Abstract
Splenic rupture in cattle is scarcely described in the literature. The aim of this work was to report the occurrence of splenic rupture in cattle in southern Brazil as well as to describe the causes of the condition. Between 2013 and 2022, 24 of the 1769 bovine necropsies performed in southern Brazil were due to splenic rupture, accounting for 1.36% of the diagnoses. Animals died due to hemoperitoneum caused by a rupture in the splenic capsule, typically associated with marked splenomegaly and a large hematoma between the capsule and the parenchyma. Clinical signs were described in a subset of cases (11 of 24 cases, 46%) and included apathy, abdominal pain, mucosal pallor, tachycardia, and respiratory distress. However, the majority (13 of 24 cases, 54%) presented as sudden death. The underlying cause of splenic rupture was established as follows: 16 cases (67%) secondary to babesiosis, 4 cases (17%) due to lymphoma, 1 case (4%) due to a thrombus, 1 case (4%) due to external trauma, 1 case due to a ruptured nodular lymphoid hyperplasia (4%), and 1 case of undetermined cause (4%). Hypovolemic shock caused by splenic rupture is an important cause of death of dairy cattle, and babesiosis and bovine leukemia virus–associated lymphoma are among the most common etiologic diagnoses (84% of cases). The description of the causes of this condition is important to clarify the pathogenesis and occurrence of splenic rupture in dairy cattle.
Splenic rupture is a well-known entity in small animals,3,6,12,37 but it is poorly described in cattle. In bovines, it most often occurs as a consequence of splenomegaly, mainly secondary to hemolytic disorders, but also due to trauma, hematoma, and neoplasia. The rupture of the splenic capsule leads to the extravasation of blood into the abdominal cavity. Depending on the size of the rupture and the species, it can cause severe hemoperitoneum and, on necropsy examination, it appears as a large amount of free blood in the abdominal cavity.14,50 In these cases, sudden death is common, with no history of clinical signs or behavioral abnormalities. 31
Splenomegaly is usually asymptomatic, especially if it occurs gradually. In most cases, clinical signs, when present, are restricted to those caused by the involvement of other organs. 9 According to the literature, the most common clinical signs may include colic, tachycardia, cold extremities, and pallor of mucous membranes, which are all suggestive of hypovolemic shock. Cattle with splenic rupture usually die within a short period of time, which makes it challenging to obtain a clinical diagnosis prior to death.9,31
One of the main causes of splenomegaly in cattle is infection with Babesia bigemina or Babesia bovis.2,9,35 Babesia spp. are intracellular protozoa disseminated most commonly by arthropods and cause hemolytic anemia. 52 In certain parts of the world, it is responsible for major economic losses.2,20,21,45 Other causes of splenic rupture in cattle reported in the literature include lymphoma31,40 and trauma. Traumatic rupture can occur in different ways, such as accidents during transportation of animals, in fights between bovines, and trampling.14,50
The aim of this work was to report the occurrence of splenic rupture in cattle diagnosed through necropsies performed between 2013 and 2022, describing the causes of this condition.
Materials and Methods
Necropsy reports (n = 1769) issued by the Veterinary Pathology Diagnostic Laboratory (LPV) at the Instituto Federal Catarinense—Campus Concórdia were reviewed. All cases (n = 24) diagnosed as splenic rupture in cattle between January 2013 and December 2022 were selected for this study. Diagnoses were obtained through field necropsies by LPV personnel. Biopsy samples received were not included in this study.
Several veterinary schools in Brazil run veterinary diagnostic laboratories. These routinely perform necropsies for teaching, as well as research and outreach, often free of charge. Field veterinarians generally rely on this service for diagnostics. The Brazilian dairy cattle herd is estimated in about 30 million animals, being the third-largest milk producer globally. Animals are predominately raised on pasture, in a semi-intensive method, receiving a moderate amount of corn silage and grain ration twice a day.17,26 Santa Catarina state is the fifth largest milk producer in the country, but where the LPV is located, in the Western part of the state, is considered the third-largest milk production area in the country. 15 This laboratory has offered free necropsy services since 2013, which are financially supported by the Brazilian government. Any veterinarian, mainly in the Western part of the state, can request a necropsy. These factors contribute to a diverse population of animals necropsied by the diagnostic laboratory, most commonly adult dairy cattle.
Samples of organs from the abdominal and thoracic cavities, as well as the locomotor and central nervous systems, were collected at necropsy, fixed in 10% buffered formalin, routinely processed, and stained with hematoxylin and eosin for microscopic evaluation. In cases where tick fever was suspected, cytologic and fixed tissue samples were subjected to Panoptic fast (Laborclin, Pinhais, Paraná) and Giemsa (Bioclin, Belo Horizonte, Minas Gerais) staining, respectively. Data such as the date of necropsy, municipality of occurrence, gross and microscopic lesions, and other epidemiological and clinical data were obtained from the reports.
Immunophenotypic analysis of neoplastic lymphocytes was carried out using the Revised European-American Lymphoma/World Health (REAL/WHO) lymphoma classification system adapted for animal use, which is based on morphological and immunophenotypic features.1,49 The immunohistochemical analysis was performed using the MACH 4TM Universal AP Polymer Kit (Biocare Medical, Pacheco, California). Primary antibodies anti-CD20 (rabbit polyclonal antibody: PA5-16701; Invitrogen, Waltham, Massachusetts; 1:400) for B lymphocytes and anti-CD3 (rabbit polyclonal antibody: A452; Dako; 1:250) for T lymphocytes were applied and incubated overnight at 25°C. To block endogenous peroxidase activity, a 10% hydrogen peroxide solution was used. Antigen retrieval for CD20 was performed in a microwave (2 cycles of 5 minutes) with Tris-ethylenediaminetetraacetic acid (EDTA) buffer (pH 9.0); antigen retrieval for CD3 was performed with 0.05% Protease type XIV (Sigma) for 15 minutes at 37°C. For both antibodies, milk was used to prevent nonspecific binding (CD3: 15 minutes; CD20: 30 minutes). Immunoreactivity was visualized using an AP-polymer kit, Mach 4 Universal (Biocare Medical) for 30 minutes and the chromogen AEC (3-amino-9-ethylcarbazole). Immunohistochemistry sections were counterstained with Harris hematoxylin. Negative control sections were incubated with Tris-buffered saline (TBS) in place of specific antibodies. Tissue sections of bovine tonsils were used as positive controls.
Results
From 2013 to 2022, a total of 1769 necropsies were performed in cattle. Of those, 24 cases were diagnosed with splenic rupture (1.36%). Table 1 shows the number and percentage of cases of splenic rupture out of the total number of bovine necropsies conducted each year. Of these cases, 84% were due to babesiosis or B-cell lymphoma (presumed bovine leukemia virus [BLV]–associated lymphoma). All cases occurred in female Holstein Friesian breeds originating from small dairy farms in the state of Santa Catarina, Brazil. The majority were adults, ranging in age from 1 to 9 years (mean = 1.7, standard deviation = 1.99), except for 2 animals, cases 6 and 13, aged 6 and 7 months, respectively.
Number of necropsies and cases of splenic rupture, babesiosis, and enzootic bovine leukosis diagnosed in the Veterinary Diagnostic Laboratory at the Instituto Federal Catarinense—Campus Concórdia between 2013 and 2022.
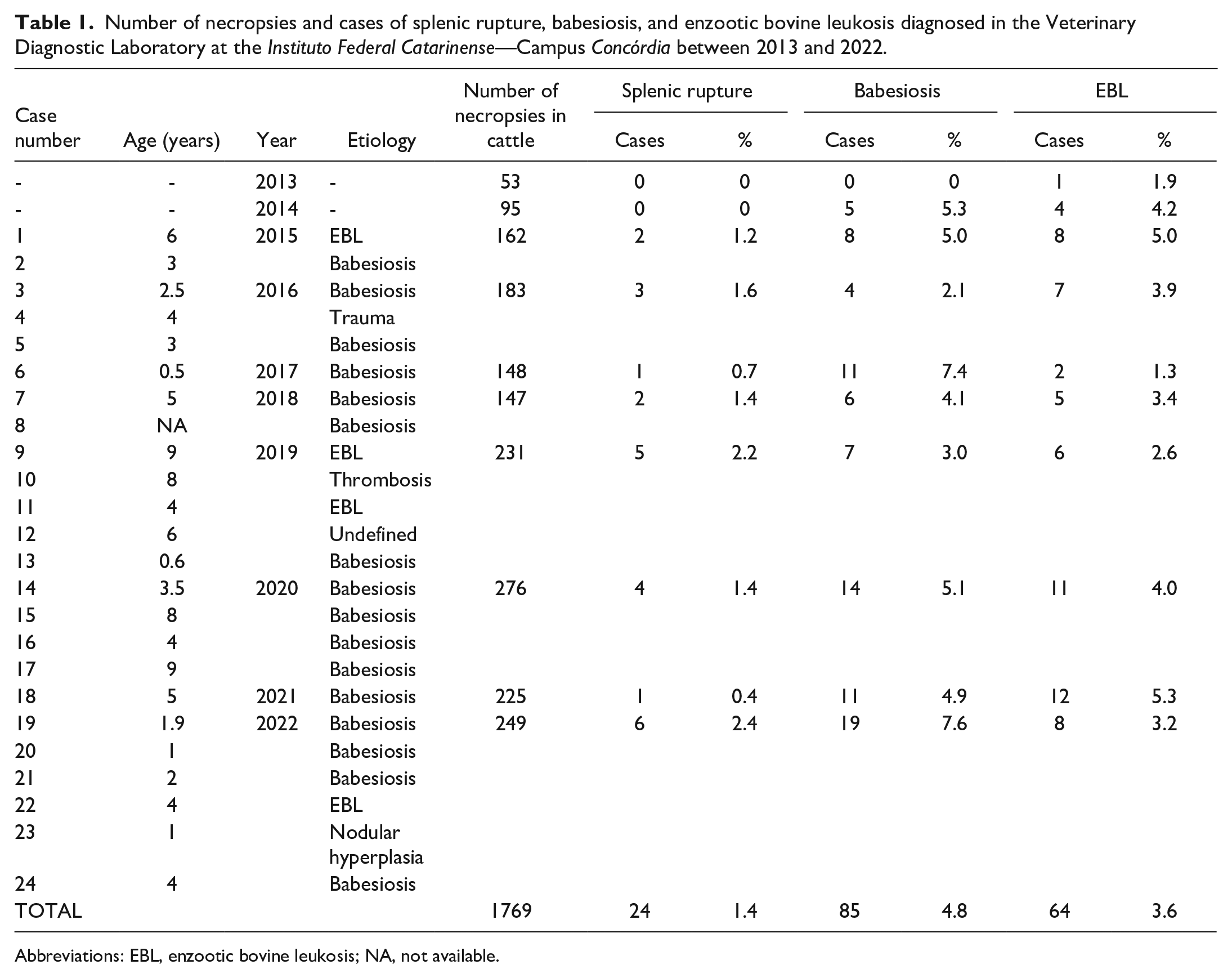
Abbreviations: EBL, enzootic bovine leukosis; NA, not available.
In 13 cases (54%), no clinical signs were observed, and the animals were found dead. When clinical signs were observed, they typically lasted a maximum of 2 hours. Nine cases (37%) showed signs of apathy and hyporexia. Two cases (9%) had longer-lasting clinical signs that included rigid walking, firm flank, fever, dehydration, mild bloating, and decreased milk production, but eventually died within approximately 12 hours. Splenic rupture was not initially considered as a differential in any of these cases.
On gross examination, all animals had severely pale mucosae and a large amount of free blood in the abdominal cavity (Fig. 1a), reaching up to approximately 20 liters. In all cases, a rupture was observed in the splenic capsule (Fig. 1b) with clots adhered to the ruptured area. There was usually only a single rupture, generally ranging between 3 and 5 cm in length and typically located in the parietal surface of the splenic capsule at the caudal border, but not affecting the splenic-ruminal area of adhesion.
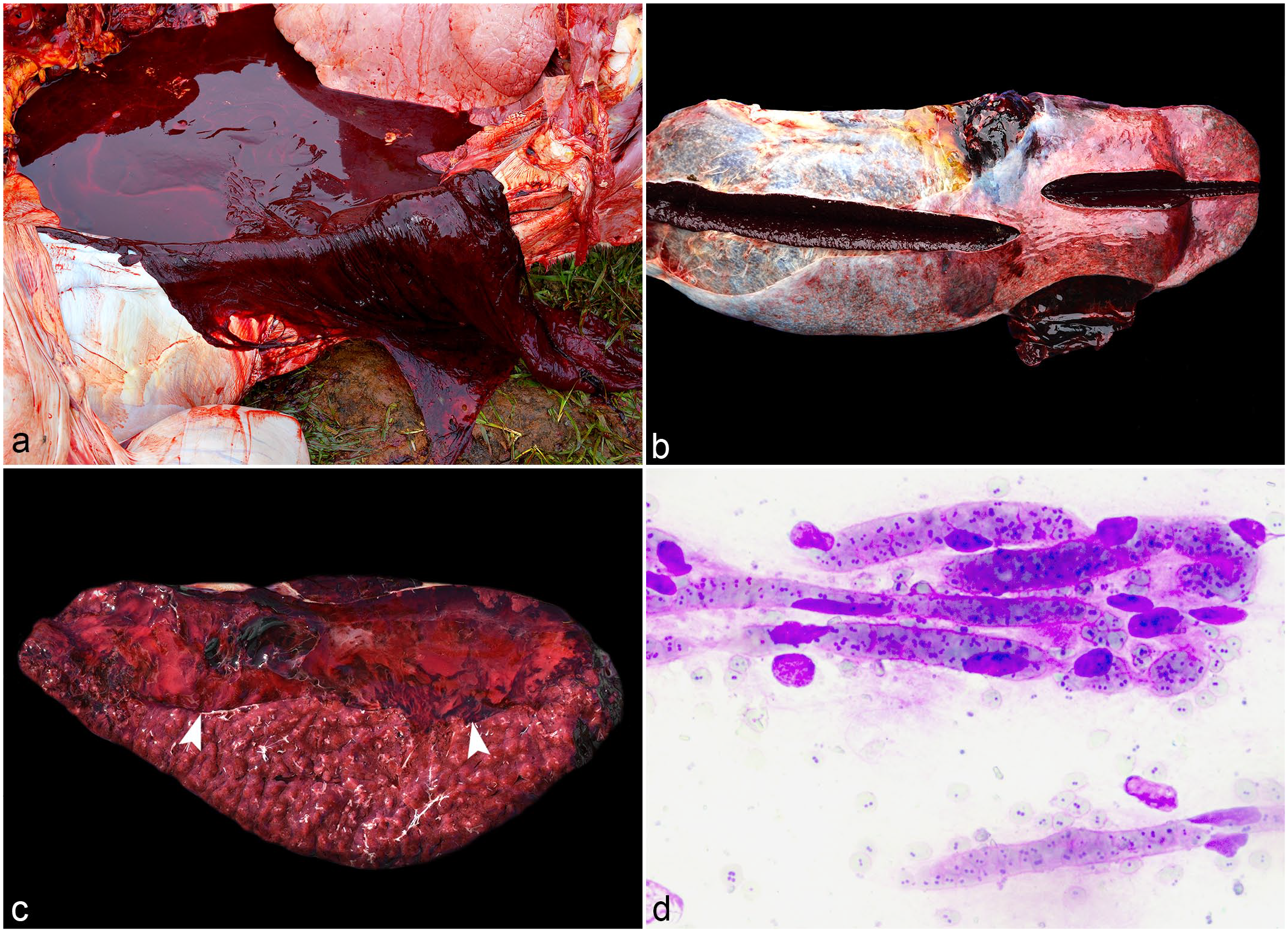
Splenic rupture in dairy cattle. (a) Abdominal cavity, large amount of blood in the abdominal cavity, with associated blood clots, case 2. (b) Spleen with severe protrusion of the splenic pulp on cut section (splenomegaly). Two areas of rupture are present at the border, one of 10 cm in length (bottom) and another of 5 cm in length (top), case 6. (c) Spleen, cut surface demonstrating a large subcapsular blood clot (arrowheads), case 7. (d) Cytologic impression smears of the brain. Capillaries are filled with erythrocytes highly parasitized by Babesia sp. organisms. Panoptic fast stain, case 16.
On the cut surface, there was moderate-to-severe protrusion of the splenic parenchyma (21 of 24 cases, 87.5%) (Fig. 1b), characterizing severe splenomegaly. In all cases except 1 (case 23), blood clots ranging from 2 to 11 cm thick were observed between the capsule and the splenic pulp (Fig. 1c).
On microscopic examination, the splenic capsule was effaced by focally extensive hemorrhage (24 of 24 cases, 100%), often with neutrophilic infiltrates (12 of 24 cases, 50%). Occasionally, below the capsule, there were fibrin thrombi (8 of 24 cases, 33%). The red pulp contained hemosiderin-laden macrophages (16 of 24 cases, 67%). In some cases, moderate numbers of intraerythrocytic parasites morphologically compatible with Babesia spp. (Fig. 1d) were observed on histologic and cytologic impression smears of the splenic parenchyma, kidney, and brain (16 of 24 cases, 67%).
In all cases diagnosed with babesiosis, animals reportedly had a history of contact with ticks on the farm. Two cases (8%) originated from farms where other cattle showed clinical signs consistent with babesiosis that improved with medical treatment. Another case (4%) originated from a farm where an outbreak of babesiosis occurred following the addition of new tick-infested hoof stock to a herd that was previously considered to be free of ectoparasites.
In 4 cases (17%), lymphoma was observed (Fig. 2a). Microscopic examination revealed proliferations of neoplastic lymphocytes arranged in dense sheets with scant cytoplasm and round to indented, 8 to 15 µm diameter, centroblastic, and euchromatic nuclei that contained vesicular and granular chromatin and multiple large, prominent, and amphophilic nucleoli. The mitotic count ranged between 19 and 82, with an average of 28, per 2.37 mm2 (equivalent to 10 FN22/40× fields). In all 4 cases, neoplastic lymphocytes demonstrated cytoplasmic immunolabeling with the CD20 antibody (Fig. 2b), hence classifying them as B lymphocytes. Most of the other cells (approximately 80%) were small non-neoplastic T cells that showed positive immunolabeling for CD3. Based on the REAL/WHO criteria, 1 all 4 neoplasms were classified as a centroblastic diffuse large B-cell lymphoma.
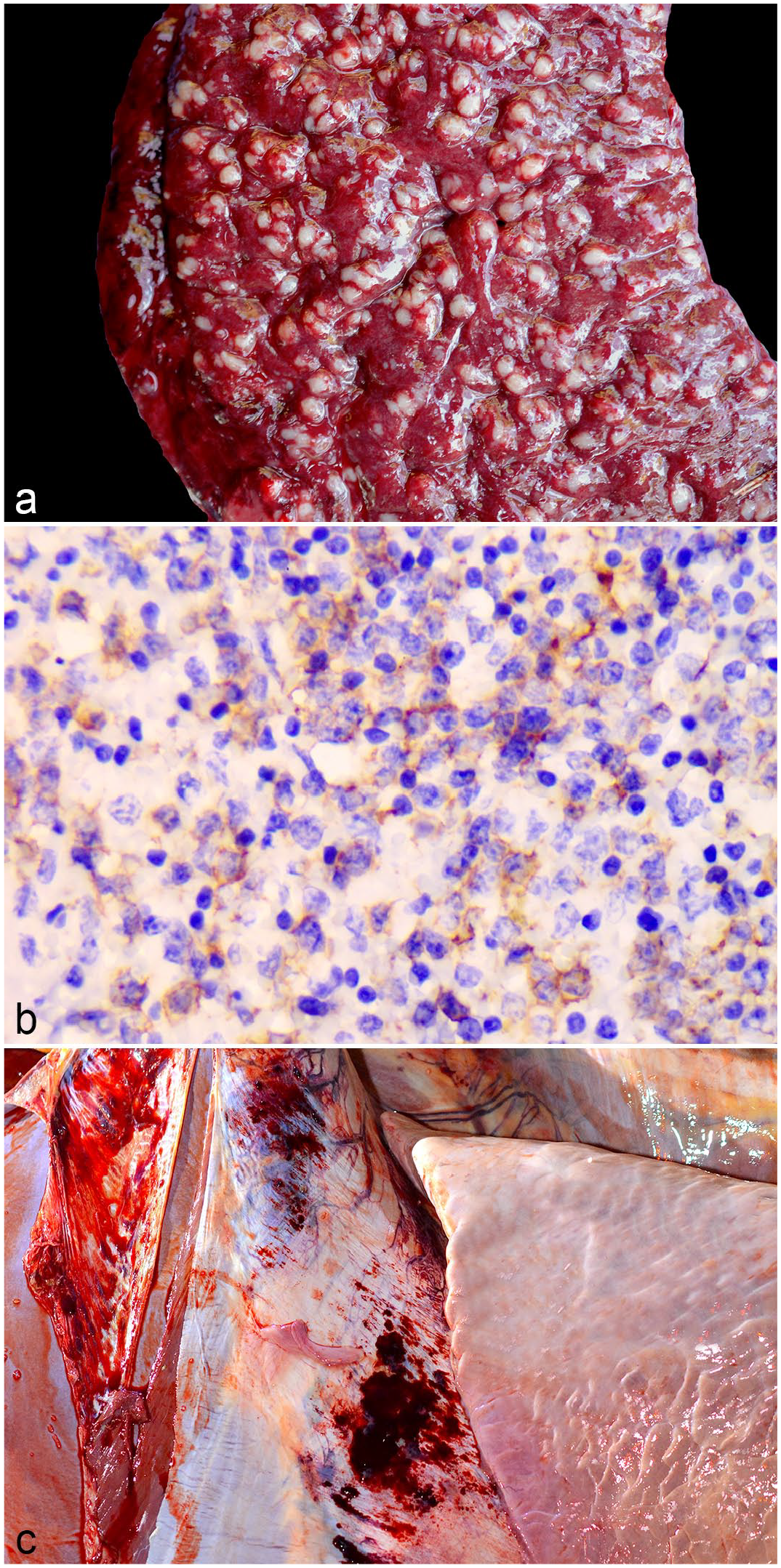
Splenic rupture in dairy cattle. (a) Spleen, cut surface with multifocal to coalescing pale yellow nodules (lymphoma). Note the subcapsular hematoma, case 1. (b) Spleen, microscopic features of bovine lymphoma in the spleen. Membranous CD20 immunolabeling of neoplastic lymphocytes, case 11. CD20 immunohistochemistry. (c) Thoracic cavity, diaphragmatic surface showing multifocal hemorrhages, case 9.
Other less common causes of splenic rupture, each represented by a single case in the sample population, included a chronic thrombus, ruptured nodular hyperplasia, and traumatic injury. In addition, a definitive cause of splenic rupture was not apparent in 1 case. In case 10, the thrombus was observed on the parietal surface of the spleen, bulging the capsule, and was composed of alternating, concentric, opaque pale yellow and red layers. No other significant gross or microscopic findings were apparent in this cow, and the underlying cause of thrombus formation was unclear. A similar nodule was found in case 23 that, on the cut surface, was grossly consistent with the surrounding splenic parenchyma. Microscopically, it was unencapsulated and composed of well-differentiated nodules of monomorphic lymphocytes lacking germinal centers and variably surrounding central arterioles (interpreted as nodular hyperplasia). Case 4 presented with a history of fighting with conspecifics. Gross examination revealed a splenic rupture, hemoperitoneum, and multiple hematomas within the subcutaneous and muscle layers of the left flank, subsequently leading to a diagnosis of traumatic splenic rupture. Finally, in case 12, a definitive cause of splenic rupture was not evident. At gross examination, the spleen was markedly enlarged, but there were no other significant gross or microscopic findings.
Discussion
In these cases, splenic ruptures were diagnosed primarily through gross examination. Etiologic diagnoses were determined in all but 1 case via histologic examination of tissues and through the use of immunohistochemistry and special stains. Regardless of the underlying cause, hemoperitoneum in cattle is generally fatal due to the lack of a feasible surgical approach in a field setting. In the literature, reports of splenic rupture in cattle are rare,31,40,46 requiring further investigation in order to have a better understanding of this condition.
Pathological or spontaneous rupture of the spleen has been described in a variety of species. Generally, in most large animal necropsies, the spleen is not typically a crucial organ to establish a cause of death except on rare occasions, such as hemolytic and hematological diseases.14,49,50
In this study, splenic rupture was fatal in all cases and clinical signs, when present, generally persisted for only a few hours. Death in all cases was the result of hypovolemic shock, caused by the large volume of blood loss through the rupture. Grossly, this presented as pale mucous membranes and a large amount of blood in the abdominal cavity. Disease progression occurred so rapidly that no clinical signs were observed in a little over half of the cases. In those that did develop clinical signs prior to death, signs were often nonspecific, highlighting the difficulty of achieving an antemortem diagnosis in the field based solely on physical examination. In addition, clinical signs only developed in cattle with babesiosis, suggesting that the signs observed were more likely associated with the infection rather than the splenic rupture itself.
In humans and animals, most cases of splenic rupture, if not surgically treated, are fatal.19,23 Very rarely, if bleeding ceases, the animal can recover and the rupture site will scar over.14,23 However, among the 1769 bovine necropsies routinely performed by LPV personnel between 2013 and 2022, we never saw large cicatricial lesions in the spleen of a bovine. The intermediate type of spleen in bovines, which does not permit rapid contraction in an attempt to control the hemorrhage after rupture, 22 may be partially responsible for the lack of evidence of scar formation in cattle.
At necropsy, the ruptured area on the spleen could be easily spotted in all cases due to the clots still attached to it and the formation of a subcapsular hematoma. The latter was described in dogs as being caused by a rupture in the red pulp that results in a localized hematoma covered by the intact splenic capsule. 50 This lesion was exclusively described in cattle diagnosed with splenic rupture at LPV during the study period.
Babesiosis was the main cause of splenic rupture in this study. In humans, splenic rupture due to Babesia spp. infection is a rare complication of the disease, with only a few cases described in the literature.13,47 It occurs secondary to phagocytosis of erythrocytes infected with Babesia spp. by splenic histiocytes in addition to platelet sequestration, resulting in thrombocytopenia. Although the exact mechanism that leads to rupture in this scenario is poorly understood, these changes lead to rapid splenomegaly and eventual spontaneous rupture.13,47 In bovine babesiosis, the splenic rupture might result from a combination of the protozoa causing hemoglobinuria, splenomegaly, and anemia by multiple mechanisms, including direct erythrocytolysis, immune-mediated recognition and removal, shortened cellular life-span caused by oxidative stress, and blood loss resulting from endothelial disruption or inadequate platelet function.5,44 To our knowledge, splenic rupture with severe hemoperitoneum in cattle naturally infected by Babesia spp. has not been previously described in the literature. We found only 1 case in an experimental trial, in which 1 bovine died due to hypovolemic shock among 36 animals. 46
Babesia spp. are a causative agent of tick fever, which is common in southern Brazil.11,30 Previously, the incidence of tick fever attributed to both Anaplasma sp. and Babesia spp. in cattle from the state of Rio Grande do Sul in Brazil between 1964 and 2008 was reported to be approximately 3.4%. 28 More recent studies have reported a slightly lower incidence at approximately 2.5% for babesiosis and 1.2% for anaplasmosis, respectively. 27 In contrast, the incidence of babesiosis and anaplasmosis cases in our diagnostic laboratory between 2013 and 2022 is approximately 4.8% (85 cases of 1769 bovine necropsies) and 3.3% (58 cases of 1769 bovine necropsies), respectively. 34 The percent difference between incidence rates of babesiosis in our diagnostic laboratory compared to that reported by Ladeira et al 27 is approximately 92% higher. Similarly, the percent difference between incidence rates of tick fever in our diagnostic laboratory (143 cases of 1769 bovine necropsies) compared to that reported by Lucena et al 28 is approximately 138% higher. This increase in cases could explain, at least partially, why babesiosis was the most common cause of splenic rupture in this study period.
Lymphoma denotes a heterogeneous group of neoplasms originating from lymphoid tissues.14,49,50 Bovine lymphomas may occur as a spontaneous disease or result from infection with BLV. 50 In cattle, it is typically found in lymph nodes, the uterus, periorbital tissues, heart, abomasum, kidney, and spinal cord.18,33 Comparatively, the spleen is considered an uncommon location for lymphoma to develop in cattle, 50 with 1 study reporting splenic involvement in only 30% of cases in cattle. 51 When the spleen is affected, the neoplastic growth occurs uniformly, causing splenomegaly due to lymphoid tissue proliferation, with possible rupture and abdominal hemorrhage,14,49,50 consistent with what was observed in 4 of our cases.
BLV-associated lymphomas are predominantly diffuse large B-cell lymphomas. Sporadic cases of precursor T-cell lymphoblastic lymphomas are not associated with BLV infection. BLV-associated lymphoma can occur in multiple forms, including multicentric in calves (between 3 and 6 months of age), within the thymus (around 1 or 2 years of age), or least commonly as an epitheliotropic lymphoma in the skin (between 2 and 3 years of age). These forms occur randomly, at a very low rate.18,42,50 As all 4 cases described in this study were represented by diffuse large B-cell lymphomas, they were classified as enzootic bovine leukosis (EBL), even without molecular techniques. Furthermore, the ages of these 4 cases ranged between 4 and 9 years old, which is characteristic of EBL and beyond the typical age range for sporadic lymphoma, as previously described.18,42 Unfortunately, molecular ancillary tests aimed at the detection of BLV in lesions were not performed due to a lack of financial support.
During the study period, 64 cases of EBL were diagnosed at the LPV by necropsy. In those cases, lymphoma was primarily associated with the abomasum, lymph nodes, and heart, although infiltration into muscle and nerves around the spinal cord, rarely with invasion into the medullary canal, was observed in a subset of cases. 34 Aside from the cases included in this study (4 of 64 cases, 6%), none of the other cases from this period had gross lesions consistent with lymphoma in the spleen. Conversely, in the cases we reported above, neoplastic lymphocytes were observed solely within the spleen in 1 case and only within the spleen and mediastinal lymph nodes in the other 3 cases. Therefore, the typical presentation of EBL was not seen, when compared with the cases diagnosed by our lab between 2013 and 2022, 34 as well as those described in the literature.14,33,49,50
Splenic rupture associated with nodular hyperplasia was observed in 1 case. Nodular hyperplasia can be subtyped based on the predominant cellular component as splenic, hematopoietic, lymphoid, or complex. The simplest type is splenic nodular hyperplasia, which is composed of an excess of red pulp. A spectrum of mixed nodular lesions can also occur that contains elements of different subtypes, which complicates attempts at classification. 49 Based on the previously described subtype classifications, the hyperplastic nodule in this case would be classified as a mixed subtype.
Nodular lymphoid hyperplasia is the most common splenic nodule found in dogs,10,43 is occasionally reported in old bulls, and is rarely seen in other species. 50 The rupture of a hyperplastic nodule can lead to hemoperitoneum3,16 and/or splenic hematoma. 8 In 1 study, approximately 7.5% of splenectomies performed in dogs occurred due to ruptured hyperplastic nodules. 7 However, to our knowledge, rupture of a hyperplastic nodule followed by the death of a bovine has not yet been described in cattle.
For the single case in which we were unable to reach a definitive diagnosis, the spleen was markedly enlarged at gross examination. In combination with a history of sudden death, this was suggestive of babesiosis, although no protozoal organisms were observed microscopically and evaluation of the urine did not reveal hemoglobinuria, a common finding in infected cattle. However, the herd it originated from had been recently treated with diminazen and tetracycline for an outbreak of tick fever, which could have lowered the protozoal burden below our threshold of detection. Thus, we speculate that this case may, in fact, represent an additional case of splenic rupture associated with babesiosis.
Severe splenomegaly can sometimes progress to splenic rupture. Aside from the potential etiologies described above, other conditions reported to cause severe splenic enlargement in cattle include salmonellosis,39,50 anthrax, and hemolytic diseases. 50 In addition, splenic amyloidosis has been described in humans.4,24
Anthrax is rarely seen in southern Brazil, mostly occurring near the border with Uruguay, and only accounts for approximately 0.05% 28 to 0.2% 41 of reported diagnoses from this region. Likewise, in the state of Santa Catarina where our cases originated, there is only 1 published anecdotal diagnosis of anthrax. 32 Furthermore, gross lesions such as bloody discharge from multiple orifices or hemorrhage within the connective tissues and microscopic observation of bacteria morphologically consistent with Bacillus anthracis9,50,53 were not seen in any of our cases.
An additional factor to be considered is that the diagnostic service in our laboratory is free of charge, and our mission focuses mainly on academic training and scientific advancement. Accordingly, even cases that already have a presumptive diagnosis from the submitting veterinarian for cause of death are necropsied by our laboratory, as opposed to other labs that may only receive cases of high diagnostic interest or relevance. These differences likely explain the changes in incidence described above for cases of babesiosis. However, they also may be more reflective of the true burden of disease within this region rather than disproportionately representing the rarer, referral-type cases more typically received by a diagnostic laboratory.
Diaphragmatic hemorrhage in cattle has previously been interpreted as an indication of exsanguination.25,48 In our experience, it is a consistent lesion in cases where death in an adult cow was due to hypovolemic shock. In 87.5% (21 of 24 cases) of the cases in this study, a severe focally extensive area of hemorrhage and/or hematoma was observed in the diaphragm (Fig. 2c). Interestingly, 2 of the cases without this lesion were the ones with longer-lasting clinical signs. Perimortem hemorrhages occur due to an increase in blood pressure in the agonal stage of death, probably in part from terminal anoxia, and are normally seen in the adrenal glands and the pericardium. 25 In these cattle, the changes are likely due to the same pathogenesis, despite not being commonly seen in the majority of cases that undergo agonal death. We have seen similar changes sporadically in cattle that die from exsanguination but also from those that die from causes unrelated to blood loss, meaning it is likely not a specific finding but rather a suggestive one. 36
Considering the impossibility of handling splenic ruptures in cattle in the field and taking into consideration the etiologies described in this report, precautions should be taken to prevent the development of infectious diseases like babesiosis and EBL, when possible. Various tools and techniques exist to accomplish this, including control of vector insects, use of individual gloves in rectal palpation of cows, special attention in the use of equipment for dehorning, castration, and the use of disposable needles and syringes. Likewise, the entry of infected animals into a herd should be avoided, and the elimination or strict control of animals already infected should be implemented.18,29,38
In summary, splenic rupture in dairy cattle in southern Brazil is a rare but serious disease. In this series, cases occurred exclusively in adult female cattle. The most common causes of splenomegaly predisposing to rupture included babesiosis and BLV-associated lymphoma. Other causes, including traumatic injury, hyperplastic nodule rupture, and thrombus rupture, were seen much less frequently. Clinical signs are generally only observed in a subset of cases and are suggestive of hypovolemic shock but tend to be nonspecific.
Footnotes
Acknowledgements
The authors are grateful to the Instituto Federal Catarinense (IFC) for supporting the Veterinary Pathology Diagnostic Laboratory as well as the numerous veterinarians and farmers who facilitated and provided information regarding the reported cases.
Authors’ Note
This article was prepared in the Uniform Requirements format.
Author Contributions
VWH, FFP, RS, EB, and REM wrote the manuscript with contributions of others. VWH, FFP, AHG, MMP, EB, MEG, TMAG, and REM conducted the necropsies. VWH, FFP, and SPP performed the IHQ. SPP, TMAG, and REM performed the histologic evaluations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Instituto Federal Catarinense (IFC) and The Brazilian National Council for Scientific and Technological Development (CNPq), project no. 307086/2022-4.
