Abstract
Cystic echinococcosis is a worldwide zoonosis caused by the cestode Echinococcus granulosus. Two types of hydatid cysts occur in intermediate hosts: fertile cysts that generate protoscoleces from the germinal layer of the cyst, and infertile cysts that do not produce protoscoleces and are unable to continue the life cycle of the parasite. The adventitial layer, a host-derived fibrous capsule surrounding the hydatid cyst, is suggested to play an important role in local immune regulation during infection and in fertility of the cysts. Fasciola hepatica, another important parasite of cattle, induces a characteristic Th2-like immune response that could modulate the immune response against E. granulosus. Natural co-infection of both parasites is common in cattle, but no reports describe the local immune response against E. granulosus with F. hepatica infection in the same host. This study analyzed the number and distribution of T and B cells in the adventitial layer of liver and lung cysts and the relationship with cyst fertility and F. hepatica co-infection. T lymphocytes were the predominant cell type in the adventitial layer of infertile hydatid cysts and were more numerous in infertile hydatid cysts. B lymphocyte numbers were not associated with hydatid cyst fertility. Mast cells were infrequent in the adventitial layer. The number of T and B cells was not associated with F. hepatica co-infection. The present study contributes to the understanding of local immune responses in bovine cystic echinococcosis.
Keywords
Cystic echinococcosis is a globally distributed parasitic disease caused by the cestode Echinococcus granulosus. 20 Higher prevalence of cystic echinococcosis in human and animals are found in countries of temperate zones, including several parts of Eurasia (Mediterranean regions, southern and central areas of Russia, central Asia, China), Australia, some regions of America (especially South America), and north and east Africa. 11 In livestock, particularly cattle and sheep, this disease has a great economic impact and affects the production of meat, wool, milk, and fertility of the infected animals. 2
The adult cestode inhabits the small intestine of carnivores (definitive host, usually canids) and produces proglottids containing infective oncospheres. After oral uptake of oncospheres by an intermediate host (normally herbivores), the metacestode develops in internal organs (mainly liver and lungs) as a fluid-filled, unilocular structure. 13 The hydatid cyst comprises two parasite-derived layers—an inner nucleated layer called the germinal layer and an outer acellular laminated layer—surrounded by a host-produced fibrous capsule called the adventitial layer. 33 In intermediate hosts, 2 types of hydatid cysts can occur: fertile cysts that develop buds and protoscoleces (the form of the parasite that is infective to definitive hosts) attached to the germinal layer and free in the hydatid fluid and infertile cysts that do not generate protoscoleces and therefore are not able to continue the parasite life cycle. 16
The biological and molecular background underlying the generation of both types of cysts have not yet been elucidated. 29 However, the host immune response is most likely involved in the fertility status of hydatid cysts. 1,18,19,23,32,34 Hydatid cysts in sheep and humans are usually surrounded by a fibrous capsule infiltrated by macrophages and B and T lymphocytes. In some cases, eosinophils are present but are always distal to the parasite. Pigs and cattle form a typical granulomatous reaction with epithelioid macrophages and multinucleated giant cells surrounding the hydatid cyst and an outer mononuclear cell infiltrate. 5 In cattle, fertile hydatid cysts with viable protoescoleces lack this granulomatous reaction, while both fertile hydatid cysts with dead protoscoleces and infertile cysts feature the aforementioned reaction, with the adventitial layer inflammatory cells infiltrating the laminated layer. 12
Fasciola hepatica is a common parasite of cattle worldwide. 22 As for many other helminth infections, F. hepatica evokes a chronic Th2-like immune response. 3 The immune response to F. hepatica affects the host reaction to other pathogens. 6 For example, cattle co-infected with F. hepatica and Mycobacterium bovis have lower IFNγ levels, altering the immune response and the progression of tuberculosis. 9 Further, we previously reported that in cattle with E. granulosus infection, F. hepatica co-infection is associated with an overall higher proportion of lung hydatid cysts and smaller hydatid cysts in the lung. 27 Alterations of the host local or systemic immune response were not reported.
However, the local immune response in both fertile and infertile cysts in cattle has been evaluated. It has been reported that early establishment-phase cysts stimulate a Th1-type immune response while established-phase cysts are associated with a Th2-type immune response. 21,33 The establishment phase of cystic echinococcosis involves infiltration of eosinophils, neutrophils, macrophages, and fibrocytes. 21,33 This generally results in a low-level inflammatory response, and aged cysts are surrounded by a fibrous layer that separates the laminated layer from host tissue. 33 Currently, there are no data on the specific immune cell populations associated with fertile versus infertile cysts or the effect of co-infection with F. hepatica. The aims of the present study were to evaluate the populations of T and B cell lymphocytes and mast cells and evaluate the effect of co-infection with F. hepatica on the cellular immune response in the adventitial layer of fertile and infertile cysts of naturally infected cattle.
Materials and Methods
Sample Collection
Tissue samples infected with E. granulosus and F. hepatica were obtained from cattle slaughtered at an abattoir in Santiago, Chile. All samples were of mixed breed cattle. Based on the antemortem and postmortem examination by the official veterinary service, lethargic animals were excluded as well as those with tuberculosis or cysticercosis. Postmortem examination comprised visual inspection and incision of viscera for the presence of hydatid cysts in liver and lungs and F. hepatica in liver. Hydatid cysts were processed and sorted as previously described. 10 Briefly, cysts were considered infertile if they showed a thin laminated layer with yellow-brown inner surface, clear hydatid fluid, and absence of protoscoleces either by macroscopic or microscopic inspection. On the contrary, hydatid cysts were considered fertile if they had a thick and whitish laminated layer and protoscoleces in the hydatid fluid or attached to the germinal layer. When protoscoleces were not visible in the hydatid fluid, the absence of protoscoleces was confirmed by microscopic examination of the germinal layer. The viability of protoscoleces was analyzed with trypan blue exclusion test. 28 All hydatid cysts included, either fertile or infertile, were at least of 1.5 cm diameter. 4,16
E. granulosus (s.l.) genotyping was determined as previously reported. 4,27 Briefly, DNA was extracted from fertile and infertile hydatid cysts. A partial COX1 tDNA (345 nt) sequence was amplified and sequenced. For Echinococcus granulosus sensu stricto, the Eg01 haplotype (GenBank Accession no. JQ250806) was used as the reference, and for Echinococcus ortleppi, the Eo01 haplotype (GenBank Accession no. KU743915) was used.
F. hepatica was diagnosed by direct visualization of adult parasites in bile ducts or compatible chronic lesions in the liver.
The samples were categorized as fertile hydatid cysts (F) with (FD+) or without (FD–) F. hepatica co-infection or infertile hydatid cysts (I) with (ID+) or without (ID–) co-infection. The University Andres Bello Bioethics Board approved the study protocol (protocol number 016/2016).
Immune Cells Infiltrating the Adventitial Layer in Fertile and Infertile Hydatid Cysts
Liver and lung hydatid cyst wall samples from both fertile and infertile cysts were fixed in Glyofix (Thermo) and embedded in paraffin, and 5-µm sections were placed on Thermo Scientific Superfrost Plus microscope slides for staining with hematoxylin and eosin (HE) 17 or Toluidine blue (for mast cells). 26 Samples with germinal, laminated, and adventitial layers in the same slide were selected for analysis. All slides images were captured using Olympus FSX100 Microscope with FSX-BSW software.
For immunohistochemical (IHC) assays, tissue sections were dewaxed in Neo-Clear, and intrinsic peroxidases were blocked with 3% H2O2 in methanol. Samples were washed 2 times with PBS 1X, and proteins were blocked for 30 minutes with ScyTek Pro-Block. Tissue sections of fertile and infertile cysts were then incubated with either 1:50 (v/v) monoclonal antibody against human CD3 (Dako, Cat No. M7254) for 30 minutes or 1:200 (v/v) polyclonal antibody against human CD20 (Thermo, Cat No. PA5-16701) at 4°C overnight. Slides were washed 2 times with PBS and incubated for 40 minutes at room temperature with EnVision + horseradish peroxidase (HRP) solution (Dako, Agilent technologies) for anti-CD20 antibody and ImmPRESS HRP Universal Antibody (Anti-Mouse IgG/Anti-Rabbit IgG, Peroxidase) for anti-CD3 antibody. After washing in PBS, antibody binding was detected with Vector ImmPACT DAB, and slides were counterstained with hematoxylin.
Adventitial Layer Immune Cell Counts
Immunohistochemical quantification was performed by Image J 1.44p software (National Institutes of Health, USA). Briefly, for each sample, a study area of 1 mm2 of adventitial layer was analyzed, using the laminated layer as a tissue reference. Images were converted to an 8-bit gray-scale format, and the threshold was set above the background staining intensity. 24,25 Positive signals were detected automatically, and the frequency of the cells was expressed as the number of positive signals/mm2.
Statistical Analysis
Data were recorded in Excel. Descriptive statistics were expressed as frequencies (qualitative data) or medians and interquartile range (IQR) for quantitative data, according to their nonnormal distribution. Inferential statistics were performed using Kruskal-Wallis test followed by Dunn’s post hoc test for pairwise comparisons using Stata V12 package. Differences were accepted as statistically significant when P values were below the .05 threshold.
Results
A total of 19 fertile hydatid cysts and 23 infertile hydatid cysts with a germinal, laminated, and adventitial layer were considered for this study. All samples corresponded to E. granulosus s.s. according to the DNA partial sequence of the COX1.
Fertile cysts had a thicker laminated layer compared to infertile cysts (Figs. 1–3). An inflammatory cell distribution pattern was observed in the adventitial layer, which was overall more intense in infertile cysts than fertile cysts (Figs. 1–4). The adventitial layer of fertile cysts consisted of fibrosis peripheral to the laminated layer and then the normal host tissue (Figs. 1, 2). In infertile and some fertile cysts, palisading macrophages were peripheral to the laminated layer (Fig. 3), followed by aggregated lymphocytes (Figs. 3 –12) and then the normal host tissue.
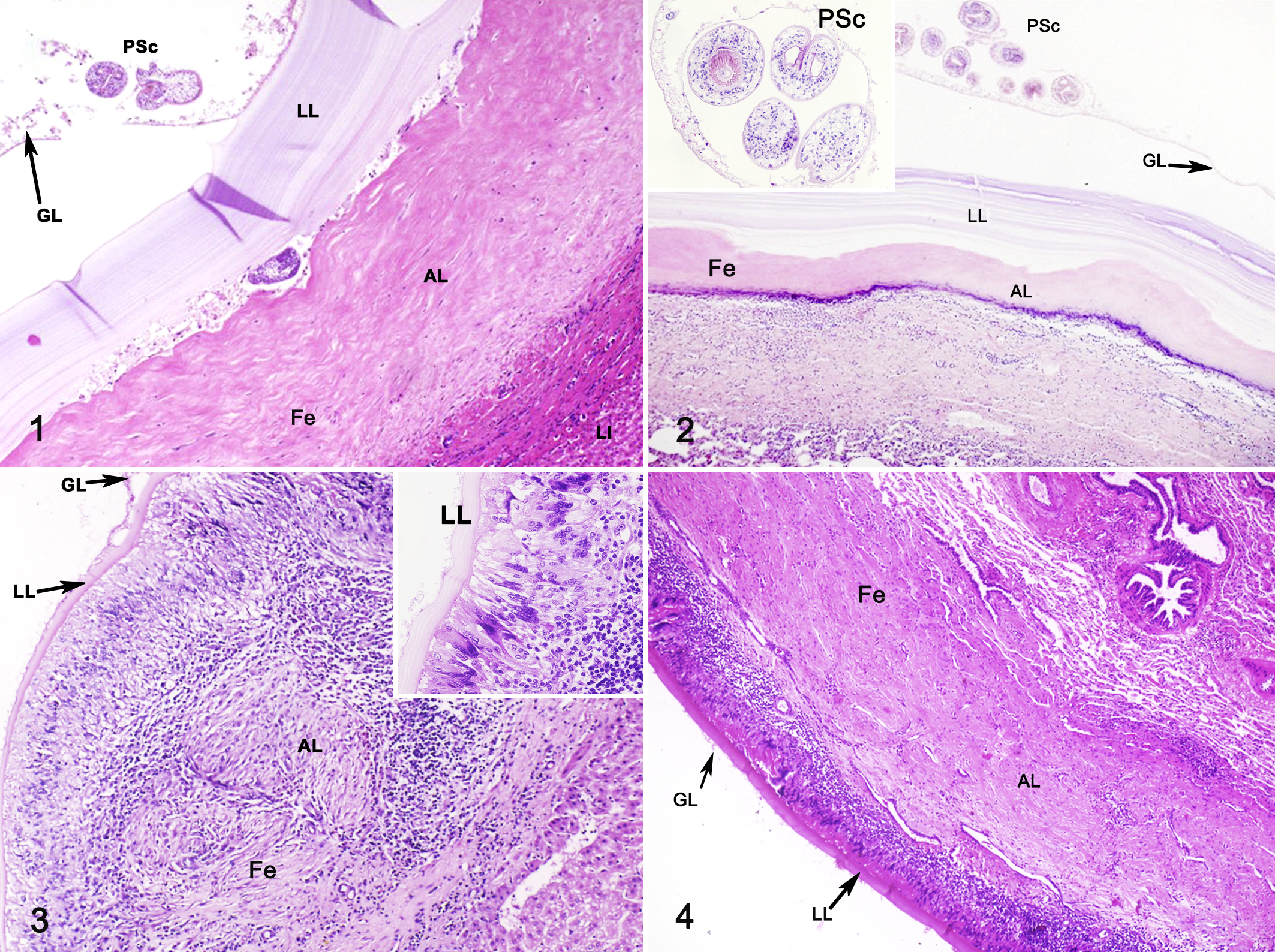
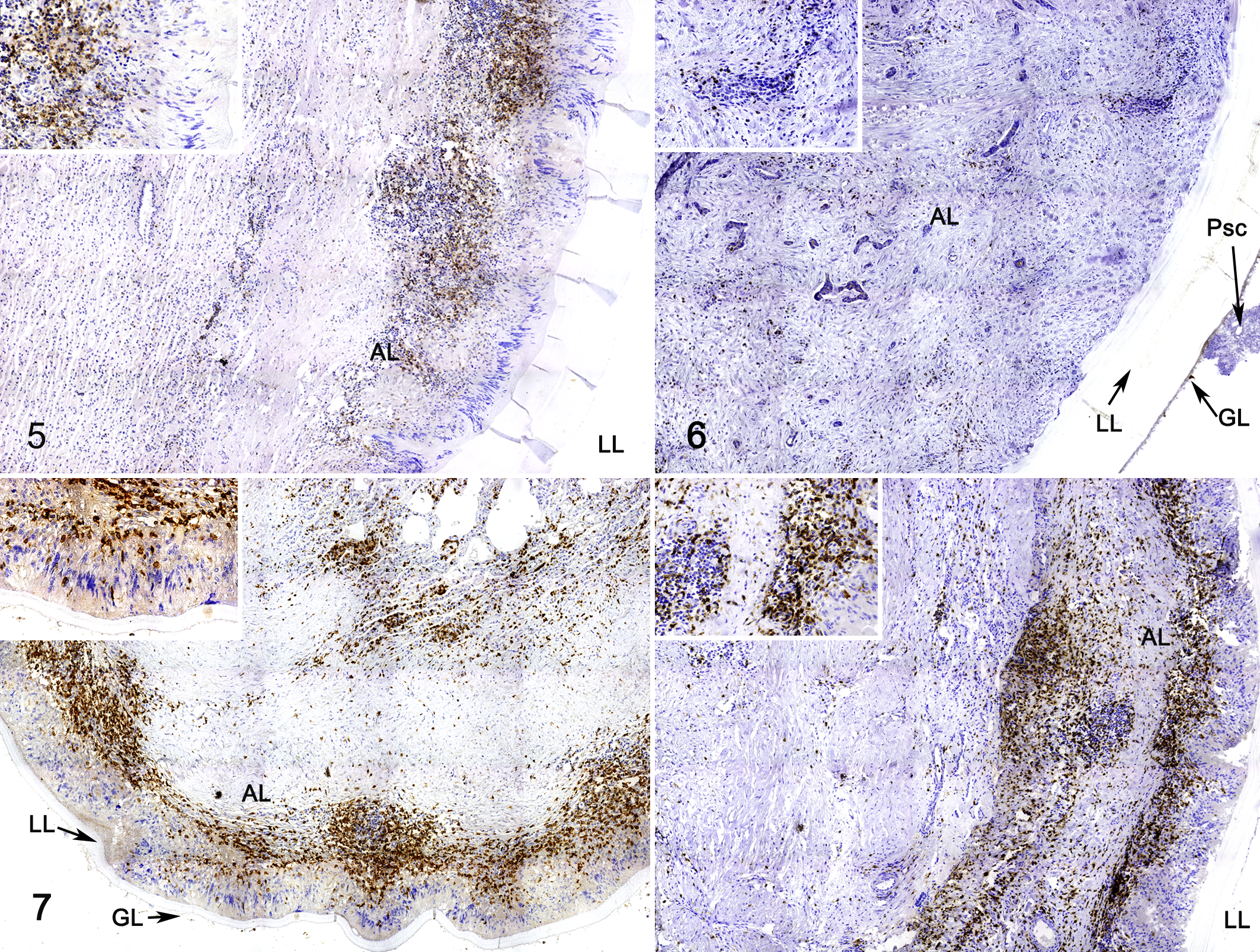
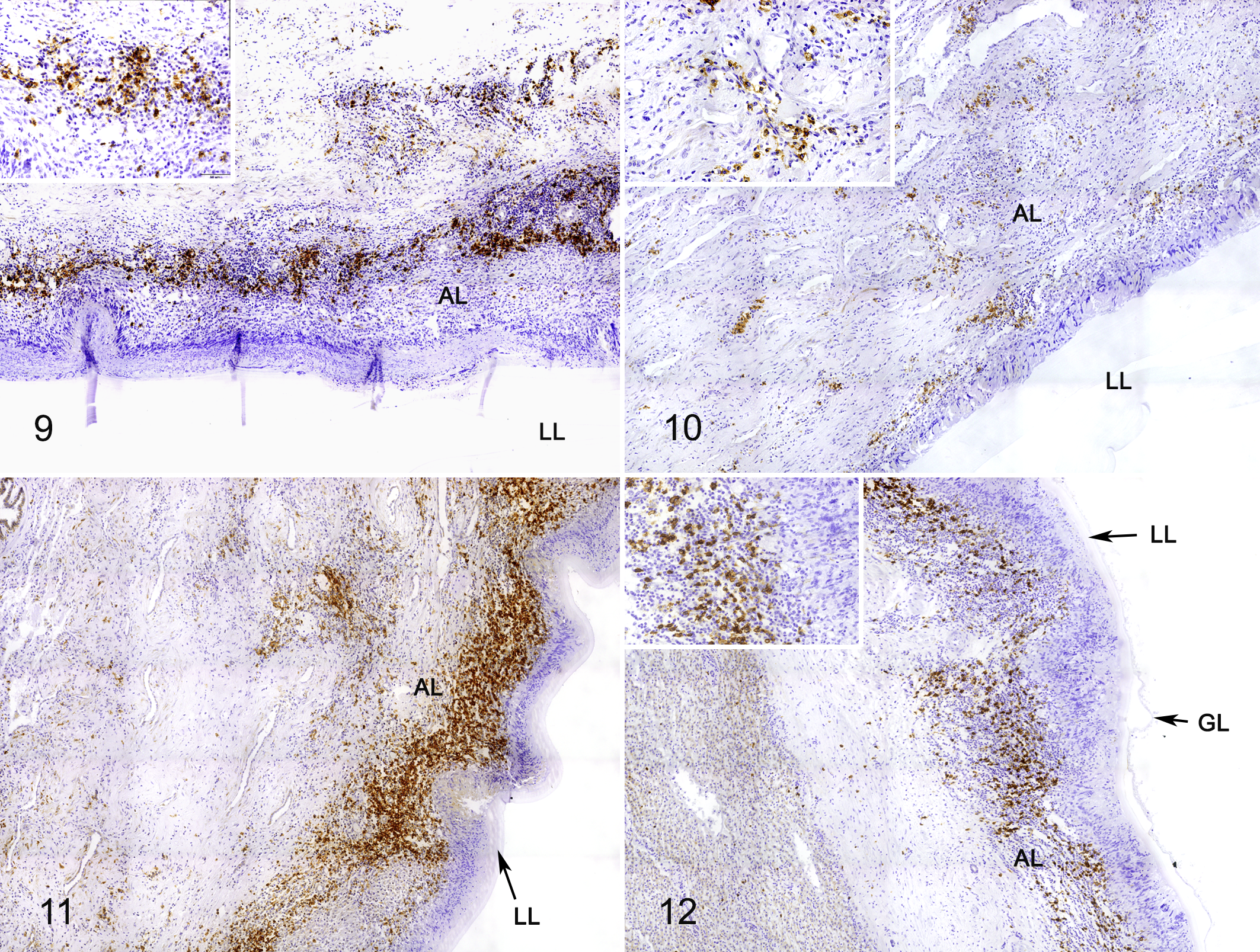
CD3-positive cells (T lymphocytes) were analyzed in 19 fertile cysts and 21 infertile cysts (8 liver cysts and 32 lung cysts), B lymphocytes (CD20 marker) were evaluated in 6 fertile cysts and 16 infertile cysts (2 liver cysts and 16 lung cysts), and mast cells were evaluated in 10 fertile cysts and 12 infertile cysts (3 liver cysts and 19 lung cysts). Due to the low number of liver cyst samples, statistical analysis was performed only for lung cysts.
CD3+ cells were peripheral to the palisading macrophages, and some were also present near the normal host tissue (Figs. 5, 7, 8). ID+ cysts had a median cell count of 2825 (IQR = 777.5) compared to 2580 (IQR = 1165) for ID– cysts. Both ID+ and ID– cysts had significantly higher CD3+ T cell populations in the adventitial layer compared to FD+ (245 median cell count; IQR = 684) and FD– (904 median cell count; IQR = 725) cysts (P < .05). Although there were no significant differences in T cells counts in adventitial layer of FD+ compared to FD– samples, T cells in FD– and ID– samples were 3.7 times higher than FD+ and ID+ samples (P < .05; Fig. 13).
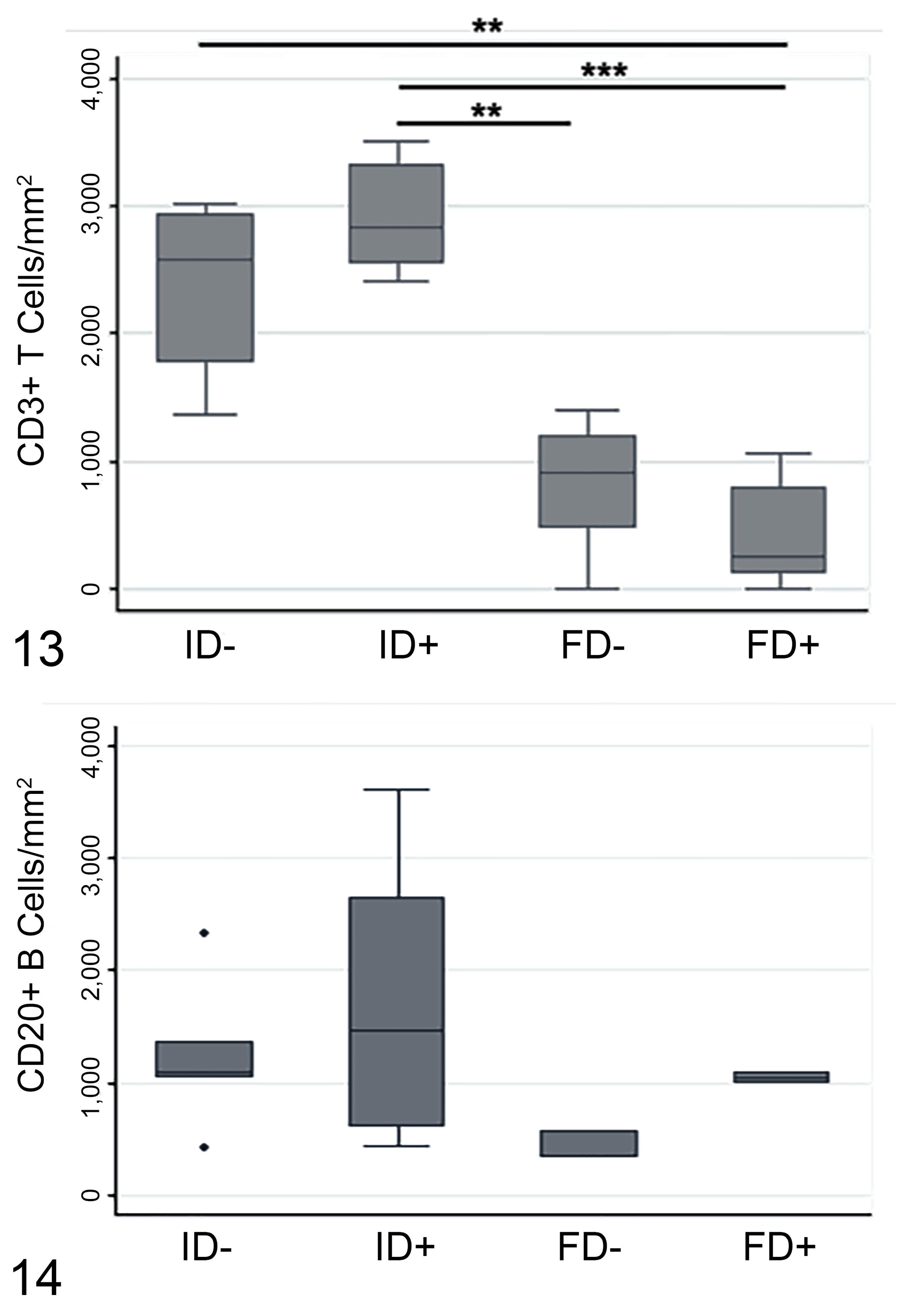
Numbers of CD3+ T lymphocytes (Figure 13) and CD20+ B lymphocytes (Figure 14) in the adventitial layer of fertile and infertile hydatid cysts of lung, with and without Fasciola hepatica co-infection. Immunolabeling was quantified as the number of positive signals per square millimeter. FD+, fertile cysts with F. hepatica co-infection (n = 9 for CD3, 2 for CD20); FD–, fertile cysts without F. hepatica co-infection (n = 5 for CD3, 3 for CD20); ID+, infertile cysts with F. hepatica co-infection (n = 8 for CD3, 6 for CD20); ID–, infertile cysts without F. hepatica co-infection (n = 9 for CD3, 5 for CD20). Data are expressed as median number of immunolabeled cells and interquartile range (IQR). *P < .05 Kruskal Wallis and Dunn’s post hoc test (adjusted P value for significance P < .0042).
CD20+ cells had a distribution similar to T cells, with a higher number located peripheral to the palisading macrophages and close to normal host tissue (Figs. 9–12). There was no significant association of cyst fertility or F. hepatica co-infection with CD20+ cell numbers. However, there was a slight increase of CD20+ cells in FD+ (2.9-fold) and ID+ (1.3-fold) samples compared with FD– and ID– samples, respectively (Figs. 9–12, 14).
Mast cells were not numerous in either fertile or infertile hydatid cysts, regardless of co-infection.
Discussion
In the present study, CD3+ and CD20+ lymphocytes were the predominant inflammatory cells in the adventitial layer of hydatid cysts in the lung of cattle. This result is in line with previous reports that show that lymphocytes have an important role in cystic echinococcosis. 21,30,31,33 A study in humans showed that CD3+ and CD20+ cells were the predominant cell type in the inflammatory infiltrate of hepatic cystic echinococcosis samples, with CD68+ cells (macrophages) also frequently present; however, cyst fertility status was not considered. 30 Likewise, infection with F. hepatica generates an important cellular immune response in livestock animals. Early in F. hepatica infection, apoptosis is induced in macrophages, lymphocytes, granulocytes, and eosinophils, which helps the survival of juvenile parasites. 7 Studies focused on co-infection between F. hepatica and other diseases demonstrated that F. hepatica excretion/secretion products reduce the airway mucus accumulation and populations of eosinophils and lymphocytes in mouse models of allergic disease, consistent with a capacity of F. hepatica to systemically modulate the immune response occurring in other diseases. 8 Fertile hydatid cysts from sheep analyzed by IHC identified lymphocyte populations surrounding established cysts with a predominance of CD3+ T cells compared to CD79+ B cells. 31 The presence of CD20+ cells in the adventitial layer in the present study was slightly higher than previously reported in cattle; large mature hydatid cysts were reported to have few B cells, with CD8+ cells as the predominant cell population in the adventitial layer with fewer CD4+ cells. 21
B cells are important immune cells that produce immunoglobulin in humoral immune responses, and they are also regarded as antigen-presenting cells. 15 In this study, a considerable proportion of inflammatory cells in the adventitial layer of both fertile and infertile cysts were CD20+, which highlights the importance of B cells in the local immune response to cystic echinococcosis in cattle. In human fertile cysts, the number of CD19+ B cells in the adventitial layer was significantly higher and were positively correlated with the number of Ki-67+ cells, indicating that B cells were proliferating in the area 14 and perhaps representing a local antibody response against the parasite. It has been previously demonstrated that infertile cysts in cattle have higher immunoglobulin G concentrations compared with fertile hydatid cysts, and these immunoglobulins are bound to the germinal layer with high affinity. Contrarily, in the germinal layer of fertile cysts, immunoglobulin G is bound with low affinity. This antigen-antibody interaction could inhibit cell proliferation for protoscolex formation, inducing apoptosis and leading to hydatid cyst infertility. 16
Our results indicate that T lymphocytes were found in greater numbers in infertile cysts than fertile cysts. There was a large population of B lymphocytes in the adventitial layer, but their numbers were not significantly associated with cyst fertility. Only low numbers of mast cells were present in the adventitial layer, suggesting that these cells do not have an important role in bovine echinococcosis. The T and B cell populations in the adventitial layer were not different in samples with or without F. hepatica co-infection.
Footnotes
Acknowledgments
We thank all the people who indirectly participated in this work, including volunteers, professionals, and technical staff of the Escuela de Medicina Veterinaria, Facultad de Ciencias de la Vida, Universidad Andres Bello.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Fondecyt-Chile, grant number 1190817 (RP); Universidad Andrés Bello, internal project DI-1388-16/I (MJ); Universidad Andrés Bello, internal project DI-1249-16/RG (RP).
