Abstract
Clostridium perfringens type D is the causative agent of enterotoxemia in sheep, goats, and cattle. Although in sheep and cattle, the disease is mainly characterized by neurological clinical signs and lesions, goats with type D enterotoxemia frequently have alterations of the alimentary system. Epsilon toxin (ETX) is the main virulence factor of C. perfringens type D, although the role of ETX in intestinal lesions in goats with type D enterotoxemia has not been fully characterized. We evaluated the contribution of ETX to C. perfringens type D enteric pathogenicity using an intraduodenal challenge model in young goats, with the virulent C. perfringens type D wild-type strain CN1020; its isogenic etx null mutant; an etx-complemented strain; and sterile, non-toxic culture medium. The intestinal tract of each animal was evaluated grossly, microscopically, and immunohistochemically for activated caspase-3. Both ETX-producing strains induced extensive enterocolitis characterized by severe mucosal necrosis, apoptosis, and diffuse suppurative infiltrates. No significant gross or microscopic lesions were observed in goats inoculated with the non–ETX-containing inocula. These results confirm that ETX is essential for the production of intestinal lesions in goats with type D disease. Also, our results suggest that the intestinal pathology of type D enterotoxemia in goats is, at least in part, associated with apoptosis.
Clostridium perfringens type D is the etiological agent of enterotoxemia in sheep, goats, and more rarely cattle.29,31,34 C. perfringens type D may be found in the intestinal tract of clinically healthy ruminants, 34 but under certain conditions, usually related to sudden dietary changes involving the ingestion of large amounts of highly fermentable carbohydrates,4,31,34 it proliferates and produces toxins that can act locally in the intestine and/or be absorbed into the bloodstream, producing enterotoxemia and exerting effects on several extra-intestinal organs.29,31,34 Epsilon toxin (ETX) produced by C. perfringens type D is responsible for the clinical signs and most lesions of enterotoxemia in sheep, goats, and probably cattle.10,11,29,31,33 However, the pathology of type D disease differs among these species. Although in goats, type D enterotoxemia is commonly associated with intestinal lesions characterized by fibrinonecrotic colitis, or more rarely enterocolitis or only enteritis,31,34 in sheep and cattle, the most striking lesions are present in the brain, lungs, and/or heart.9,11,13,15,31,34 Brain lesions in goats with type D enterotoxemia are rare and so are intestinal lesions in sheep and cattle with this disease.24,31,34 The reasons for the differences in the type of lesions in sheep, cattle, and goat type D enterotoxemia remain unknown.
We performed a detailed description of the gastrointestinal pathology in goats challenged with C. perfringens type D strain CN1020 (wild type), its ext null mutant, the complemented strain in which the etx gene was reintroduced, or sterile, non-toxic culture media, using tissues collected during a previous study. 11
Materials and Methods
Tissues, Bacterial Strains, and Experimental Procedures
Formalin-fixed, paraffin-embedded samples of rumen, reticulum, omasum, abomasum, small intestine, cecum, and colon from 14, 3- to 4-month-old, male or female Boer or Toggenburg goats were available from the referred study. 11 All animals were between 20 and 30 kg of body weight and none had been vaccinated against clostridial or other diseases. In the referred study, 4 groups composed of 3, 3, 6, and 2 animals had been experimentally inoculated intraduodenally with the C. perfringens type D wild-type strain CN1020 (WT), its etx knock out mutant strain JIR4981 (KO), the etx mutant complemented in trans with the wild-type etx gene strain (JIR12604), or with sterile, non-toxic tryptone glucose yeast (TGY) medium (control), respectively. The construction of the mutant strains and the details of the inoculation procedures, a summary of the clinical and pathology data, tissue collection, and initial processing, were published previously.11,18 Briefly, all goats challenged with the WT or the complemented etx strains developed acute clinical disease, and died or were euthanized within 24 hours postinoculation. Gross and microscopic changes consisting of necrotizing enterocolitis were observed in goats inoculated with the WT or complemented strains. No clinical alterations or gross or microscopic lesions were observed in any goat receiving the KO strain or TGY medium. All procedures involving animals were reviewed and approved by the University of California-Davis Institutional Animal Care and Use Committee (permit # 16383).
Gross and Microscopic Changes
The gross changes of the whole gastrointestinal tract from all goats were analyzed (Table 1). Sections of all tissues that had been fixed in 10%, buffered, pH 7.2 formalin; embedded in paraffin; and processed routinely to obtain 4-µm-thick hematoxylin and eosin sections were available for microscopic analysis. For this study, 14 sections of jejunum and 14 sections of spiral colon (1 each per animal) were analyzed microscopically and subsequently stained with Gram stain and phosphotungstic acid hematoxylin (PTAH) for fibrin detection. In addition, all sections of colon were stained with Alcian blue to identify the presence of mucus. A semiquantitative score system ranging from 0 (no lesions) to 4 (severe lesions) (Supplemental Table S1) was used to evaluate the severity of lesions in hematoxylin and eosin-stained sections of the gastrointestinal tract of each animal, and a mean score (± standard deviation [SD]) was calculated for each group (Table 2). The following specific parameters were qualitatively evaluated in each small intestinal and colonic section: enterocyte attenuation (characterized by flattening of the remaining enterocytes after epithelial surface loss in an attempt to preserve the integrity of the lining epithelium), enterocyte death, enterocyte desquamation, lamina propria contraction (villous atrophy), inflammatory infiltrate, dilated lymphatics, submucosal edema, pseudomembrane formation, and mucus in the lumen. An overall severity average score for microscopic lesions for each intestinal segment was calculated for each group of goats (Table 2). In addition, villus blunting and fusion (atrophy), and villous/crypt ratio were evaluated in sections of the small intestine (Table 2). For the latter, the height of 4 villi was measured from the tip to the crypt-villus junction and the depth of 4 crypts was measured from the crypt-villus junction to their base. These measurements were repeated in 3 randomly selected microscopic fields per section at 100×, and a mean ± SD was calculated. The presence of gram-positive rods in the small and large intestine was evaluated using a score from 0 (not observed) to 4 (large numbers observed). A score of 0 (not observed) to 4 (large amount observed) was also employed to evaluate the presence of fibrin stained with PTAH in the intestinal and vascular lumen. The presence of free mucus in the intestinal lumen and goblet cells of Alcian blue-stained sections of colon was scored in 5 fields per section, including 2 sections per goat, using a score from 0 (no free mucus/goblet cells stained) to 4 (large amount of free mucus/goblet cells stained). The presence of gram-positive rods, fibrin, and mucus/goblet cells in the small intestine and colon of each inoculated group was calculated as an average score ± SD (Table 3).
Gross lesions in the small intestine and colon of goats inoculated with Clostridium perfringens type D wild-type strain CN1020 (WT); its isogenic etx null mutant JIR4081 (etx KO); its complemented derivative JIR12604 (etx compl); or TGY, sterile, non-toxic medium (control).
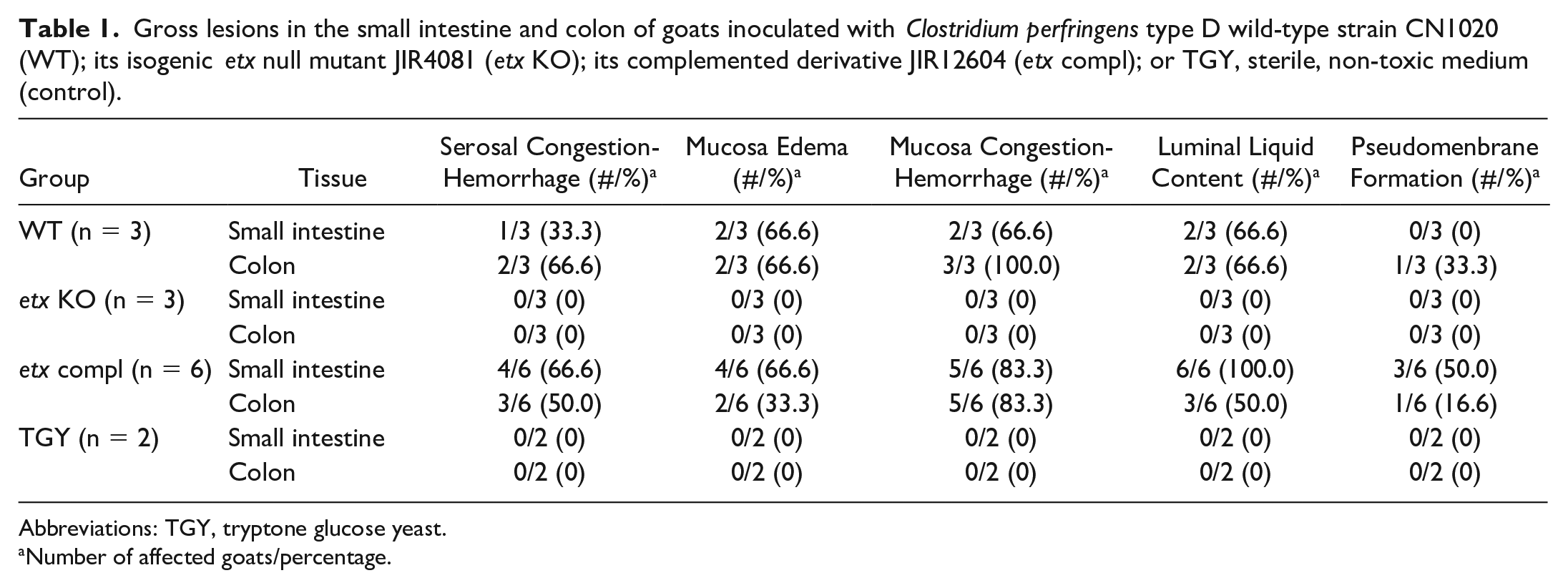
Abbreviations: TGY, tryptone glucose yeast.
Number of affected goats/percentage.
Score of histologic lesions in small intestine and colon of goats inoculated with Clostridium perfringens type D wild-type strain CN1020 (WT), its isogenic etx null mutant JIR4081 (etx KO); its complemented derivative JIR12604 (etx compl); or TGY, sterile, non-toxic medium (control). a
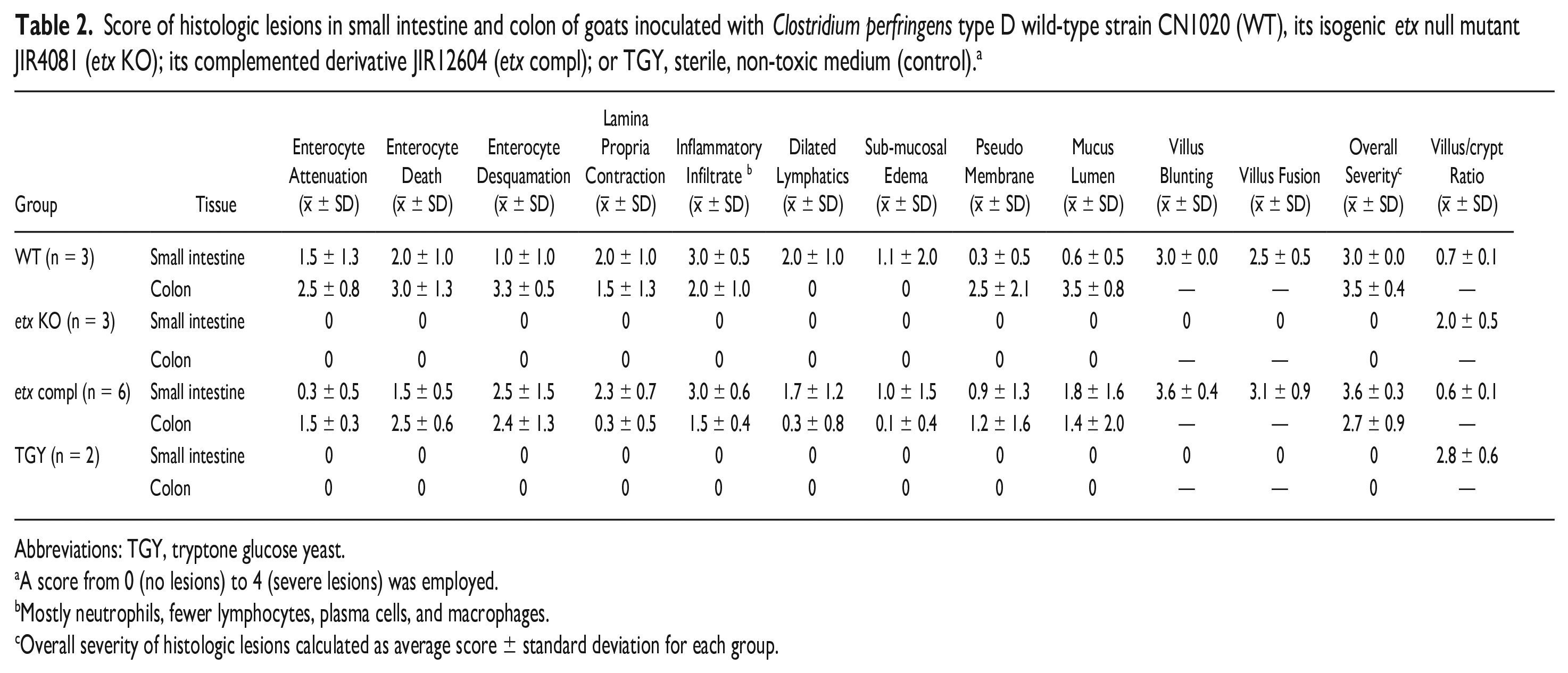
Abbreviations: TGY, tryptone glucose yeast.
A score from 0 (no lesions) to 4 (severe lesions) was employed.
Mostly neutrophils, fewer lymphocytes, plasma cells, and macrophages.
Overall severity of histologic lesions calculated as average score ± standard deviation for each group.
Presence of gram-positive rods, Clostridium perfringens, and activated caspase-3 immunohistochemistry (IHC), PTAH-stained fibrin, mucus in the lumen, and goblet cells (Alcian blue) in the small intestine and colon of goats inoculated with C. perfringens type D wild-type strain CN1020 (WT); an isogenic etx null mutant JIR4081 (etx KO); its complemented derivative JIR12604 (etx compl); or TGY, sterile, non-toxic medium (control).
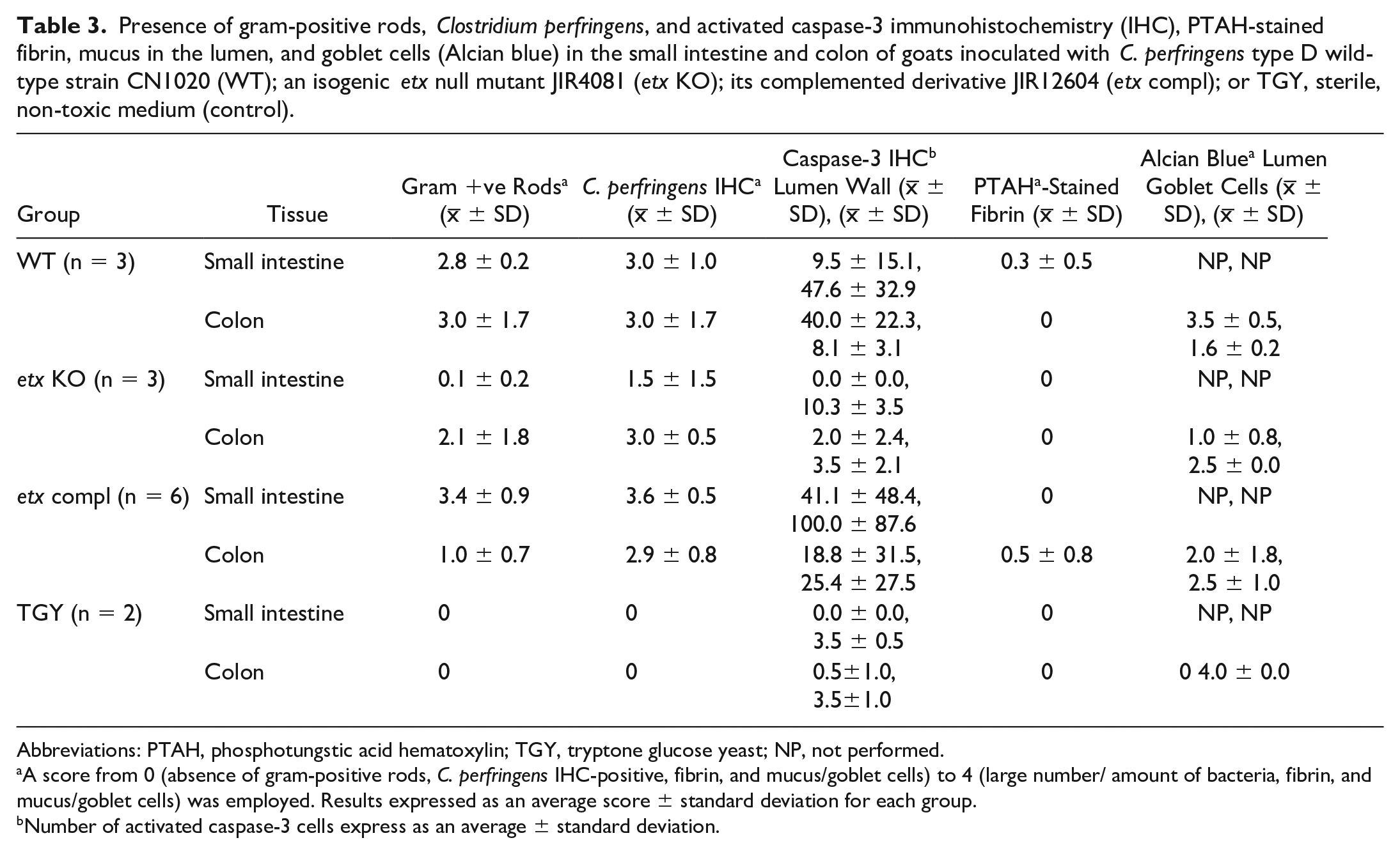
Abbreviations: PTAH, phosphotungstic acid hematoxylin; TGY, tryptone glucose yeast; NP, not performed.
A score from 0 (absence of gram-positive rods, C. perfringens IHC-positive, fibrin, and mucus/goblet cells) to 4 (large number/ amount of bacteria, fibrin, and mucus/goblet cells) was employed. Results expressed as an average score ± standard deviation for each group.
Number of activated caspase-3 cells express as an average ± standard deviation.
Clostridium perfringens Immunohistochemistry
Immunohistochemistry (IHC) for C. perfringens was performed on sections of small intestine and colon from all animals, as previously described, 7 using the EnVision Kit (Dako, Carpinteria, California) according to the instructions of the manufacturer. Briefly, a rabbit polyclonal anti-C. perfringens antibody was used as the primary reagent (Gen Way Bio, San Diego, California) and the immunoreactivity was visualized with the Nova Red chromogen (Vector laboratories Inc., Burlingame, California). Small intestine from a goat inoculated experimentally with C. perfringens type D and from which this microorganism had been isolated was used as positive control. Small intestine from a healthy goat that was culturally negative for C. perfringens was used as negative control. Additional negative controls consisted of sections of the test tissues incubated with normal rabbit serum instead of anti-C. perfringens antibodies. The IHC analysis was evaluated using a scale from 0 (no positively stained rods) to 4 (large numbers of positively stained rods), and the results were expressed as an average score ± SD (Table 3).
Activated Caspase-3 Immunohistochemistry
IHC for activated caspase-3 was performed as a hallmark of apoptosis and the major effector caspase initiating the cell death cascade in sections of small intestine and colon from all animals. The EnVision Kit was employed according to the instructions of the manufacturer. Antigen retrieval was performed with citrate buffer. A rabbit polyclonal anti-cleaved caspase-3 antibody (ASP175, Cell Signaling Technology, Inc., Danvers, Massachusetts) was used as the primary reagent at a 1:100 dilution, and the immunoreactivity was visualized with the Nova Red chromogen. A lymph node from a cow with lymphoma that contained abundant apoptotic lymphoid cells was employed as a positive control. Sections of all the tissues in which the primary antibody was replaced by normal rabbit immunoglobulin G (IgG) were used as negative controls. For the analysis of activated caspase-3 IHC, the positive cells in the lumen (desquamated epithelial cells) and the wall (epithelial cells attached to the basement membrane) were counted in 5 fields per section, including 2 sections of each intestinal segment (jejunum and spiral colon) at 20X, using a 10×/22 eyepiece, equivalent to a total field of view of 0.55 mm in diameter per section. The results were expressed as an average ± SD of positive caspase-3 IHC cells (Table 3).
Statistical Analyses
The Shapiro-Wilk and Bartlett tests were used to assess the normality of distribution and variance homogeneity, respectively, of the investigated parameters. Since all parameters in our study were non-normally distributed, the non-parametric Kruskal-Wallis test was used to analyze differences between groups, followed by Dunn’s multiple comparison test as post hoc analysis. Values of P < .05 were considered statistically significant. Data were expressed as mean ± SD. Statistical analysis was done using R version 4.3.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Gross changes, including serosal and mucosal congestion and hemorrhage, luminal liquid content, mural edema, and/or the presence of pseudomembranes were observed in the small intestine of most goats inoculated with the WT strain (Fig. 1a), but not with the KO strain (Fig. 1b). Inoculation of the etx-complemented strain produced changes identical to those described for the WT strain (Fig. 1c). Similarly, these macroscopic changes were observed in the colon of most animals inoculated with the WT (Fig. 1d), but not with the KO strain (Fig. 1e), whereas inoculation of the etx-complemented strain recapitulated the gross characteristics of its WT counterpart (Fig. 1f).
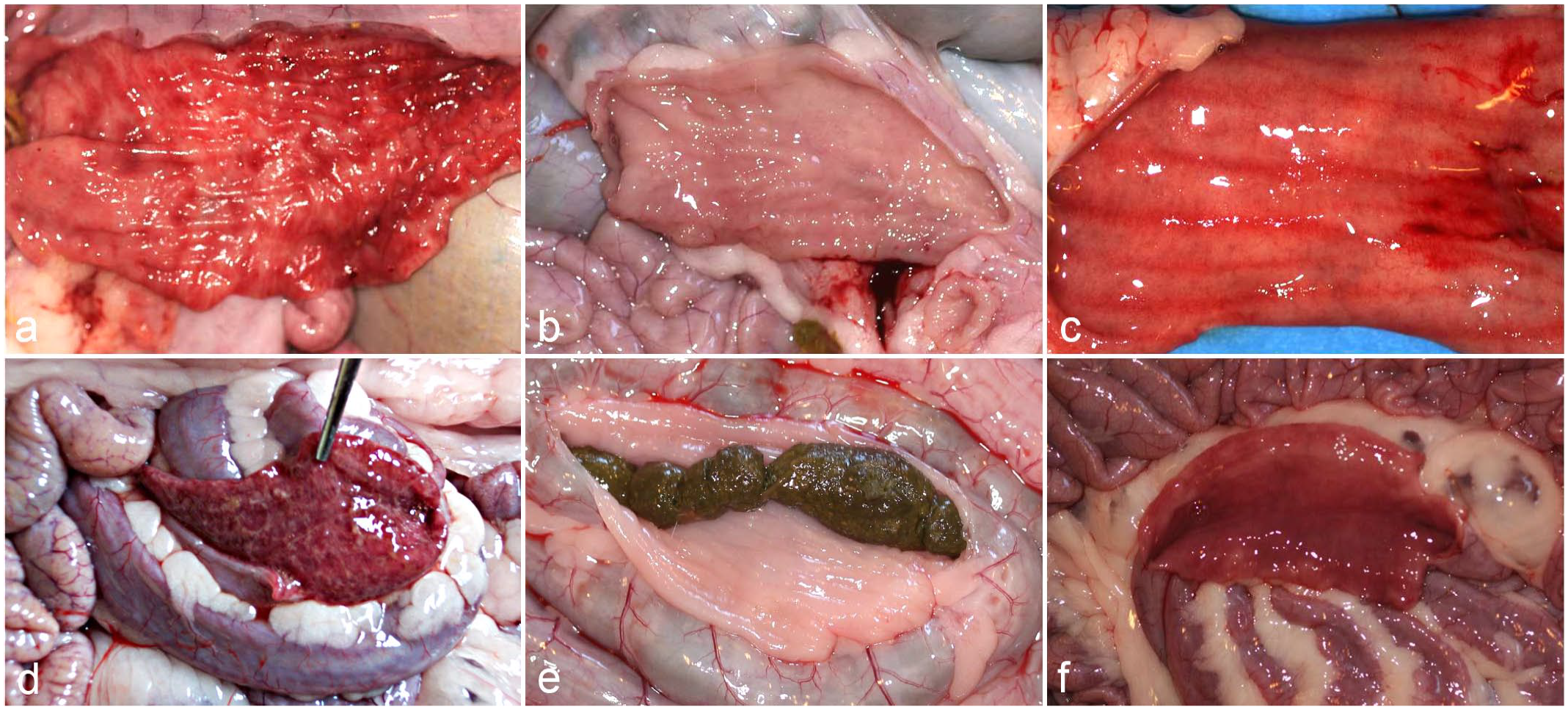
Gross changes in the small intestine and colon of goats inoculated intraduodenally with C. perfringens type D and its mutants. (a) Small intestine, C. perfringens type D wild-type strain CN1020. Diffuse bright-red discoloration of the mucosa. (b) Small intestine, C. perfringens type D etx null mutant JIR4081(etx-KO). Mucosa with normal appearance. (c) Small intestine, C. perfringens type D etx-complemented strain JIR12604 (etx-complemented). Diffuse bright-red discoloration of the mucosa. (d) Colon, C. perfringens type D wild-type strain CN1020. Diffuse bright-red discoloration of the mucosa which is partly covered by a pseudomembrane. (e) Colon, C. perfringens type D etx null mutant JIR4081(etx-KO). Mucosa with normal appearance. (f) Colon, C. perfringens type D etx-complemented strain JIR12604 (etx-complemented). Diffuse bright-red discoloration of the mucosa.
Microscopically, enteritis was observed in all the goats inoculated with the WT strain (Fig. 2a), but this change was absent in animals receiving the KO strain (Fig. 2b). Enteritis was also associated with the inoculation of the etx-complemented strain (Fig. 2c), but not with TGY alone (Fig. 2d). Microscopic lesions in the small intestine of goats challenged with both ETX-producing strains consisted of mild to severe necrotizing enteritis characterized by villous atrophy; mucus in the lumen; desquamation; death and/or attenuation of enterocytes; infiltration of neutrophils and fewer lymphocytes, plasma cells and macrophages in the lamina propria; dilated lymphatics; submucosal edema; and/or pseudomembrane formation (Table 2). Microscopic lesions in the colon of goats inoculated with the WT strain consisted of severe necrotizing colitis (Fig. 2e) characterized by changes similar to those described for the small intestine, except for villus changes. No microscopic changes were observed in the colon of animals receiving the KO strain (Fig. 2f); however, necrotizing colitis was observed again when animals received the ext-complemented strain (Fig. 2g). Inoculation of TGY alone did not produced lesions in the colon (Fig. 2h) or in the prestomachs and abomasum of any animal in the 4 groups. No significant differences in overall lesion severity were observed in the small intestine and colon of goats inoculated with the 2 different ETX-producing strains (Fig. 3a). The villous height to crypt ratio in the small intestine of the goats inoculated with the WT or the complemented etx mutant strains was statistically lower than in goats inoculated with the KO strain or TGY (Fig. 3b). In addition, empty goblet cells and large amount of free, extruded mucus in the intestinal lumen were prominent microscopic features in the colon of goats inoculated with both ETX-producing strains (Fig. 4). Gram-positive rods were immunolabeled for C. perfringens by IHC were present on the surface of colonic mucosa or, more frequently, mixed with the pseudomembrane and desquamated epithelial cells in goats challenged with the WT, KO, or complemented strains (Fig. 5a-c, respectively), and absent in animals receiving TGY alone (Fig. 5d).
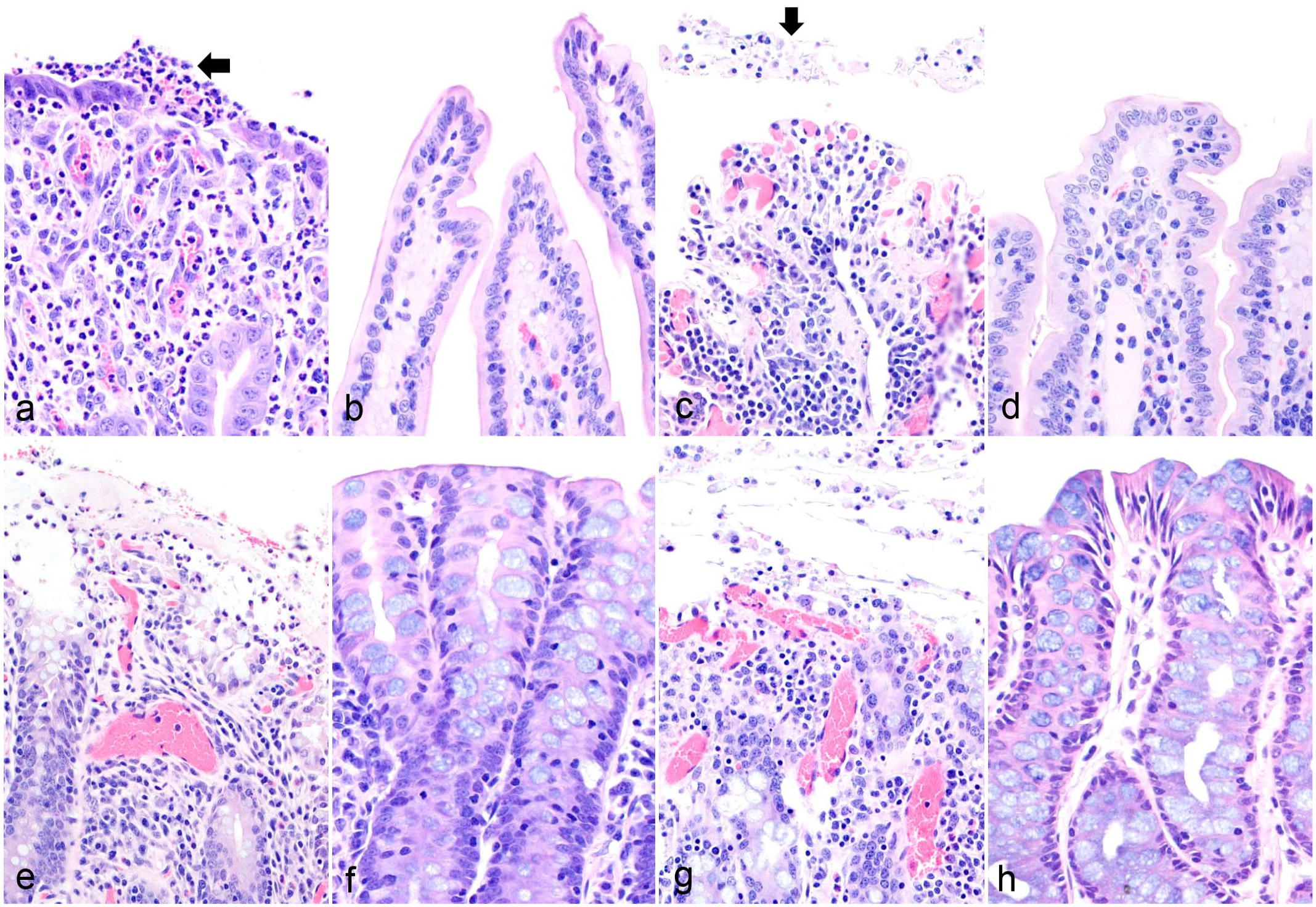
Histopathologic features of intraduodenal challenge in goats with C. perfringens type D. Hematoxylin and eosin. (a) Small intestine, C. perfringens type D wild-type strain CN1020. Villus blunting, congestion, and neutrophilic infiltration of the lamina propria and incipient pseudomembrane formation (arrow). (b) Small intestine, C. perfringens type D etx null mutant JIR4081 (etx-KO strain). Mucosal villa with normal appearance. (c) Small intestine, C. perfringens type D etx-complemented strain JIR12604 (etx-complemented). Mucosal ulceration, villus blunting, pseudomembrane formation (arrow), and leukocytic infiltration. (d) Small intestine, TGY sterile, non-toxic medium (control). Mucosal villa with normal appearance. (e) Colon, C. perfringens type D wild-type strain CN1020. Pseudomembranous colitis. (f) Colon, C. perfringens type D etx null mutant JIR4081 (etx-KO strain). Mucosa with normal appearance. (g) Colon, C. perfringens type D etx-complemented strain JIR12604 (etx-complemented). Pseudomembranous colitis. (h) Colon, TGY sterile, non-toxic medium (control). Mucosa with normal appearance.
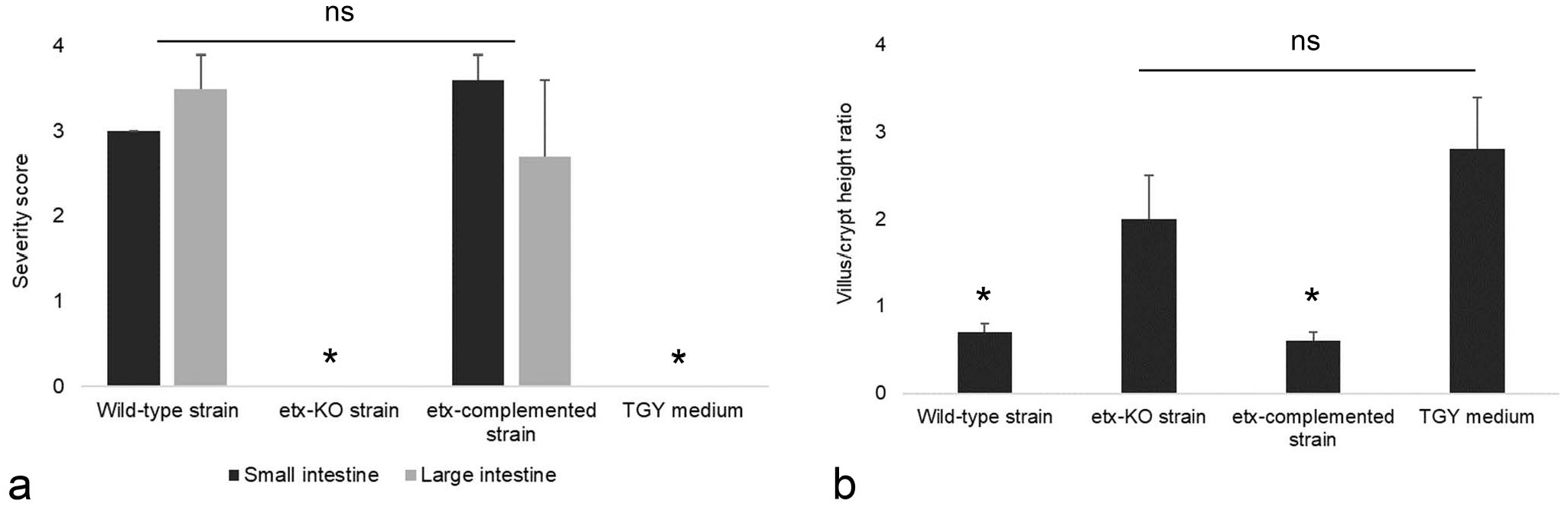
Microscopic evaluation of small and large intestine of goats inoculated with C. perfringens type D wild-type strain CN1020, its isogenic etx null mutant JIR4081 (etx-KO), its complemented derivative JIR12604 (etx-complemented) or TGY, sterile, non-toxic medium (control). (a) Overall severity score. Significant differences (P < .05) between the small and large intestine of goats inoculated with both ETX-producing strains versus the corresponding intestinal sections of goats inoculated with etx-KO or TGY are indicated with asterisks. (b) Score of villous/crypt height ratio in the small intestine. Significant differences (P < .05) between both ETX-producing strains versus etx-KO strain and TGY are indicated with asterisks. ns: not significant.
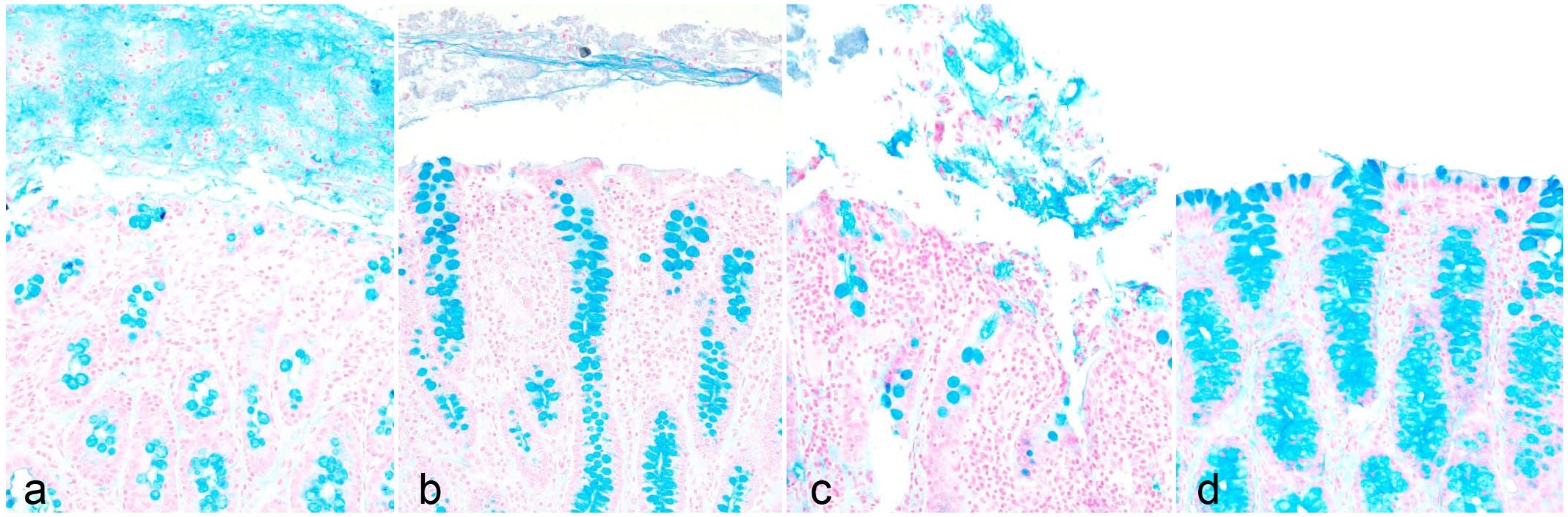
Evaluation of mucus secretion in the large intestine of goats inoculated with C. perfringens type D. Alcian blue. (a) C. perfringens type D wild-type strain CN1020. Excess of free mucus mixed with desquamated epithelial cells in the lumen, and empty goblet cells in the mucosa. (b) C. perfringens type D etx null mutant JIR4081 (etx-KO). Mild amount of free mucus in the lumen. (c) C. perfringens type D etx-complemented strain JIR12604 (etx-complemented). Moderate amount of free mucus mixed with desquamated epithelial cells in the lumen, and empty goblet cells in the mucosa. (d) TGY sterile, non-toxic medium (control). Absence of free mucus in the lumen.
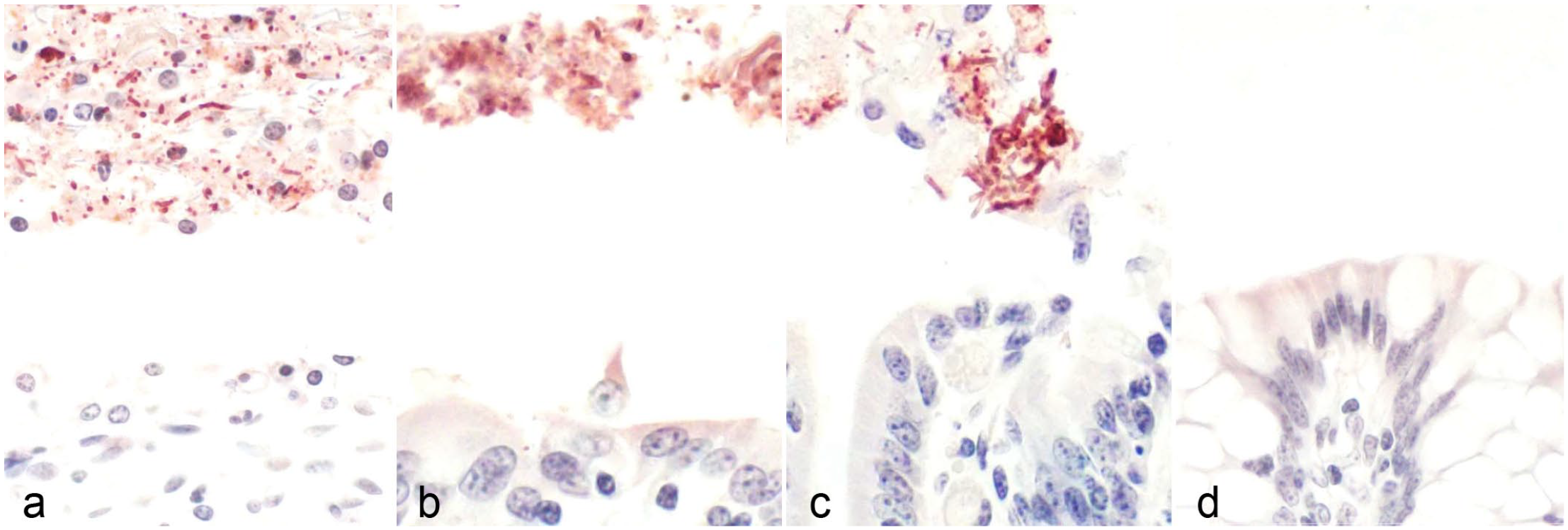
Immunohistochemical evaluation of the large intestine of goats inoculated with C. perfringens type D. C. perfringens immunohistochemistry. (a) C. perfringens type D wild-type strain CN1020. Positive rods present in association with desquamated epithelial cells in the lumen. (b) C. perfringens type D etx null mutant JIR4081(etx-KO). Positive rods free in the lumen. (c) C. perfringens type D etx-complemented strain JIR12604 (etx-complemented). Positive rods present in association with desquamated epithelial cells in the lumen. (d) TGY sterile, non-toxic medium (control). Absence of positive rods.
A variable number of gram-positive rods, many of which immunolabeled by C. perfringens IHC, were present in the lumen of the small intestine, in close association with the surface of the mucosa in WT, complemented, and KO strains-inoculated animals (Table 3). No gram-positive or C. perfringens IHC-immunolabeled rods were found in the small intestine of goats inoculated with TGY alone (Table 3). A small amount of fibrin was seen in the lumen of PTAH-stained sections of small intestine of WT strain-inoculated animals (Table 3).
The comparative results of IHC for activated caspase-3 in the small intestine and colon of goats challenged with the different inocula are presented in Table 3. Positivity for activated caspase-3 IHC was observed in enterocytes free in the lumen and those still attached to the basement membrane (Fig. 6). The labeling for activated caspase-3 was observed in the nucleus and cytoplasm, but it was stronger in the former. The number of activated caspase-3 positive cells in the small intestine and colon of goats inoculated with WT and complemented mutant strains was higher (P < .05) than in goats inoculated with non–ETX-containing inocula (Fig. 7). The immunolabeling for activated caspase-3 was higher (P < .05) in the small intestine than in the colon of goats inoculated with the complemented mutant strain, but no significant differences in activated caspase-3 immunolabeling were observed between the small intestine and colon of goats inoculated with the WT strain (Fig.7).
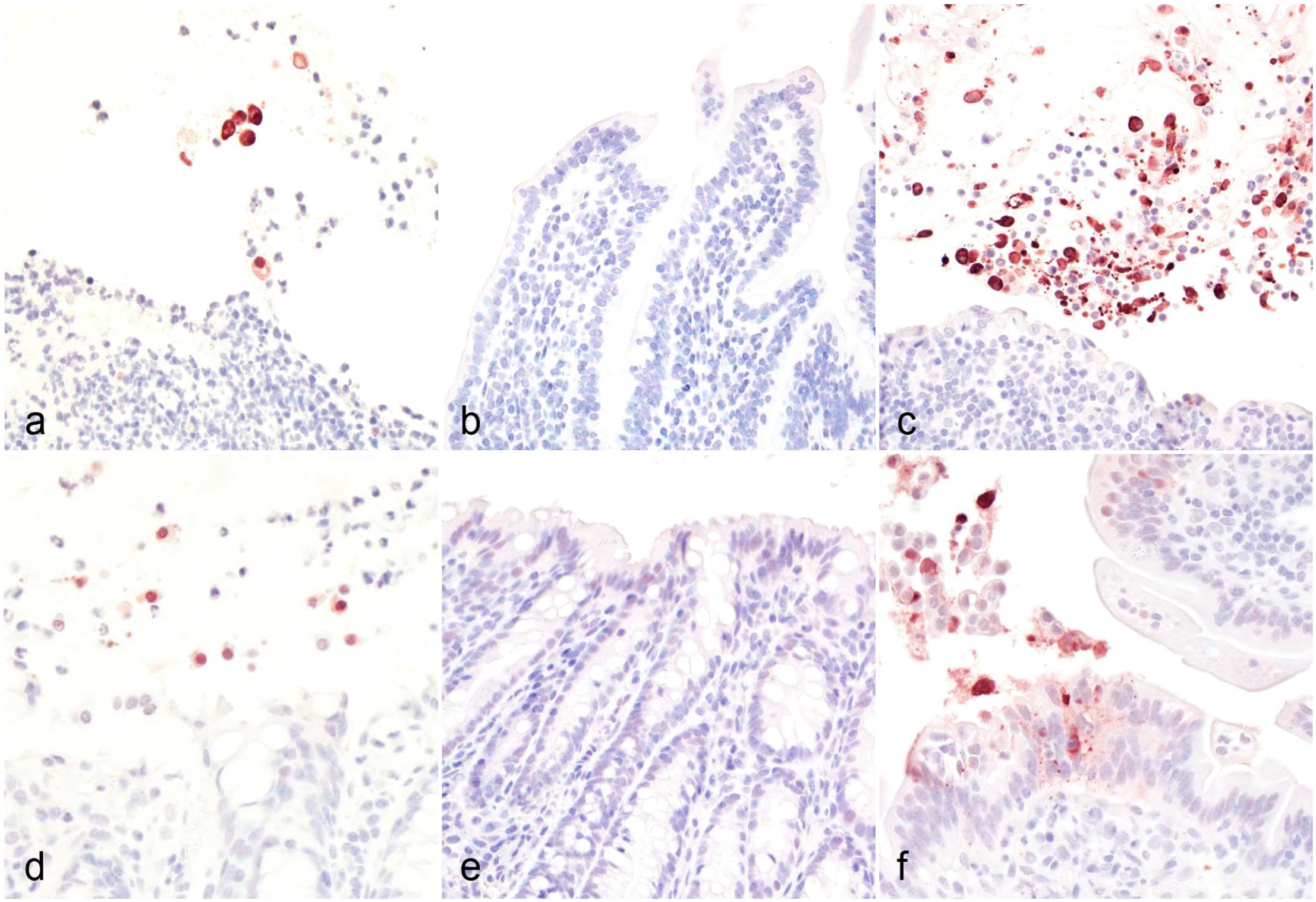
Immunohistochemistry (IHC) for activated caspase-3 of the (a, b, c) small and (d, e, f) large intestine of goats inoculated with (a, d) C. perfringens type D wild-type strain CN1020, (b, e) its isogenic etx null mutant JIR4081 (etx-KO), or (c, f) its complemented derivative JIR12604 (etx-complemented).
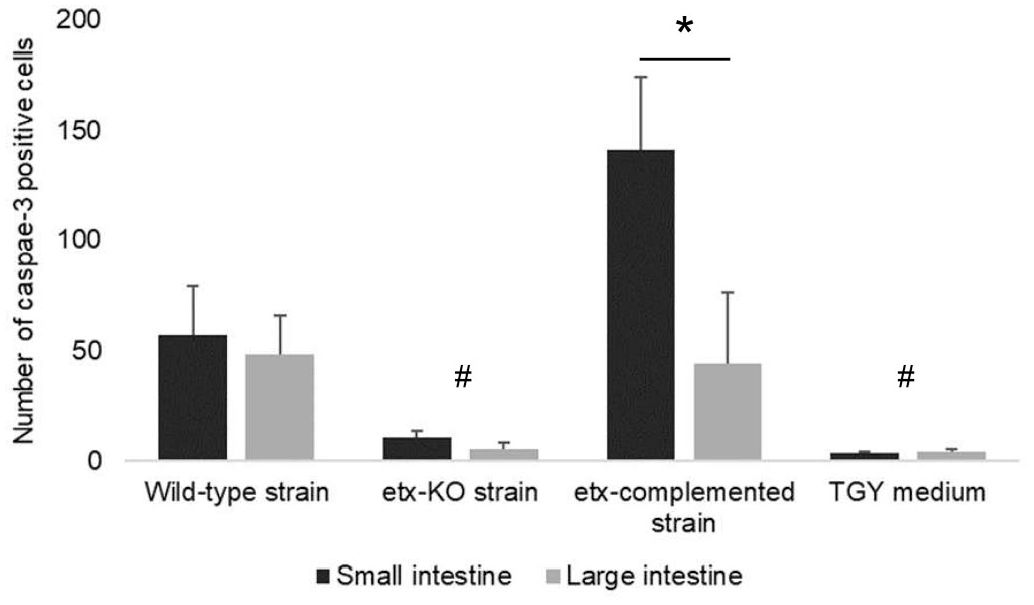
Quantification of activated caspase-3 positive cells by immunohistochemistry in the small and large intestine of goats inoculated with C. perfringens type D wild-type strain CN1020; its isogenic etx null mutant JIR4081 (etx-KO); its complemented derivative JIR12604 (etx-complemented); or TGY, sterile, non-toxic medium (control). Significant differences (P < .05) between etx KO strain and TGY versus both ETX- producing strains are indicating with #. Significant differences between the small and large intestine of goats inoculated with the etx-complemented strain are indicated with an asterisk.
Discussion
Most of the clinical, gross, and microscopic changes observed in naturally occurring cases of enterotoxemia by C. perfringens type D have been reproduced in sheep and goats by intraduodenal inoculation of cultures of this microorganism30,33 or ETX.10,11 However, the pathogenesis of this disease in goats remains to be fully elucidated. In our study, lesions were observed in the small intestine and colon of all goats inoculated with the ETX-producing strains, but not in those inoculated with non–ETX-containing inocula. These results, coupled with those of a previous study that fulfilled the so-called molecular Koch’s postulates, 11 confirm that ETX is essential for the pathogenesis of intestinal lesions in type D caprine enterotoxemia.
The most consistent lesion described in spontaneous cases of type D enterotoxemia in goats is fibrinonecrotizing (pseudomembranous) colitis, with a few animals showing enterocolitis, and rarely only enteritis.4,31,34 In this study, all animals inoculated with ETX-producing strains had enterocolitis and no significant differences in the overall severity of lesions were observed between the small intestine and colon of goats challenged with either ETX-producing strain. This is different from most published reports on spontaneous caprine enterotoxemia in which the most prevalent lesion is colitis.31,34 It is possible that the high prevalence of lesions in the small intestine in these cases was associated with the artificial route of inoculation (intraduodenal) and the large dose of inoculum used.
Several studies have been performed trying to explain the differences in the pathogenesis of type D enterotoxemia between sheep and goats.10,11,30,32,33 However, the reason why severe lesions are usually observed in the intestine of goats with type D enterotoxemia but not in sheep with the same disease is not known. Only a few cell lines are sensitive to the effects of ETX, suggesting the existence of specific ETX receptors on those cells.1,21,25,26 It is possible that those still unidentified receptors are also present in different concentrations in the small intestinal and colonic caprine enterocytes. ETX can bind to mouse enterocytes; however, it remains unclear if the toxin can bind to enterocytes of other species. 16 Differential binding specificity between species might explain the difference in lesions observed in the intestine of sheep and goats with type D enterotoxemia. Myelin and lymphocyte protein (MAL) is a receptor for ETX that has been found in multiple tissues of mice and several cell lines, and it has been suggested that MAL is required for both ETX binding and cytotoxicity.14,27 However, no information is available about the presence and distribution of this protein in tissues of ruminants. It is possible that MAL is present in the intestine of goats but not sheep. Although it is likely that host factors are responsible for the difference in lesions seen in ovine and caprine enterotoxemia, we cannot rule out that strain-associated factors also contribute to this variation in lesions. Comparing the genetic arsenal of type D strains isolated from cases of caprine and ovine enterotoxemia might help to elucidate this.
Goblet cells are modified epithelial cells that secret mucus, which forms a protective gel-like layer over the intestinal epithelium and protects against bacterial invasion. 19 In this study, most of the goblet cells in the colon of goats inoculated with the ETX-producing strains were empty, having discharged their mucus content into the lumen. This agrees with previous reports of experimental and spontaneous type D enterotoxemia,10,30 and it is believed to be a non-specific change associated with increased mucus secretion, which is stimulated by the presence of pathogenic bacteria, pro-inflammatory cytokines, or others irritants. 19 In this case, since these cells were replete with mucus in the colon of goats inoculated with non–ETX-containing inocula, it is reasonable to assume that factors other than C. perfringens (the organism itself) were responsible for this difference. ETX was most likely responsible for the emptying of the goblet cells in goats inoculated with ETX strains. Cytokines produced in response to the effect of ETX in the intestine may have also contributed to the emptying of goblet cells.
In vitro, some C. perfringens type D strains produce sialidases (NanH, NanI, and/or NanJ) that promote the adhesion of the bacteria to the enterocytes increasing the sensitivity of these cells to ETX. 20 The mechanism by which sialidases promote adhesion is not fully understood, but it has been suggested that some sialidases (particularly NanI) might increase the exposure of ETX receptors on the host cell surface to this toxin and might modify the host cell surface charge to increase toxin binding and cytotoxicity. 20
Apoptosis helps maintaining the homeostasis of cell populations in tissues, but it can also be triggered by diverse stimuli, including tumor necrosis factor, bacterial lipopolysaccharide, ischemia-reperfusion injury, trauma, parasitic infections, and several toxins. 35 A number of studies have associated pathological intestinal cell shedding with the activation of caspases,17,22,23,36 which are among the main components of the apoptosis cascade. In this study, we found strong immunolabeling for activated caspase-3 in epithelial cells of the small and large intestine of goats challenged with both ETX-producing strains. The number of apoptotic cells positively labeled for activated caspase-3 was higher in the small intestine than in the colon of goats challenged with the ETX-complemented strain (Fig. 6b), suggesting that ETX-related apoptosis is more prevalent in the small intestine than in the colon. This however, must be considered cautiously as these animals were inoculated in the small intestine with large doses of bacteria, something that is unlike to occur in natural cases of enterotoxemia. Studies performed in vitro demonstrated that ETX binds to specific receptors on the Madin-Darby Canine Kidney (MDCK) cell surface and form large pores in the plasma membrane causing rapid loss of intracellular K+ and an increase of Cl− and Na+, whereas the increase in Ca++ occurs later.25,29 This seems to be the first event leading to cell death by necrosis and is apparently followed by adenosine triphosphate (ATP) depletion, adenosine monophosphate (AMP)-activated protein kinase stimulation, mitochondrial membrane permeabilization, and mitochondrial-nuclear translocation of apoptosis-inducing factor, the latter being a potent caspase-independent cell death factor.6,25
In our study, significant activation of caspase-3 was demonstrated, indicating apoptotic cell death in many enterocytes. However, if ETX acts on intestinal epithelium in a fashion similar to that described in MDCK cells, it is reasonable to assume that necrosis also occurs in enterocytes exposed to ETX. Other clostridial toxins (eg C. perfringens enterotoxin) cause apoptosis in low doses, but necrosis 5 or necroptosis 28 when present in high concentration. Most goats inoculated with ETX-producing strains showed severe, acute inflammatory responses in the small and large intestine. ETX elicits a rapid cytokine response in mice and it is possible that a similar event occurs in goats. 12 The inflammatory response may also be a response to necrosis, because although significant apoptosis was observed in enterocytes, we cannot rule out that necrosis or pyroptosis also occurred given the histologic features observed in this study.
The score for gram-positive rods was lower than the score of the rods immunolabeled by C. perfringens IHC in the small and large intestine of goats inoculated with the WT, etx-complemented, or etx null mutant strains (Table 3). These results could be explained by the gram positivity variability that frequently occurs in several gram-positive bacteria, including clostridia, under stressful conditions. In this situation, the peptidoglycan of the cell wall is affected and the bacteria can turn gram-negative.2,3 However, the antigenic determinants are conserved and this could explain why more rods immunolabeled for C. perfringens IHC than those that were gram-positive.
The results of this study show that ETX is essential for the production of intestinal lesions in goats with type D enterotoxemia. In addition, our results indicate that in goats, apoptosis is a significant mechanism of intestinal cell death by ETX, in addition to necrosis (and/or necroptosis).
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241273122 – Supplemental material for Intestinal pathology in goats challenged with Clostridium perfringens type D strain CN1020 wild-type and its genetically modified derivatives
Supplemental material, sj-pdf-1-vet-10.1177_03009858241273122 for Intestinal pathology in goats challenged with Clostridium perfringens type D strain CN1020 wild-type and its genetically modified derivatives by Eleonora L. Morrell, Mauricio A. Navarro, Jorge P. Garcia, Juliann Beingesser and Francisco A. Uzal in Veterinary Pathology
Footnotes
Author Contributions
FAU and JB designed and performed the experiments. JPG contributed to the experimental design. ELM and MAN performed histologic evaluations. MAN performed statistical analysis. The manuscript was written by ELM, MN, and FAU with contribution from the other authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant R01 AI056177 from the National Institute of Allergy and Infectious Diseases (NIAID). ELM was supported by INTA, Argentina (Resol 1177/14). MAN was supported by ANID, Chile (Fondecyt Project 11230617).
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
