maxillary and cats with mandibular SCCs.
Summary of Clinical, Epidemiologic, Histologic, and Immunohistochemical Variables With Respect to Survival Time in Cats With Oral SCC.
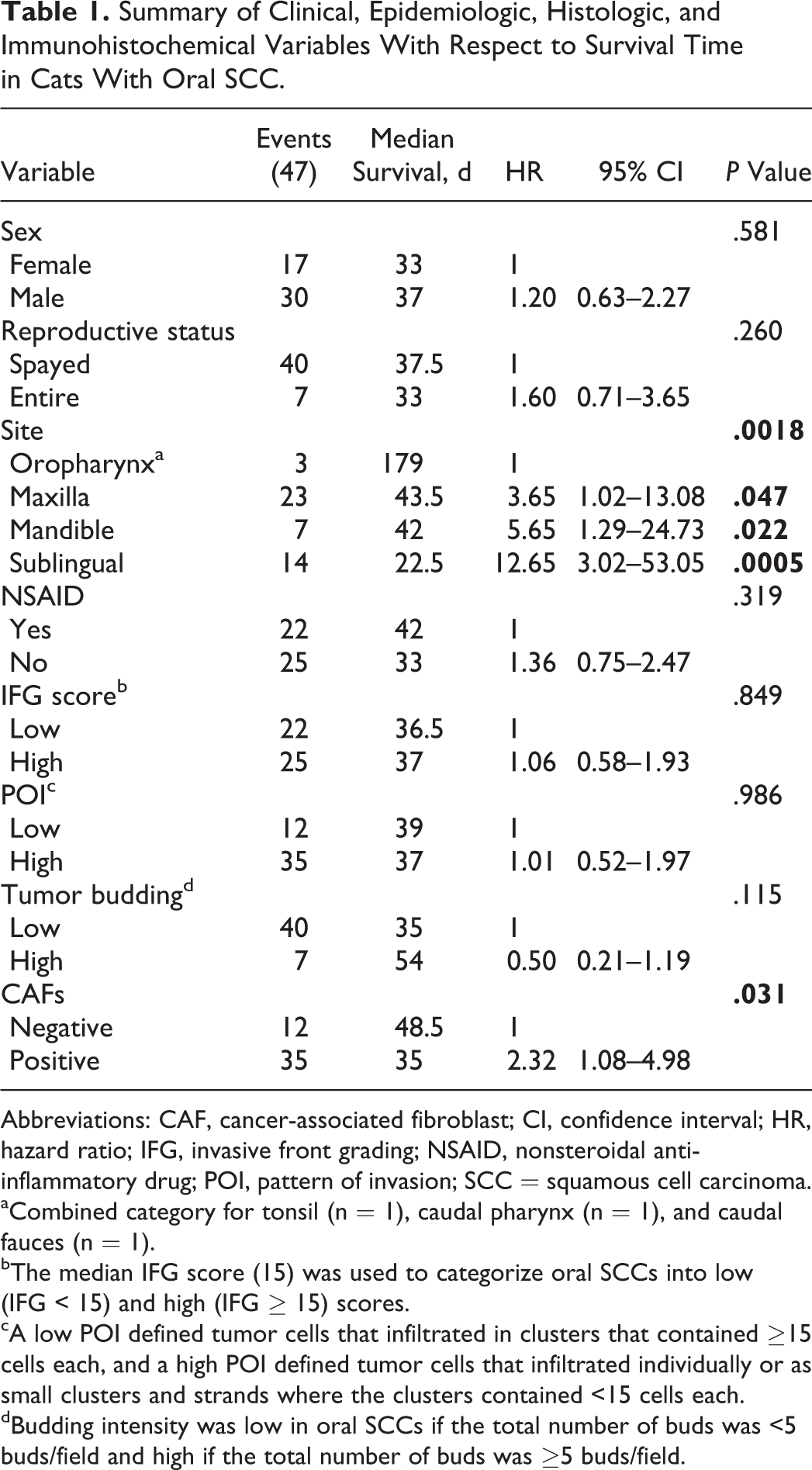
Abbreviations: CAF, cancer-associated fibroblast; CI, confidence interval; HR, hazard ratio; IFG, invasive front grading; NSAID, nonsteroidal anti-inflammatory drug; POI, pattern of invasion; SCC = squamous cell carcinoma.
aCombined category for tonsil (n = 1), caudal pharynx (n = 1), and caudal fauces (n = 1).
bThe median IFG score (15) was used to categorize oral SCCs into low (IFG < 15) and high (IFG ≥ 15) scores.
cA low POI defined tumor cells that infiltrated in clusters that contained ≥15 cells each, and a high POI defined tumor cells that infiltrated individually or as small clusters and strands where the clusters contained <15 cells each.
dBudding intensity was low in oral SCCs if the total number of buds was <5 buds/field and high if the total number of buds was ≥5 buds/field.
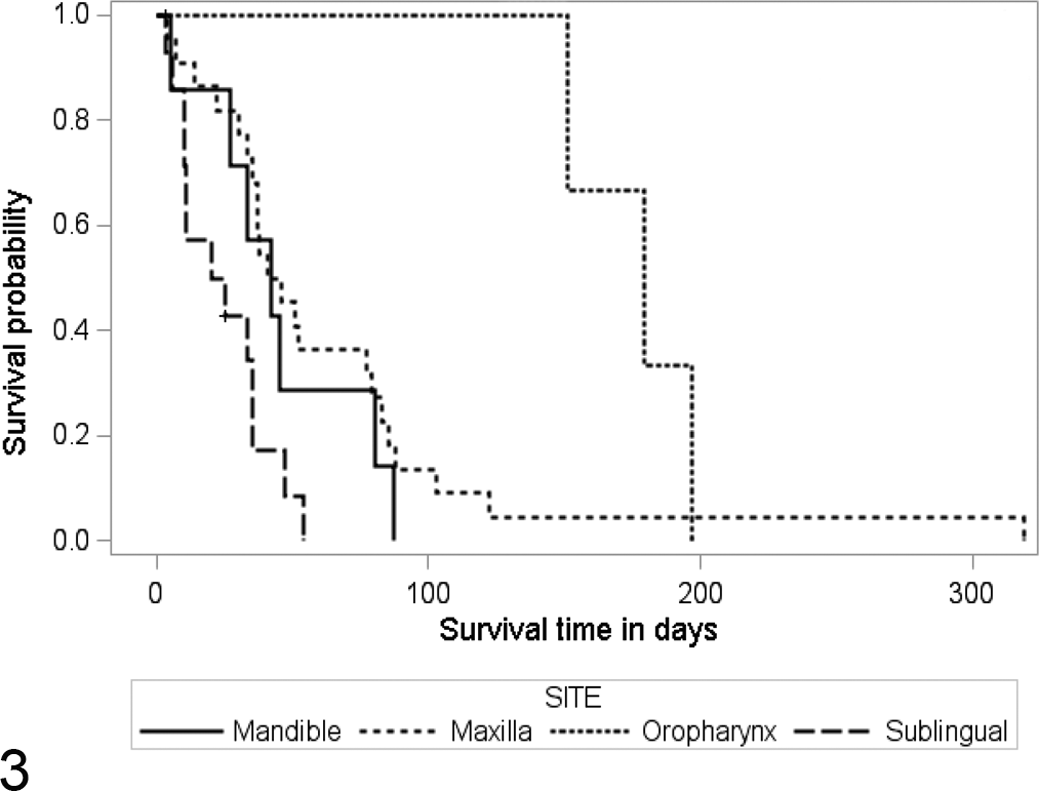
Kaplan-Meier survival curves for 47 cats with oral squamous cell carcinoma, illustrating the differences in survival time (ST) among sublingual, mandibular, maxillary, and oropharyngeal sites, with median survival times of 22.5, 42, 43.5, and 179 days, respectively (P = .0018).
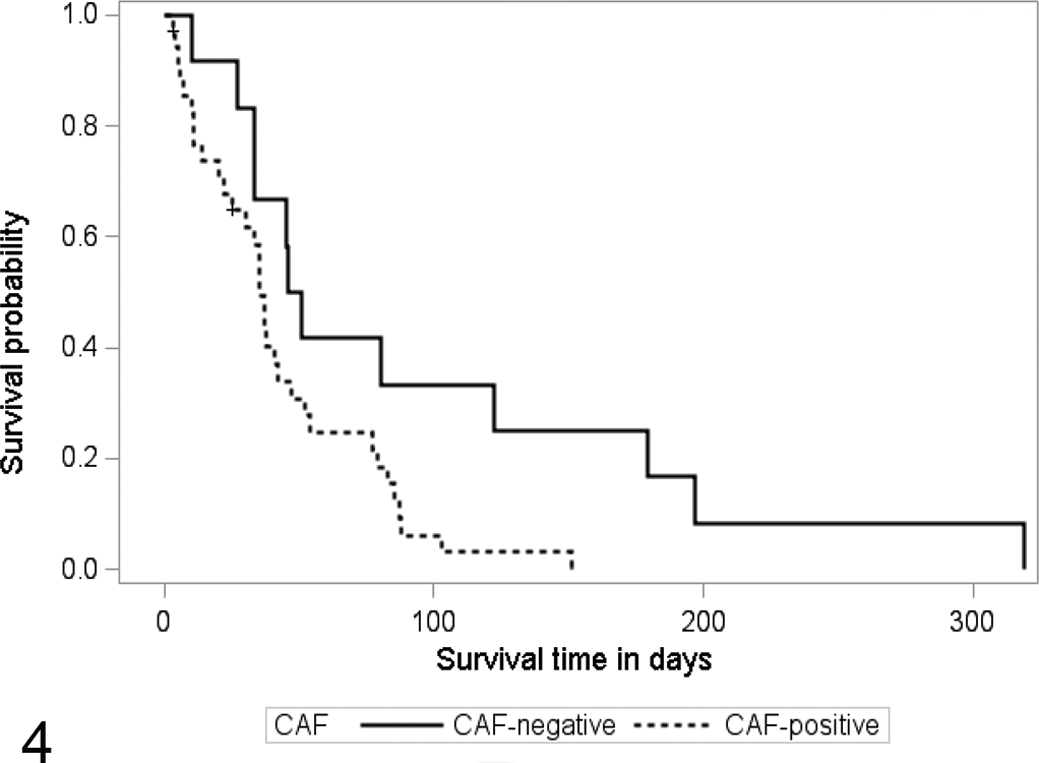
Kaplan-Meier survival curves for 47 cats with oral squamous cell carcinoma, illustrating the difference in survival time (ST) between specimens that were negative for stromal cancer-associated fibroblasts (CAFs) and those that were positive for CAFs (P = .031). The median ST for CAF-positive tumors was 35 days, compared with 48.5 days for CAF-negative tumors.
The distribution of CAF-positive and CAF-negative cases varied with the site of the oral SCC and the POI (P = .025 and P = .005, respectively) (Table 2). In order of decreasing frequency, the number of CAF-positive cases was highest within sublingual SCCs (92.9%), then maxillary (78.3%), mandibular (42.9%), and oropharyngeal (33.3%) SCCs, and 85.7% of SCCs that exhibited high POIs were CAF positive. There was also a statistically significant association between site and POI (P = .045).
The Correlation Between CAFs and the Histological Pattern of Invasion and Site of Oral Squamous Cell Carcinoma.a
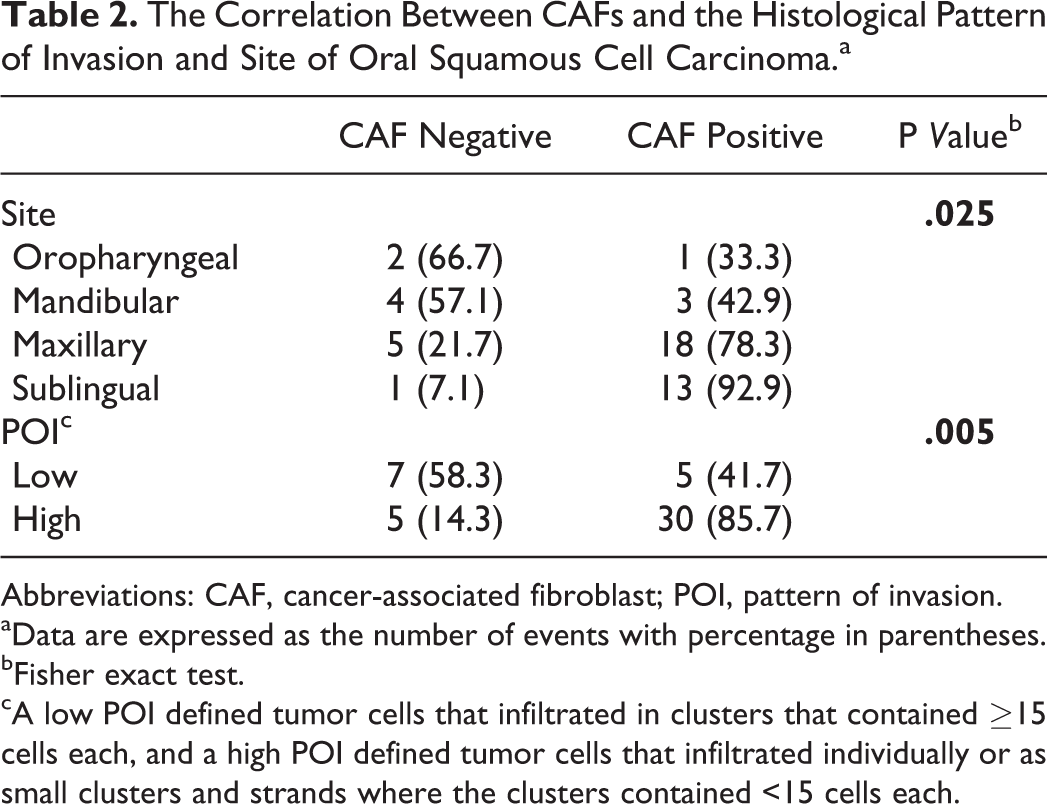
Abbreviations: CAF, cancer-associated fibroblast; POI, pattern of invasion.
aData are expressed as the number of events with percentage in parentheses.
bFisher exact test.
cA low POI defined tumor cells that infiltrated in clusters that contained ≥15 cells each, and a high POI defined tumor cells that infiltrated individually or as small clusters and strands where the clusters contained <15 cells each.
The IFG score ranged from 11 to 18, and no significant associations were found between IFG score and ST (P = .85). No significant associations were identified between ST and POI (P = .99) or between ST and the intensity of budding within the SCCs (P = .12).
Discussion
α-SMA immunostaining identified a CAF-positive tumor stroma in 74.5% of feline oral SCCs. In humans, CAFs are present in the stroma surrounding about 80% of oral SCCs. 3 Therefore, the rate of detection of CAFs in the feline samples is similar to that observed within human oral SCCs. In the present study, cats with CAF-positive oral SCCs had a modest, but significantly shorter ST compared with cats with CAF-negative SCCs. This inverse association between the presence of CAFs within the stroma and ST has also been reported in human oral SCCs. 16,18,33 The results of the present study therefore suggest that, as in human oral SCCs, the presence of CAFs in the tumor stroma could potentially be used as a marker to predict the prognosis of feline oral SCC.
In the present study, a CAF was defined as any fibroblast-like stromal cell that was immunopositive for α-SMA. However, studies in people have revealed that although most CAFs contain α-SMA, some CAFs may be negative for α-SMA but instead express different molecules, such as fibroblast-specific protein and platelet-derived growth factor receptor–α. 10,31 This variation in protein expression is thought to be due to the presence of different subpopulations of CAFs within a neoplasm. The existence of different CAF subpopulations is further supported by the widely accepted hypothesis that CAFs arise from multiple different precursors. CAFs have been shown to transdifferentiate from resident fibroblasts, bone marrow–derived mesenchymal stem cells, and epithelial-mesenchymal transition. 17,24,29 Because only the presence of α-SMA was evaluated in the present study, it is possible that the neoplasms could have contained additional CAFs that only expressed fibroblast-specific protein or platelet-derived growth factor receptor–α.
CAFs are thought to promote an invasive and malignant tumor phenotype. Their presence in the stroma of human oral SCCs is associated with a more invasive growth pattern, deeper invasion, increased rates of locoregional lymph node metastasis, and increased levels of the cell proliferation markers (Ki67). 14 –16,18 These findings suggest that CAFs are likely to influence ST by promoting a more malignant behavior of the neoplasm. In this study, there was a strong association between POI and CAFs, where 86% of biopsies that exhibited high POIs were also positive for CAFs. This supports findings that CAFs are associated with an invasive growth pattern, and given the evidence from human studies, promoting a more malignant phenotype may be one of the mechanisms whereby CAFs influence ST in feline oral SCCs. Furthermore, the strong association between POI and CAFs may account for why the POI and site of the neoplasm was only weakly associated.
Differences in ST were observed among the 4 different SCC sites. Oropharyngeal SCCs had the longest ST compared with maxillary and mandibular SCCs, while sublingual SCCs had the shortest. Results for site-specific differences in ST in cats with oral SCC are variable. Oropharyngeal SCCs in cats have been previously associated with both shorter 28 and longer 11 STs compared with nonoropharyngeal SCCs. Furthermore, other studies have not detected an association between the site of a feline oral SCC and ST. 13,38 The variability in these results may be attributed to variations in site classification, treatment, small sample size, or misclassification of the original site due to advanced presentation of the tumor.
In humans, differences between ST and the location of the oral SCC are well established. Some of these differences are probably due to differences in the ease of surgical excision and different rates of metastasis of the oral SCC. In addition, SCCs in different locations tend to have different causes, and this may affect prognosis. Human papillomavirus–induced SCCs, typically being oropharyngeal, have a better prognosis than non–human papillomavirus–induced SCCs. 12 In cats, no cause for oral SCC has been established, with current evidence suggesting that these tumors are not significantly associated with either exposure to environmental tobacco smoke or papillomavirus infection. 4,20,21 The variation in ST observed in this study could indicate that feline oral SCCs do have different causes at different sites, and this warrants further investigation.
The present study showed an association between site and the presence of CAFs. Sublingual SCCs had the shortest median ST and contained the highest proportion of CAF-positive SCCs; conversely, oropharyngeal SCCs had the longest median ST and contained the lowest proportion of CAF-positive SCCs. It is possible that the differences in ST between the oral sites can be attributed, in part, to their association with CAFs. Furthermore, these results may suggest that the development of CAFs within the stroma could be dependent on the site of the SCC in the oral cavity. An association between site and CAFs has not been reported in humans, probably because most studies investigate oral SCCs from a single site. However, it is known that cancer cells and CAFs engage in molecular crosstalk and that the association between site and CAFs could actually be a reflection of the tumor phenotype. Further understanding of the biology of feline oral SCCs at different sites is required to appreciate why site and CAFs are associated. When the effect of CAFs on ST within each site was evaluated, there were no statistically significant differences between CAF-positive and CAF-negative tumors. The absence of significant differences within groups was suspected to be due to the smaller numbers of SCCs within each site category.
No significant associations were found between ST and the remaining epidemiologic, clinical, and histologic parameters. In humans, a high histologic grade, an invasive POI, and the presence of tumor budding are all associated with a worse prognosis of an oral SCC. 1,7,8 Although the POI was associated with CAFs, the assessment of this parameter, along with histologic grading and tumor budding, needs to be performed at the invasive front and this requires representative biopsy samples. It is possible that the quality and size of biopsy specimens in the present study did not contain histologically representative sections that truly reflected the behavior of the SCC. However, the results of the present study suggest that detection of CAFs within the stroma is a simpler and more reliable predictor of ST in cats with oral SCCs compared with histologic grading, POI, or assessment of tumor budding.
The retrospective nature of this study presents limitations to the data and statistical analysis. The biopsy samples were of variable size and depth, and samples may not have represented the true stage of development of the tumor. Furthermore, ST was calculated from the time of biopsy and not necessarily from the time of tumor initiation. Feline oral SCCs frequently present at an advanced stage, and this is likely to have an effect on the true ST. The effect of euthanasia as an end point is an important limitation in the present study. Although this factor is difficult, if not impossible, to eliminate, it must always be accounted for when interpreting survival data in veterinary species. The case material in this study was obtained from various veterinary clinics and veterinarians throughout New Zealand, and although treatment was limited to palliative care, there will be an unavoidable degree of variation in ST due to the differences in the clinical factors chosen by clients to determine the appropriate time for euthanasia.
Conclusions
This study demonstrates the potential prognostic value of assessing CAFs within the stroma supporting feline oral SCCs. This study also demonstrates that ST varies depending on the location of the oral SCC; cats with oropharyngeal SCCs survived up to 5 months longer compared with cats with sublingual and gingival SCCs. Three quarters of samples contained CAFs, suggesting that tumor-induced transdifferentiation of CAF precursors is common in feline oral SCCs. Cats with SCCs that did not contain a significant population of CAFs had a ST 2 weeks longer than cats with CAF-positive SCCs. This modest difference in survival needs to be weighed against the cost of immunohistochemical grading, but if future studies support the value of assessing α-SMA within the tumor stroma, it will become an easily accessible, simple, and inexpensive tool for providing veterinary practitioners with additional prognostic information.
Footnotes
Acknowledgements
We acknowledge Richard Campbell, Raewynne Pearson, Danielle Wiseman, Dr Adrienne French, Dr Mike Hardcastle, Dr Bernie Vaatstra, Dr Shen-Yan Hea, and the numerous veterinarians who facilitated and provided information regarding the reported cases.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
