Abstract
The present study aimed to evaluate the histologic, histochemical, and immunohistochemical changes in buffalo livers with cystic echinococcosis. Noninfected and infected livers were collected from the freshly slaughtered buffalo at the Aligarh abattoir. Small pieces of both infected and noninfected livers (n = 5) were cut and processed for histologic and histochemical studies. Immunohistochemistry was performed using rabbit anti-CD3, CD19, and CD117 antibodies. The results revealed the presence of brood capsules and germinal and laminated membranes surrounded by a fibrous adventitial layer, followed by moderate and diffused infiltration of eosinophils, monocytes, neutrophils, lymphocytes, and marked focal infiltration of mast cells. The infected livers also had mild dilation of central veins and sinusoids, mild and focal necrosis of hepatic tissue, and congestion of central and portal veins. Periodic acid-Schiff reaction revealed marked glycogen depletion in the infected liver. Masson’s trichrome stain showed marked deposition of collagen fibers in the portal area, adventitial layer, and between the hepatocytes compared with the noninfected liver, where deposition was found only in the portal area. The T-cell response was more pronounced than the B-cell response in infected liver. Thus, it can be concluded that hydatid cyst infection causes several pathological and biochemical changes and increased infiltration of inflammatory cells in the infected livers, suggesting the involvement of nonspecific immune responses against hydatid cysts. The T-cell response was more pronounced than B-cells, indicating the involvement of cell-mediated immunity against cystic echinococcosis. These findings may help to understand the local immune responses to cystic echinococcosis.
Introduction
Cystic echinococcosis (CE) is a cosmopolitan zoonotic disease caused by the larval stage of the taeniid tapeworm belonging to the species complex Echinococcus granulosus sensu lato (s. l.) and is amongst the list of severe parasitic diseases of humans. 43 E. granulosus sensu lato (s. l.) consists of many genotypes, among which G3 is most commonly prevalent in Indian water buffalo (Bubalus bubalis).8,19,20,36 The tapeworm’s life cycle involves dogs and wild canines as definitive hosts and various herbivorous and omnivorous mammals as intermediate hosts. Humans may acquire infection through direct contact with the definitive host or by consuming contaminated food and water. They are considered aberrant hosts as they are not involved in perpetuating the natural life cycle. 19 The eggs are released into the environment with the feces of the definitive host. When ingested by the intermediate host, the oncospheres are liberated from the eggs, penetrate intestinal mucosa through lytic secretion, enter the circulatory system (venous and lymphatic), and may lodge in any organ, preferably the liver and lungs, 25 where they develop into the fluid-filled metacestode (CE cysts).
The CE cyst comprises an inner nucleated germinal layer and an outer thick elastic acellular laminated layer of variable thickness, followed by a host-produced fibrous adventitial layer. 40 The clinical and economic importance of E. granulosus is confined to the metacestode infection. 39 In the initial primary infection stage, small cysts (<5.0 cm) do not produce any pathology and remain asymptomatic. After a prolonged incubation period, the growing cysts exert pressure and other space-occupying effects on adjacent vital organs, causing pain and other degenerative changes that result in clinical disease of the affected host. The clinical signs and symptoms of the disease depend upon the affected organs, size and number of cysts, complications due to cyst rupture, concurrent bacterial infections, and interaction between the expanding cyst and the adjacent organs. 26
The lifespan of hydatid cysts has been reported as long as 16 years in horses 33 and 53 years in humans. 37 Such prolonged survival of the parasite in its hosts indicated that the parasite uses several strategies to subvert or avoid the protective immune responses of the hosts. Two mechanisms are employed by the parasite to evade the host immune response: passive escape, wherein the parasite circumvents the detrimental effects of immune response by developing into hydatid cysts, and immunomodulation, whereby the parasite actively interacts with the host immune system to minimize its impact. 44 The immune responses to CE infection have been categorized into pre-encystment and post-encystment phases, distinguished by the formation of the laminated layer around the developing oncospheres. 31 During the early establishment phase, the cyst stimulates a Th1-type immune response, which is responsible for killing the metacestode at the initial stage of development.38,44 However, the established-phase cyst stimulates a Th2-type immune response, which favors the establishment of parasites by involving interleukin-10, which inhibits the killing of parasites. 45
CE causes mild to acute pathological changes like hepatocellular degeneration, biliary hyperplasia, necrosis, and sinusoid congestion.2,5–7,14 Furthermore, rupture of the cysts releases the cyst fluid with protoscoleces, which may be responsible for disease recurrence and anaphylactic shock. The host-derived fibrous adventitious layer around the cysts is thought to play an essential role in regulating local immune response during infection. 12 Many workers have reported the presence of lymphocytes, eosinophils, neutrophils, plasma cells, macrophages, and fibroblasts, mainly above the adventitial layer, whereas in a few cases, above the cyst wall in the infected liver and lungs of sheep and camel.1–3,5–7,24,35 The local immune responses against the hydatid cyst infection were also investigated by identifying different immune cells using their markers. Many workers have identified T-cells by using CD3, CD4, and CD8 markers and B-cells by CD19 and CD79 markers in sheep,16,42 cattle,18,34 and human hosts17,41 as well as in experimentally infected mice 14 with hydatid cyst, but no study is available on buffaloes. Therefore, in the present study, histologic, histochemical, and immunohistochemical studies were carried out to investigate the damage caused by the hydatid cysts and the host’s responses against G3 genotype infecting buffalo. These pathological changes in the host will provide an insight into disease manifestation.
Materials and Methods
Sample Collection and Preparation
A total of 50 livers were collected from Indian water buffalo freshly slaughtered at the Aligarh abattoir, brought to the laboratory, and analyzed for the presence of hydatid cyst. Among these, 15 livers had hydatid cyst, out of which 5 samples were analyzed in the present study. The infected liver tissue adjacent to the hydatid cyst was cut into 1 × 1 cm pieces, washed with phosphate-buffered saline (PBS), and fixed in 10% neutral-buffered formalin. After fixation, blocks were paraffin-embedded. Small pieces of noninfected livers (n = 5) were also processed in the same way as described above and used as a control.
Histology
Serial sections of 2 to 5 µm in thickness of both infected and noninfected livers were cut on a rotary microtome (York Scientific Industries, Delhi), floated on albumin-coated glass slides, stretched, and were used for histologic and histochemical studies using previously described methods. 27 The tissue sections were stained with hematoxylin and eosin for histologic assessments and with toluidine blue, Periodic acid-Schiff, and Masson’s trichrome for the localization of mast cells, glycogen, and collagen, respectively. The histologic changes were graded as mild, moderate, and marked on the basis of degree of changes in the tissue and as focal and diffused based on distribution/pattern of changes.
Immunohistochemistry
For the immunohistochemical assays, 3 to 5 µm thick sections were floated on poly-L lysine-coated glass slides, dewaxed, and rehydrated. Antigen retrieval was performed by heating the slides in 10 mM sodium-citrate buffer (pH = 6.0) with 0.05% Tween 20 at 95°C in a microwave oven for 30 minutes and cooled at room temperature. Endogenous peroxidase blocking was performed by incubating the slides in 3% H2O2 for 30 minutes at room temperature and then incubating the slides in Tris buffer (pH = 7.2) for 5 minutes. The primary antibodies against CD3 (rabbit monoclonal antibody, Epredia), CD19 (rabbit monoclonal antibody, PathnSitu Biotechnologies), and CD117 (rabbit polyclonal antibody, PathnSitu Biotechnologies) were diluted in PBS (pH = 7.2) in the ratio of 1:100. These primary antibodies were then added to the slides and incubated overnight in a humidity chamber. After incubation, slides were washed with Tris buffer (pH = 7.2) and a secondary antibody (antirabbit antibody conjugated with horseradish peroxidase, PathnSitu Biotechnologies) was added to the slides, incubated for 30 minutes, and washed 3 times for 3 minutes each with Tris buffer (pH = 7.2). The antigen-antibody reaction was visualized using 3,3’ diaminobenzidine (DAB) chromogen (Biogenix Technologies), diluted in DAB buffer at a ratio of 1:50. The slides were counterstained by hematoxylin for 30 seconds, washed with distilled water, dehydrated, cleared, and finally mounted in DPX (Sisco Research Laboratories Pvt. Ltd.). The total number of immune positive cells was counted in 10 microscopic field using a 40× lens (0.344 mm2 field of view), and the percent labeled immune cells per total cells was expressed as mild (1%-10% of cells), moderate (11%-50% of cells), and marked (>51% of cells) reactions. Negative and positive controls were also run simultaneously using lymph nodes as positive control, whereas in negative control, PBS was used instead of primary antibody. All microphotographs were taken on the light microscope (Olympus BX51DIC, Japan) attached to a digital camera, Olympus DP74, Japan.
Results
Histologic changes were evaluated in hematoxylin and eosin-stained slides. The infected livers contained increased sinusoid and central vein dilation; portal and central vein congestion; mild, focal necrosis of hepatic tissue; cytoplasmic vacuolation; and increased inflammatory cell infiltration in the portal areas (Fig. 1a) compared to the noninfected livers, which had no histologic lesions (Fig. 1b). In the infected livers, there was a thin inner germinal layer containing a brood capsule with protoscoleces surrounded by a laminated layer and an outer fibrous adventitial layer (Fig. 1c, d). Intense cellular infiltrates above the adventitial layer were composed of eosinophils, monocytes, neutrophils, and lymphocytes (Fig. 1e). However, only a few immune cells were present in the hepatic plates of control livers (Fig. 1f).
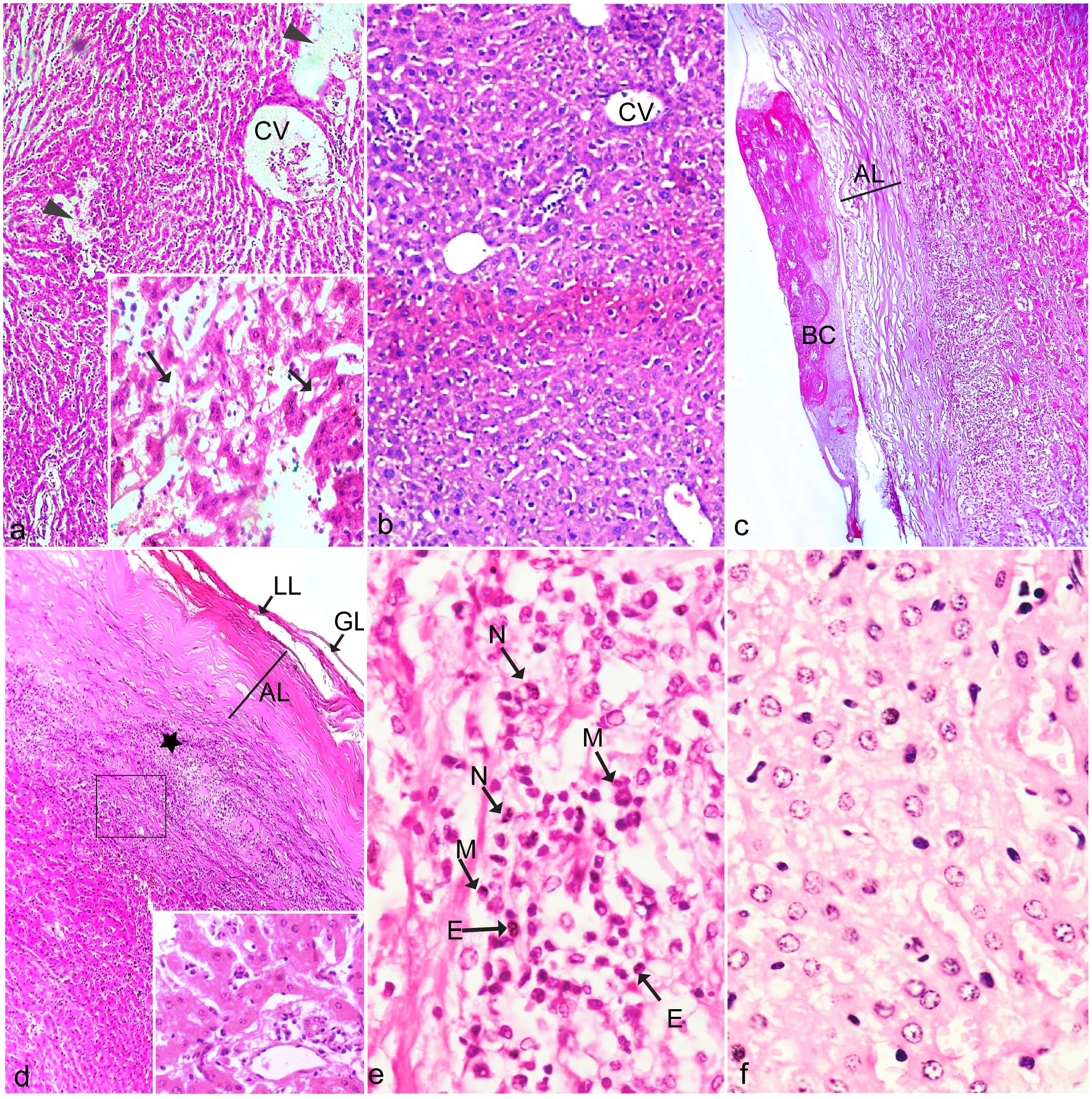
Histologic sections of noninfected and infected liver with cystic echinococcosis. Hematoxylin and eosin. (a) Infected liver showing dilation and congestion of central vein (CV) and necrosis of hepatic tissue (arrowhead). Inset: Higher magnification showing cytoplasmic vacuolation (arrows). (b) Noninfected liver showing the normal structure of central vein (CV) and hepatocytes. (c) Infected liver showing a brood capsule (BC) and fibrous adventitial layer (AL). (d) Infected liver showing germinal layer (GL), laminated layer (LL), and moderate cellular infiltration (asterisk) around the adventitial layer (AL). The inset represents a higher magnification view of the area within the box. (e) Infected liver showing intense infiltration of monocytes (M), neutrophils (N), and eosinophils (E). (f) Noninfected liver with few lymphocytes.
The histochemical stains revealed the differential distribution of glycogen and collagen fibers in infected and noninfected livers. A strong periodic acid-Schiff reaction was present in the noninfected livers, indicating abundant hepatocellular glycogen, whereas a weak reaction in the infected livers indicated marked depletion of glycogen (Fig. 2a, b). Collagen was found only in the portal areas in the noninfected livers, whereas in the infected livers, marked deposition of collagen fibers was found in the fibrous adventitial layer of the cyst and dissecting between hepatocytes (Figs. 2c, d). Staining of the sections by toluidine blue revealed the absence of mast cells in noninfected livers (Fig. 2e), whereas there was marked infiltration of mast cells in the infected livers (Fig. 2f). This was further confirmed by anti-CD117 immunohistochemistry, which immunolabels mast cells. CD117 immunohistochemistry indicated scattered cells with weak immunolabeling in the noninfected livers (Fig. 3a), whereas the infected livers contained increased numbers of cells with strong immunolabeling (Fig. 3b). Furthermore, T- and B-cells were also localized in infected and noninfected livers based on the immunolabeling patterns of antibodies against CD3 for T-cells and CD19 for B-cells. A low number of T-cells were diffusely distributed throughout the noninfected livers (Fig. 3c), whereas the infected livers had localized increased numbers of T-cells adjacent to the cyst (Fig. 3d). In contrast, both noninfected and infected livers were observed to have rare scattered cells immunolabeled for CD19 (Fig. 3e,f).
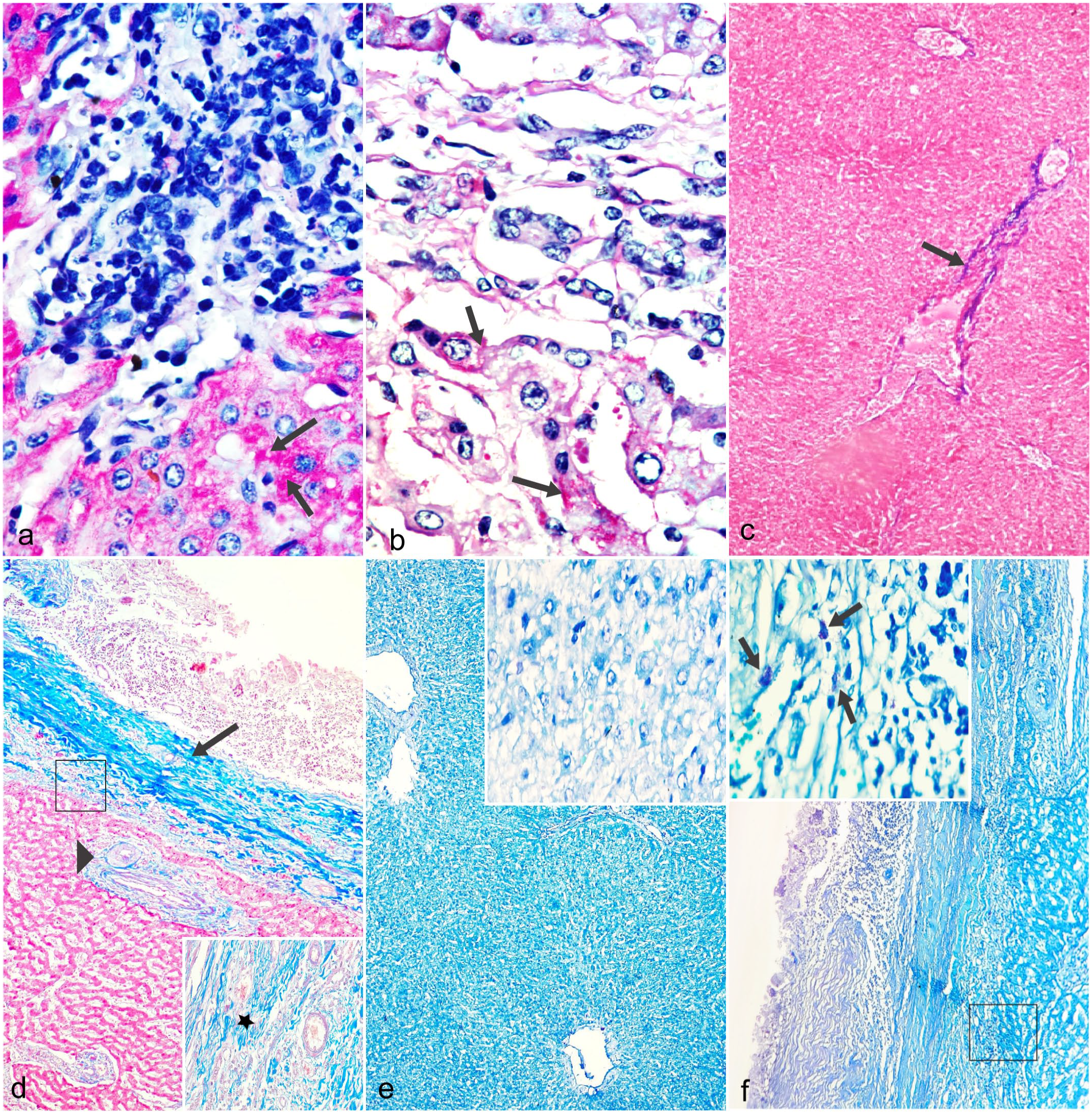
Histochemical localization of glycogen, collagen, and mast cells in noninfected and infected livers with cystic echinococcosis. (a) Noninfected liver showing abundant intrahepatocellular glycogen (arrows). Periodic acid-Schiff reaction (PAS). (b) Infected liver showing marked depletion of glycogen (arrows) (PAS). (c) Noninfected liver showing collagen deposition only in the portal area (arrow). Masson’s trichrome. (d) Infected liver showing increased deposition of collagen in the portal area (arrowhead) and adventitial layer of the brood capsule (arrow). Inset: higher magnification showing collagen in between hepatocytes (asterisk). Masson’s trichrome. (e) Noninfected liver showing absence of mast cells. Inset: higher magnification. Toluidine blue. (f) Infected liver showing numerous mast cells. The black square represents the location of the inset that shows a higher magnification image of mast cells (arrows). Toluidine blue.
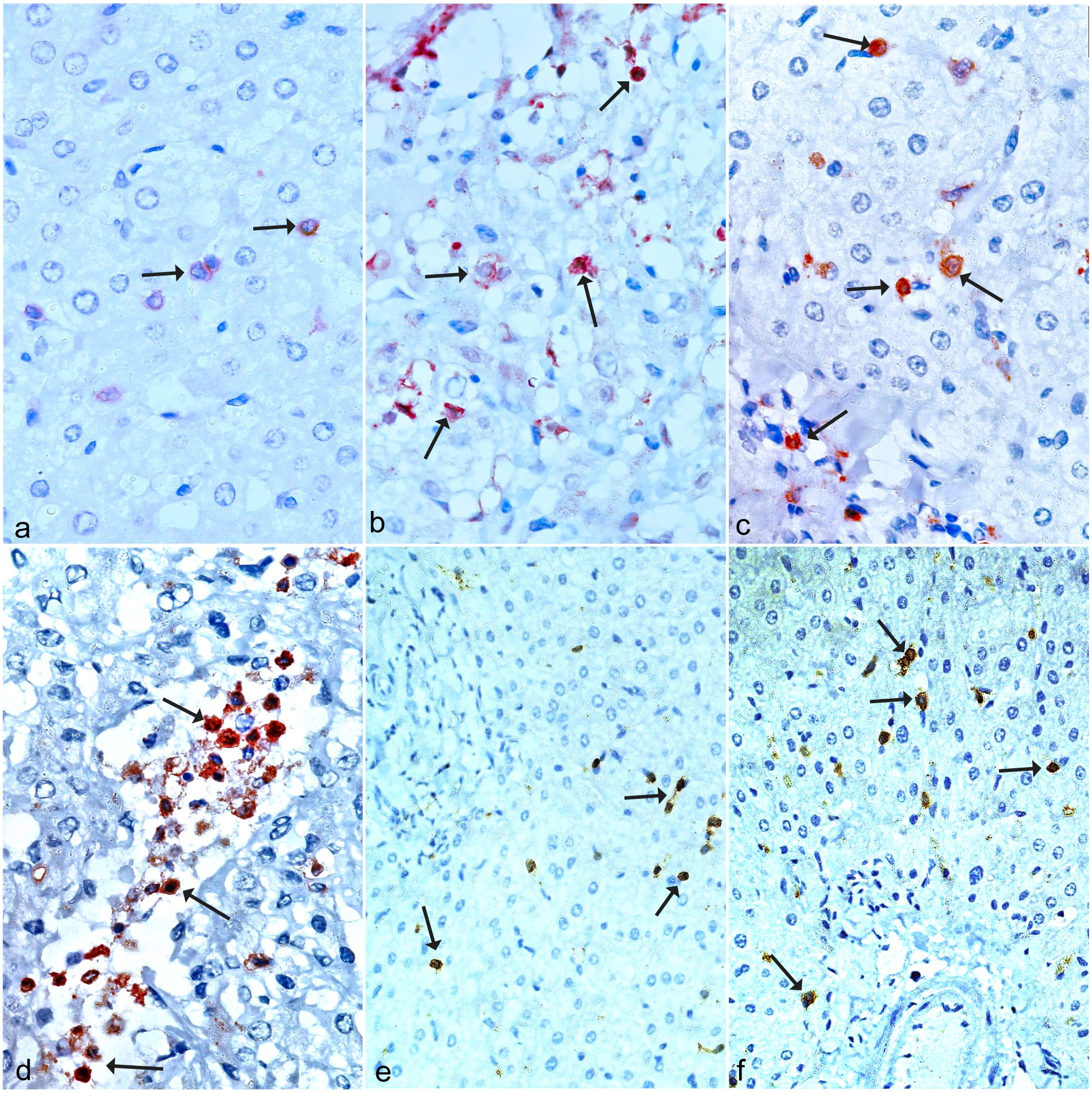
Immunohistochemical localization of CD117, CD3, and CD19 in noninfected and infected livers with cystic echinococcosis. (a) Noninfected liver with few scattered CD117-immunolabeled cells (arrows). CD117 immunohistochemistry (IHC). (b) Infected liver with focally increased numbers of CD117-immunolabeled mast cells adjacent to the cyst (arrows). CD117 IHC. (c) Noninfected liver with few scattered, individual CD3-immunolabeled cells (arrows). CD3 IHC. (d) Infected liver with focally increased numbers of CD3-immunolabeled cells (arrows). CD3 IHC. (e, f) Few scattered CD19-immunolabeled cells in (e) noninfected and (f) infected livers (arrows). CD19 IHC.
Discussion
CE causes mild to severe pathology depending upon the organs involved, the size and number of cysts, and their interaction with the adjacent organs. 13 In the present study, congestion of portal veins, dilation of central veins and sinusoids, and degeneration of the hepatic tissue were observed in the infected livers with hydatid cysts. Such changes could be explained by the fact that the growing cysts exert pressure and other space-occupying effects, causing the replacement of the host’s tissue and other degenerative changes in the affected organs. Similarly, many investigators have reported hepatocellular degeneration, portal vein congestion, sinusoid dilation, necrosis, and fibrosis in portal areas of infected livers of sheep,3,16,23,35 camels, 24 and experimentally infected mice, 14 where these changes were correlated with the size of cysts and pressure exerted by the growing cyst. Beige et al5–7 reported cellular degeneration, necrosis, cirrhosis, and dilation of sinusoids in the infected liver and congestion, hemorrhage, peribronchiolar epithelial hyperplasia, alveolar dilation, and calcification in the infected lungs of sheep with hydatid cysts and suggested that the severity of histopathologic changes was inversely related to the distance of tissues from the cyst.
Increased infiltration of monocytes, neutrophils, eosinophils, lymphocytes, and fibroblasts was observed around the fibrous adventitious layer, which indicates the host’s inflammatory response against the parasites. The damage caused by the parasite to the host tissue triggers the release of vasoactive and chemotactic factors, which increase the permeability of blood vessels and blood flow to the affected area, leading to an influx of macrophages, eosinophils, basophils, and mast cells from the blood to the damaged site and thus causing inflammation. 3 Similarly, many investigators have reported infiltration of monocytes, neutrophils, eosinophils, lymphocytes, and fibroblasts above the fibrous adventitial layer in the infected livers of sheep and camels and correlated this with the host inflammatory response against the parasite.2,3,5–7. In experimentally infected sheep, Petrova 28 reported necrosis and infiltration of neutrophils and macrophages around the oncosphere within 3 to 5 days post-infection. In vitro, experimental studies have also shown that neutrophils, with the help of antibodies, kill the oncospheres of E. granulosus through antibody-dependent cell-mediated cytotoxicity. 32
Increased infiltration of mast cells was observed around the hydatid cysts, which could be a host’s defense response against parasites, as it was shown that mast cells are involved in expelling parasites from the intestine by increasing epithelial paracellular permeability. 22 It is presumed that the hydatid cyst infection may induce the recruitment of mast cells at the site of infection that release chemotactic factors that attract and enhance phagocytic cells’ function, thus providing defense against parasites.11,45 However, it is not clear whether infiltration of mast cells occurs primarily against the migrating oncospheres or due to the exudation of hydatid fluid from enlarging cysts. 6 Therefore, further studies are required to identify the triggering agent of mast cell activation due to hydatid infection. Similar to our findings, an increased number of mast cells have been reported in infected livers of sheep and mice with CE,3,6,7,14 in abomasal tissues of goats infected with Haemonchus contortus, 30 and in the intestine and gills of fishes infected with acanthocephalan (Echinorhynchus truttae) and copepod (Ergasilus sieboldi), respectively. 10 These studies have correlated the increased number of mast cells with local type-I hypersensitivity and the host’s defense response.
Depletion of glycogen was noticed in the infected livers, which could be due to its utilization by the parasite for their survival, as suggested by Al-Sabawi et al, 3 who also found less glycogen in the livers of sheep infected with hydatid cysts. Cheng and Snyder 9 reported the depletion of glycogen content in the hepatopancreatic cells of snails (Helisoma trivolvis), and the appearance of polysaccharides within the developing cercariae of Glypthelmins pennsylvaniensis, and correlated this with the utilization of the host’s glycogen by the parasite. They postulated that the developing cercariae might secrete a glycogen-digesting enzyme, which hydrolyzes the glycogen of hepatopancreatic cells of snails, and absorbs the simpler sugar, which is then utilized by them to resynthesize glycogen.
More deposition of collagen in the portal area, around the adventitial layer, and in between the hepatocytes was noticed in the infected livers, indicating increased synthesis of collagen by the parasitized organ because of continuous irritation caused by the developing cysts and gradual and slow exosmosis occurring in the cyst, which could be a protective response of the host that leads to the formation of cysts wall and confinement of the parasite. Increased collagen deposition has been reported in hepatic hydatid cysts of sheep, goats, and cattle,3,6,7,16,29 where a similar suggestion has been made. Anwar et al 4 reported that fibrosis and cirrhosis in infected sheep livers with hydatid cysts may be due to immunological reactions of the host tissue.
Immunohistochemical studies were performed to identify infiltrated cells in noninfected and infected liver tissues with hydatid cysts. Increased CD3-positive T-cells were observed in the parasitized than the nonparasitized livers of buffalo, whereas few CD19-immunolabeled B-cells were identified in both infected and noninfected livers. Such differential response suggests the involvement of T cell-mediated immunity against hydatid cysts. Similarly, many researchers have reported marked expression of CD3 markers (T-cells) in the infected tissue of lungs and liver of sheep, cattle, humans, and mice with CE2,14,16,17,42 and suggested the involvement of a T-cell response against hydatid cysts. Hou et al 15 reported a predominant occurrence of CD4-positive T-cells in hepatic tissues of humans and mice infected with hydatid cysts and suggested that T-cells are involved in forming an immune environment.
Thus, it can be summarized that hydatid cyst infection induces several pathological and biochemical changes along with increased infiltration of inflammatory cells in the liver of water buffalo (Supplemental Figure S1), suggesting the involvement of immune responses against hydatid cysts. The T-cell response was more pronounced than the B-cell response, indicating the involvement of cell-mediated immunity against CE (Supplemental Figure S1). These findings may be helpful in understanding the local immune responses against CE and designing control strategies. However, more studies are required on the involvement of these cells in organs other than the liver, and hosts infected with different genotypes of Echinococcus, as genotypes vary in host specificity, pathogenicity, antigenicity, transmission dynamics, and control of hydatid disease.8,21
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241309399 – Supplemental material for Histologic, histochemical, and immunohistochemical changes in buffalo liver with cystic echinococcosis
Supplemental material, sj-pdf-1-vet-10.1177_03009858241309399 for Histologic, histochemical, and immunohistochemical changes in buffalo liver with cystic echinococcosis by Humera Aslam, Hana Hussein M. S. Wardah, Kafil Akhtar, Sayeedul Hasan Arif and Malik Irshadullah in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank the Chairperson, Department of Zoology and Department of Pathology, Aligarh Muslim University, Aligarh, for providing the laboratory facilities.
Author Contributions
HA and MI contributed to the study’s conception, design and methodology validation. HA collected material and performed data analysis, and HH helped in the collection and processing of samples. SHA and KA helped in section cutting, imaging, and analysis of histopathological and immunohistochemical studies. HA wrote the first draft of the manuscript, and MI performed the editing and supervision. All authors have commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author (HA) is thankful to the University Grants Commission, New Delhi (575-CSIR-UGC NET JUNE 2018) for providing financial assistance.
Consent to Publish
Authors gave their consent to publish.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
